Introduction
Gastric cancer (GC) is the fourth most common lethal
neoplasm and the second most common cause of cancer-associated
mortality worldwide (1). The primary
cause of GC with a poor prognosis is the GC being diagnosed at an
advanced stage (2). In order to
improve the outcome of GC, it is an urgent requirement to identify
genetic events regulating GC cell proliferation.
An increasing number of studies have indicated that
microRNAs (miRNAs/miRs) are a class of small, short non-coding RNAs
(18–24 nucleotides) that have emerged as important
post-transcriptional regulators by targeting the 3′-untranslated
region (3′-UTR) of the mRNA of their target genes (3–5). It was
reported that certain miRNAs act as oncogenes or tumor suppressors.
miR-761 was reported to promote the progression and metastasis of
non-small cell lung cancer by targeting ING4 and TIMP2 (6). miR-761 was revealed to be upregulated
and to regulate tumorigenesis in hepatocellular carcinoma by
targeting Mitofusin-2 (7). The
results of a study undertaken by Shi et al (8) indicated that miR-761 acted as a tumor
suppressor that inhibited tumor progression by targeting MSI1 in
ovarian carcinoma (8). However, the
molecular mechanisms underlying miR-761 in GC remains largely
unknown. The results of the present study demonstrated that miR-761
promoted GC cell proliferation via targeting the 3′-UTR of GSK3β.
The results provided novel insight into the mechanisms of GC tumor
development mediated by miR-761.
Materials and methods
Clinical specimens
A total of 8 gastric carcinoma (GC) tissues [4 male
and 4 female patients, age range 35–65 years (mean age, 40±2
years)] and two normal gastric mucosal tissues [1 female (age 36)
and 1 male (age 50) patients] were obtained from the Department of
Gastroenterology, Huaihe Hospital (North campus), Henan University
(Kaifeng, China) between 1 February 2015 and 1 October 2015. The
present study was approved by the Ethics Committee of Huaihe
Hospital (North campus), Henan University (Kaifeng, China). All
participants provided written informed consent. Tissue samples were
stored in frozen liquid nitrogen following collection.
Cell culture
Human gastric cancer SGC-7901, MGC-803, MKN-45 and
AGS cell lines were provided by the American Type Culture
Collection (Manassas, VA, USA), and maintained in Dulbecco's
modified Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum
(FBS; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany, USA), 100 U/ml
penicillin-streptomycin (Invitrogen; Thermo Fisher Scientific,
Inc.), and human gastric epithelial cells (HGECs) were purchased
from Wuhan PriCells Biomedical Technology Co., Ltd. (Wuhan, China)
and maintained in PriCells medium (Wuhan PriCells Biomedical
Technology Co., Ltd.). All cells were cultured at 37°C in a
humidified incubator with 5% CO2.
Plasmids and transfection
Transfection of the cells with 2 µM miRNA-761 mimics
or miR-761 inhibitors (miR-761-in; GeneCopoeia, Inc., Rockville,
MD, USA) and their negative controls was performed using
Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocols. SGC-7901 cells were
infected with GSK3β si-RNAs, which were designed and synthesized by
GeneCopoeia, Inc. Transfection of siRNAs was performed using
Lipofectamine 2000, according to the manufacturer's protocols.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from clinical tissues and
cells using TRIzol reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) according to the manufacturer's protocols. The miRNA Q-PCR
Detection kit (GeneCopoeia, Inc.) was used for quantification of
miRNA levels according to the manufacturer's protocols. U6 was used
as an internal control. The 2−ΔΔCq method was used to
quantify relative RNA expression. All procedures were performed in
triplicate (9).
MTT assays and colony formation
Cell proliferation assays were conducted using MTT
assays, SGC-7901 cells (3×103 cells/well) were seeded
onto 96-well plates with 100 µl DMEM supplemented with 10% FBS.
Following incubation of cells for 1, 2, 3, 4, 5 and 6 days, 20 µl 5
mg/ml MTT solution (Sigma-Aldrich; Merck KGaA) was added each well
and incubated for 4 h, and then medium was removed and 150 µl DMSO
(Sigma-Aldrich; Merck KGaA) was added. Next, the absorbance of each
well was measured using a microplate reader set at 490 nm.
