Introduction
As one of the most common malignant tumors, lung
cancer is characterized by high morbidity and mortality rates
(1). There are many types of lung
cancer, and the number of patients with non-small cell lung cancer
(NSCLC) is the largest, accounting for approximately 80% of the
total number of patients with lung cancer (2). Due to the complex diagnosis of lung
cancer and imperceptible early symptoms, 70–80% of patients with
lung cancer are initially diagnosed at an advanced stage (3).
The p53 gene was originally considered a tumor
suppressor gene that exerts an antitumor effect by regulating cell
growth and apoptosis. After mutation, the wild-type p53 gene
becomes the mutant-type p53 gene with an apoptosis-inhibiting
effect, which can stimulate and promote the growth of tumor cells,
losing the normal antitumor effect (4). Previous findings have confirmed that p53
gene mutates in 50% of human tumor tissues, and can be used as a
tumor biomarker (5). Matrix
metalloproteinase (MMP) is a zinc ion-dependent protease family, of
which MMP-2 is a major member (6,7). MMP-2 is
mainly secreted by tumor cells and interstitial cells in the form
of zymogen. After hydrolysis and activation, MMP-2 can degrade type
IV collagen in the basement membrane. When the collagen in the
basement membrane is degraded and destroyed, MMP-2 further affects
the hindering effect of the basement membrane on tumor cells
(8). MMP-2 has been shown to play a
key role in degrading the extracellular matrix and promoting tumor
invasion and metastasis (9,10).
In recent years, surgery after adjuvant chemotherapy
has become a new strategy for the treatment of lung cancer, but
there are no unified criteria for the chemotherapeutic effect
clinically at present. Therefore, in this study, the effects of
interventional chemotherapy on the protein expressions of p53 and
MMP-2 in tumor tissues of NSCLC patients, and their correlations
with tumor cell apoptosis in patients and chemotherapeutic effect
were investigated. The mRNA expression levels of p53 and MMP-2 in
tumor tissues before and after chemotherapy were detected via
reverse transcription-quantitative polymerase chain reaction
(RT-qPCR). Changes in the protein expressions of p53 and MMP-2 in
tumor tissues and tissue cell apoptosis before and after
chemotherapy were further studied, and the correlations of the
expression of p53 and MMP-2 in tumor tissues of patients with
chemotherapeutic effect before chemotherapy were analyzed combined
with clinical data.
Materials and methods
Materials
Ribonucleic acid (RNA) extraction kit (Invitrogen,
Carlsbad, CA, USA); primer synthesis, RT kit, real-time
fluorescence quantitative PCR kit (Takara Biotechnology Co., Ltd.,
Dalian, China); p53, MMP-2, glyceraldehyde-3-phosphate
dehydrogenase (GAPDH) primary antibodies and horseradish peroxidase
(HRP)-labeled secondary antibodies (Proteintech, Wuhan, China);
terminal deoxynucleoitidyl transferase-mediated dUTP nick
end-labeling (TUNEL) apoptosis assay kit, DAPI staining solution
and Triton X-100 (Nanjing KeyGen Biotech, Nanjing, China); protein
extraction kit, and bicinchoninic acid (BCA) protein assay kit
(Beyotime Biotechnology, Nantong, China) were used in the present
study.
A total of 80 elderly patients with NSCLC aged ≥65
years, admitted to the Department of Surgery, The Second Hospital
of Dalian Medical University (Dalian, China) were selected,
including 44 males and 36 females with an average age of 75.5
years. Tissue specimens were obtained from all the patients via
bronchoscopy and computed tomography (CT)-guided biopsy, and the
patients were diagnosed with NSCLC via pathological examination but
received no chemotherapy. Patients were treated with navelbine +
cisplatin chemotherapy. At 2 weeks after chemotherapy, blood and
urine routine, hepatic and renal functions, cardiac function and
chest CT were reviewed. Patients underwent surgery at 2 weeks after
chemotherapy, if there were no surgical contraindications.
Specimens after chemotherapy were used as postoperative
pathological specimens; one part of the specimens before and after
chemotherapy was fixed with formalin and embedded into paraffin,
while the other part was directly cryopreserved at −80°C. In the
study, the acquisition of all the specimens was approved by the
Clinical Ethics Committee of The Second Hospital of Dalian Medical
University (Dalian, China). All the patients enrolled or their
families signed the informed consent.
