Introduction
Pleomorphic hyalinizing angiectatic tumors (PHATs)
were first described by Smith et al (1) in 1996, and are rare mesenchymal tumors
of uncertain lineage and intermediate malignancy (1–3). PHATs are
defined by the presence of clusters of variably-sized thin-walled
ectatic vessels that are infiltrated and surrounded by amorphous
fibrin-rich hyaline material (1,2). The
angiectatic vessels characteristic of PHATs are surrounded by
spindle-shaped, plump and round pleomorphic cells commonly arranged
in sheets or, more rarely, in fascicles (1). Often, a subset of cells adjacent to the
vessels contains intracytoplasmic hemosiderin. The pleomorphic
cellular population is composed of large single and multinucleate
cells with abundant cytoplasm, enlarged irregular nuclei and, in
certain cases, large intranuclear inclusions (1). Mitotic figures are rare (1). PHATs contain a mixed chronic
inflammatory infiltrate that is notable for the presence of mast
cells, but also includes lymphocytes, plasma cells and eosinophils
(1,3).
The main mass of a PHAT may be accompanied by a partially myxoid
spindle-cell component on the periphery, a potential precursor
lesion to a classical PHAT that was described as an early PHAT by
Folpe and Weiss in 2004 (2). The
first cytogenetic PHAT data published revealed an unbalanced
translocation of chromosomes 1 and 3 and chromosomes 1 and 10, with
breakpoints mapped to transforming growth factor-β receptor 3
(TGFBR3) and meningioma-expressed antigen 5 (MGEA5) (4). Subsequent fluorescence in situ
hybridization and single nucleotide polymorphism analyses of PHATs
demonstrated that some, but not all, are characterized by TGFBR3
and MGEA5 gene rearrangements (5–7).
There are few published reports describing the
imaging appearance of PHATs. A PHAT typically appears as a soft
tissue mass without osseous changes or calcification (8–10). In
cases in which magnetic resonance imaging (MRI) is utilized, the
PHAT presents as a soft-tissue mass that may be accompanied by
edema or hematoma. The PHAT appears hypointense-isointense on
T1-weighted sequences, heterogeneously isointense-hyperintense on
T2-weighted sequences, and exhibits enhancement following
administration of intravenous contrast (4,8,11–18). The
current study presents an assessment of two patients with similar
PHATs, and describes the key imaging, histological and
immunophenotypical findings of these tumors.
Case reports
Case A
A 50-year old male presented in 2016 to our
institution with an 18-month history of an enlarging right buttock
mass. The patient first noticed the mass following trauma and
subjectively considered that the mass grew to be the size of a
grapefruit. The patient denied experiencing any pain, except when
sleeping on the right side; at the time of presentation, the
patient was taking no pain medication and denied any radicular
symptoms, night pain or weight loss. The patient also had normal
muscle strength in the right lower extremity. A physical
examination revealed a palpable, non-tender mobile mass in the
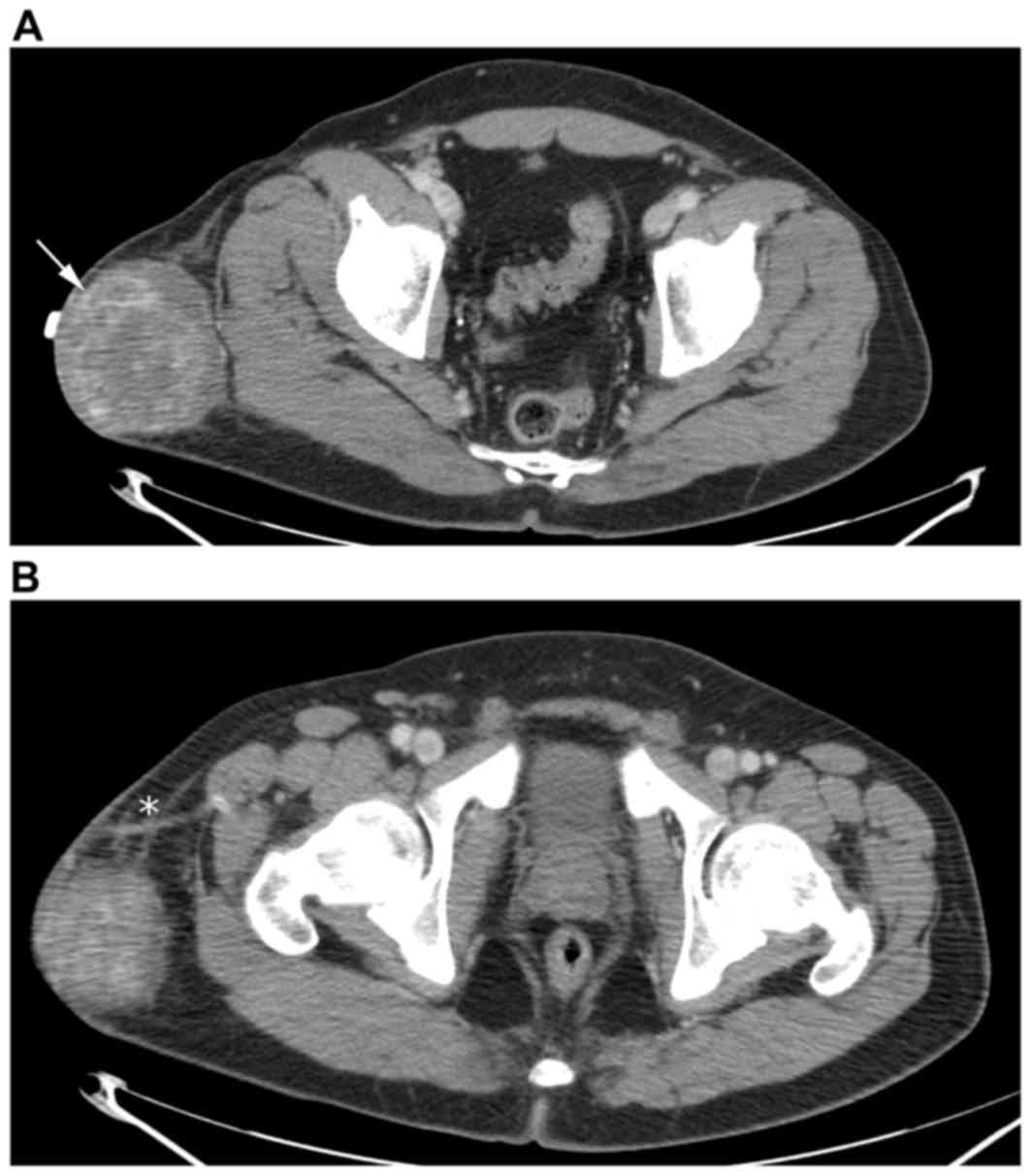
right buttock. A contrast-enhanced computed tomography (CT) scan of
the pelvis revealed an 8.0×7.7×8.6 cm heterogeneously enhancing
subcutaneous soft tissue mass overlying the right gluteus maximus
(Fig. 1A). There was a prominent
feeding artery noted as arising from the right profunda artery
(Fig. 1B). MRI was performed using a
Siemens Verio 3T MRI machine (Siemens AG, Munich, Germany).