For the colony formation assay, transfected SGC-7901
cells (1×103 cells/well) were added to each well of a
6-well plate and incubated for ~2 weeks until the colony was
clearly formed. Next, the cells were fixed with 4% methanol at room
temperature for 30 min and stained with 0.5% crystal violet for 10
min at room temperature. Visible colonies were manually
counted.
Cell cycle assays by flow
cytometry
For analysis of the cell cycle, SGC-7901 cells were
harvested after 48 h transfection, prior to being washed with PBS
and then fixed in ice-cold 70% ethanol at 4°C overnight. The next
day, the cell were incubated with RNase A at 37°C for 30 min, and
then stained with propidium iodide (PI; Sigma-Aldrich; Merck KGaA)
at 4°C for 30 min in the dark, prior to the cells being analyzed by
a flow cytometer using the CellQuest Pro software version 5.1 (BD
Biosciences, Franklin Lakes, NJ, USA).
Luciferase assays
The GSK3β 3′-UTR and the GSK3β 3′-UTR mutant were
amplified and cloned into the downstream of pGL3/luciferase vector
(Promega Corporation, Madison, WI, USA). Cells were co-transfected
with miR-761 mimics, miR-761-in or the relative miR-NC control and
GSK3β 3′UTR or the mutant 3′UTR using Lipofectamine 2000 reagent
(Invitrogen; Thermo Fisher Scientific, Inc.). Following
transfection for 48 h, firefly and Renilla luciferase
activities were performed sing the dual-luciferase assay system
(Promega Corporation).
Western blotting
Cells were harvested and lysed with
radioimmunoprecipitation assay buffer, and the protein
concentration was measured using a bicinchoninic acid assay kit
(cat. no. #23227; Thermo Fisher Scientific, Inc.). Equal amounts
proteins (50 µg) were separated by 10% SDS-PAGE gels and
transferred onto polyvinylidene difluoride membranes. The membranes
were blocked with 5% skimmed milk at room temperature for 1 h and
then probed with 1:1,000 diluted anti-GSK3β (cat. no. ab32391),
anti-CyclinD1 (cat. no. ab134175) and anti-P27 (cat. no. ab32034)
primary antibodies (Abcam, Cambridge, MA, USA) at 4°C overnight.
The membrane was subsequently probed with a horseradish
peroxidase-conjugated secondary antibody rabbit anti-mouse IgG
(cat. no. P0023D; 1:5,000; Beyotime Institute of Biotechnology,
Haimen, China) for 2 h at room temperature. β-actin (dilution
1:5,000; cat. no. ab8227; Abcam) was used as the internal control
and was incubated at 4°C overnight. Signals were visualized using
enhanced chemiluminescent substrates (EMD Millipore, Billerica, MA,
USA), according to the manufacturer's protocols, and was analyzed
using the Quantity One software version 4.6 (Bio-Rad Laboratories,
Inc., Hercules, CA, USA).
Statistical analysis
All data are presented as the mean ± standard
deviation and all statistical analyses were performed using SPSS
18.0 software (SPSS, Inc., Chicago, IL, USA). All experiments were
repeated at least three times independently. Student's t-test was
used for pair-wise comparisons and one-way analysis of variance,
followed by Tukey's post hoc test was used for multiple
comparisons. P<0.05 was considered to indicate a statistically
significant difference.