Detection of mRNA expression of p53
and MMP-2 in tumor tissue specimens in patients before and after
chemotherapy via RT-qPCR
Approximately 100 mg tumor tissue specimens before
and after chemotherapy were taken from each patient, and the total
RNA was extracted from the tissues to be tested according to the
protocol of the RNA extraction kit. Specimens with an absorbance
(A) 260/A280 ratio of 1.8–2.0 were selected for subsequent
experiments. Reverse transcription was performed according to the
protocol of the RT kit to obtain the complementary DNA (cDNA). With
cDNA as a template, the mRNA expression of p53 and MMP-2 was
detected via quantitative PCR, and GAPDH was used as an internal
control. Primer sequences are shown in Table I. Reaction conditions were as follows:
95°C for 10 min, 95°C for 15 sec, 60°C for 1 min, and amplification
for 35 cycles. The cycle threshold (Cq) value was output from the
instrument software, and the relative expression level was
calculated according to the formula: ΔCq (target gene) = Cq (target
gene) - Cq (control gene).
 | Table I.RT-qPCR primer sequences. |
Table I.
RT-qPCR primer sequences.
| Gene | Primer | Primer sequence |
|---|
| name | name |
| p53 | F: |
5′-TGCGTGTGGAGTATTTGGATG-3′ |
|
| R: |
5′-TGGTACAGTCAGAGCCAACCTC-3′ |
| MMP-2 | F: |
5′-CTCATCGCAGATGCCTGGAA-3′ |
|
| R: |
5′-TTCAGGTAATAGGCACCCTTGAAGA-3′ |
| GAPDH | F: |
5′-GCACCGTCAAGGCTGAGAAC-3′ |
|
| R: |
5′-TGGTGAAGACGCCAGTGGA-3′ |
Detection of protein expression of p53
and MMP-2 in tumor tissue specimens in patients before and after
chemotherapy via western blot analysis
An appropriate number of frozen tissue specimens of
patients were taken, lysed using a tissue lysis buffer, and
centrifuged at 3000 × g at 4°C for 15 min, prior to the supernatant
being collected. The concentration of protein extracted was
determined using a BCA kit, and 50 µg protein was subjected to
sodium dodecyl sulfate polyacrylamide gel electrophoresis
(SDS-PAGE), transferred onto a polyvinylidene fluoride (PVDF)
membrane using the wet method, sealed using the blocking solution
at room temperature for 2 h, and added with rabbit anti-human p53
and MMP-2 primary monoclonal antibodies (1:1,000; cat. nos. 2527
and 87809, respectively; both obtained from Cell Signaling
Technology, Inc., Danvers, MA, USA) for incubation at 4°C
overnight. The membrane was washed with TTBS 3 times and added with
anti-rabbit IgG, HRP-linked secondary polyclonal antibody (1:2,000;
cat. no. 7074; Cell Signaling Technology, Inc.) for incubation at
room temperature for 2 h. Color was developed using the
electrochemiluminescence (ECL) solution in the dark, followed by
scanning and recording with a gel imager. The gray scale was
analyzed and compared with GADPH as an internal reference.
Detection of protein expression of p53
and MMP-2 in tumor tissue specimens in patients before and after
chemotherapy via IHC
Tissue sections were routinely dewaxed, followed by
antigen retrieval using the citrate buffer solution via microwave.
Normal serum blocking solution was dropwise added to block the
sections, p53 and MMP-2 primary antibodies (diluted at 1:100) were
added, and the sections were placed in a refrigerator at 4°C
overnight. The sections were washed with phosphate-buffered saline
(PBS) 3 times, added with the biotin-labeled secondary antibodies
for incubation for another 15 min, and washed again with PBS 3
times, followed by color development in the dark with DAB solution,
re-staining with hematoxylin, sealing via gum, observation and
photography under a microscope (TE2000-U, Nikon Corp., Tokyo,
Japan).
Staining results were evaluated in randomly selected
areas: the expression intensity was graded and scored according to
the staining depth and percentage of positive cells. Staining
intensity was: no staining (0 point), pale yellow (1 point), brown
yellow (2 points) and dark brown (3 points); the number of positive
cells in high-power fields (×400) was: <5% (0 point), 5–25% (1
point), 26–50% (2 points) and >50% (3 points). If the two points
were added up, a score of >3 points indicated a positive
expression, while that of ≤2 points indicated a negative
expression. Results were statistically analyzed.
