Introduction
We previously reported that the immunoreactivity of
voltage-dependent anion channel 1 (VDAC1) is significantly
increased in uterine cervical cancer tissues compared with that in
normal tissues (1). Cell viability
decreased following VDAC1 gene silencing in SiHa and CaSki cervical
cancer cells. In addition, cancer tissues with increased VDAC1
immunoreactivity exhibited deep stromal invasion and an increase in
the tumor size. The N-terminal region of VDAC1 is the target of an
anti-apoptotic member of the B-cell lymphoma-2 (Bcl-2) family
(2,3). Bcl-2 may protect against the apoptosis
of cervical cancer cells via its interaction with the N-terminal
part of VDAC1 (4). Therefore, it is
hypothesized that Bcl-2 inhibition may reduce the progression of
cervical cancer.
The Bcl-2 family consists of the pro-apoptotic
members, Bcl-2-associated X protein (Bax) and Bcl-2 homology domain
3 (BH3)-only proteins, in addition to the anti-apoptotic members,
including Bcl-2, Bcl-xL and Bcl-w (5,6). ABT-737
is a well-characterized BH3 mimetic that activates the
pro-apoptotic Bcl-2 like protein (7). As an inhibitor of Bcl-2, ABT-737
predominately interacts with Bcl-2 and further enhances the effect
of apoptosis by activating pro-apoptotic proteins, including
Bcl-2-associated death (Bad) promoter, and therefore induces cell
death mostly through the intrinsic pathway of apoptosis (7–9).
A previous study showed the efficacy of ABT-737
administration in vivo by itself (10); however, a number of preclinical
investigations demonstrated the effectiveness of ABT-737 in
conjunction with chemotherapy and radiotherapy (11–13).
ABT-737 was an effective adjuvant to radiotherapy in head and neck
squamous cell carcinoma (14).
Uterine cervical cancer is the second most common
type of gynecological cancer in Taiwan, based on the 2013 annual
cancer registry report. In Taiwanese women in 2013, cervical cancer
was the seventh most common cancer, with 1,579 cases, and was also
ranked seventh with regard to the number of cancer-associated
mortalities (15). Radiotherapy is a
cornerstone of treatment of cervical cancer, especially for the
locally advanced stages (16). To
the best of our knowledge, there is only one study that has
reported the effect of combining ABT-737 and irradiation on
cervical cancers (17). ABT-737 may
improve the radiation sensitivity of cervical cancer HeLa cells and
thereby promote apoptosis (17).
Histologically, HeLa cells are of adenocarcinoma cell histology.
However, the majority of cervical cancer types present with a
squamous cell carcinoma (SCC) histology. Therefore, the present
study was conducted to elucidate the combined effect of ABT-737 and
irradiation on SCC uterine cervix cancer cells using the SiHa and
CaSki cell lines, and to evaluate whether ABT-737 could strengthen
the effect of irradiation on cervical cancer cells.
Materials and methods
The cancer genome atlas (TCGA)
Based on the cervical cancer data from The Cancer
Genome Atlas (18) (https://tcga-data.nci.nih.gov/tcga/), which
corresponds to the cervical squamous cell carcinoma and
endocervical adenocarcinoma (CESC) dataset (n=286) from the Broad
GDAC Firehose (http://gdac.broadinstitute.org/). Scatter plots of the
expression values were generated with respect to the pathological
tumor stage for Bcl-2 using Prism software (GraphPad Prism, version
6.0, GraphPad Software). The Bcl-2 expression of patients with
advanced stage was compared with that of patients with stage I
cancer. TCGA was used to determine whether an association existed
between uterine cervical cancer Tumor-Node-Metastasis stage
(19) and Bcl-2 expression. The
present study was approved by The Institutional Review Board of
Chung Shan Medical University Hospital (Taichung, Taiwan).
Cell culture
Human uterine cervical cancer CaSki and SiHa cell
lines were purchased from The American Type Culture Collection.