Results
miR-761 was upregulated in human GC
tissues and GC cell lines
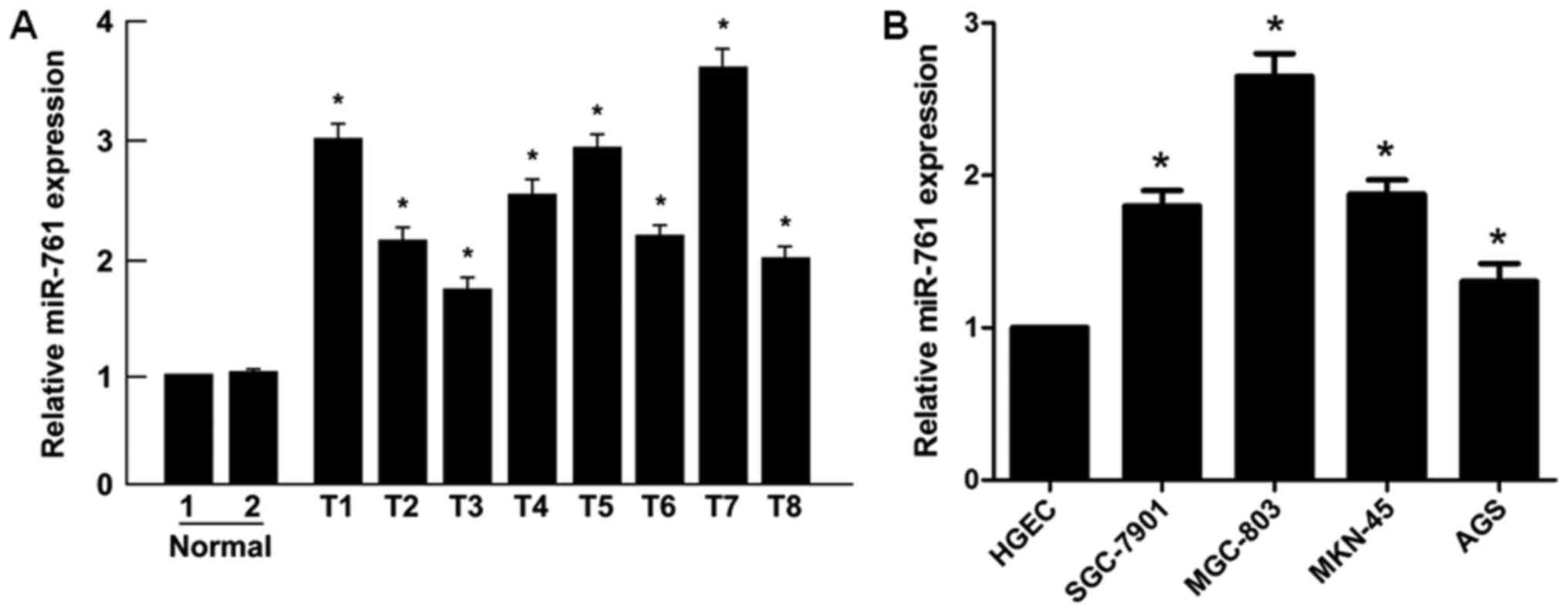
Initially, it was revealed that the expression
levels of miR-761 were significantly upregulated in GC tissues
compared with those in normal gastric mucosal tissues (P<0.05;
Fig. 1A). Similarly, significantly
higher levels of miR-761 were detected in the human GC SGC-7901,
MGC-803, MKN-45 and AGS cell lines compared with those in the human
gastric epithelial cells (HGEC; P<0.05; Fig. 1B). Taken together, these results
indicated that miR-761 is upregulated in GC primary tumors and cell
lines.