Detection of changes in apoptosis of
tumor cells before and after chemotherapy via TUNEL assay
Sections were completely immersed in 4%
paraformaldehyde fixing solution for fixation for 30 min,
permeabilized with 1% Triton X-100 and rinsed 3 times. DNase I
reaction solution (100 µl) already prepared was dropwise added onto
the sections selected for incubation at 37°C for 30 min, and the
sections were rinsed with PBS 3 times. The sections were then added
with 50 µl TdT reaction solution, placed into a warm box, incubated
in the dark at 37°C for 60 min, and rinsed again with PBS 3 times
in the dark. The sections were added with streptavidin-tetraethyl
rhodamine isothiocyanate (TRITC) labeling solution, placed into a
wet box, incubated in the dark at 37°C for 30 min, and rinsed again
with PBS 3 times in the dark. After re-staining via DAPI staining
solution, the sections were incubated in the dark for 10 min, and
rinsed with PBS 3 times in the dark, followed by observation and
photography under a fluorescence microscope (Olympus Corporation,
Tokyo, Japan).
Correlation of p53 and MMP-2 protein
expressions with chemotherapeutic effect
At 2 weeks after surgery, the chemotherapeutic
effect was evaluated according to the therapeutic evaluation
criteria of solid tumor of WHO. The effects were divided into four
types: complete remission, partial remission, stable disease and
progressive disease. Complete remission and partial remission
indicated the effectiveness, while stable disease and progressive
disease indicated the ineffectiveness, followed by data statistics
and analysis.
Statistical analysis
Statistical Product and Service Solutions (SPSS)
17.0 software (International Business Machines Corporation, Armonk,
NY, USA) was used for data processing in the present study.
Measurement data were presented as mean ± standard deviation, and
Student's t-test was used for the intergroup comparison; Chi-square
analysis was used for the comparison of enumeration data between
two groups. P≤0.05 suggested that the difference was statistically
significant.
Results
mRNA expression of p53 and MMP-2 in
tissue specimens of patients before and after chemotherapy
Frozen tumor tissues and paracarcinoma normal
tissues were taken from patients before and after chemotherapy to
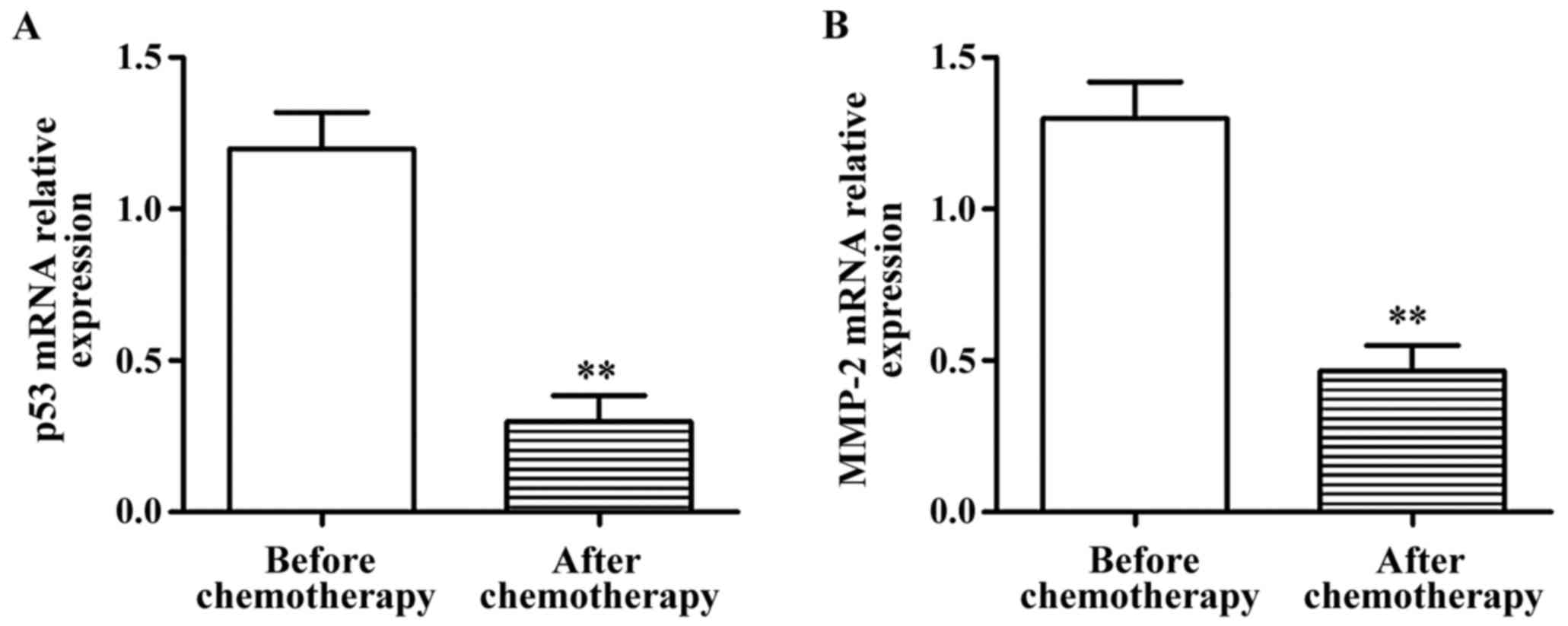
detect the mRNA expression of p53 and MMP-2 via RT-PCR. The results
showed that the mRNA expression of p53 and MMP-2 in lung cancer
tissues after chemotherapy were significantly lower than those
before chemotherapy, and the differences were statistically
significant (p<0.01) (Fig. 1).
