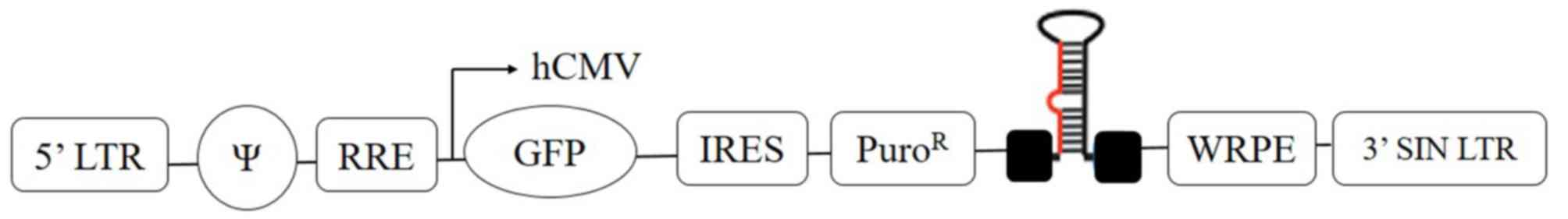
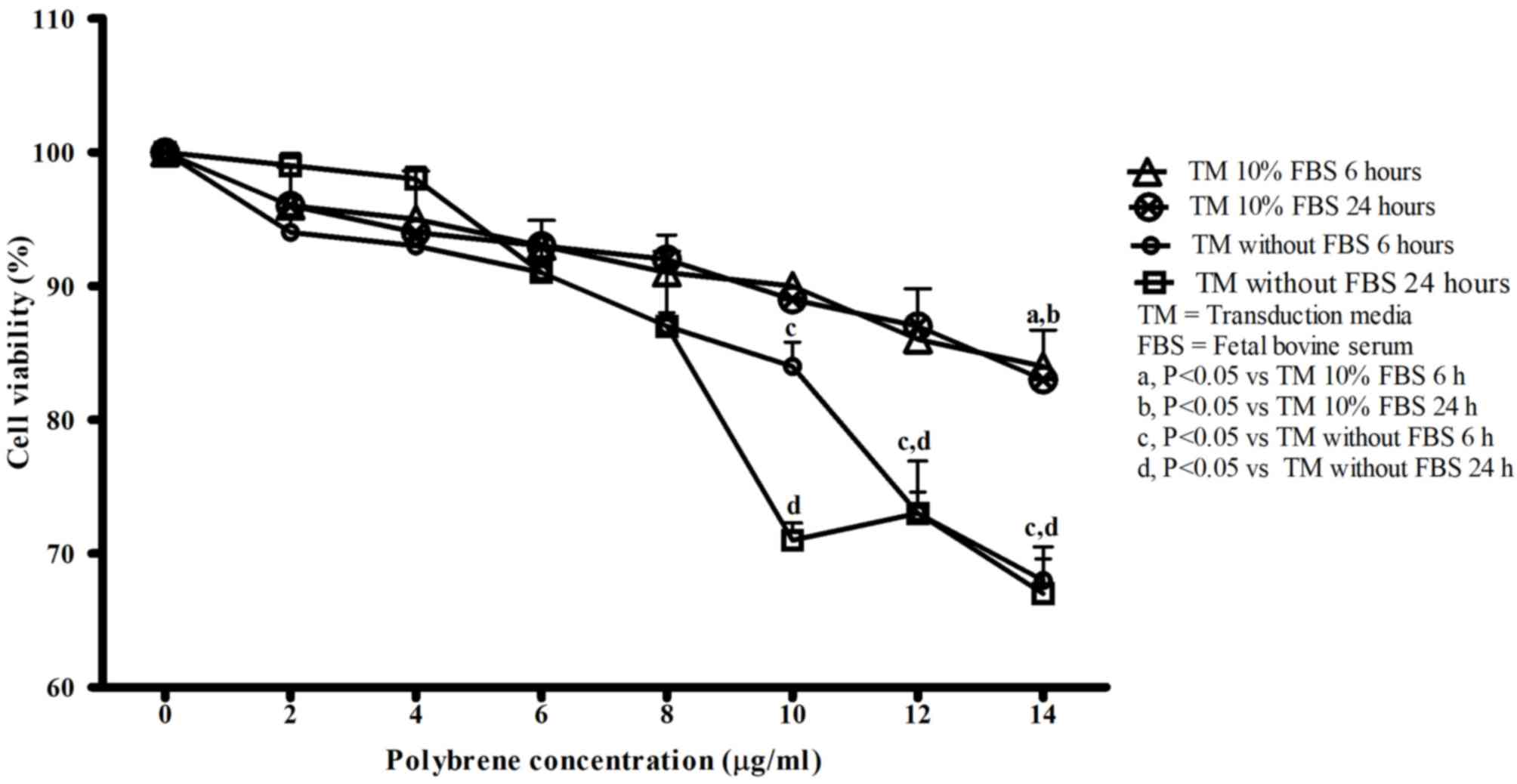
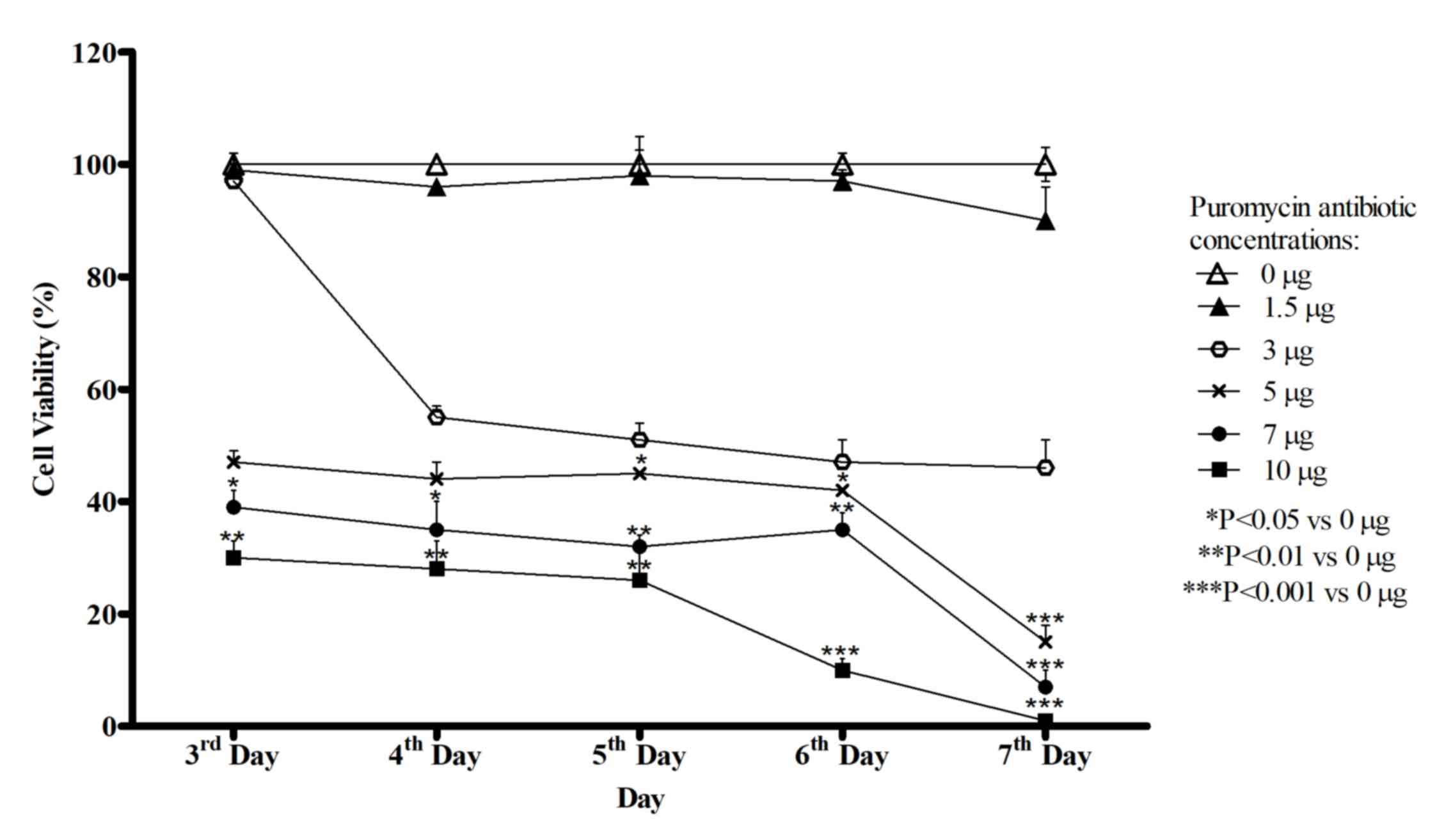
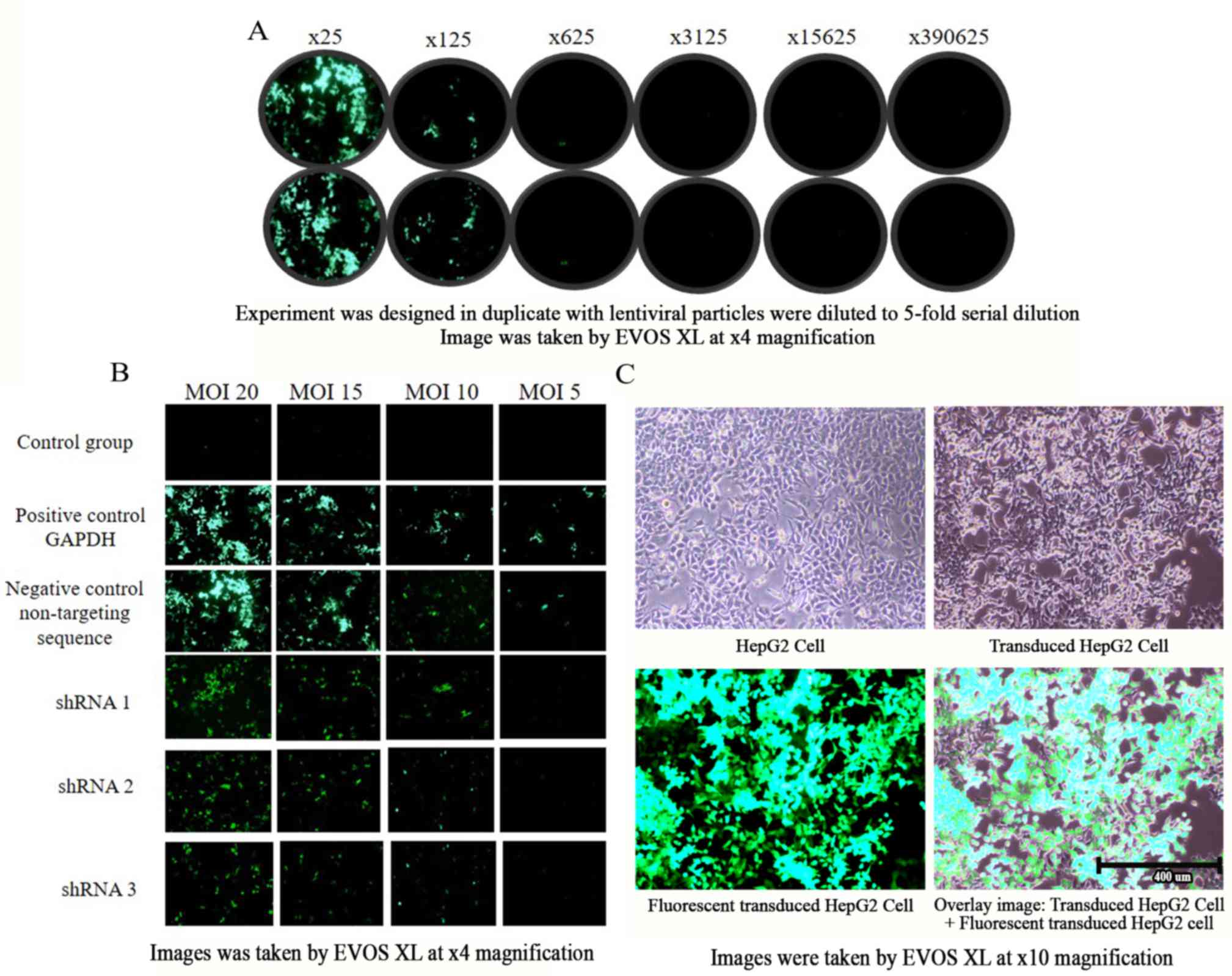
|
1
|
Manjunath N, Haoquan W, Sandesh S and
Premlata S: Lentiviral delivery of short hairpin RNAs. Adv Drug
Deliv Rev. 61:732–745. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Aigner A: Gene silencing through RNA
interference (RNAi) in vivo: Strategies based on the direct
application of siRNAs. J Biotechnol. 124:12–25. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
López-Terrada D, Cheung SW, Finegold MJ
and Knowles BB: HepG2 is a hepatoblastoma-derived cell line. Hum
Pathol. 40:1512–1515. 2009. View Article : Google Scholar