miR-761 promoted, while miR-761-in
suppressed GC cell proliferation
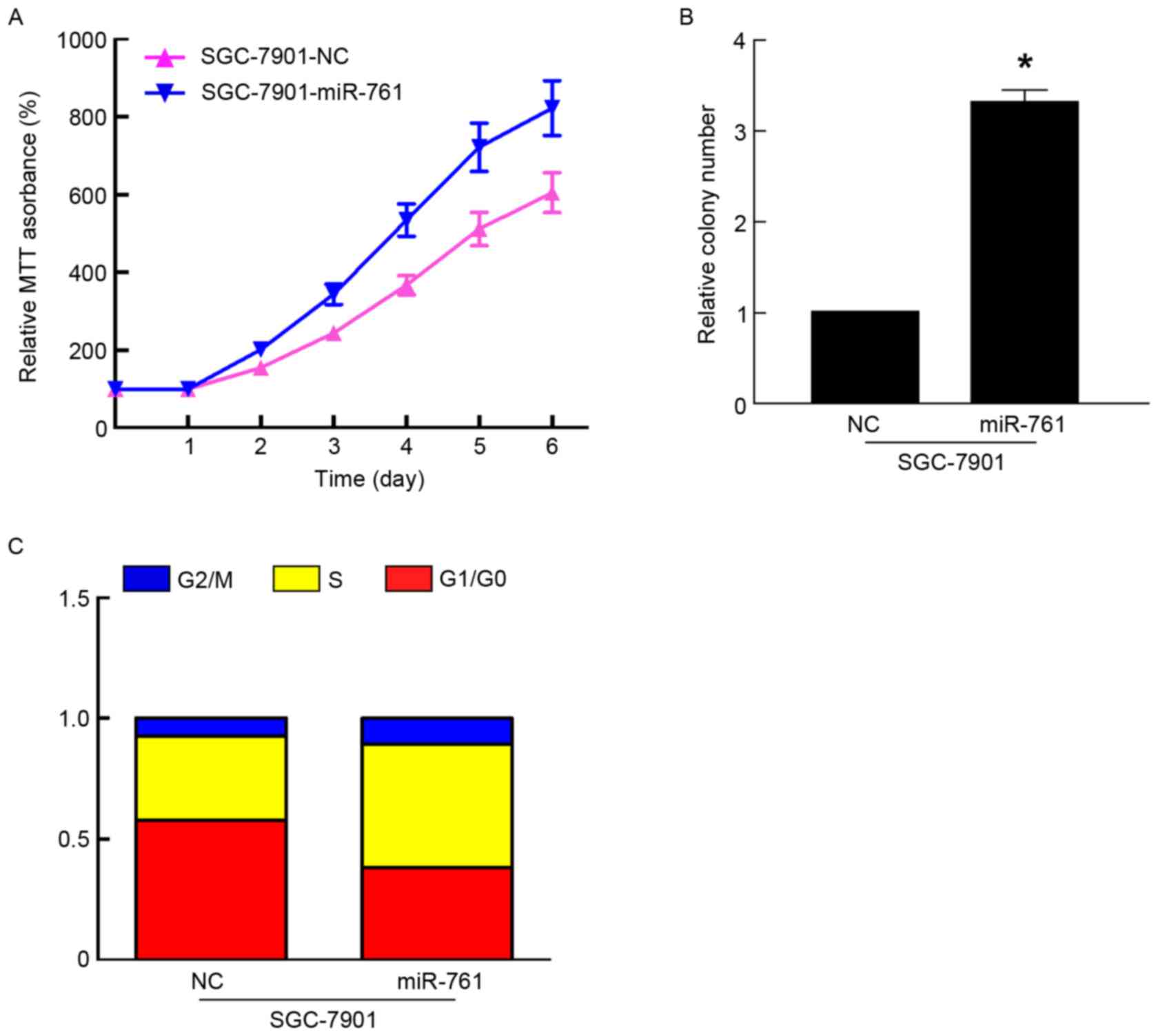
To further characterize the functional importance of
miR-761 in GC progression, its effect on the proliferation of GC
cells was assessed using MTT and colony formation assays. The
results of the MTT and colony formation assays indicated that
overexpression of miR-761 in SGC-7901 cells significantly promoted
cell proliferation and colony formation (P<0.05; Fig. 2A and B). As proliferation is directly
associated with the cell cycle, the effect of miR-761 on cell cycle
progression was detected in SGC-7901 cells. Compared with the
