Introduction
Benign and malignant lymphadenopathy appear similar
in imaging studies, and therefore may be difficult to diagnose.
Diagnostic imaging studies, including those using ultrasound,
computed tomography and magnetic resonance imaging, are required to
whole-body assess patients for potentially malignant
lymphadenopathy. Imaging modalities including ultrasound, computed
tomographic scan and positron emission tomography have become more
advanced in the previous 10 years (1,2). However,
while diagnostic imaging may help to identify metastases in
numerous patients with cancer, in the presence of inflammation or
multiple tumors, differentially diagnosing patients may be
challenging for multiple reasons, including the presence of
metastatic lymph nodes of unknown origin (3–5). In
addition, deciding if lymphadenopathy that appears benign should be
followed conservatively without treatment using the diagnostic
imaging results alone may be challenging.
Histopathologically diagnosing a suspected
metastatic lymph node may substitute for diagnosing the primary
cancer when tissue samples from this primary cancer may not be
collected for histopathological analysis. Open thoracic surgery,
laparotomy, or other procedures including mediastinoscopy or
laparoscopy were required; however, these procedures were carried
out under general anesthesia, and therefore required a lot of time
and expense. These procedures are also associated with causing
tissue damage, which may cause delay to the start of therapy.
Patients exhibiting more serious medical conditions (including
hypotension and low performance status) are also exposed to greater
anesthetic and surgical associated risks. If imaging and puncturing
may be performed under endoscopic ultrasound (EUS) guidance, the
enlarged lymph node tissue may be collected for diagnosis by
EUS-guided fine needle aspiration (FNA). EUS-FNA material was used
for cytology and histological review. This collected tissue may be
used to determine malignancy and identify the origin of the
metastasis (3,6,7). The
present study retrospectively evaluated the usefulness of EUS-FNA
for patients with lymphadenopathy treated at Saitama Medical
University International Medical Center (Saitama, Japan).
Materials and methods
Patients
The present study included 72 patients (42 males; 30
females; mean age, 67 years; age range, 24–85 years) who underwent
EUS-FNA between July 2013 and December 2016 for lymphadenopathy
that could not be diagnosed based solely on imaging, at the
Department of Gastroenterology, Saitama Medical University
International Medical Center. The inclusion criteria were as
follows: Lymphadenopathy >10 mm in maximal diameter;
lymphadenopathy indicated by computed tomography to be approachable
by EUS-FNA from the esophagus, stomach, duodenum or lower
gastrointestinal tract; and suspected metastatic lymph nodes with a
primary cancer for which tissue could not be collected. The
exclusion criteria were as follows: Lymphadenopathy permitting a
superficial approach to collect tissue, e.g., superficial
lymphadenopathy; treatment with anti-thrombotic agents that may not
be discontinued; and the presence of unavoidable blood vessels in
the puncture route.
The present study complied with the Declaration of
Helsinki, as revised in Brazil 2013. All patients provided written
informed consent for EUS-FNA. The Institutional Review Board of
Saitama Medical University International Medical Center approved
the present study.
Methods
A linear echoendoscope (GF-UCT260; Olympus Optical
Co., Ltd., Tokyo, Japan) was used for EUS-FNA. A 22- or 25-gauge
needle (Expect™ Slimline; Boston Scientific Corporation,
Marlborough, MA, USA) was used for the EUS-guided procedure.
Following lymph node aspiration and stylet removal, negative
pressure was provided using a 20 ml syringe. After 20 rapid suction
strokes, the negative pressure was released and the needle was
removed. The aspiration sample was pushed out onto a slide glass by
re-inserting the stylet or by creating positive pressure using air.
The sample was examined macroscopically to identify whether it was
white, as white indicates histologic core specimen (8), not the vascular content. If macroscopic
results demonstrated an increase in blood components, the negative
pressure level used for the subsequent needle aspiration procedures
was decreased as necessary. The material, which included both the
tissue and blood components, was stored in 10% formalin (room
temperature) for histopathological diagnosis and cytology. Since
Saitama Medical University International Medical Center does not
perform on-site cytology, the EUS-FNA procedure was repeated until
the sample volume was macroscopically considered adequate for
histopathological diagnosis, including for immunostaining, as long
as the procedure could be continued. It is important to note that
due to the use of automated immunohistochemical staining, the
present study could not clarify the number of repeats required. The
present study performed EUS-FNA and samples were analyzed by
pathologists from the Department of Pathology, Saitama Medical
University International Medical Center (Saitama, Japan). If
complications including major bleeding occurred during the
procedure, the procedure would be stopped immediately. All patients
were hospitalized for the procedure and received follow-up until
the following day. Patients were discharged if they exhibited no
complications and were asked to return for an outpatient visit ~1
week later.
Cytological examination
Smears and needle rinses were alcohol-fixed (95%
ethanol, room temperature for 15 min) and subsequently Papanicolau
stained in the laboratory. All specimens were examined by a
cytopathologist to render a final diagnosis according to a five
tier diagnostic system: Non-diagnostic, negative for malignancy,
atypical, suspicious for malignancy and positive for
malignancy.