|
|
4
|
Pang RTK, Poon TCW, Wong N, Lai PBS, Wong
NLY, Chan CML, Yu JSW, Chan ATC and Sung JJY: Comparison of protein
expression patterns between hepatocellular carcinoma cell lines and
a hepatoblastoma cell line. Clin Proteomics. 1:313–331. 2004.
View Article : Google Scholar
|
|
5
|
Le Bihan O, Chèvre R, Mornet S, Garnier B,
Pitard B and Lambert O: Probing the in vitro mechanism of action of
cationic lipid/DNA lipoplexes at a nanometric scale. Nucleic Acids
Res. 39:1595–1609. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cesaratto L, Vascotto C, D'Ambrosio C,
Scaloni A, Baccarani U, Paron I, Damante G, Calligaris S,
Quadrifoglio F, Tiribelli C and Tell G: Overoxidation of
peroxiredoxins as an immediate and sensitive marker of oxidative
stress in HepG2 cells and its application to the redox effects
induced by ischemia/reperfusion in human liver. Free Radic Res.
39:255–268. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sazli Abdul Rahman F, Jubri Z, Rahman
Abdul M, Karsani SA, Top Md AG and Ngah Wan WZ: Gamma-tocotrienol
treatment increased peroxiredoxin-4 expression in HepG2 liver
cancer cell line. BMC Complement Altern Med. 15:642015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ito R, Takahashi M, Ihara H, Tsukamoto H,
Fujii J and Ikeda Y: Measurement of peroxiredoxin-4 serum levels in
rat tissue and its use as a potential marker for hepatic disease.
Mol Med Rep. 6:379–384. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Denning W, Das S, Guo S, Xu J, Kappes JC
and Hel Z: Optimization of the transductional efficiency of
lentiviral vectors: Effect of sera and polycations. Mol Biotechnol.
53:308–314. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Logan AC, Nightingale SJ, Haas DL, Cho GJ,
Pepper KA and Kohn DB: Factors influencing the titer and
infectivity of lentiviral vectors. Hum Gene Ther. 15:976–988. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Davis HE, Rosinski M, Morgan JR and
Yarmush ML: Charged polymers modulate retrovirus transduction via
membrane charge neutralization and virus aggregation. Biophys J.