SGC-7901 cells transfected with miR-NC, miR-761 decreased the
percentage of G1/G0 phase cells, and increased the percentage of S
phase cells (Fig. 2C). However,
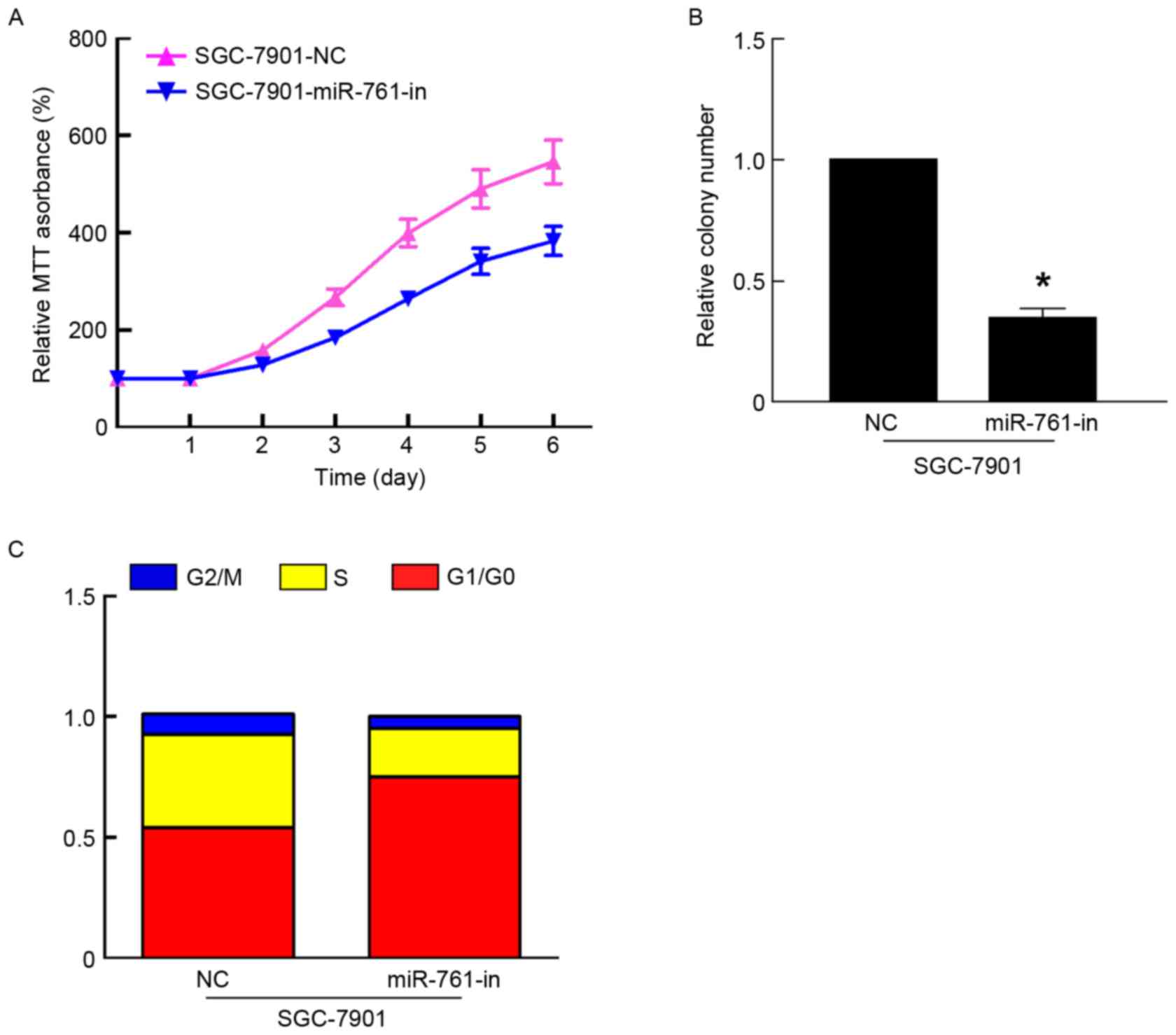
miR-761-in significantly suppressed the proliferation of GC cells
using MTT and colony formation assays (P<0.05; Fig. 3A and B). In addition, flow cytometry
revealed that miR-761-in increased the percentage of SGC-7901 cells
in the G1/G0 phase, and decreased the percentage of SGC-7901 cells
in the S phase (Fig. 3C). Taken
together, these results suggested that miR-761 promoted GC growth
by regulating the cell cycle.