SiHa cells were cultured in Dulbecco's modified Eagle's medium
(Gibco; Thermo Fisher Scientific, Inc.), and CaSki cells were
cultured in RPMI-1640 medium (Gibco; Thermo Fisher Scientific,
Inc.). All media were supplemented with 2 mM glutamine, 100 µM
sodium pyruvate, 100 µM non-essential amino acids, 1%
penicillin-streptomycin and 10% fetal bovine serum (Gibco; Thermo
Fisher Scientific, Inc.). Cells were grown in a humidified
atmosphere with 5% CO2 at 37°C.
Cell viability assay
Cell viability was examined by an MTT assay. In
total ~5×103 of CaSki or SiHa cells were seeded per well
in a 96-well plate and cultured for 4 days. MTT was added into each
well to a final concentration of 0.5 mg/ml. The insoluble formazan
was collected and dissolved in dimethylsulfoxide, and the optical
density value was measured with a scanning spectrophotometer at a
wavelength of 570 nm.
Mitochondrial membrane potential (MMP)
assay
In total, ~5×105 CaSki or SiHa cells were
seeded in 6-cm dishes and treated with ABT-737 (2.5 or 5.0 µM)
(Cayman Chemical Company) combined with irradiation (10 or 20 Gy)
for 48 h. Untreated control was defined as ABT-737 0 µM and
irradiation 0 Gy. At 30 min prior to harvesting, the cells were
stained at 37°C with a 2.5-µM final concentration of
5,5,6,6′-tetrachloro-1,1,3,3′-tetraethylbenzimi-dazolylcarbocyanine
iodide (JC-1) dye (Invitrogen; Thermo Fisher Scientific, Inc.) to
detect the MMP by fluorescence microscopy and flow cytometry using
CellQuest 5.1 software (BD Biosciences). Membrane-permeant JC-1 dye
is widely used in apoptosis study to monitor MMP and can be used as
an indicator of MMP in various cell types (20,21).
Changes in MMP were assessed by the intensity of red and green
fluorescence signals detected by flow cytometry. Red fluorescence
[light-emitting material (lem), 590 nm] indicated JC-1 aggregation
in the mitochondria with an increased MMP in healthy cells, whereas
green fluorescence (lem, 527 nm) indicated JC-1 monomers in the
cytoplasm with a decreased MMP in apoptotic cells (22).
Irradiation treatment
CaSki or SiHa cells were plated at a density of
5×105 cells per dish in 6-cm dishes. After 16 h of
incubation at 37°C to allow sufficient time for the cells to
completely adhere to the surface, the cells were treated with
radiation at doses of 10 or 20 Gy by Elekta Axesse™ instrument.
Western blotting
Cells were lysed in a buffer containing 50 mM Tris
(pH 7.4), 150 mM NaCl, 2 mM EDTA, 1mM Na3VO4,
10 mM NaF, 10 mg/ml aprotinin, 10 mg/ml leupeptin, 1 mM
phenylmethylsulfonyl fluoride and 1% Triton-100. The protein
concentrations were determined using Bio-Rad Protein assay (Bio-Rad
Laboratories, Inc.). Equal amounts of protein (20 µg) were
subjected to gel electrophoresis on a 10% gel and then transferred
to polyvinylidene difluoride membranes. Blots were incubated in a
Tris-buffered saline solution at pH 7.6 containing 5% skimmed dry
milk and 0.1% (v/v) Tween-20 for 1 h at 25°C. The membranes were
incubated overnight at 4°C with primary antibodies, washed with
PBS-Tween-20, and incubated with horseradish peroxidase-conjugated
anti-rabbit secondary antibody (cat. no. 7074; dilution 1:5,000;
Cell Signaling Technology, Inc.) for 1 h at room temperature. An
enhanced chemiluminescence kit (PerkinElmer, Inc.) was used to
detect the target proteins. Primary antibodies against β-actin
(cat. no. 3700; dilution 1:5,000), Poly ADP ribose polymerase
(PARP; cat. no. 9542; dilution 1:1,000), cleaved caspase-7 (cat.
no. 9491; dilution 1:500), retinoblastoma (RB; cat. no. 9309;
dilution 1:1,000) and phosphorylated RB (pRB; cat. no. 9308;
dilution 1:1,000) were purchased from Cell Signaling Technology,
Inc. Thymidylate synthase (TS; cat. no. 113289; dilution 1:1,000)
and cyclin-dependent kinase 6 (CDK6; cat. no. 103992; dilution
1:1,000) were purchased from GeneTex, Inc.