86:1234–1242. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Morgan JR, LeDoux JM, Snow RG, Tompkins RG
and Yarmush ML: Retrovirus infection: Effect of time and target
cell number. J Virol. 69:6994–7000. 1995.PubMed/NCBI
|
|
13
|
Andreadis S, Lavery T, Davis HE, Le Doux
JM, Yarmush ML and Morgan JR: Toward a more accurate quantitation
of the activity of recombinant retroviruses: Alternatives to titer
and multiplicity of infection. J Virol. 74:1258–1266. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dokka S, Toledo D, Shi X, Ye J and
Rojanasakul Y: High-efficiency gene transfection of macrophages by
lipoplexes. Int J Pharm. 206:97–104. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang B, Metharom P, Jullie H, Ellem KA,
Cleghorn G, West MJ and Wei MQ: The significance of controlled
conditions in lentiviral vector titration and in the use of
multiplicity of infection (MOI) for predicting gene transfer
events. Genet Vaccines Ther. 2:62004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Taxman DJ, Livingstone LR, Zhang J, Conti
BJ, Iocca HA, Williams KL, Lich JD, Ting JP and Reed W: Criteria
for effective design, construction, and gene knockdown by shRNA
vectors. BMC Biotechnol. 6:72006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Takasaki S, Kotani S and Konagaya A: An
effective method for selecting siRNA target sequences in mammalian
cells. Cell Cycle. 3:790–795. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Luria SE: Cell susceptibility to viruses.
Ann N Y Acad Sci. 61:852–855. 1955. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yang L, Bailey L, Baltimore D and Wang P:
Targeting lentiviral vectors to specific cell types in vivo. Proc
Natl Acad Sci USA. 103:11479–11484. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gupta S, Schoer RA, Egan JE, Hannon GJ and
Mittal V: Inducible, reversible, and stable RNA interference in
mammalian cells. Proc Natl Acad Sci USA. 101:1927–1932. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Healthcare G: Technical manual: GIPZ
Lentiviral shRNA. Thermo Sci. 2015.
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Moore CB, Guthrie EH, Huang MT and Taxman
DJ: Short hairpin RNA (shRNA): Design, delivery, and assessment of
gene knockdown. Methods Mol Biol. 629:141–158. 2010.PubMed/NCBI
|
|
24
|
Svoboda P: Off-targeting and other
non-specific effects of RNAi experiments in mammalian cells. Curr
Opin Mol Ther. 9:248–257. 2007.PubMed/NCBI
|
|
25
|
Huppi K, Martin SE and Caplen NJ: Defining
and assaying RNAi in mammalian cells. Mol Cell. 17:1–10. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Whither RNAi. Nat Cell Biol. 5:489–490.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goncz KK, Gruenert DC and Colosimo A:
Expression vector system and a method for optimization and
confirmation of DNA delivery and quantification of targeting
frequency. Journal. 2005.
|
|
28
|
Feng Y, Nie L, Thakur MD, Su Q, Chi Z,
Zhao Y and Longmore GD: A multifunctional lentiviral-based gene
knockdown with concurrent rescue that controls for off-target
effects of RNAi. Genomics Proteomics Bioinformatics. 8:238–245.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jackson AL and Linsley PS: Noise amidst
the silence: Off-target effects of siRNAs? Trends Genet.
20:521–524. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Snøve O Jr and Holen T: Many commonly used
siRNAs risk off-target activity. Biochem Biophys Res Commun.
319:256–263. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Matveeva O, Nechipurenko Y, Rossi L, Moore
B, Saetrom P, Ogurtsov AY, Atkins JF and Shabalina SA: Comparison
of approaches for rational siRNA design leading to a new efficient
and transparent method. Nucleic Acids Res. 35:e632007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
ter Brake O, 't Hooft K, Liu YP, Centlivre
M, von Eije KJ and Berkhout B: Lentiviral vector design for
multiple shRNA expression and durable HIV-1 inhibition. Mol Ther.