T1-weighted sequences (repetition time (TR)/echo time (TE), 700/24
ms; slice thickness, 3 mm; interslice gap, 0.9 mm; acquisition
matrix, 448×336); short-tau inversion recovery (STIR) sequences
(TR/TE, 3400/48 ms; slice thickness, 3 mm; interslice gap, 0.9 mm;
and acquisition matrix, 256×192); and T1-weighted contrast-enhanced
sequences with fat saturation (TR/TE, 638/23 ms; slice thickness, 4
mm; interslice gap, 0.4 mm; and acquisition matrix 320×256) were
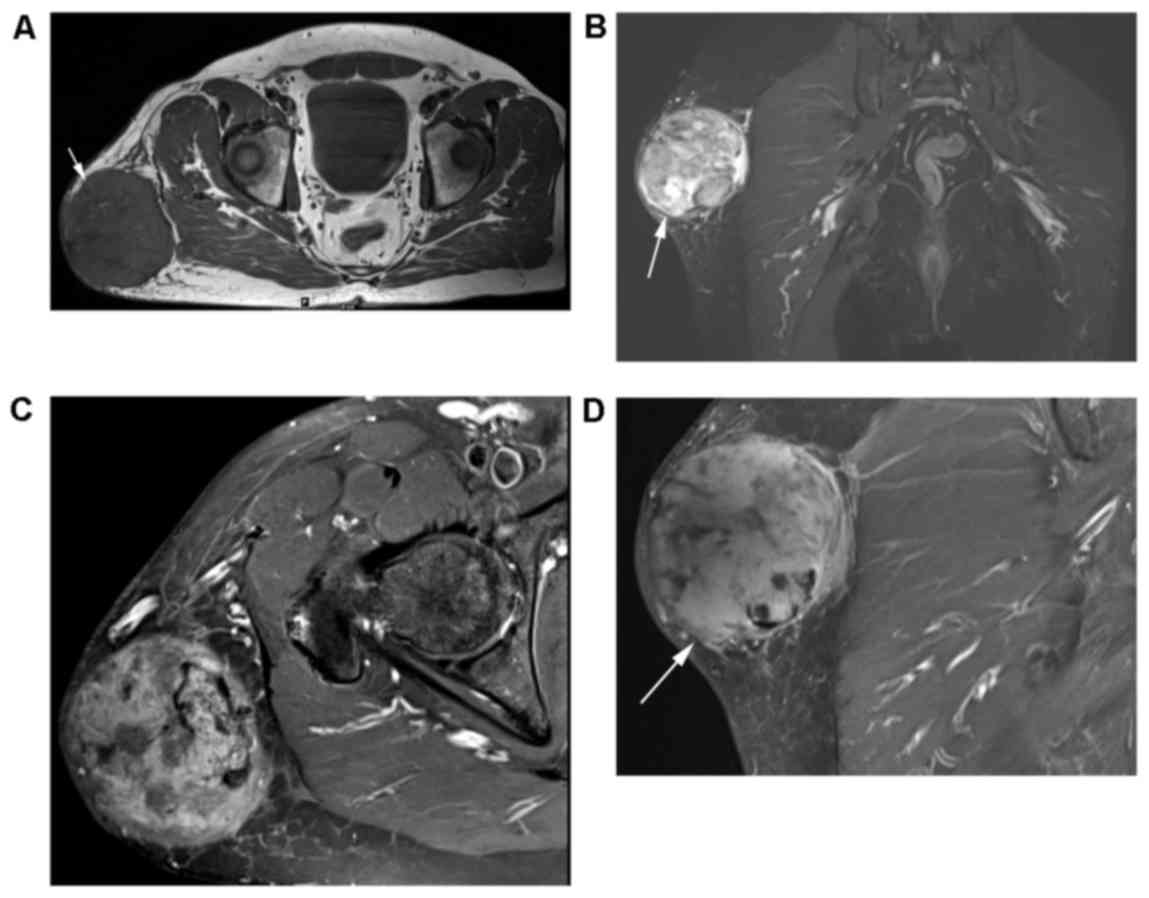
obtained. T1-weighted imaging revealed the presence of a
heterogeneous predominantly isointense mass (Fig. 2A) with multiple areas of T1 signal
hyperintensity (possibly representing areas of hemorrhage) and
multiple areas of T1 signal hypointensity. The lesion was
superficial to the right gluteus maximus fascia and present in the
subcutaneous tissues (Fig. 2B).
Multiple flow voids consistent with small arteries were observed in
the lesion (Fig. 2C). There was no
lymphadenopathy and no osseous involvement. The mass exhibited
heterogeneous enhancement following administration of intravenous
contrast (Fig. 2D). The lesion was
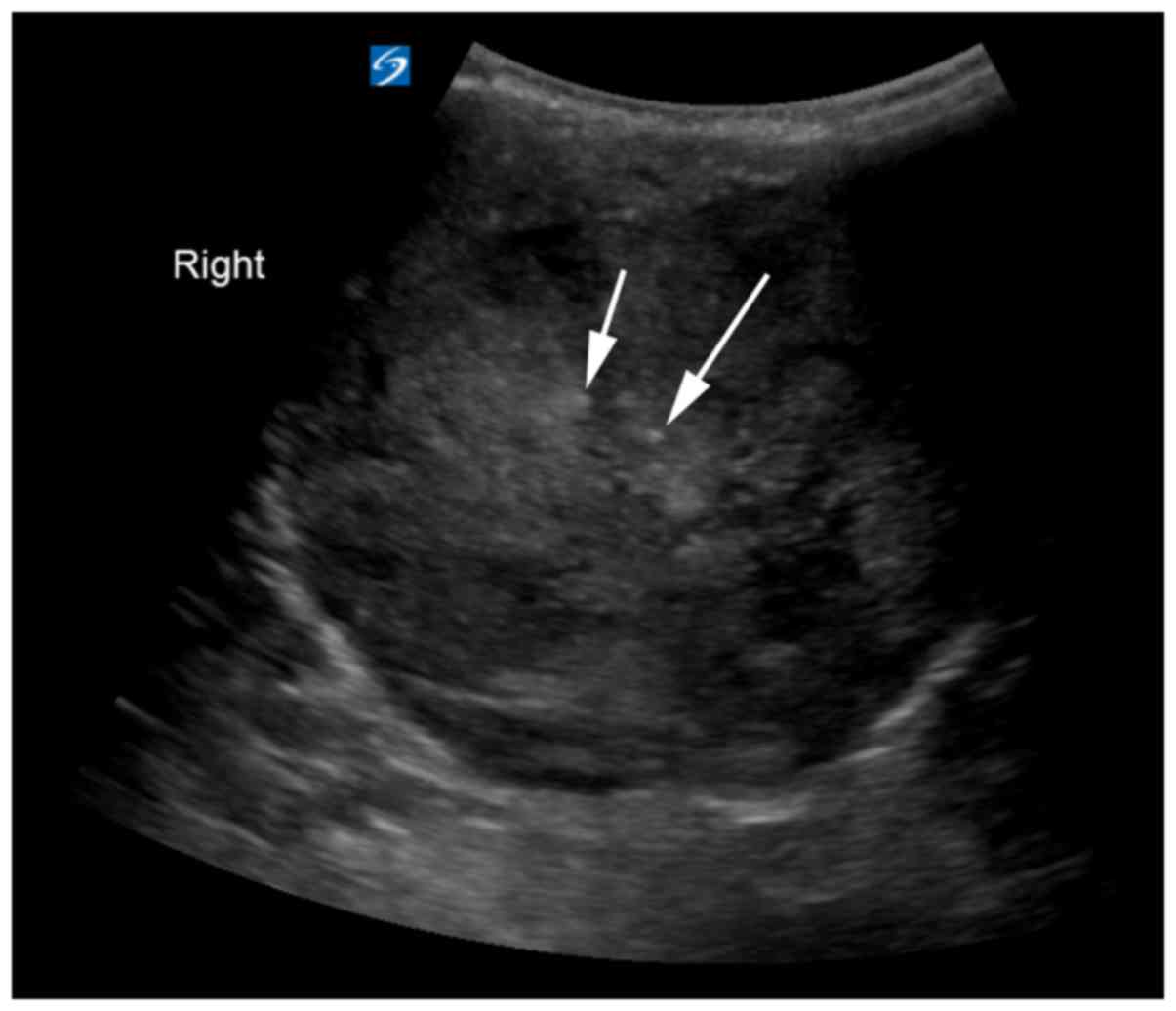
heterogeneously hypoechoic with internal globular and punctate
hyperechoic foci when evaluated by ultrasound (Fig. 3).