Pathological examination
Biopsy cores were fixed in 10% neutral buffered
formalin (room temperature, 20 h), embedded in paraffin and cut
into 4 µm thick serial sections for hematoxylin and eosin (Η&Ε)
staining. Slides were evaluated using a BX51 microscope
(magnification, ×2, ×4, ×10, ×20, ×40 and ×60; Olympus, Japan). The
protocol for the H&E staining in the pathological examination
was as follows; deparaffinized sections twice with xylene to remove
the paraffin for complete rehydration, 10 min each, rehydrate
samples 3 times with 100, 95 and 80% ethanol, 5 min each, followed
by incubation with 95% ethanol for 2 min and 70% ethanol for 2 min.
Samples were then washed with distilled water and stained with
Carrazzi's hematoxylin solution for 10 min and then rinsed under
tap water for 10 min. Samples were then counterstained with
eosin-phloxine solution for 5 min and dehydrated 4 times with 80,
95, 100 and 100% ethanol 5 min each for complete dehydration.
Samples were treated twice with xylene for 5 min each for
dealcoholization, and then mounted with xylene based mounting
medium. This protocol was performed at room temperature in all
stages.
Immunohistochemical analysis
Immunohistochemical analysis was performed on the
formalin-fixed, paraffin-embedded sections. The 4 µm thick sections
were mounted on poly-L-lysine-coated slides and deparaffinized and
dehydrated through graded alcohol series and water. Antigen
retrieval was performed, and sections were immunostained using the
LSAB universal kit in the Benchmark system (Ventana Medical Systems
Inc.; Roche Diagnostics, Basel, Switzerland). The antibodies that
we used were listed in (Table I). The
heating temperature was room temperature. Washing regent was EZ
prep and no rehydration was performed. An automated
immunohistochemistry protocol was used as follows:
Deparaffinization, 75°C; cell conditioning, 95–100°C; primary
antibody, 37°C; incubation 16–32 min. Endogenous peroxidase
blocking was carried out with I-VIEW inhibitor for 4 min.
Endogenous biotin blocking was performed using blocker A for 4 min
and blocker B for 4 min. Samples were incubated with secondary
antibody (I-VIEW BIOTIN Ig) for 8 min. Samples were visualized
using I-VIEW DAB and I-VIEW H2O2, incubated for 8 min, and I-VIEW
COPPER incubated for 4 min. Slides were evaluated using a BX51
microscope (×2, ×4, ×10, ×20, ×40, ×60)
 | Table I.Antibodies that were used for
immunohistochemistry. |
Table I.
Antibodies that were used for
immunohistochemistry.
| Antibodies | Source r/m | Clone | Company | Dilution | Heat-induced epitope
retrieval M/S/E | Vis method |
|---|
| CD3 | m | 2GV6 | Roche | ×1 | S | VENTANA |
| CD5 | m | 4C7 | Leica | ×25 | S | iVIEW |
|
|
|
|
|
|
| DAB Detection
kit |
|
|
|
|
|
|
| VENTANA |
|
|
|
|
|
|
| Automated
immune-histochemistry |
| CD10 | m | 56C6 |
| ×40 | S |
|
| CD20cy | m | L26 | DAKO | ×400 | S |
|
| MUM-1 | m | MUM1p |
| ×50 | M |
|
| Bcl-1 (CyclinD1) | m | DSC-6 |
| ×50 | M |
|
| Bcl-2 | m | 124 |
| ×100 | S |
|
| Bcl-6 | m | PG-B6 |
| ×20 | S |
|
| CK7 | m | OV-TL 12/30 |
| ×100 | E |
|
| CK20 | m | Ks20.8 |
| ×100 | M |
|
| TTF-1 | m | 8G7G3/1 |
| ×50 | M |
|
| Pax8 | r | Polyclonal | Proteintech | ×200 | M |
|
| ER |
|
|
|
|
|
|
| Estrogen
receptor | m | SP-1 | Roche | ×1 | S |
|
| PSA |
|
|
|
|
|
|
|
Prostate-specific | m | ER-PR8 | DAKO | ×100 | M |
|
| Antigen |
|
|
|
|
|
|
| S-100 | r | Polyclonal |
| ×500 | M |
|
| p40 | r | Polyclonal | Calbiochem | ×500 | S |
|
| CDX-2 | m | CDX2-88 | BioGenex | ×50 | S |
|
| DOG-1 | m | K9 | Leica | ×100 | S |
|
| Chromogranin A | r | Polyclonal | DAKO | ×100 | M |
|
| Synaptophysin | m | 27G12 | Nichirei | ×1 | M |
|
| CD117, c-kit
Oncoprotein | r | Polyclonal | DAKO | ×20 | M |
|
| Desmin | m | D33 |
| ×1 | S |
|
Lesions were evaluated using several primary
antibodies. The staining protocol of each antibody was performed
according to each data sheet. For detection of lesions, the
sections were incubated with 3,3′-diaminobenzidine for 8 min at
room temperature. With each set of staining processes, known
positive and negative control samples were included. Positive
control tissues were as follows: CD3, CD5, CD10, CD20cy, MUM-1,
Bcl-1 (Cyclin D1), Bcl-2, and Bcl-6 were tonsil; CK7, CK20, and
CDX-2 were tissues of Cytokeratin (esophagus, colon and urinary
bladder); ER (Estrogen Receptor) was mammary gland; P40 was tissues
of Squamous cell carcinoma; DOG-1 and CD117 c-kit Oncoprotein were
tissues of Gastrointestinal stromal tumor; TTF-1, Pax8, PSA, S-100,
Chromogranin A, Synaptophysin, and Desmin were multi-control
tissues (Cerebrum, thyroid, lung, liver, pancreas, kidney,
prostate). Cells within the positive control tissues that are known
to be negative for each protein were used as negative controls in
the present study. All specimens were diagnosed by a pathologist in
Department of Pathology, Saitama Medical University International
Medical Center (Saitama, Japan).