16:557–564. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nasri M, Karimi A and Farsani
Allahbakhshian M: Production, purification and titration of a
lentivirus-based vector for gene delivery purposes. Cytotechnology.
66:1031–1038. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Nash KL, Jamil B, Maguire AJ, Alexander GJ
and Lever AM: Hepatocyte-specific gene expression from integrated
lentiviral vectors. J Gene Med. 6:974–983. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nishitsuji H, Ikeda T, Miyoshi H, Ohashi
T, Kannagi M and Masuda T: Expression of small hairpin RNA by
lentivirus-based vector confers efficient and stable
gene-suppression of HIV-1 on human cells including primary
non-dividing cells. Microbes Infect. 6:76–85. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rao DD, Vorhies JS, Senzer N and
Nemunaitis J: siRNA vs. shRNA: Similarities and differences. Adv
Drug Deliv Rev. 61:746–759. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zamule SM, Strom SC and Omiecinski CJ:
Preservation of hepatic phenotype in lentiviral-transduced primary
human hepatocytes. Chem Biol Interact. 173:179–186. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lim LP, Lau NC, Garrett-Engele P, Grimson
A, Schelter JM, Castle J, Bartel DP, Linsley PS and Johnson JM:
Microarray analysis shows that some microRNAs downregulate large
numbers of target mRNAs. Nature. 433:769–773. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li L, Lin X, Khvorova A, Fesik SW and Shen
Y: Defining the optimal parameters for hairpin-based knockdown
constructs. RNA. 13:1765–1774. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
McIntyre GJ, Arndt AJ, Gillespie KM, Mak
WM and Fanning GC: A comparison of multiple shRNA expression
methods for combinatorial RNAi. Genet Vaccines Ther. 9:92011.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Westerink WM and Schoonen WG: Phase II
enzyme levels in HepG2 cells and cryopreserved primary human
hepatocytes and their induction in HepG2 cells. Toxicol In Vitro.
21:1592–1602. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wilkening S and Bader A: Influence of
culture time on the expression of drug-metabolizing enzymes in
primary human hepatocytes and hepatoma cell line HepG2. J Biochem
Mol Toxicol. 17:207–213. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Prijic S and Sersa G: Magnetic
nanoparticles as targeted delivery systems in oncology. Radiol
Oncol. 45:1–16. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Song H and Yang PC: Construction of shRNA
lentiviral vector. N Am J Med Sci. 2:598–601. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wotherspoon S, Dolnikov A, Symonds G and
Nordon R: Susceptibility of cell populations to transduction by
retroviral vectors. J Virol. 78:5097–5102. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Johnston JC, Gasmi M, Lim LE, Elder JH,
Yee JK, Jolly DJ, Campbell KP, Davidson BL and Sauter SL: Minimum
requirements for efficient transduction of dividing and nondividing
cells by feline immunodeficiency virus vectors. J Virol.
73:4991–5000. 1999.PubMed/NCBI
|
|
47
|
Orang Valinezhad A, Safaralizadeh R and
Kazemzadeh-Bavili M: Mechanisms of miRNA-mediated gene regulation
from common downregulation to mRNA-specific upregulation. Int J
Genomics. 2014:9706072014.PubMed/NCBI
|
|
48
|
Zeng Y and Cullen BR: Efficient processing
of primary microRNA hairpins by Drosha requires flanking
nonstructured RNA sequences. J Biol Chem. 280:27595–27603. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Anderson L and Seilhamer J: A comparison
of selected mRNA and protein abundances in human liver.
Electrophoresis. 18:533–537. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Schwanhäusser B, Busse D, Li N, Dittmar G,
Schuchhardt J, Wolf J, Chen W and Selbach M: Global quantification
of mammalian gene expression control. Nature. 473:337–342. 2011.
View Article : Google Scholar : PubMed/NCBI
|