Protein expression of p53 and MMP-2 in
tissue specimens of patients before and after chemotherapy
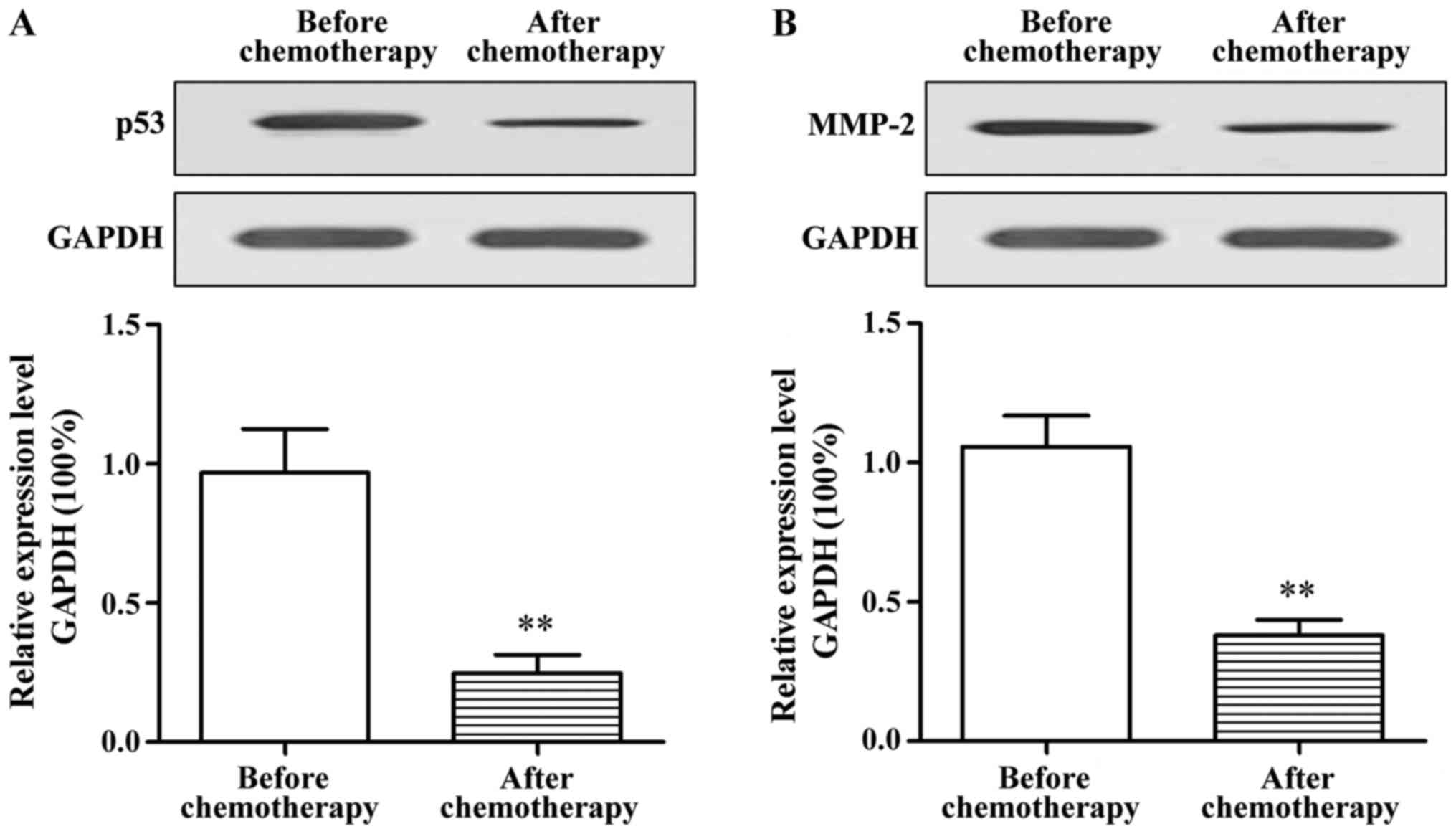
The protein expression of p53 and MMP-2 in tissue
specimens of patients before and after chemotherapy were further
detected via western blot analysis. The results showed that the
protein expression of p53 and MMP-2 in tumor tissues of elderly
patients with lung cancer after chemotherapy was significantly
decreased compared with those before chemotherapy (p<0.01)
(Fig. 2).