miR-761 directly targets GSK3β by
binding to its 3′-UTR and altering levels of proteins associated
with cell proliferation and the cell cycle in GC cells
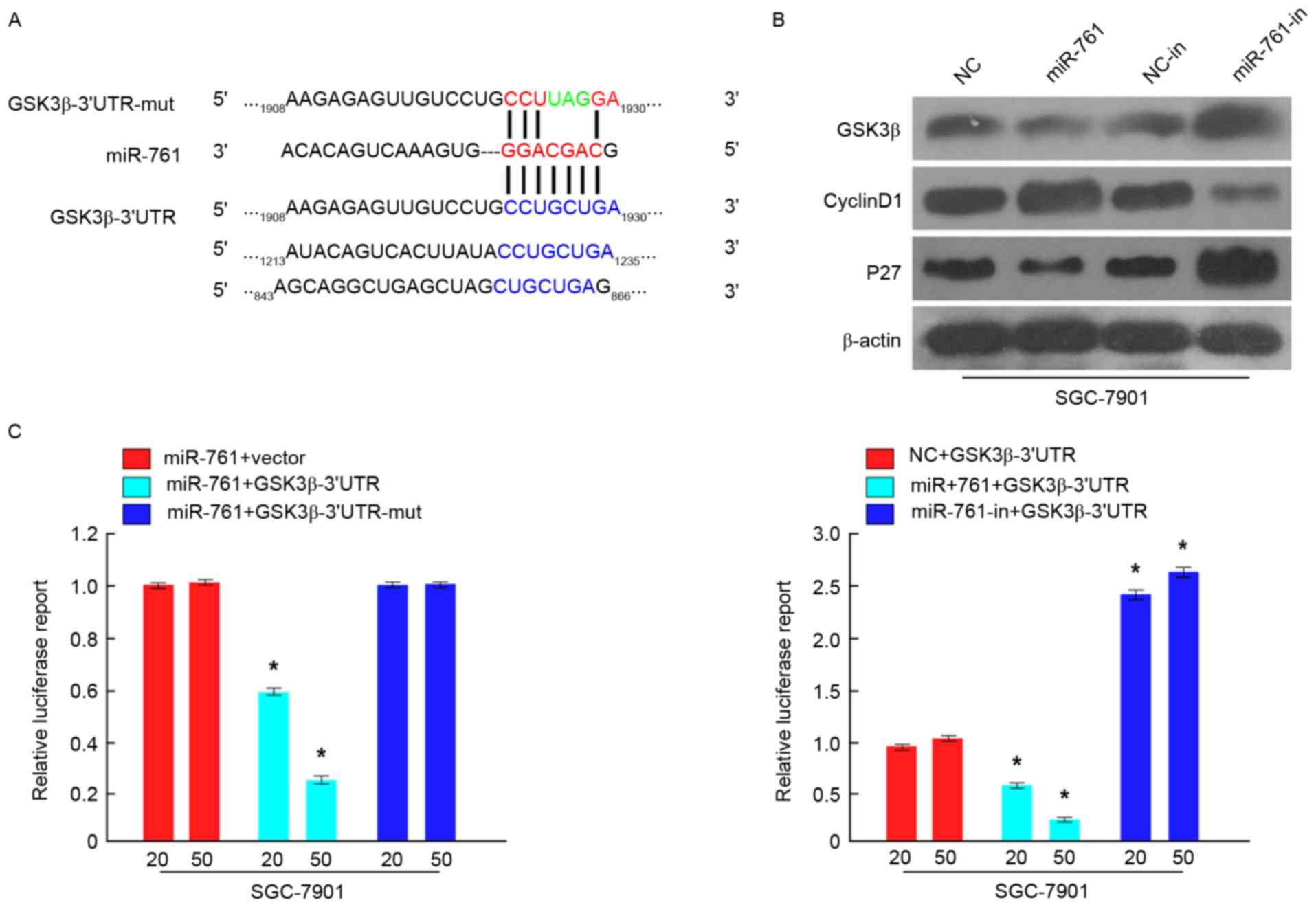
To investigate the molecular mechanisms by which
miR-761 promoted GC cell proliferation, GSK3β was predicted as a
potential target of miR-761 using TargetScan (http://www.targetscan.org/) (Fig. 4A). To confirm potential miR-761
binding sites in the 3′-UTR of GSK3β, luciferase assays were used.
As demonstrated in Fig. 4B, the
results indicated that compared with the negative control, miR-761
attenuated the luciferase activity of the wild-type GSK3β-3′-UTR,
but had no effect on the luciferase activity of the
GSK3β-3′-UTR-mut. Furthermore, miR-761-in increased wild-type
3′-UTR GSK3β luciferase activity (Fig.
4C). Additionally, western blot analysis indicated that GSK3β
expression was significantly downregulated in SGC-7901 cells
following transfection with miR-761 and upregulated in
miR-761-in-transfected SGC-7901 cells (P<0.05; Fig. 4C). Cell proliferation and cell cycle
regulatory genes, including Cyclin D1 and P27, were also detected
by western blot analysis. The results revealed that Cyclin D1
expression was increased, while P27 expression was decreased in
miR-761-overexpressing SGC-7901 cells, while miR-761-in exhibited
the opposite effect (Fig. 4B).