The present study evaluated the sensitivity,
specificity, positive and negative predictive value, overall
accuracy, helpfulness for determining the management of
lymphadenopathy, and the EUS-FNA-associated complications. The type
of cancer was predicted prior to surgery by blood examination,
computed tomography or magnetic resonance imaging.
The final diagnosis used the EUS-FNA-based tissue
diagnosis or postoperative tissue diagnosis and the clinical course
in patients who underwent surgery. Lymphadenopathy was used to
diagnose EUS-FNA-based tissue as benign, with whole-body
assessments were performed, including imaging modalities after 6
months. If the image results and clinical course were without
aggravation, they were diagnosed as benign. If insufficient EUS-FNA
sample volume was gained surgical biopsy was performed.
Of the 72 patients enrolled in the present study, 24
(33.3%) had a history of a different type of cancer (Table II). The median longest diameter of
the examined lymph node was 21 mm (10–90 mm). Transesophageal,
transgastric, transduodenal and transcolorectal FNA were performed
in 5, 58, 8 and 1 patient, respectively. A 22-gauge needle was used
in 67 patients, while a 25-gauge needle was used in 5 patients. The
mean number of puncture attempts was 3.8 (range, 1–8).
 | Table II.Clinicodemographic characteristics of
the study population (n=72). |
Table II.
Clinicodemographic characteristics of
the study population (n=72).
|
Characteristics | Number of
patients |
|---|
| Male/female | 42/30 |
| Mean age
(range) | 67 years
(24–85) |
| History of
different cancer |
|
| Gastric
cancer | 5 |
| Colon
cancer | 4 |
|
Pancreatic cancer | 4 |
| Bile
duct cancer | 3 |
|
Hepatocellular carcinoma | 2 |
| Lung
cancer | 2 |
|
Esophageal cancer | 1 |
|
Cervical cancer | 1 |
| Renal
pelvis cancer | 1 |
|
Malignant lymphoma | 1 |
| Median longest
diameter of the lymph node, mm (range) | 21 (10–90) |
| Puncture site, n
(%) |
|
|
Esophagus | 5 (6.9) |
|
Stomach | 58 (80.6) |
|
Duodenum | 8 (11.1) |
|
Rectum | 1 (1.4) |
| Aspiration needle
size, n (%) |
|
22-gauge | 67 (93.1) |
|
25-gauge | 5 (6.9) |
| Mean number of
puncture attempts (range) | 3.8 (1–8) |
| Malignant/benign,
n | 64/8 |
Results
The final diagnosis was malignancy in 64 patients
and benign lymphadenopathy in 8 patients (Table II). The 64 malignancies included
malignant lymphoma in 33 patients (B-cell lymphoma in 30 patients,
T-cell lymphoma in 3 patients), bile duct cancer in 6 patients,
malignancy of unknown origin in 5 patients, pancreatic cancer in 4
patients, gastric cancer in 3 patients, lung cancer in 3 patients,
testicular cancer in 2 patients, ovarian cancer in 2 patients,
prostate cancer in 1 patient, esophageal cancer in 1 patient, colon
cancer in 1 patient, cancer of the duodenal papilla in 1 patient,
adrenal paraganglioma in 1 patient and a gastrointestinal stromal
tumor in 1 patient (Table III). Of
the 33 patients with a final diagnosis of lymphoma, the disease was
subtyped according to the classification of lymphoid neoplasms
(World Health Organization; 2016) (9)
via EUS-FNA in 23 patients (69.7%).
 | Table III.Final diagnoses of the
lymphadenopathies (n=72). |
Table III.
Final diagnoses of the
lymphadenopathies (n=72).