Reactive oxygen species (ROS)
detection
Cell-permeant 2′,7′-dichlorodihydrofluorescein
diacetate (H2DCFDA; Invitrogen Thermo Fisher Scientific,
Inc.) is a widely used ROS indicator. The reduced non-fluorescent
fluorescein H2DCFDA can be oxidized and converted into
fluorescent 2′,7′-dichlorofluorescein (DCF) by intracellular ROS.
H2DCFDA (10 µM) was used to label intracellular ROS at
4°C for 30 min and the DCF intensity was detected by flow
cytometry.
Analysis of cell cycle and
apoptosis
Cells were treated with or without ABT-737/radiation
for 48 h, and the cell cycle distribution was analyzed using flow
cytometry. A total of 5×105 cells were trypsinized,
washed with PBS and fixed in 80% ethanol at 4°C for 30 min, then
washed with PBS and incubated with 100 µg/ml RNase A
(Sigma-Aldrich; Merck KGaA) at 37°C for 30 min, prior to being
stained with propidium iodide (PI; 50 µg/ml; Sigma-Aldrich; Merck
KGaA) at 37°C for 10 min, and analyzed using flow cytometry on a BD
FACSCalibur flow cytometer (BD Biosciences). The percentage of
cells in different phases of the cell cycle was analyzed using
Cell-FIT software version 2.0 (Becton Dickinson Instruments).
Apoptosis in CaSki and SiHA cells was confirmed by BD FACSCalibur
flow cytometry to detect phosphatidylserine expression on cell
surfaces by using fluorescein isothiocyanate (FITC)-labeled Annexin
V and PI (FITC-Annexin V Apoptosis Detection kit I; BD Pharmingen™;
BD Biosciences).
Statistical analysis
Data were analyzed by an analysis of variance
followed by Dunnett's post hoc test using Predictive Analytics
software (version 18; IBM Corp.) to evaluate the significance of
differences between the untreated control group and the other
groups. Data are presented as the mean ± standard deviation based
on three independent repeats. P<0.05 was considered to indicate
a statistically significant difference.
Results
Comparison of Bcl-2 expression among
cancer tissues of different stages
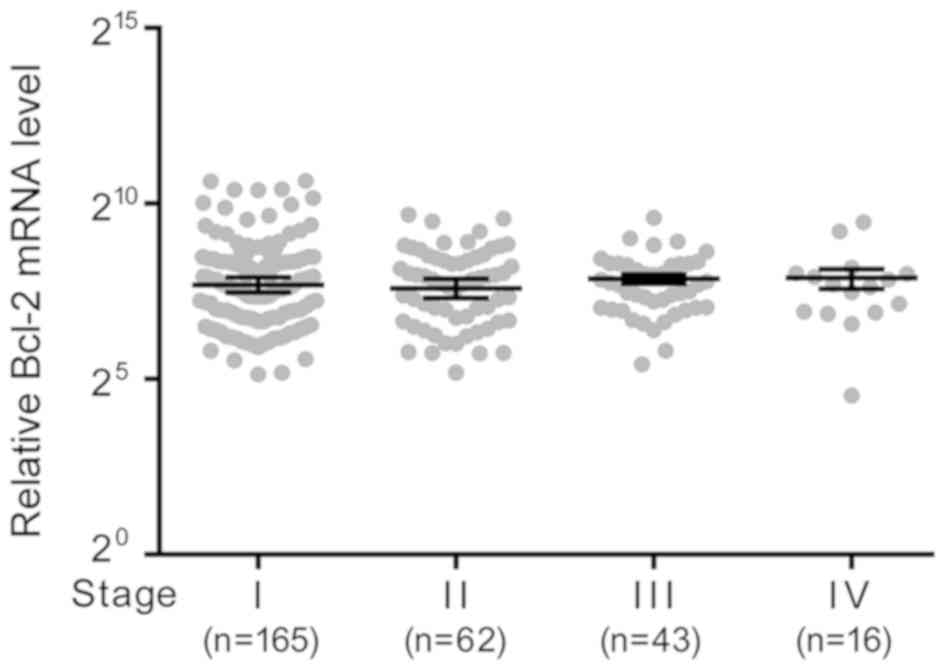
Based on TCGA data, the mRNA expression levels of
Bcl-2 among cancer tissues of different stages was compared. There
was no significant difference in expression levels between the
stages (Fig. 1).