Discussion
Numerous studies have demonstrated that miRNAs may
act as activators or inhibitors of tumor proliferation (10–13). An
increasing number of studies have revealed that aberrant expression
of miRNAs serves essential roles in the carcinogenesis of cancer
(14–16). miR-152 was reported to inhibit gastric
cancer cell proliferation and motility by targeting CD151 (17). miR-935 was revealed to promote gastric
cancer cell proliferation by regulating SOX7 (18). The results of a study undertaken by Gu
et al (19) indicted that
miRNA-183 suppressed apoptosis and promoted proliferation and
invasion of gastric cancer cells by targeting PDCD4. miR-761 was a
newly discovered oncogenic miRNA, which was upregulated and
regulates tumorigenesis in hepatocellular carcinoma (7). However, the detailed biological function
of miR-761 in GC remains largely unclear.
The results of the present study demonstrated that
miR-761 expression levels in GC tissues and cells were
significantly upregulated compared with those in normal gastric
mucosal tissues and human gastric epithelial cells. Furthermore,
ectopic miR-761 expression led to a lower percentage of cells in
the G1/G0 phase and a higher percentage of cells in the S phase,
and subsequently promoted GC cell proliferation and increased GC
cell colony formation, while miR-761-in exhibited the opposite
effect. Further experimentation was used to investigate the
molecular mechanism of miR-761 in promoting cell proliferation and
the cell cycle in GC. Bioinformatics analysis indicated that GSK3β,
a crucial modulator of the Wnt/β-catenin signaling pathway
(20), was the direct target of
miR-761. miR-224 was reported to sustain Wnt/β-catenin signaling
and to promote the aggressive phenotype of colorectal cancer by
suppressing the expression of GSK3β/SFRP2 (21). Deng et al (22) indicated that miR-519 promoted the
progression of colorectal cancer through regulating Orai1 via
regulating the Akt/GSK3β signaling pathway. In the present study,
the results of the luciferase reporter assay and western blot
analysis confirmed that miR-761 suppressed GSK3β expression by
targeting the 3′-UTR of GSK3β. Additionally, the cell proliferation
and cell cycle regulatory genes, including Cyclin D1 and P27, were
detected by western blot analysis. Cyclin D1 expression was
increased, while P27 expression was decreased by miR-761 in
SGC-7901 GC cells. Taken together, the results of the present study
revealed that miR-761 functionally suppressed GSK3β expression, and
subsequently regulated cellular proliferation and cell cycle
regulators, Cyclin D1 and P27, which suggests that miR-761 is
associated with cell proliferation and the cell cycle in GC.
In conclusion, miR-761 was upregulated and
functioned as an oncogene in GC. Furthermore, it was revealed that
miR-761 promoted cell proliferation and the cell cycle by targeting
GSK3β. The present study therefore demonstrated that miR-761 could
be considered as a novel therapeutic target for patients with
GC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed in this study are
included in this article.
Authors' contributions
XS conceived and designed the experiments. XS, HH
and KL performed the experiments. KL and MZ collected the samples
and analyzed the data. All authors wrote and approved the final
manuscript.
Ethics statement and consent to
participate
The present study was approved by the Ethics
Committee of Huaihe Hospital (North campus), Henan University
(Kaifeng, China). All participants provided written informed
consent.
Patient consent for publication
All patients provided informed consent for the
publication of their data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pasechnikov V, Chukov S, Fedorov E,
Kikuste I and Leja M: Gastric cancer: Prevention, screening and
early diagnosis. World J Gastroenterol. 20:13842–13862. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shen F, Cai WS, Feng Z, Li JL, Chen JW,
Cao J and Xu B: MiR-492 contributes to cell proliferation and cell
cycle of human breast cancer cells by suppressing SOX7 expression.