| Type of cancer | Number of
patients |
|---|
| Malignant | 64 |
| Benign | 8 |
| Malignant
lymphoma | 33 |
| Reactive
lymphadenopathy | 8 |
| Bile duct
cancer | 6 |
| Origin unknown | 5 |
| Pancreatic
cancer | 4 |
| Gastric cancer | 3 |
| Lung cancer | 3 |
| Testicular
cancer | 2 |
| Ovarian cancer | 2 |
| Prostate
cancer | 1 |
| Esophageal
cancer | 1 |
| Colon cancer | 1 |
| Cancer of the
duodenal papilla | 1 |
| Adrenal
paraganglioma | 1 |
| Gastrointestinal
stromal tumor | 1 |
The origin was identified using EUS-FNA in 87.5%
(56/64) of metastatic lymph nodes. The histopathological diagnosis
differed from that which had been expected based on the
pre-procedural images in 9 patients and the cancer management for
these patients was subsequently changed (Table IV), for example, alternate anticancer
drugs were administered. The present study pre-procedurally
predicted that hepatocellular carcinoma would be diagnosed in a
77-year-old male patient, as the patient had undergone surgery for
hepatocellular carcinoma 4 years previously, and the
lymphadenopathy was due to recurrence (Table IV). In 8 cases, for which the present
study pre-procedurally predicted lymphoma, there was no history of
cancer or multiple lymphadenopathies. Furthermore, certain patients
exhibited an elevated soluble interleukin-2 receptor level. In a
57-year-old male patient (Table IV),
the presence of multiple lymphadenopathies suggestive of malignant
lymphoma was identified to be metastatic prostate cancer based on
the histopathology examination following EUS-FNA, and therefore the
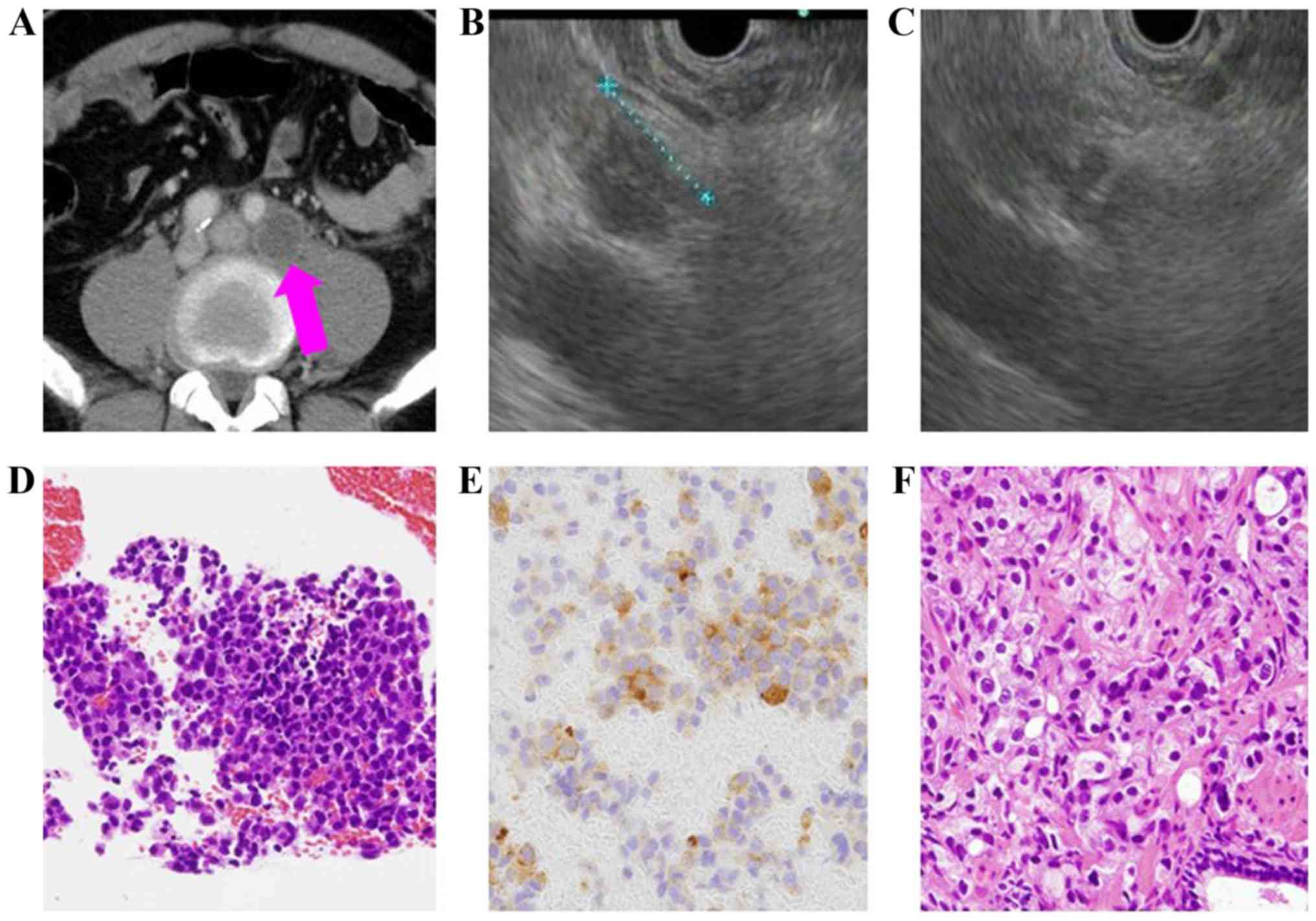
cancer management for this patient was subsequently changed. This
patient was administered degarelix (antihormon drug; Fig. 1).
 | Table IV.Diagnostic changes following
endoscopic ultrasound-guided fine needle aspiration in 9
patients. |
Table IV.