Histologic sections 5 µm thickness were
deparaffinized and stained at room temperature with hematoxylin (5
min) and eosin (40 sec) on a Tissue Tek Prisma Automatic Stainer
(Sakura Finetek USA, Inc., Torrance, CA, USA). On examination with
light microscopy (variable objectives, magnification range
25–400×), the sections revealed a spindle cell lesion with sparsely
sampled, hyalinized vessels. The cellular component was largely
bland and spindled, with intermixed hemosiderin-laden macrophages
and scattered large, atypical cells. Immunohistochemistry on
formalin-fixed, paraffin-embedded tissue was performed using a
Leica Bond-IIITM instrument using the Bond Polymer Refine Detection
System according to the manufacturer's protocols (DS9800; Leica
Microsystems, Inc., Buffalo Grove, IL, USA). Heat-induced epitope
retrieval was performed for 20 min with ER1 or ER2 solution (Leica
Biosystems) according to the manufacturer's protocols. The tissues
were then incubated with the following primary antibodies at room
temperature for 15 min: CD34 (ready to use dilution; cat no.
PA0354; Leica Biosystems GmbH, Wetzlar, Germany), pancytokeratin
(dilution 1:75; cat no. MU357-UC; BioGenex Laboratories, Inc., San
Ramon, CA, USA), S100 (ready to use dilution; cat no. IR50461;
Dako; Agilent Technologies, Inc., Santa Clara, CA, USA), smooth
muscle actin (dilution, 1:600; cat no. M0851; Dako), epithelial
membrane antigen (dilution, 1:100, cat no. M0613, Dako) and
collagen IV (dilution 1:100; cat no. M0785; Dako). All
interpretation of immunohistochemical stains was conducted with
routine light-microscopy (variable magnification, ×25-400). On
initial biopsies, immunohistochemical studies were inconclusive
with respect to line of differentiation. No high-grade features
were seen. Given the small sample size and the lack of definitive
histological features, the non-specific diagnosis of ‘spindle-cell
neoplasm’ was determined, with a differential diagnosis that
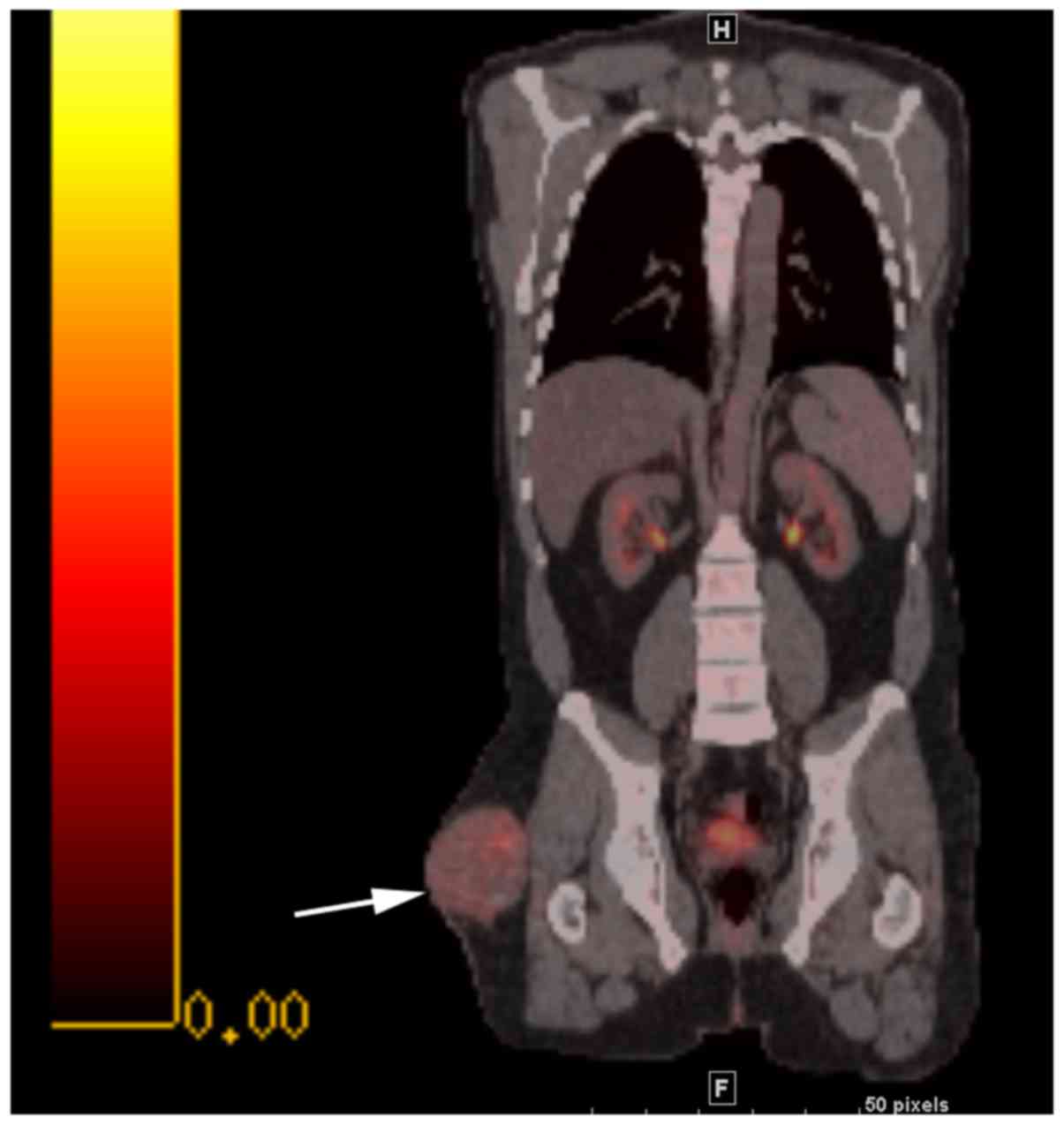
included PHAT. Positron-emission tomography/computed tomography
(PET/CT) demonstrated a mildly hypermetabolic lesion (maximum
standardized uptake value of 4.2) with no evidence of
hypermetabolic metastases (Fig. 4).