Effect of ABT-737 and irradiation on
cell viability in CaSki and SiHa uterine cervical cells
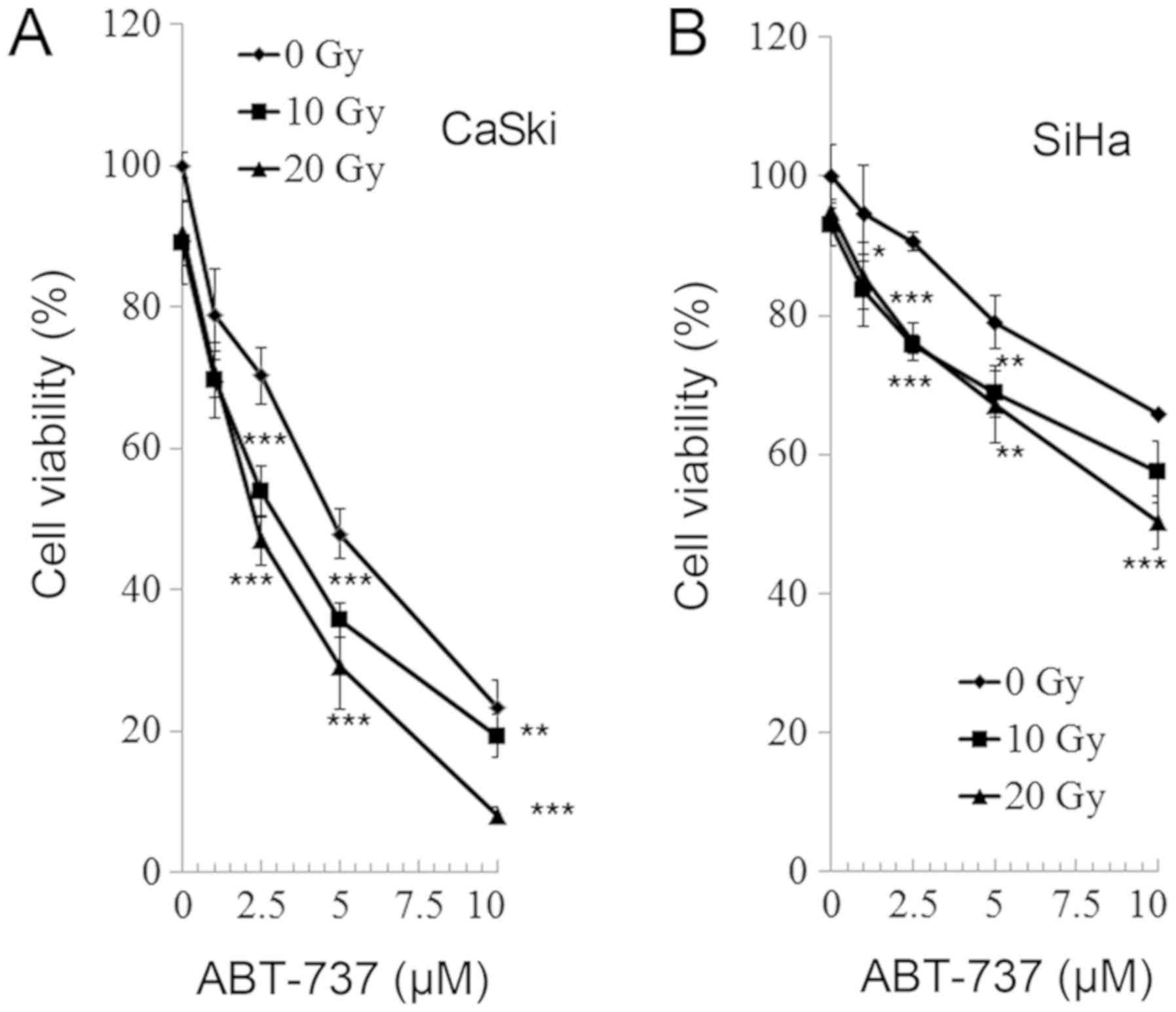
CaSki and SiHa cells were treated with different
concentrations of Bcl-2 inhibitor ABT-737 (0, 1, 2.5, 5, and 10 µM)
and different doses of irradiation (0, 10, and 20 Gy) for 48 h.