Diagnostic changes following
endoscopic ultrasound-guided fine needle aspiration in 9
patients.
| Sex | Age, years | Punctured lymph
node | Diameter (mm) | Cytology
histopathology | Pre-procedural
prediction | Final
diagnosis |
|---|
| Female | 50 | Para-aortic | 42 | Atypical
paraganglioma | Lymphoma | Paraganglioma |
| Male | 77 | Para-aortic | 12 | Positive |
|
|
|
|
|
|
| Adenocarcinoma | HCC | Metastasis of
unknown origin |
| Female | 80 | Para-aortic | 30 | Positive |
|
|
|
|
|
|
| Adenocarcinoma | Lymphoma | Metastasis of
ovarian cancer |
| Male | 24 | Proximate to the
abdominal aorta | 50 | Positive |
|
|
|
|
|
|
| Germ cell
tumor | Lymphoma | Metastasis of
testicular cancer |
| Male | 57 | Para-aortic | 21 | Positive |
|
|
|
|
|
|
| Adenocarcinoma | Lymphoma | Metastasis of
prostate cancer |
| Male | 74 | Proximate to the
celiac artery | 35 | Positive |
|
|
|
|
|
|
| Adenocarcinoma | Lymphoma | Metastasis of bile
duct cancer |
| Male | 40 | Para-aortic | 15 | Positive |
|
|
|
|
|
|
| Germ cell
tumor | Lymphoma | Metastasis of
testicular cancer |
| Male | 73 | Para-aortic | 26 | Positive |
|
|
|
|
|
|
| GIST | Lymphoma | Metastasis of
GIST |
| Female | 75 | Para-aortic | 26 | Atypical |
|
|
|
|
|
|
| Serous
carcinoma | Lymphoma | Metastasis of
duodenal papilla |
Of the 64 patients with a final diagnosis of
malignancy, the diagnosis was based on EUS-FNA alone in 61
patients. Of the remaining 3 patients whose malignancy could not be
diagnosed using EUS-FNA alone, the EUS-FNA sample volume was
insufficient in 1 patient with current lung and esophageal cancer.
This particular patient received fluorouracil (FP; 1200 mg dose
between days 1 to 5) and cisplatin (120 mg dose on day 1) treatment
every 4 weeks. This patient received 6 courses of FP therapy for
lung cancer as this was more advanced compared with the esophageal
cancer. The remaining 2 patients, who were suspected to exhibit
malignant lymphoma, underwent laparotomy biopsy due to insufficient
EUS-FNA sample volume. These patients were diagnosed with lymphoma
and received rituximab (375 mg/m2 of body surface area
on day 1), cyclophosphamide (750 mg/m2 of body surface
area on day 1), doxorubicin hydrochloride (50 mg/m2 of
body surface area on day 1), vincristine sulfate (1.4
mg/m2 of body surface area on day 1), and prednisolone
(100 mg dose between days 1 to 5) treatment every 3 weeks. Both
patients received 5 courses. All 8 patients with a benign diagnosis
exhibited reactive lymphadenopathy, which was not aggravated during
the 6-month follow-up. In 72 cases, 8 patients were diagnosed as
benign and 64 as malignant. The EUS-FNA diagnosis of benign and
malignancy was 11 patients and 61 patients. The sensitivity,
specificity, positive predictive value, negative predictive value
and accuracy of malignancy diagnosed by EUS-FNA were 95.3% (61/64),
100% (8/8), 100% (61/61), 72.7% (8/11) and 95.8% (69/72).
EUS-FNA helped to identify the origin of the cancer
in the majority of the patients (56/64; 87.5%). The procedure also
assisted in determining the appropriate cancer management plan;
specifically, EUS-FNA assisted with cancer staging, diagnosing