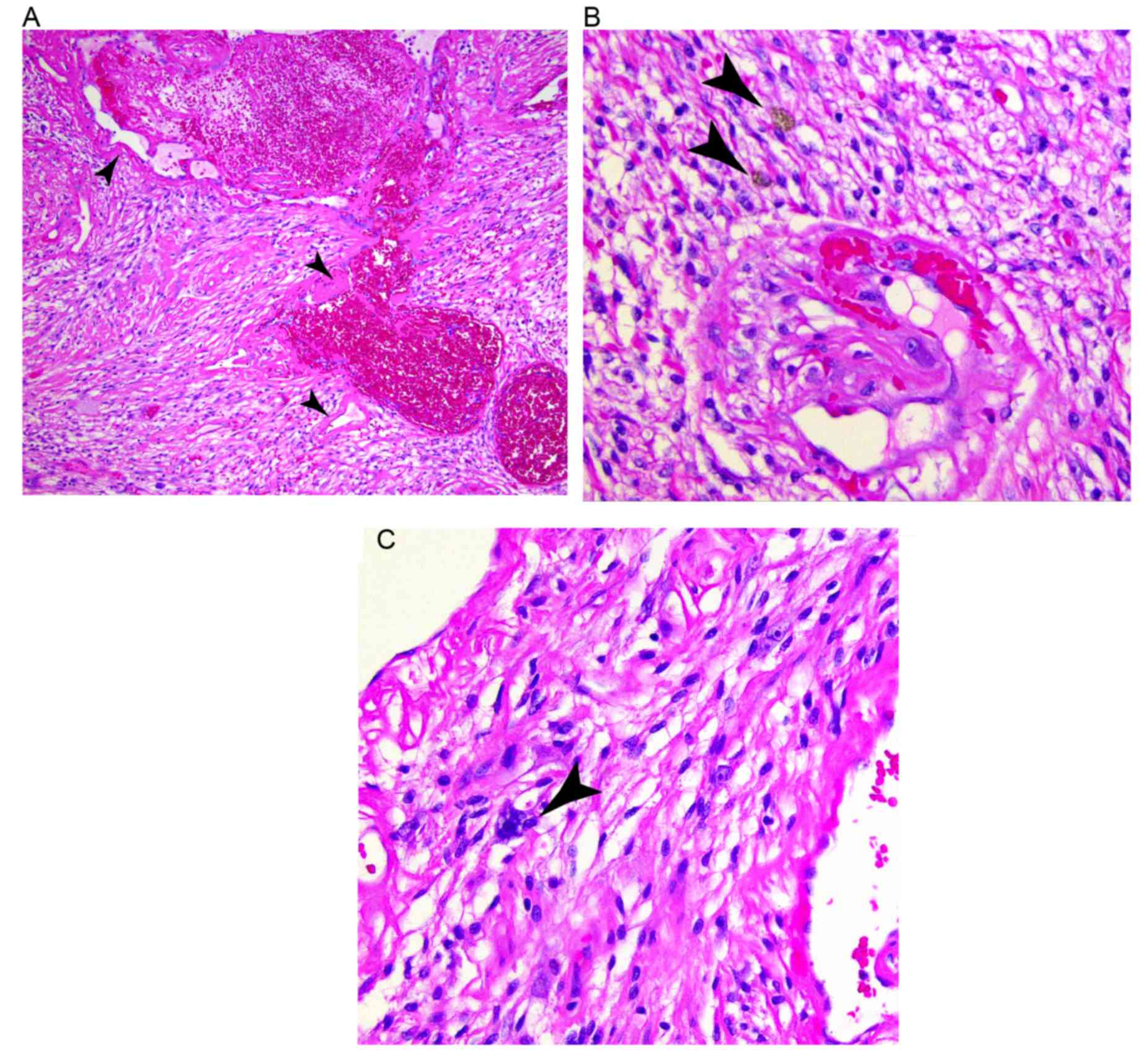
The lesion was resected with wide surgical margins. The excised
specimen contained a circumscribed tumor with prominent
thin-walled, hyalinized vessels (Fig.
5). There were areas of hemorrhage and vascular thrombosis. An
intermediate density of bland ovoid cells with rounded nuclear
contour and occasional small nucleoli, along with admixed
hemosiderin-laden cells and scattered large single- and
multi-nucleated atypical cells was also observed. Mitotic activity
was low (<1/10 mitoses per high-power field). On the basis of
these morphological findings, the patient was diagnosed with
PHAT.
Case B
A 72-year-old male presented to our institution in
2013 with a mass along the right buttock that had been slowly
growing for almost 30 years. The mass was mildly uncomfortable when
the patient sat. The patient also reported no unintentional weight
loss or lymphadenopathy. A physical examination revealed a
palpable, mobile, non-tender mass measuring approximately 10 cm in
size. MRI was performed using a Siemens Espree 3T MRI (Siemens AG).
T2-weighted sequences (TR/TE, 6,721/167 ms; slice thickness, 5 mm;
interslice gap, 1 mm; and acquisition matrix, 320×320) and
T1-weighted gradient imaging with fat saturation following
administration of intravenous contrast (TR/TE, 170/2.37 ms; slice
thickness, 5 mm; interslice gap, 1 mm; and acquisition matrix,
256×218) were obtained. The lesion was superficial to the fascia of
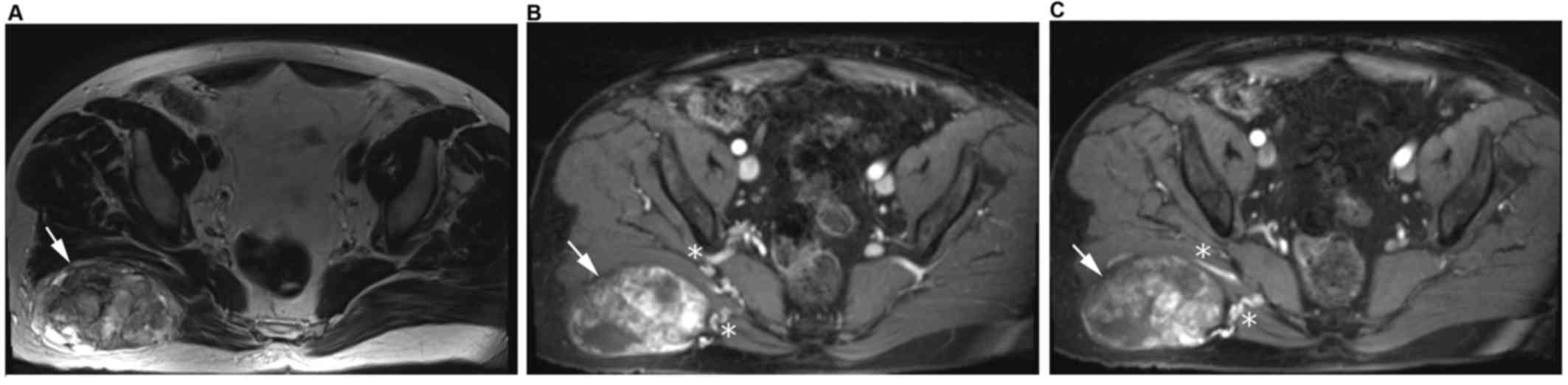
the right gluteus maximus and measured ~6.3×10.4×10.2 cm. The
lesion exhibited a heterogeneously hyperintense T2 intensity signal
(Fig. 6A) and heterogeneous
enhancement following administration of intravenous contrast; there
were two prominent arterial vessels originating from the right
internal iliac artery supplying the lesion (Fig. 6B and C). The patient underwent gross
total resection of the tumor with negative margins. The
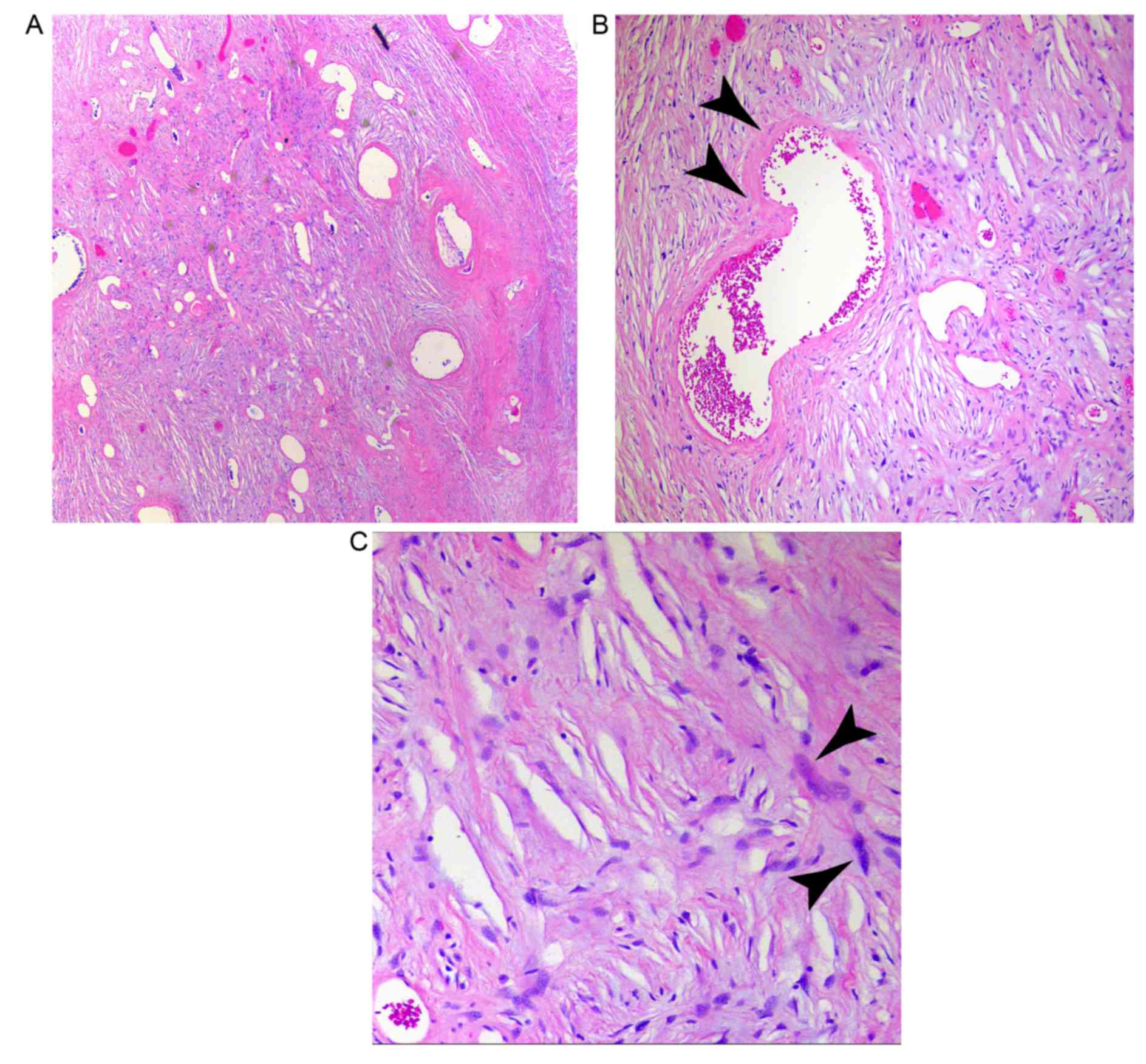
histological findings were similar to those in case A, although
this patient had reduced cellularity and more prominent areas of
myxoid stroma (Fig. 7). As in case A,
the tumor was composed of a mixture of bland spindle cells with
occasional large irregular cells that had retained cytoplasm and
large atypical nuclei. There was no discernable mitotic activity or
necrosis (Fig. 7). In the
immunohistochemical studies, the spindled tumor cells were positive
for cluster of differentiation CD34 expression, focally positive
for pancytokeratin expression, and negative for the expression of
S100, smooth muscle actin, epithelial membrane antigen and collagen
IV. The final histological diagnosis was a PHAT.
The requirement for patient consent for publication
of the study was waived and ethical approval was provided by the
Institutional Review Board of the University of Pennsylvania
(Philadelphia, PA, USA).
Discussion
To date, <100 cases of PHAT have been reported
worldwide (14,19). The World Health Organization currently
classifies PHAT as a benign neoplasm of uncertain differentiation
(3). No reports of metastatic PHATs
exist; however, it has been known to recur as sarcoma in rare cases
(2,20). Owing to its locally aggressive
behavior and high recurrence rate (33–50%), it has been argued that
a PHAT should be classified as a mesenchymal tumor of intermediate
malignancy (2).
PHAT was first described by Smith et al
(1) in 1996. The etiology of PHAT is
not well understood and a number of competing theories persist
regarding the formation of classic PHAT (1–3). The
original description of the tumor by Smith et al (1) posited that the encroachment of a
developing PHAT onto the vessels of healthy tissue leads to the
release of the plasma contents of damaged endothelial cells, and
precipitates a process of organization that results in the tumor's
characteristic perivascular hyalinization (1). Smith et al (1) further proposed that the release of
vasoactive mast cell products in response to tissue injury from a
developing PHAT may be a cause of increased vessel permeability
during development (1). Alternately,
Folpe and Weiss (2) argue that early
PHAT, characterized by low-medium spindle cell density, rare
pleomorphic cells and myxoid extracellular matrix, is a precursor
lesion to classic PHAT (2). PHAT
typically presents in the subcutaneous tissues of the lower
extremities, but may also develop intramuscularly, and has been
documented in the arm, axilla, back, buccal mucosa, buttock, chest,
hand, perineum, popliteal fossa and retroperitoneum (2,3,13,21). The
most common locations of PHATs, based on the published literature,
include the leg (18/97; 18.6%), thigh (17/97; 17.5%), foot (14/97;
14.4%) and the ankle (10/97; 10.3%) (1–41). PHAT