Cell viability was analyzed by an MTT assay. As presented in
Fig. 2, the cell viability of the
CaSki and SiHa cells was significantly reduced following treatment
with all doses of ABT-737 and at all levels of irradiation compared
with the untreated control (ABT-737 0 µM and irradiation 0 Gy). The
effect of treatment and irradiation on cell viability was
dose-dependent and there was an enhanced effect on cell viability
when cells were treated with ABT-737 and irradiation
synergistically.
Effect of ABT-737 combined with
irradiation on MMP in CaSki and SiHa cells
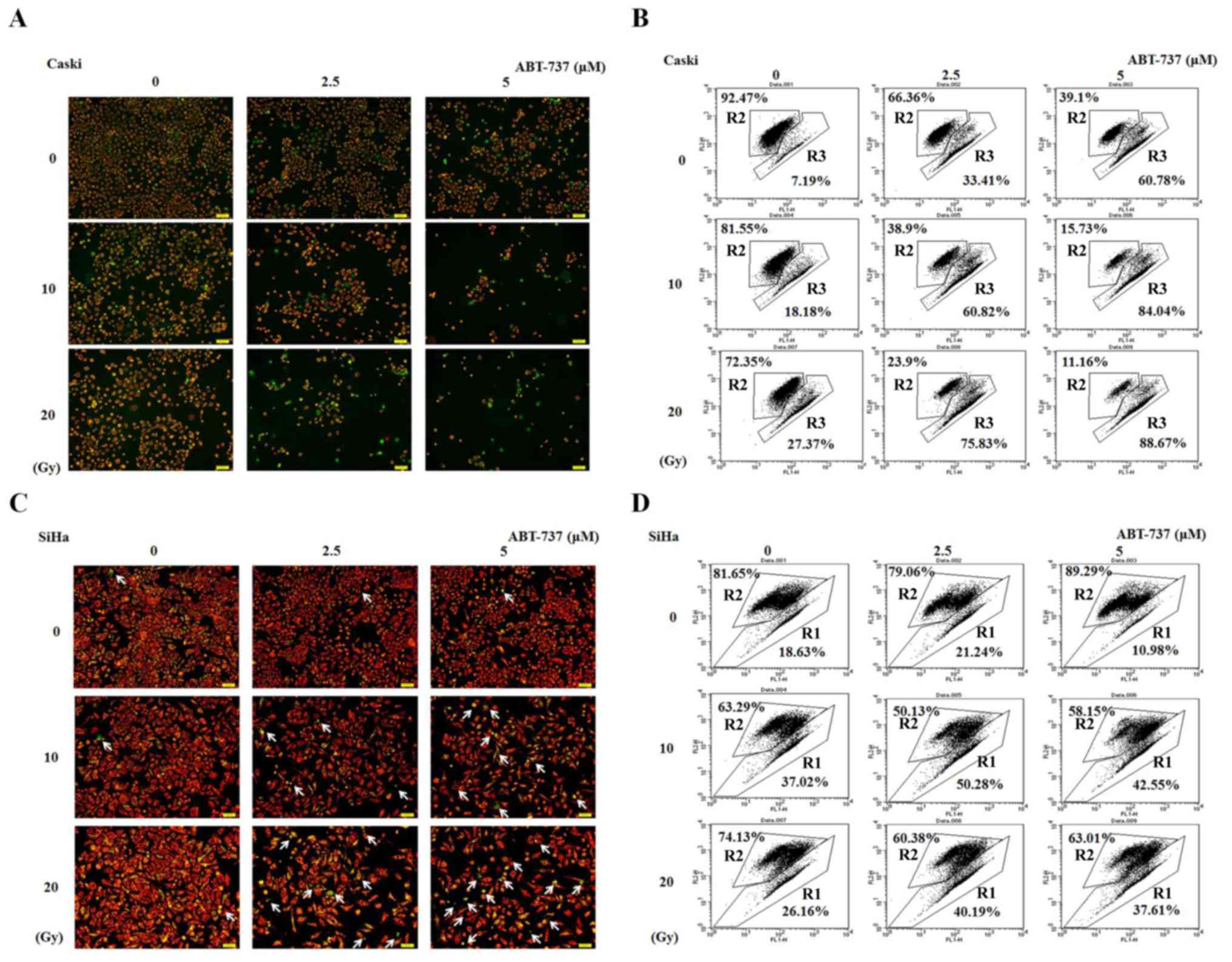
CaSki and SiHa cells were treated with different
concentrations of ABT-737 and different doses of irradiation for 48
h. The cells were stained with JC-1 dye to detect the presence of
MMP by both fluorescent microscopy and flow cytometry. The live
cells stained with JC-1 fluoresced red, whereas the apoptotic cells
fluoresced green. The number of cells showing green fluorescence as
detected by fluorescent microscope increased in CaSki and SiHa
cells treated with ABT-737 and irradiation compared with untreated
control (ABT-737 0 µM and irradiation 0 Gy) (Fig. 3A and C). The effect was more evident
in CaSki cells than in SiHa cells. Similar results were shown in