recurrence (Table V) and diagnosing
using the metastatic lymph node when collecting samples from the
original cancer was impractical (Table
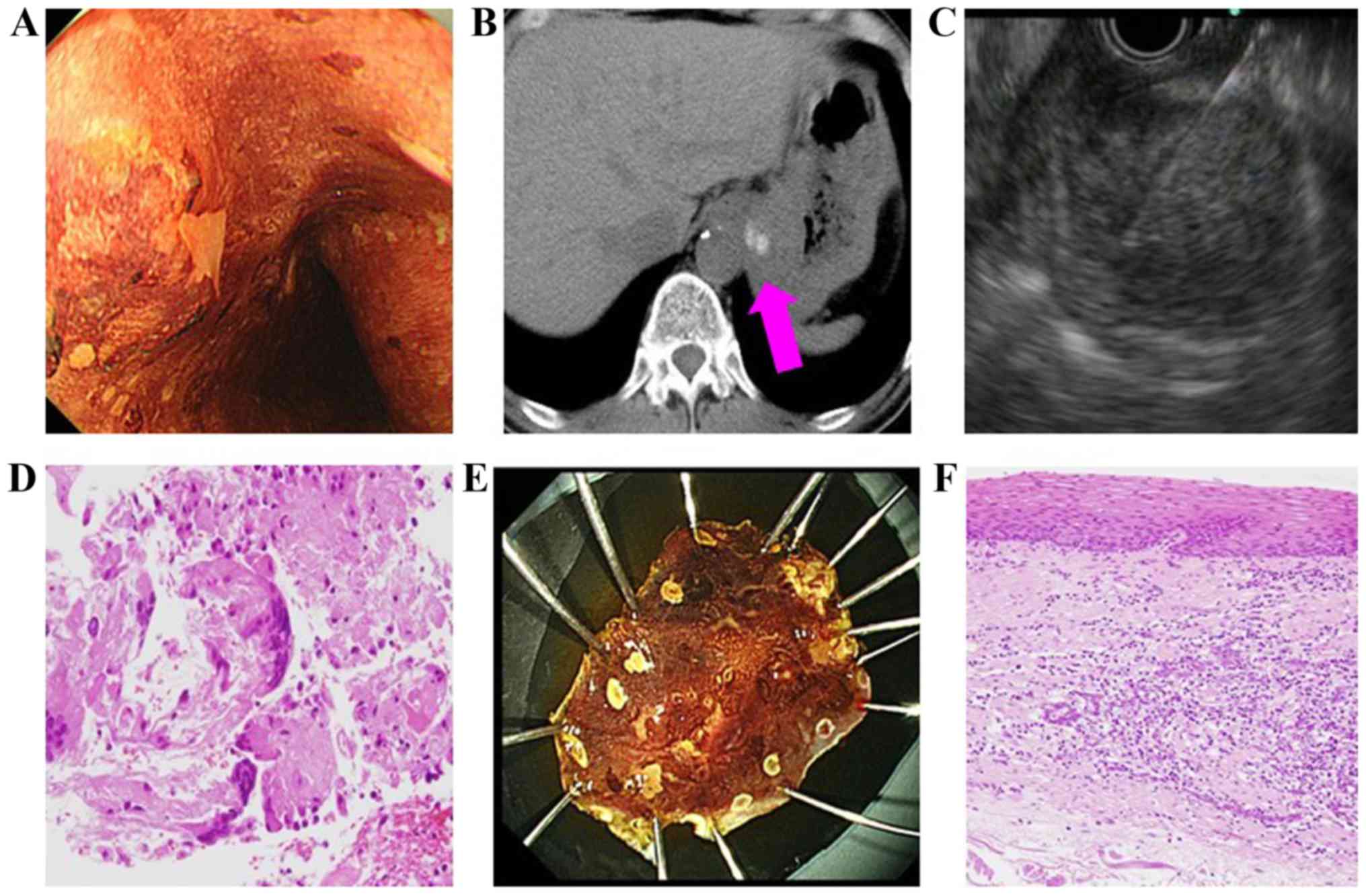
VI). In 1 patient (a 63-year-old male; Table VI) with suspected esophageal cancer,
EUS-FNA was performed on an enlarged cardiac lymph node as no
definitive diagnosis could be made following repeated biopsies and
the collection of original cancer tissue was impossible. EUS-FNA
subsequently revealed a diagnosis of squamous cell carcinoma
(Fig. 2). No patient that underwent
EUS-FNA reported any complications.
 | Table V.Patients for whom endoscopic
ultrasound-guided fine needle aspiration helped diagnose
recurrence. |
Table V.
Patients for whom endoscopic
ultrasound-guided fine needle aspiration helped diagnose
recurrence.
| Sex | Age, years | Origin | Punctured lymph
node | Diameter (mm) | Cytology
histopathology | Duration of
recurrence (months) |
|---|
| Male | 73 | Lung | Mediastinal | 22 | Positive Squamous
cell carcinoma | 20 |
| Male | 77 | Stomach | Lesser
curvature | 11 | Positive
Adenocarcinoma | 42 |
| Male | 74 | Stomach | Proximate to the
common hepatic artery | 15 | Positive
Adenocarcinoma | 8 |
| Female | 83 | Bile duct | Proximate to the
superior mesenteric artery | 20 | Positive
Adenocarcinoma | 38 |
| Female | 68 | Pancreas | Proximate to the
common hepatic artery | 20 | Positive Atypical
cell | 11 |
| Male | 75 | Colon | Mediastinal | 22 | Positive
Adenocarcinoma | 27 |
| Male | 73 | Lung | Mediastinal | 12 | Positive Squamous
cell carcinoma | 20 |
 | Table VI.Endoscopic ultrasound-guided fine
needle aspiration of metastatic lymph nodes in patients with no
available sample of the original cancer tissue. |
Table VI.
Endoscopic ultrasound-guided fine
needle aspiration of metastatic lymph nodes in patients with no
available sample of the original cancer tissue.
| Sex | Age, years | Cancer origin | Reason for
unavailable tissue sample | Punctured lymph
node | Diameter (mm) | Cytology
histopathology |
|---|
| Male | 70 | Stomach | Unable to collect
via biopsy | Proximate to the
common hepatic artery | 20 | Negative
Adenocarcinoma |
| Male | 63 | Esophagus | Unable to collect
via biopsy | Gastric cardia | 39 | Positive Squamous
cell carcinoma |
| Male | 74 | Stomach | Unable to collect
via biopsy | Proximate to the
common hepatic artery | 41 | Positive
Adenocarcinoma |
| Male | 77 | Pancreas | Gastrointestinal
stenosis present | Proximate to the
celiac artery | 10 | Positive
Adenocarcinoma |
Discussion
The proposed EUS image-based diagnostic criteria for
malignant lymphadenopathy include round or oval cross-sections,
sharp demarcations, internal hypoechoic features and >10 mm
largest diameter (3). Overall, only
EUS image-based diagnostic accuracy is 80% when all criteria are
met (3). However, differentially
diagnosing inflammation and metastasis, and determining the most
appropriate cancer management using EUS of lymphadenopathy alone is
challenging (3–5). Although contrast EUS has become
increasingly diagnostically accurate, EUS results alone remain
insufficient for diagnosing patients (4,5).
EUS-FNA is useful when EUS alone is not reliable.