typically presents in adults; the cases presented in the published
literature (97 patients) range in age from 10–89 years, with a
median age of 57. PHAT demonstrates a slight predilection for
females over males; the literature reviewed in the present study
demonstrates a male to female ratio of 3:4 out of 97 reported cases
(2,22,23). No
information is currently available concerning race and, as <10%
of reported cases include this metric, no claims can be made
regarding a predilection. Table I
presents a list of all previously published cases of PHAT,
including data on age, sex, race, tumor size, location and
treatment (1,2,4,5,8–41).
 | Table I.List of published studies of
PHAT. |
Table I.
List of published studies of
PHAT.
| Author, year | Sex | Age | Race | Location | Treatment | Imaging | Size, cm | Pre-operative
duration | (Refs.) |
|---|
| Smith et al,
1996 | M | 33 | NR | Shoulder | Wide excision +
XRT | NR | 3.5 | NR | (1) |
|
| F | 78 | NR | Leg | Local excision then
amputation | NR | 8 | NR |
|
|
| F | 62 | NR | Chest wall | Simple excision +
XRT | NR | 2.5 | NR |
|
|
| F | 33 | NR | Ankle | Excision + XRT | NR | 5.5 | NR |
|
|
| M | 70 | NR | Thigh | Simple
excision | NR | 4.5 | NR |
|
|
| F | 32 | NR | Leg | Simple
excision | NR | NR | NR |
|
|
| F | 59 | NR | Thigh | Anucleation | NR | 6 | NR |
|
|
| M | 72 | NR | Thigh | Simple
excision | NR | 4.5 | NR |
|
|
| M | 60 | NR | Thigh | Excision | NR | 6 | NR |
|
|
| F | 58 | NR | Chest wall | NR | NR | ≥2 | NR |
|
|
| F | 78 | NR | Arm | Simple
excision | NR | Recurrence, 6 | NR |
|
|
| M | 57 | NR | Buttock | Excision | NR | NR | NR |
|
|
| M | 83 | NR | Thigh | Excision | NR | 2.3 | NR |
|
|
| F | 51 | NR | Ankle | Excision | NR | Recurrence,
≥2.5 | NR |
|
| Folpe and Weiss,
2004 | F | 39 | NR | Hand | NA | NR | NR | Unknown | (2) |
|
| F | 30 | NR | Ankle | Local excision | NR | NR | Unknown |
|
|
| M | 42 | NR | Forearm | Wide excision and
XRT | NR | NR | Many years |
|
|
| F | 46 | NR | Foot | Wide excision | NR | 13 | Unknown |
|
|
| M | 10 | NR | Foot | Wide excision | NR | NR | 1 year |
|
|
| F | 51 | NR | Ankle | Local excision,
then wide excision and XRT | NR | NR | Unknown |
|
|
| F | 44 | NR | Ankle | Local excision | NR | ‘Large’ | Unknown |
|
|
| M | 67 | NR | Leg | NA | NR | 4 | Unknown |
|
|
| F | 32 | NR | Leg | NA | NR | 6 | Unknown |
|
|
| M | 36 | NR | Thigh | Local excision | NR | 7 | Unknown |
|
|
| F | 50 | NR | Ankle | Local excision | NR | 2.5 | 2 months |
|
|
| F | 53 | NR | Foot | NA | NR | 7 | Unknown |
|
|
| F | 84 | NR | Leg | NA | NR | 0.3 | Unknown |
|
|
| F | 57 | NR | Foot | Wide excision | NR | 10.2 | 16 years |
|
|
| F | 49 | NR | Leg | Local excision | NR | NR | Unknown |
|
|
| M | 56 | NR | Axilla | NA | NR | NR | Unknown |
|
|
| M | 49 | NR | Thigh | Local excision | NR | NR | Unknown |
|
|
| M | 79 | NR | Pelvis | Local excision,
then wide excision and XRT | NR | 14 | Unknown |
|
|
| M | 60 | NR | Ankle | NA | NR | 5 | 2–3 years |
|
|
| M | 27 | NR | Thigh | NA | NR | 5.6 | Many years |
|
|
| M | 66 | NR | Ankle | Local excision,
then wide excision and XRT | NR | 7 | 6 months |
|
|
| M | 54 | NR | Foot | Local excision
followed by amputation following recurrence | NR | 3.2 | Unknown |
|
|
| F | 32 | NR | Leg | Local excision and
re-excision | NR | 5 | 2 years |
|
|
| F | 46 | NR | Leg | Local excision and
re-excision | NR | 2 | Unknown |
|
|
| F | 77 | NR | Thigh | Local excision and
re-excision | NR | 14 | Unknown |
|
|
| F | 79 | NR | Foot | Wide excision | NR | 1.8 | Unknown |
|
|
| F | 77 | NR | Thigh | NA | NR | 4.2 | 3 months |
|
|
| M | 67 | NR | Arm | NA | NR | 5.8 | Unknown |
|
|
| M | 89 | NR | Leg | Local excision and
re-excision | NR | 3.5 | Unknown |
|
|
| M | 74 | NR | Buttock | Local excision and
re-excision | NR | 5.5 | Unknown |
|
|
| F | 75 | NR | Thigh | NA | NR | NR | 2 years |
|
|
| M | 49 | NR | Inguinal | NA | NR | 4 | Many years |
|
|
| M | 63 | NR | Inguinal | NA | NR | 11 | 3 years |
|
|
| F | 38 | NR | Back | NA | NR | 3 | Unknown |
|
|
| F | 51 | NR | Foot | NA | NR | 6.5 | Unknown |
|
|
| M | 32 | NR | Arm | NA | NR | 6 | Unknown |
|
|
| M | 44 | NR | Buttock | Local excision and
re-excision | NR | ‘Large’ | 11 years |
|
|
| F | 67 | NR | Ankle | Local excision and
re-excision | NR | 6.1 | Unknown |
|
|
| F | 59 | NR | Leg | Local excision,
then wide excision and XRT | NR | 6.5 | Unknown |
|
|
| F | 42 | NR | Foot | Local excision
followed by wide excision following recurrence | NR | 2 | Unknown |
|
|
| M | 43 | NR | Leg | Wide excision | NR | 20 | 20 years |
|
| Wei et al,
2012 | F | 37 | Caucasian | Foot | Surgical resection
followed by two subsequent re-excisions and external beam radiation
to a total of 60 Gy in 30 fractions for recurrences | MRI | 5 | NR | (4) |
| Mohajeri et