Detection of the protein expression of
p53 and MMP-2 in tissue specimens of patients before and after
chemotherapy via IHC
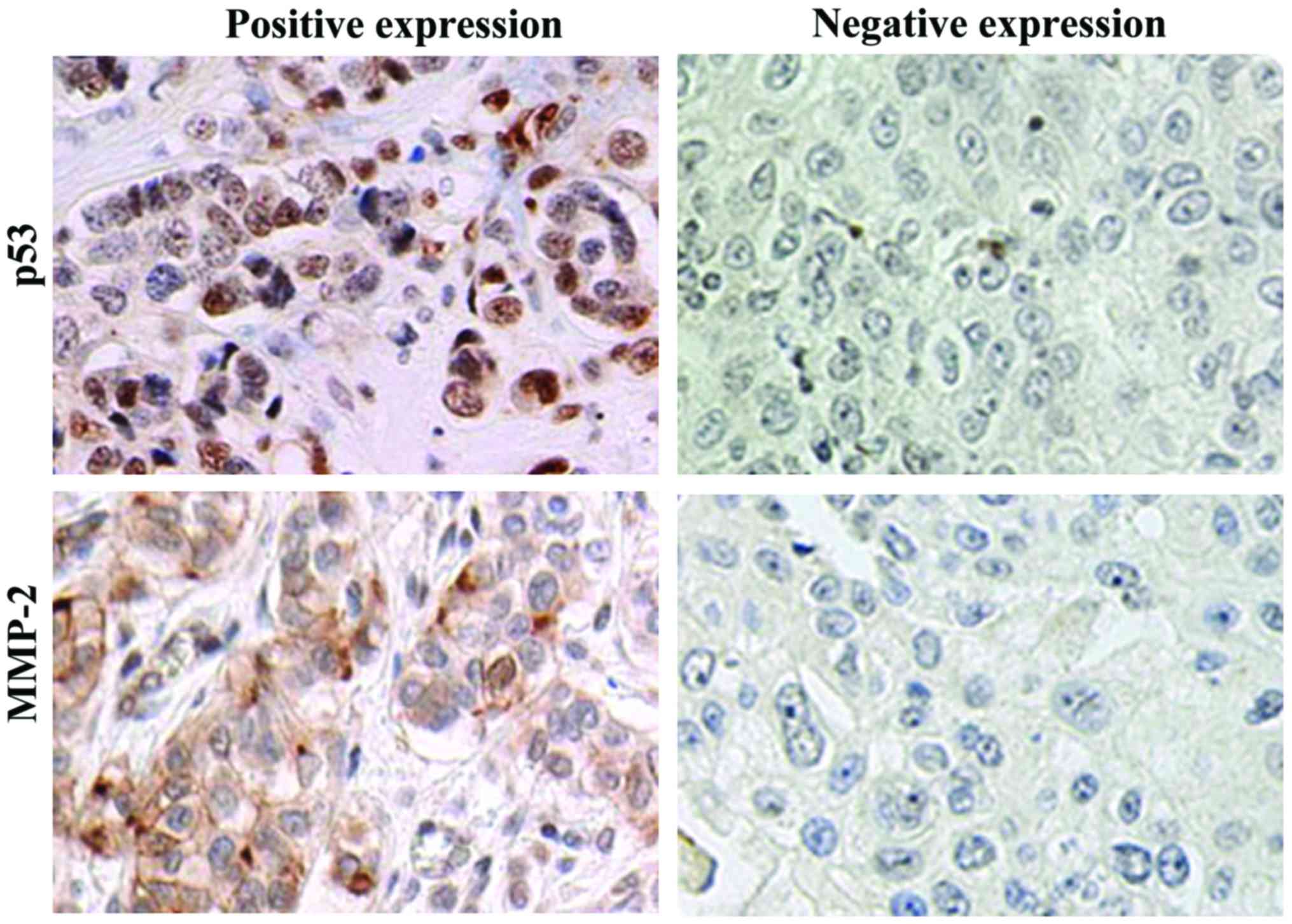
IHC results revealed that p53 protein was located in
the nucleus, while MMP-2 protein was located in the cytoplasm,
showing the diffuse or scattered distribution (Fig. 3). Statistical analysis showed that the
positive expression rates of p53 and MMP-2 in lung cancer tissues
of 80 patients were 76.25% (61/80) and 71.25% (57/80) before
chemotherapy, respectively, and 27.50% (22/80) and 23.75% (19/80)
after chemotherapy, respectively. The positive expression rates of
p53 and MMP-2 after chemotherapy were significantly lower than
those before chemotherapy, and the differences were statistically
significant (p<0.01).