Already a part of general practice, EUS-FNA for lymphadenopathy
helps to identify metastasis and diagnose inflammatory diseases,
including tuberculosis (5–7). Previous studies have reported that
concurrent EUS and EUS-FNA increase the overall diagnostic accuracy
(3,6,7). Recently,
multiple skills including ‘funning technique’ or ‘slow-pull
technique’ were reported to achieve successful EUS-FNA results
(10). Nakahara et al
(11) reported a 96% overall
diagnostic accuracy of EUS-FNA for abdominal lymphadenopathy of
unknown origin in 57 patients. EUS-FNA is used for mediastinal
lymphadenopathy, with a sensitivity of 82–93% and a specificity of
89–100% (12–14). In addition, EUS-FNA is used with a
lower gastrointestinal tract approach for pelvic lymphadenopathy
(15). This procedure has been
reported to be useful for urological cancer types, including
prostate and bladder cancer, with a sensitivity of 94.4% (15). EUS-FNA was also useful for diagnosing
malignant lymphoma. Yasuda et al (16) subtyped lymphoma according to the World
Health Organization classification in 44 of their 48 patients, who
could subsequently receive multiple tailored treatments, including
chemotherapy. They also used a 19-gauge needle to perform EUS-FNA,
which was reportedly safely performed, and complications occurred
in 1% of patients.
In accordance with these previous studies, the
present study concluded that EUS-FNA is minimally invasive and
accurate for diagnosing endoscopically approachable
lymphadenopathy. In the present study, a 25-gauge needle was used
in 5 cases. In 3 cases, the lesions were punctured with a tight
angular scope position. In 2 cases, small blood vessels were
present within the lesions. In these cases, a thinner (25-gauge)
needle was used. The present study performed EUS-FNA for
lymphadenopathy in 72 patients, with an increased tissue collection
rate and without any ensuing complications. The present study
attributed the absence of complications to the use of a 25-gauge
needle for lesions with a bleeding risk due to the small blood
vessels located within the lesion. The outcomes of EUS-FNA were
similar to those reported in previous studies, with a sensitivity,
specificity, positive predictive value, negative predictive value
and overall accuracy of 95.3 (61/64), 100 (8/8), 100 (61/61), 72.7
(8/11) and 95.8% (69/72), respectively. The rate of subtyping
malignant lymphoma according to the WHO classification (9) was 69.7% (23/33); this was low compared
with the rate reported by Yasuda et al (16). To increase the rate of subtyping,
subsequent studies should increase the sample volume, perhaps by
using a 19-gauge needle, as reported in another previous study
A prior study suggested that although thinner (22-
or 25-gauge) needles provide a decreased volume of cellular
material compared with that which larger (19-gauge) needles
provide, the specimens from the former are less contaminated by
blood and are therefore easier to evaluate (17). In addition, thinner needles may be
easier to use due to increased flexibility, particularly for
locations that require the scope to bend (17). Therefore, the majority of studies on
EUS-FNA have been performed using 22-gauge needles (11,12,15). As
aforementioned, a 25-gauge needle is suitable for lesions that
require puncturing in a tight angulated scope position and lesions
that contain small blood vessels. For subtyping malignant lymphoma,
a 19-gauge needle may be suitable.
Immunostaining was revealed to be useful for
diagnosing based on the sample collected using EUS-FNA in the
present study. If the specimen was histologically similar in
regards to EUS-FNA and surgical results, recurrent cancer would be
diagnosed.
Providing accurate clinical information to the
pathologist ensures appropriate differential diagnosis. Sharing
information with the pathologist is important for identifying
specific types of cancer from EUS-FNA samples (18). Definitively diagnosing patients with
suspected cancer from whom collecting biopsy or original cancer
tissue is impractical due to gastrointestinal stenosis may be
challenging. However, no previous reports on EUS-FNA of metastatic
lymph nodes in such patients were identified in the present study.
As the usefulness of EUS-FNA has been confirmed, a proactive use of
EUS-FNA is recommended for cases in which clinicians' suspect
metastatic lymphadenopathy.
EUS-FNA identified the origin of the metastases in
87.5% (56/64) of the metastatic lymph nodes. The histopathological
diagnosis differed from that which was expected based on the
pre-procedural images in 9 patients; the cancer management strategy
for these patients was subsequently changed. This result suggested
that these patients may have received inappropriate treatment had
they been diagnosed solely using the imaging results. Accordingly,
the present study emphasized how important definitively diagnosing
using EUS-FNA may be. However, the cancer was diagnosed without
identifying the origin in certain patients and definitively
diagnosing using EUS-FNA may be difficult in patients for whom the
origin may not be identified by prior whole-body assessments. In
such patients, further whole-body examinations will be necessary to
identify the cancer origin, indicating the importance of detailed
whole-body assessments in these patients.
To conclude, the present study found that EUS-FNA
was not associated with complications in these patients, and that
it was useful for diagnosing lymphadenopathy that could not be
diagnosed solely based on images. In addition to EUS-FNA
techniques, prior whole-body examinations including blood
examination and imaging modalities are important for the diagnosis
of lymphadenopathy.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mochizuki K, Gabata T, Kozaka K, Hattori
Y, Zen Y, Kitagawa H, Kayahara M, Ohta T and Matsui O: MDCT
findings of extra pancreatic nerve plexus invasion by pancreas head
carcinoma: Correlation with en bloc pathological specimens and
diagnostic accuracy. Eur Radiol. 20:1757–1767. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Raman SP, Chen Y and Fishman EK:
Cross-sectional imaging and the role of positron emission
tomography in pancreatic cancer evaluation. Semin Oncol. 42:40–58.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bhutani MS, Hawes RH and Hoffman BJ: A
comparison of the accuracy of echo features during endoscopic
ultrasound (EUS) and EUS-guided fine-needle aspiration for
diagnosis of malignant lymph node invasion. Gastrointest Endosc.