al, 2012 | F | 58 | NR | Foot | Surgical excision
with a marginal margin | NR | 0.3×1.5×1.5 | 1 year | (5) |
|
| M | 33 | NR | Thigh | Surgical excision
with a narrow margin | NR | 0.5×3.5×2.5 | NR |
|
| Suzuki et
al, 2014 | F | 68 | NR | Thigh | Wide resection was
performed for the internal lesion, whereas an intralesional
resection was performed for the outer lesion | Radiography,
MRI | 10.5×4.5×3.5 | 2 months | (8) |
| Cimino-Mathews and
Ali, 2011 | M | 46 | NR | Leg | Wide local surgical
excision | Radiograph | 3×1×1 | 2 months | (9) |
| Illueca et
al, 2012 | F | 37 | NR | Ankle | Surgical
excision | X-ray | 3.5×2×1.5 | 2 months | (10) |
| Lin and Crapanzano,
2005 | F | 45 | NR | Thigh | Surgical
excision | MRI | 3.5×2.8×1.5 | 3 months | (11) |
| Subhawong et
al, 2012 | M | 46 | Hispanic | Leg | Radical
excision | MRI | 1.9×1.7×2.1 | NR | (12) |
|
| F | 49 | African
American | Foot | Radical
resection | MRI | 9.5×3.4×7.0 | 4 years |
|
|
| F | 87 | NR | Arm | Surgical
resection | MRI | 4.8×4.6×5.8 | 3 years |
|
| Chu et al,
2016 | F | 26 | NR | Pelvis | Radical
excision | MRI, CT | 9.4×9.1×9.5 | Unknown | (13) |
| Brazio et
al, 2016 | M | 22 | NR | Forearm | Excise without wide
margins (to retain hand function) | MRI | 16.7×24.2×26 | 2 years | (14) |
| Felton and
Ramlogan, 2015 | M | 62 | NR | Leg | NR | MRI | 5×6 | 5 years | (15) |
| Lee et al,
2005 | M | 63 | NR | Breast | Total mastectomy
radiology | MRI, | 25.6×8.3×26.3 | 4 years | (16) |
| Fujiwara et
al, 2004 | M | 69 | Asian
(Japanese) | Back | Surgical
resection | MRI | 7 | 23 years | (17) |
| Fukunaga and
Ushigome, 1997 | M | 58 | Asian
(Japanese) | Axilla | Marginal
Excision | Ultrasound, MRI,
X-Ray | 8 | 2 months | (18) |
| Changchien et
al, 2014 | M | 76 | Hungarian | Arm | Surgical
removal | NR | 4.6×4.5×3 | 5 months | (19) |
| Kazakov et
al, 2007 | F | 76 | NR | Axilla | Surgical
excision | NR | 3.5 | Unknown | (20) |
| Ide et al,
2004 | F | 86 | NR | Buccal mucosa | Surgical
excision | NR | 3×3×2 | 2.5 years | (21) |
| El-Tal and
Mehregan, 2005 | F | 60 | NR | Foot | Surgical
excision | NR | 2.2×2.1×1.1 | NR | (22) |
| Matsumoto and
Yamamoto, 2002 | F | 83 | NR | Thigh | Surgical
resection | NR | 5.0×5.0×2.0 | 10 years | (23) |
| Tallarigo et
al, 2009 | M | 75 | NR | Breast | Radical mastectomy
and axillary node clearance | Mammograph,
ultrasound | 8 | Several years | (24) |
| Labanaris et
al, 2011 | M | 68 | NR | Scrotum | Inguinal radical
orchiectomy with high ligation of the spermatic cord | Ultrasound, CT | 18×15 | NR | (25) |
| Capovilla et
al, 2006 | M | 66 | NR | Buttock | Surgical
excision | NR | 4 | NR | (26) |
| Groisman et
al, 2000 | F | 41 | NR | Leg | Simple excision for
diagnosis then wide local excision | NR | NR | 3 years | (27) |
|
| F | 44 | NR | Leg | Simple excision for
diagnosis then wide local excision | NR | NR | 1 year |
|
| Gallo et al,
1997 | F | 88 | NA | Thigh | NA | NA | NA | NA | (28) |
| Silverman and Dana,
1997 | F | 59 | NR | Foot | Local Excision | NR | 2 | Unknown | (29) |
| Husek and Veselý,
2001 | M | 63 | NA | Forearm | Surgical
excision | NA | 4.0×3.5×2.5 | NA | (30) |
| Jaggon and Aitken,
2007 | F | 77 | NR | Flank | Wide local
excision | NR | 8×6 | 2 years | (31) |
| Parameshwarappa
et al, 2010 | F | 65 | NR | Forearm | Wide local
excision | NR | 5×4 | 13 years | (32) |
| Peng et al,
2010 | M | 49 | NR | Buttock | Surgical excision
followed by wide excision for recurrence | CT of
recurrence | 14×6×3.5 | NR | (33) |
| Idrees et
al, 2013 | F | 71 | Caucasian | Renal Hilum | Right radical
nephrectomy and limited retroperitoneal lymph node dissection | CT | 4.2×6 | NR | (34) |
| Kuang, 2013 | F | 35 | NR | Neck | Surgical
removal | CT | 3.4×4×5 | 6 months | (35) |
| Rekhi and Aggarwal,
2013 | F | 63 | NR | Leg | Surgical
resection | NR | 4×3×3 | 3–4 years | (36) |
| Fan et al,
2015 | F | 51 | NR | Chest wall | NR | NR | 2×1 | 1 year | (37) |
| Morency et
al, 2015 | F | 55 | NR | Foot | NA | NA | NA | NA | (38) |
| Yorita et
al, 2015 | M | 56 | Asian
(Japanese) | Thigh | Surgical
resection | CT | 6.5×3.8×3.5 | 8 months | (39) |
| Kane et al,
2016 | M | 35 | NA | Hand | Surgical
excision | NA | NA | NA | (40) |
| Onyemkpa and
Oyasiji, 2016 | F | 79 | African
American | Inguinal | Wide local | CT excision | 4.9×7.2×10.7 | 7 years | (41) |
PHAT often rises to medical attention following
several years of slow growth (1–3). Upon
clinical examination, PHAT typically presents grossly as an
oval-round lobulated mass that varies in color from tan to maroon,
and which may be associated with bruising or surrounding hematoma
(2,9,10). PHAT
may present as partially or predominantly cystic (1,12), may be
painless, painful or tender upon examination, and may be
accompanied by necrosis in rare cases (1,14).