Changes in cell apoptosis in patients
before and after chemotherapy
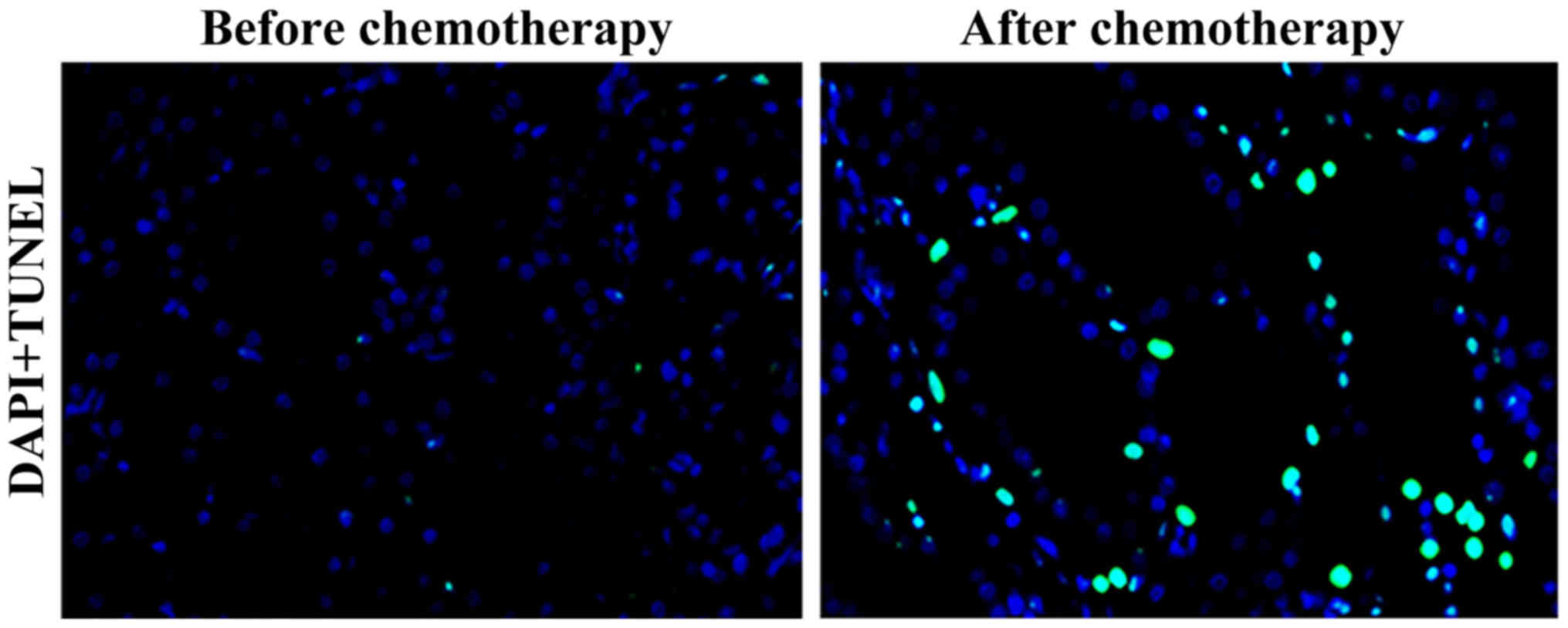
Results of the TUNEL assay showed that the green
fluorescence-stained nucleus indicated the positive cells, namely
apoptotic cells. Compared with that before chemotherapy, the number
of apoptotic cells in tumor tissues of elderly patients with lung
cancer was significantly increased after chemotherapy (Fig. 4).
Correlations of p53 and MMP-2
expressions in lung cancer tissues before chemotherapy with
chemotherapeutic effect
The effective rates of chemotherapy in patients with
a negative expression of p53 and MMP-2 in lung cancer tissues
before chemotherapy were 78.95 and 78.26%, respectively, while
those in patients with positive expressions of p53 and MMP-2 in
lung cancer tissues before chemotherapy were 52.46 and 50.88%,
respectively. The effective rates of chemotherapy in patients with
negative expressions of p53 and MMP-2 in lung cancer tissues before
chemotherapy were significantly higher than those in patients with
positive expressions, and the differences were statistically
significant (p<0.01) (Table
II).
 | Table II.Correlations of p53 and MMP-2
expressions in lung cancer tissues before chemotherapy with
chemotherapeutic effect [n (%)]. |
Table II.