45:474–479. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kanamori A, Hirooka Y, Itoh A, Hashimoto
S, Kawashima H, Hara K, Uchida H, Goto J, Ohmiya N, Niwa Y and Goto
H: Usefulness of contrast-enhanced endoscopic ultrasonography in
the differentiation between malignant and benign lymphadenopathy.
Am J Gastroenterol. 101:45–51. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Song HJ, Kim JO, Eun SH, Cho YD, Jung IS,
Cheon YK, Moon JH, Lee MS, Shim CS, Kim BS and Jin SY: Endoscopic
ultrasonograpic findings of benign mediastinal and abdominal
lymphadenopathy confirmed by EUS-guided fine needle aspiration. Gut
Liver. 1:68–73. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Eloubeidi MA, Wallace MB, Reed CE,
Hadzijahic N, Lewin DN, Van Velse A, Leveen MB, Etemad B, Matsuda
K, Patel RS, et al: The utility of EUS and EUS-guided fine needle
aspiration in detecting celiac lymph node metastasis in patients
with esophageal cancer: A single center experience. Gastrointest
Endosc. 54:714–719. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen VK and Eloubeidi MA: Endoscopic
ultrasound-guided fine needle aspiration is superior to lymph node
echo features: A prospective evaluation of mediastinal and
peri-intestinal lymphadenopathy. Am J Gastroenterol. 99:628–633.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Iwashita T, Yasuda I, Mukai T, Doi S,
Nakashima M, Uemura S, Mabuchi M, Shimizu M, Hatano Y, Hara A and
Moriwaki H: Macroscopic on-site quality evaluation of biopsy
specimens to improve the diagnostic accuracy during EUS-guided FNA
using a 19-gauge needle for solid lesions: A single-center
prospective pilot study (MOSE study). Gastrointest Endosc.
81:177–185. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Swerdlow SH, Campo E, Pileri SA, Harris
NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz
AD and Jaffe ES: The 2016 revision of the World Health Organization
classification of lymphoid neoplasms. Blood. 127:2375–2390. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bhatia V and Varadarajulu S: Endoscopic
ultrasonography-guided tissue acquisition: How to achieve
excellence. Dig Endosc. 29:417–430. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nakahara O, Yamao K, Bhatia V, Sawaki A,
Mizuno N, Takagi T, Shimizu Y, Koshikawa T, Yatabe Y and Baba H:
Usefulness of endoscopic ultrasound-guided fine needle aspiration
(EUS-FNA) for undiagnosed intra-abdominal lymphadenopathy. J
Gastroenterol. 44:562–567. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Srinivasan R, Bhutani MS, Thosani N,
Săftoiu A, Rice DC, Ioncică AM, Eapen GA, Gupta P, Jaganmohan S,
Artifon EL and Zwischenberger JB: Clinical impact of EUS-FNA of
mediastinal lymph nodes in patients with known or suspected lung
cancer or mediastinal lymph nodes of unknown etiology. J
Gastrointestin Liver Dis. 21:145–152. 2012.PubMed/NCBI
|
|
13
|
Nguyen TQ, Kalade A, Prasad S, Desmond P,
Wright G, Hart D, Conron M and Chen RY: Endoscopic ultrasound
guided fine needle aspiration (EUS-FNA) of mediastinal lesions. ANZ
J Surg. 81:75–78. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Micames CG, McCrory DC, Pavey DA, Jowell
PS and Gress FG: Endoscopic ultrasound-guided fine-needle
aspiration for non-small cell lung cancer staging: A systematic
review and metaanalysis. Chest. 131:539–548. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gleeson FC, Clain JE, Karnes RJ, Rajan E,
Topazian MD, Wang KK and Levy MJ: Endoscopic ultrasound-guided
tissue sampling facilitates the detection of local recurrence and
extra pelvic metastasis in pelvic urologic malignancy. Diagn Ther
Endosc. 2012:2195212012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yasuda I, Tsurumi H, Omar S, Iwashita T,
Kojima Y, Yamada T, Sawada M, Takami T, Moriwaki H and Soehendra N:
Endoscopic ultrasound guided fine-needle aspiration biopsy for
lymphadenopathy of unknown origin. Endoscopy. 38:919–924. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Polkowski M, Larghi A, Weynand B,
Boustière C, Giovannini M, Pujol B and Dumonceau JM; European
Society of Gastrointestinal Endoscopy (ESGE), : Learning,
techniques, and complications of endoscopic ultrasound (EUS)-guided
sampling in gastroenterology: European society of gastrointestinal
endoscopy (ESGE) technical guideline. Endoscopy. 44:190–206. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Iglesias-Garcia J, Lariño-Noia J,
Abdulkader I and Domínguez-Muñoz JE: Rapid on-site evaluation of
endoscopic-ultrasound-guided fine-needle aspiration diagnosis of
pancreatic masses. World J Gastroenterol. 20:9451–9457. 2014.
View Article : Google Scholar : PubMed/NCBI
|