The imaging appearance of PHAT is non-specific.
Typical evaluation of any soft-tissue mass includes radiography of
the region, followed by contrast-enhanced MRI evaluation and then
by biopsy if clinically warranted. Limited imaging studies of PHAT
exist in the literature and the majority of data are in the form of
single case reports. In cases in which radiographs are published,
there is consensus that PHAT typically appears as a soft tissue
mass without osseous changes or calcifications; however, a single
case with calcifications has been reported (8–10,14,24). To
the best of our knowledge, only three examples of the ultrasound
appearance of PHAT have been described in the literature (18,24,25). All
three describe PHAT as a hypoechoic lesion with an internal
hyperechogenic appearance, characterized by hyperechoic tissue
interrupted by hypoechoic bands, which are presumably blood vessels
(18,24,25). In
reports of PHAT using MRI, the tumor presents as a soft tissue mass
that may be accompanied by edema or hematoma (8,11–15). PHAT appears hypointense to isointense
with T1 weighting, heterogeneously hyperintense to isointense with
T2 weighting, and exhibits heterogeneous enhancement following the
intravenous administration of gadolinium-based contrast (4,8,11–13,16–18).
PHATs with well-demarcated and infiltrative borders have previously
been described in the literature (9,12). The two
cases presented in the current study exhibited prominent borderline
ectatic arterial vessels supplying the tumors and well-demarcated
tumor borders. The imaging differential diagnosis includes
subcutaneous metastasis, melanoma or a soft tissue sarcoma.
PHAT is notable for the presence of clusters of
ectatic vessels surrounded by fibrin-rich hyaline material and
cuffed by lamellae of collagen that extend into the mass of the
tumor and trap individual cells, potentially forming pockets of
hyalinized material (1,2). PHATs contain spindle-shaped, plump and
round pleomorphic cells (1,2). Cells exhibit hyperchromasia, nuclear
pleiomorphism and a lack of cytoplasmic differentiation; they may
also contain hemosiderin, nuclear inclusions and cytoplasmic
vimentin filaments (9,26). Immunohistochemical analysis of PHATs
typically reveal the presence of CD34 and may also detect
expression of vimentin, B-cell lymphoma 2, factor XIIIa, vascular
endothelial growth factor and CD99; PHAT lacks expression of S-100
protein, CD31, von Willebrand Factor, smooth muscle actin, desmin
and epithelial membrane protein (1,2,11,22,27). PHAT
cells stained for Ki-67 rarely exhibit positive staining,
consistent with the histological observation of few mitotically
active cells (1,22). Histological differential diagnoses
include neurilemmoma, undifferentiated pleomorphic sarcoma (UPS),
giant-cell angiofibroma and solitary fibrous tumor (SFT) (1,3,11,22,23). PHAT
can be distinguished from neurilemmoma by the absence of S-100,
lack of encapsulation, and the absence of Antoni A and B areas
(11,22). PHAT can be distinguished from UPS by
its infrequent mitotic figures and the presence of nuclear
inclusions (1,9,22). PHAT
can be distinguished from by the presence of clusters of
thin-walled vessels, atypical cells with bland cytoplasm and
intranuclear inclusions (11).
Wide local excision with tumor free margins is the
recommended treatment for PHAT (1,2,16) as they may occur as a collision tumor
with sarcomas such as myxofibrosarcomas (20). Surgical excision may be combined with
low dose radiation to reduce the risk of local recurrence (1,2,4). The patients were followed with serial
imaging, as these lesions have a tendency to locally recur. One
patient was followed lost to follow up after one year, and the
other remains under active imaging surveillance (follow up of 1
year).
The present study reported and discusses the
ultrasound, CT, MRI and PET/CT imaging appearance and correlating
histology of two histologically verified cases of PHAT. Biopsy
remains a requirement because imaging features are
non-specific.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
PHAT
|
pleomorphic hyalinizing angiectatic
tumors
|
|
CT
|
computed tomography
|
|
MRI
|
magnetic resonance imaging
|
|
PET/CT
|
positron emission tomography/computed
tomography
|
References
|
1
|
Smith ME, Fisher C and Weiss SW:
Pleomorphic hyalinizing angiectatic tumor of soft parts. A
low-grade neoplasm resembling neurilemoma. Am J Surg Pathol.
20:21–29. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Folpe AL and Weiss SW: Pleomorphic
hyalinizing angiectatic tumor: Analysis of 41 cases supporting
evolution from a distinctive precursor lesion. Am J Surg Pathol.
28:1417–1425. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fletcher CDM; World Health Organization
and International Agency for Research on Cancer, : WHO
Classification of Tumours of Soft Tissue and Bone. 4th. IARC Press;
Lyon: 2013
|
|
4
|
Wei S, Pan Z, Siegal GP, Winokur TS,
Carroll AJ and Jhala D: Complex analysis of a recurrent pleomorphic
hyalinizing angiectatic tumor of soft parts. Hum Pathol.
43:121–126. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mohajeri A, Kindblom LG, Sumathi VP,
Brosjö O, Magnusson L, Nilsson J, Nord KH and Mertens F: SNP array
and FISH findings in two pleomorphic hyalinizing angiectatic
tumors. Cancer Genet. 205:673–676. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Antonescu CR, Zhang L, Nielsen GP,
Rosenberg AE, Dal Cin P and Fletcher CD: Consistent t(1;10) with
rearrangements of TGFBR3 and MGEA5 in both myxoinflammatory
fibroblastic sarcoma and hemosiderotic fibrolipomatous tumor. Genes
Chromosomes Cancer. 50:757–764. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Carter JM, Sukov WR, Montgomery E,
Goldblum JR, Billings SD, Fritchie KJ and Folpe AL: TGFBR3 and
MGEA5 rearrangements in pleomorphic hyalinizing angiectatic tumors
and the spectrum of related neoplasms. Am J Surg Pathol.