Correlations of p53 and MMP-2
expressions in lung cancer tissues before chemotherapy with
chemotherapeutic effect [n (%)].
|
| p53 | MMP-2 |
|---|
|
|
|
|
|---|
| Chemotherapeutic
effect | Positive |
| Negative | Positive |
| Negative |
|---|
| Effective | 32 (52.46) |
| 15 (78.95) | 29 (50.88) |
| 18 (78.26) |
| Ineffective | 29 |
| 4 | 28 |
| 5 |
| χ2 |
| 4.19 |
|
| 5.07 |
|
| p-value |
| <0.05 |
|
| <0.05 |
|
Discussion
Lung cancer has a high morbidity rate, and its
mortality rate ranks first in malignant tumors in China (11). There are no obvious clinical symptoms
of lung cancer in the early stage, thus patients have been in the
advanced stage when diagnosed. Approximately 40% of patients with
newly diagnosed lung cancer are elderly, aged above 65 years
(12).
The p53 gene is located on human chromosome 17p13,
which is a kind of tumor suppressor gene and pro-apoptosis gene.
Wild-type p53 plays a key role in cellular gene transcription, cell
cycle regulation, apoptosis, cell proliferation and differentiation
(13–15). Previous findings showed that the
mutation of p53 gene is closely related to the occurrence and
development of liver cancer (15,16). The
content of wild-type p53 protein is very low in normal cells, and
it has a short half-life period, and cannot be detected via IHC and
other commonly used methods. However, the mutant-type p53 protein
has a longer half-life period and a higher expression level, and is
easily detected (17,18).
The MMP-2 gene is located on human chromosome 16q21,
which can degrade, not only type IV collagen in basement membrane,
but also type V, VI and X collagen and gelatin following
activation. Extracellular matrix and basement membrane act as
natural barriers in tumor invasion and diffusion processes; thus,
passing through this layer of tissue barrier is a key step in the
tumor cell metastasis (19).
Additionally, tumor cells can specifically express MMP-2 highly to
degrade type IV collagen, and destroy the tissue barrier, thereby
promoting tumor cell invasion and metastasis (20).
In this study, the effects of interventional
chemotherapy on the protein expression of p53 and MMP-2 in tumor
tissues of NSCLC patients, and their correlations with tumor cell
apoptosis in patients and chemotherapeutic effect were
investigated. The mRNA expression levels of p53 and MMP-2 in tumor
tissues before and after chemotherapy were detected via RT-qPCR and
the results showed that the mRNA expression levels of p53 and MMP-2
in lung cancer tissues after chemotherapy were significantly lower
than those prior to chemotherapy. The protein expression levels of
p53 and MMP-2 in tumor tissues before and after chemotherapy were
further studied. The results showed that the protein expression of
p53 and MMP-2 in tumor tissues of elderly patients with lung cancer
after chemotherapy were significantly decreased, and the positive
expression rates of p53 and MMP-2 after chemotherapy were
significantly lower than those before chemotherapy. In addition,
the effect of chemotherapy on apoptosis of tissue cells was studied
via TUNEL assay. The results revealed that the number of apoptotic
cells in tumor tissues of elderly patients with lung cancer was
significantly higher after chemotherapy than those before
chemotherapy. It was also found combined with clinical data
analysis that the effective rates of chemotherapy in patients with
negative expressions of p53 and MMP-2 in lung cancer tissues before
chemotherapy were significantly higher than those in patients with
positive expressions.
In conclusion, the results of the present study
preliminarily demonstrate that the expression levels of p53 and
MMP-2 can be used to predict the sensitivity of elderly patients
with lung cancer to chemotherapy drugs, and both p53 and MMP-2 may
serve as molecular markers for predicting the sensitivity of lung
cancer to chemotherapy, better serving the clinical treatment.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XSZ and KYW performed PCR. JQG and RJL were
responsible for western blot analysis. QBG and LS helped with IHC.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
In the study, the acquisition of all the specimens
was approved by the Clinical Ethics Committee of The Second
Hospital of Dalian Medical University (Dalian, China). All the
patients enrolled or their families signed the informed
consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen QY, Zheng Y, Jiao DM, Chen FY, Hu HZ,
Wu YQ, Song J, Yan J, Wu LJ and Lv GY: Curcumin inhibits lung
cancer cell migration and invasion through Rac1-dependent signaling
pathway. J Nutr Biochem. 25:177–185. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Feng B, Zhang K, Wang R and Chen L:
Non-small-cell lung cancer and miRNAs: Novel biomarkers and
promising tools for treatment. Clin Sci (Lond). 128:619–634. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johnson DH: Locally advanced, unresectable
non-small cell lung cancer: New treatment strategies. Chest. 117
Suppl 1:S123–S126. 2000. View Article : Google Scholar
|
|
4
|
Deveraux QL and Reed JC: IAP family
proteins - suppressors of apoptosis. Genes Dev. 13:239–252. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Holcik M, Gibson H and Korneluk RG: XIAP:
Apoptotic brake and promising therapeutic target. Apoptosis.