Tumour Biol. 36:1913–1921. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang X, Ke X, Pu Q, Yuan Y, Yang W, Luo
X, Jiang Q, Hu X, Gong Y, Tang K, et al: MicroRNA-410 acts as
oncogene in NSCLC through downregulating SLC34A2 via activating
Wnt/β-catenin pathway. Oncotarget. 7:14569–14585. 2016.PubMed/NCBI
|
|
5
|
Cheng CM, Shiah SG, Huang CC, Hsiao JR and
Chang JY: Up-regulation of miR-455-5p by the TGF-β-SMAD signalling
axis promotes the proliferation of oral squamous cancer cells by
targeting UBE2B. J Pathol. 240:38–49. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yan A, Yang C, Chen Z, Li C and Cai L:
MiR-761 promotes progression and metastasis of non-small cell lung
cancer by targeting ING4 and TIMP2. Cell Physiol Biochem. 37:55–66.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhou X, Zhang L, Zheng B, Yan Y, Zhang Y,
Xie H, Zhou L, Zheng S and Wang W: MicroRNA-761 is upregulated in
hepatocellular carcinoma and regulates tumorigenesis by targeting
Mitofusin-2. Cancer Sci. 107:424–432. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shi C and Zhang Z: miR-761 inhibits tumor
progression by targeting MSI1 in ovarian carcinoma. Tumour Biol.
37:5437–5443. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yao K, He L, Gan Y, Zeng Q, Dai Y and Tan
J: MiR-186 suppresses the growth and metastasis of bladder cancer
by targeting NSBP1. Diagn Pathol. 10:1462015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wei P, Qiao B, Li Q, Han X, Zhang H, Huo Q
and Sun J: MicroRNA-340 suppresses tumorigenic potential of
prostate cancer cells by targeting high-mobility group
nucleosome-binding domain 5. DNA Cell Biol. 35:33–43. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang C, Liu K, Li T, Fang J, Ding Y, Sun
L, Tu T, Jiang X, Du S, Hu J, et al: miR-21: A gene of dual
regulation in breast cancer. Int J Oncol. 48:161–172. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Deng D, Wang L, Chen Y, Li B, Xue L, Shao
N, Wang Q, Xia X, Yang Y and Zhi F: MicroRNA-124-3p regulates cell
proliferation, invasion, apoptosis, and bioenergetics by targeting
PIM1 in astrocytoma. Cancer Sci. 107:899–907. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Song H, Zhang Y, Liu N, Wan C, Zhang D,
Zhao S, Kong Y and Yuan L: miR-92b regulates glioma cells
proliferation, migration, invasion, and apoptosis via PTEN/Akt
signaling pathway. J Physiol Biochem. 72:201–211. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lu YC, Chang JT, Chan EC, Chao YK, Yeh TS,
Chen JS and Cheng AJ: miR-196, an emerging cancer biomarker for
digestive tract cancers. J Cancer. 7:650–655. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang F, Li B and Xie X: The roles and
clinical significance of microRNAs in cervical cancer. Histol
Histopathol. 31:131–139. 2016.PubMed/NCBI
|
|
17
|
Zhai R, Kan X, Wang B, Du H, Long Y, Wu H,
Tao K, Wang G, Bao L, Li F and Zhang W: miR-152 suppresses gastric
cancer cell proliferation and motility by targeting CD151. Tumour
Biol. 35:11367–11373. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang M, Cui G, Ding M, Yang W, Liu Y, Dai
D and Chen L: miR-935 promotes gastric cancer cell proliferation by
targeting SOX7. Biomed Pharmacother. 79:153–158. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gu W, Gao T, Shen J, Sun Y, Zheng X, Wang
J, Ma J, Hu XY, Li J and Hu MJ: MicroRNA-183 inhibits apoptosis and
promotes proliferation and invasion of gastric cancer cells by
targeting PDCD4. Int J Clin Exp Med. 7:2519–2529. 2014.PubMed/NCBI
|
|
20
|
Taelman VF, Dobrowolski R, Plouhinec JL,
Fuentealba LC, Vorwald PP, Gumper I, Sabatini DD and De Robertis
EM: Wnt signaling requires sequestration of glycogen synthase
kinase 3 inside multivesicular endosomes. Cell. 143:1136–1148.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li T, Lai Q, Wang S, Cai J, Xiao Z, Deng
D, He L, Jiao H, Ye Y, Liang L, et al: MicroRNA-224 sustains
Wnt/β-catenin signaling and promotes aggressive phenotype of
colorectal cancer. J Exp Clin Cancer Res. 35:212016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Deng W, Wang J, Zhang J, Cai J, Bai Z and
Zhang Z: Orai1, a direct target of microRNA-519, promotes
progression of colorectal cancer via Akt/GSK3β signaling pathway.
Dig Dis Sci. 61:1553–1560. 2016. View Article : Google Scholar : PubMed/NCBI
|