38:1182–1192. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Suzuki K, Yasuda T, Hori T, Oya T,
Watanabe K, Kanamori M and Kimura T: Pleomorphic hyalinizing
angiectatic tumor arising in the thigh: A case report. Oncol Lett.
7:1249–1252. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cimino-Mathews A and Ali SZ: Pleomorphic
hyalinizing angiectatic tumor: Cytomorphologic features on
fine-needle aspiration. Diagn Cytopathol. 39:214–217. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Illueca C, Machado I, Cruz J, Almenar S,
Noguera R, Navarro S and Llombart-Bosch A: Pleomorphic hyalinizing
angiectatic tumor: A report of 3 new cases, 1 with sarcomatous
myxofibrosarcoma component and another with unreported soft tissue
palpebral location. Appl Immunohistochem Mol Morphol. 20:96–101.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin O and Crapanzano JP: Fine-needle
aspiration cytology of pleomorphic hyalinized angiectatic tumor: A
case report. Diagn Cytopathol. 32:238–242. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Subhawong TK, Subhawong AP, Montgomery EA
and Fayad LM: Pleomorphic hyalinizing angiectatic tumor: Imaging
findings. Skeletal Radiol. 41:1621–1626. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chu ZG, Liu MQ, Zhu ZY, Lv FJ and Ouyang
Y: Pelvic retroperitoneal pleomorphic hyalinizing angiectatic tumor
(PHAT) of soft tissue: A case report. BMC Med Imaging. 16:282016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Brazio PS, Morrison AL, Oh M, Goldberg N
and Boutros CN: Large pleomorphic hyalinizing angiectatic tumor of
the forearm: A multidisciplinary perspective. Surgery.
159:1471–1473. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Felton SJ and Ramlogan D: Spontaneous
bruising overlying a subcutaneous swelling: Pleomorphic hyalinizing
angiectatic tumor, a relatively new entity of intermediate
malignancy. Int J Dermatol. 54:204–206. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee JC, Jiang XY, Karpinski RH and Moore
ED: Pleomorphic hyalinizing angiectatic tumor of soft parts.
Surgery. 137:119–121. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fujiwara M, Yuba Y, Wada A, Ozawa T and
Tanaka T: Pleomorphic hyalinizing angiectatic tumor of soft parts:
Report of a case and review of the literature. J Dermatol.
31:419–423. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fukunaga M and Ushigome S: Pleomorphic
hyalinizing angiectatic tumor of soft parts. Pathol Int.
47:784–788. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Changchien YC, Bocskai P, Kovács I,
Hargitai Z, Kollár S and Török M: Pleomorphic hyalinizing
angiectatic tumor of soft parts: Case report with unusual
ganglion-like cells and review of the literature. Pathol Res Pract.
210:1146–1151. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kazakov DV, Pavlovsky M, Mukensnabl P and
Michal M: Pleomorphic hyalinizing angiectatic tumor with a
sarcomatous component recurring as high-grade myxofibrosarcoma.
Pathol Int. 57:281–284. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ide F, Shimoyama T and Horie N:
Pleomorphic hyalinizing angiectactic tumor of the buccal mucosa. J
Oral Pathol Med. 33:451–453. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
El-Tal AE and Mehregan D: Pleomorphic
hyalinizing angiectatic tumor of soft parts: Case report and
literature review. J Cutan Pathol. 33:361–364. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Matsumoto K and Yamamoto T: Pleomorphic
hyalinizing angiectatic tumor of soft parts: A case report and
literature review. Pathol Int. 52:664–668. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tallarigo F, Squillaci S, Putrino I, Zizzi
N and Bisceglia M: Pleomorphic hyalinizing angiectatic tumor of the
male breast: A heretofore unreported occurrence. Pathol Res Pract.
205:69–73. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Labanaris AP, Zugor V, Witt JH and Kuhn R:
Pleomorphic hyalinizing angiectatic tumor of the scrotum. Urol J.
8:248–250. 2011.PubMed/NCBI
|
|
26
|
Capovilla M, Birembaut P, Cucherousset J,
Ploton D, de Saint-Maur PP, Fléjou JF and Lesec G: Pleomorphic
hyalinizing angiectatic tumor of soft parts: Ultrastructural
analysis of a case with original features. Ultrastruct Pathol.
30:59–64. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Groisman GM, Bejar J, Amar M and Ben-Izhak
O: Pleomorphic hyalinizing angiectatic tumor of soft parts:
Immunohistochemical study including the expression of vascular
endothelial growth factor. Arch Pathol Lab Med. 124:423–426.
2000.PubMed/NCBI
|
|
28
|
Gallo C, Murer B and Roncaroli F:
Pleomorphic hyalinizing angiectasic soft-tissue tumor. Description
of a case. Pathologica. 89:531–535. 1997.PubMed/NCBI
|
|
29
|
Silverman JS and Dana MM: Pleomorphic
hyalinizing angiectatic tumor of soft parts: Immunohistochemical
case study shows cellular composition by CD34+ fibroblasts and
factor XIIIa+ dendrophages. J Cutan Pathol. 24:377–383. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Husek K and Veselý K: Pleomorphic
hyalinizing angiectatic tumor. Cesk Patol. 37:177–181.
2001.PubMed/NCBI
|
|
31
|
Jaggon JR and Aitken RD: Pleomorphic
hyalinizing angiectatic tumour of soft parts: A case report and
review of the literature. West Indian Med J. 56:544–546.
2007.PubMed/NCBI
|
|
32
|
Parameshwarappa S, Rodrigues G, Nagpal N
and Rao L: Pleomorphic hyalinizing angiectatic tumor of soft parts
involving the upper limb. Indian J Surg. 72:263–264. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Peng HC, Huang MT, Chen DJ, Leung TK and
Chu JS: Pleomorphic hyalinizing angiectatic tumor of soft parts. J
Formos Med Assoc. 109:616–620. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Idrees MT, Kieffer T and Badve S:
Pleomorphic hyalinizing angiectatic tumor of renal hilum. Ann Diagn
Pathol. 16:489–493. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kuang P: CT findings of pleomorphic
hyalinizing angiectatic tumor (PHAT) of soft parts of the neck. Jpn
J Radiol. 31:204–207. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rekhi B and Aggarwal S: Pleomorphic
hyalinizing angiectatic tumor exhibiting intricate branching
vasculature: An interesting pattern in a rare tumor. Indian J
Pathol Microbiol. 56:321–323. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fan C, Lin X, Yang L and Wang E: A
subcutaneous pleomorphic hyalinizing angiectatic tumor of soft
parts of the right chest wall: Report of a rare case. Int J Clin
Exp Pathol. 8:11804–11808. 2015.PubMed/NCBI
|
|
38
|
Morency E, Laskin W and Lin X: Cytologic
and histologic features of pleomorphic undifferentiated sarcoma
arising in a hybrid hemosiderotic fibrolipomatous tumor and
pleomorphic hyalinizing angiectatic tumor: Report of an unusual
case with a literature review. Acta Cytol. 59:493–497. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yorita K, Ishihara A, Tokumitsu T,
Minematsu E, Ohno A, Ikejiri H and Kataoka H: A case of a
pleomorphic hyalinizing angiectatic tumor of soft parts with
intracytoplasmic hemosiderin pigment apparent upon fine-needle
aspiration cytology. Diagn Cytopathol. 43:407–411. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kane PM, Gaspar MP, Whiting BB and Culp
RW: Pleomorphic hyalinizing angiectatic tumor arising in the hand:
A Case Report. Hand (N Y). 11:NP20–NP23. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Onyemkpa C and Oyasiji T: Pleomorphic
hyalinizing angiectatic tumor arising in the groin: A case report.
Int J Surg Case Rep. 28:227–230. 2016. View Article : Google Scholar : PubMed/NCBI
|