6:253–261. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chia CY, Kumari U and Casey PJ: Breast
cancer cell invasion mediated by Gα12 signaling involves expression
of interleukins-6 and −8, and matrix metalloproteinase-2. J Mol
Signal. 9:62014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stoeltzing O, Ahmad SA, Liu W, McCarty MF,
Wey JS, Parikh AA, Fan F, Reinmuth N, Kawaguchi M, Bucana CD, et
al: Angiopoietin-1 inhibits vascular permeability, angiogenesis,
and growth of hepatic colon cancer tumors. Cancer Res.
63:3370–3377. 2003.PubMed/NCBI
|
|
8
|
Zhang W, Wang F, Xu P, Miao C, Zeng X, Cui
X, Lu C, Xie H, Yin H, Chen F, et al: Perfluorooctanoic acid
stimulates breast cancer cells invasion and up-regulates matrix
metalloproteinase-2/-9 expression mediated by activating NF-κB.
Toxicol Lett. 229:118–125. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Iochmann S, Bléchet C, Chabot V, Saulnier
A, Amini A, Gaud G, Gruel Y and Reverdiau P: Transient RNA
silencing of tissue factor pathway inhibitor-2 modulates lung
cancer cell invasion. Clin Exp Metastasis. 26:457–467. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Safranek J, Pesta M, Holubec L, Kulda V,
Dreslerova J, Vrzalova J, Topolcan O, Pesek M, Finek J and Treska
V: Expression of MMP-7, MMP-9, TIMP-1 and TIMP-2 mRNA in lung
tissue of patients with non-small cell lung cancer (NSCLC) and
benign pulmonary disease. Anticancer Res. 29:2513–2517.
2009.PubMed/NCBI
|
|
11
|
Shi Y and Sun Y: Medical management of
lung cancer: Experience in China. Thorac Cancer. 6:10–16. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gridelli C, Aapro M, Ardizzoni A, Balducci
L, De Marinis F, Kelly K, Le Chevalier T, Manegold C, Perrone F,
Rosell R, et al: Treatment of advanced non-small-cell lung cancer
in the elderly: Results of an international expert panel. J Clin
Oncol. 23:3125–3137. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu W, Wang F, Tang J, Liu X, Yuan Z, Nie C
and Wei Y: Proapoptotic protein Smac mediates apoptosis in
cisplatin-resistant ovarian cancer cells when treated with the
anti-tumor agent AT101. J Biol Chem. 287:68–80. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hussain AR, Uddin S, Ahmed M, Bu R, Ahmed
SO, Abubaker J, Sultana M, Ajarim D, Al-Dayel F, Bavi PP, et al:
Prognostic significance of XIAP expression in DLBCL and effect of
its inhibition on AKT signalling. J Pathol. 222:180–190. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shangary S and Wang S: Targeting the
MDM2-p53 interaction for cancer therapy. Clin Cancer Res.
14:5318–5324. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shangary S, Qin D, McEachern D, Liu M,
Miller RS, Qiu S, Nikolovska-Coleska Z, Ding K, Wang G, Chen J, et
al: Temporal activation of p53 by a specific MDM2 inhibitor is
selectively toxic to tumors and leads to complete tumor growth
inhibition. Proc Natl Acad Sci USA. 105:3933–3938. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Scoumanne A and Chen X: Protein
methylation: A new mechanism of p53 tumor suppressor regulation.
Histol Histopathol. 23:1143–1149. 2008.PubMed/NCBI
|
|
18
|
Lu C and El-Deiry WS: Targeting p53 for
enhanced radio- and chemo-sensitivity. Apoptosis. 14:597–606. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fernandez-Garcia B, Eiró N, Marín L,
González-Reyes S, González LO, Lamelas ML and Vizoso FJ: Expression
and prognostic significance of fibronectin and matrix
metalloproteases in breast cancer metastasis. Histopathology.
64:512–522. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Parsons SL, Watson SA, Collins HM, Griffin
NR, Clarke PA and Steele RJ: Gelatinase (MMP-2 and −9) expression
in gastrointestinal malignancy. Br J Cancer. 78:1495–1502. 1998.
View Article : Google Scholar : PubMed/NCBI
|