Fig. 3B detected by flow cytometry.
The results found at 20 Gy and 5 µM do not appear to follow the
same pattern of increase in the SiHa cell line (Fig. 3D). Potential reasons for this could
be due to the treated SiHa cells being dead for too long a time and
breaking up into cell debris or they were not lysed adequately
enough to be detected by flow cytometry. So the increased pattern
was not observed. R2 represents live cells, whereas R1 or R3
represent apoptotic cells. These results indicate that the MMP was
decreased in CaSki and SiHa cells treated with ABT-737 and
irradiation.
Effect of ABT-737 and irradiation on
cell apoptosis in CaSki and SiHa cells
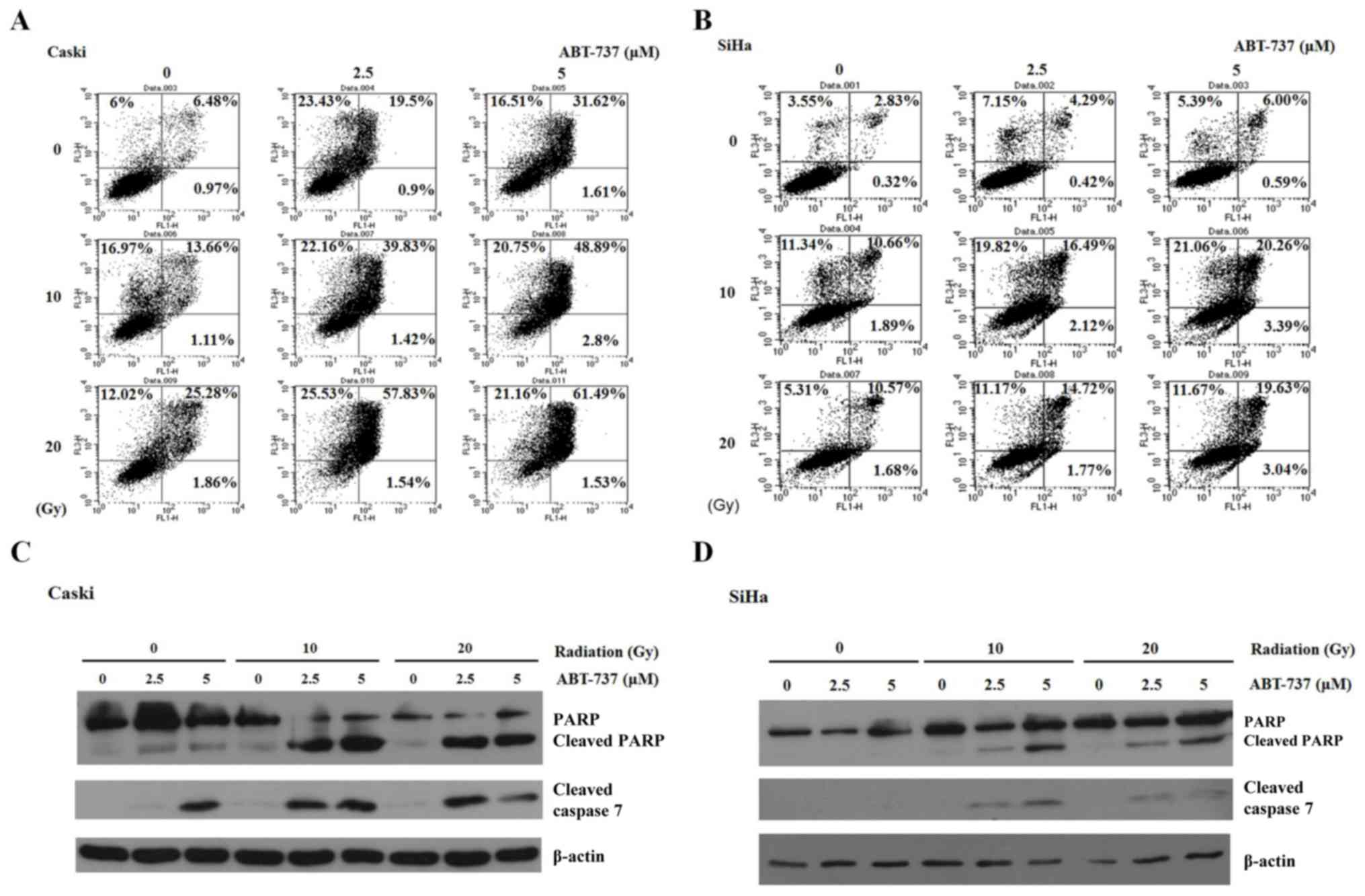
CaSki and SiHa cells were treated with different
concentrations of Bcl-2 inhibitor ABT-737 (2.5 and 5 µM) and
different doses of irradiation (10 and 20 Gy) for 48 h. The cells
were stained with Annexin V-FITC and PI to detect cell apoptosis by
flow cytometry. Annexin V-FITC-positive (early apoptosis, right
lower panel) and Annexin V-FITC/PI-positive (late apoptosis, right
upper panel) were quantified as apoptotic cells. The percentage of
apoptotic cells increased significantly in CaSki and SiHa cells
treated with ABT-737 and irradiation (Fig. 4A and B). However, the apoptotic
percentage in SiHa cells treated with ABT-737 5 µM and irradiation
20 Gy was less compared with the cells treated with 5 µM ABT-737
and 10 Gy irradiation. Potential reasons for this are discussed
above, including prolonged cell death or improper lysing.
Additionally, 10 Gy irradiation may be a toxicity threshold of SiHa
cells. As such, the increased apoptotic pattern was not observed.
The increase in apoptosis was evident in cells treated with ABT-737
and irradiation. Apoptosis was also determined by the presence of
cleaved caspase-7 by western blotting. When ABT-737 concentration
and irradiation dose were increased, the cleaved bands of PARP and
caspase-7 became visible (Fig. 4C and
D). These results demonstrate that ABT-737 and irradiation
could enhance cell apoptosis in CaSki and SiHa cells.
Effect of ABT-737 combined with
irradiation on ROS production in CaSki and SiHa cells
Following treatment of cells with ABT-737 and
irradiation, the cells were stained with H2DCFDA dye to
detect ROS by flow cytometry. As shown in Fig. 5, the augmented ROS production caused
by ABT-737 and irradiation was more notable in CaSki cells than in
SiHa cells.
Effect of ABT-737 and irradiation on
the cell cycle and phosphorylation of RB in CaSki and SiHa
cells
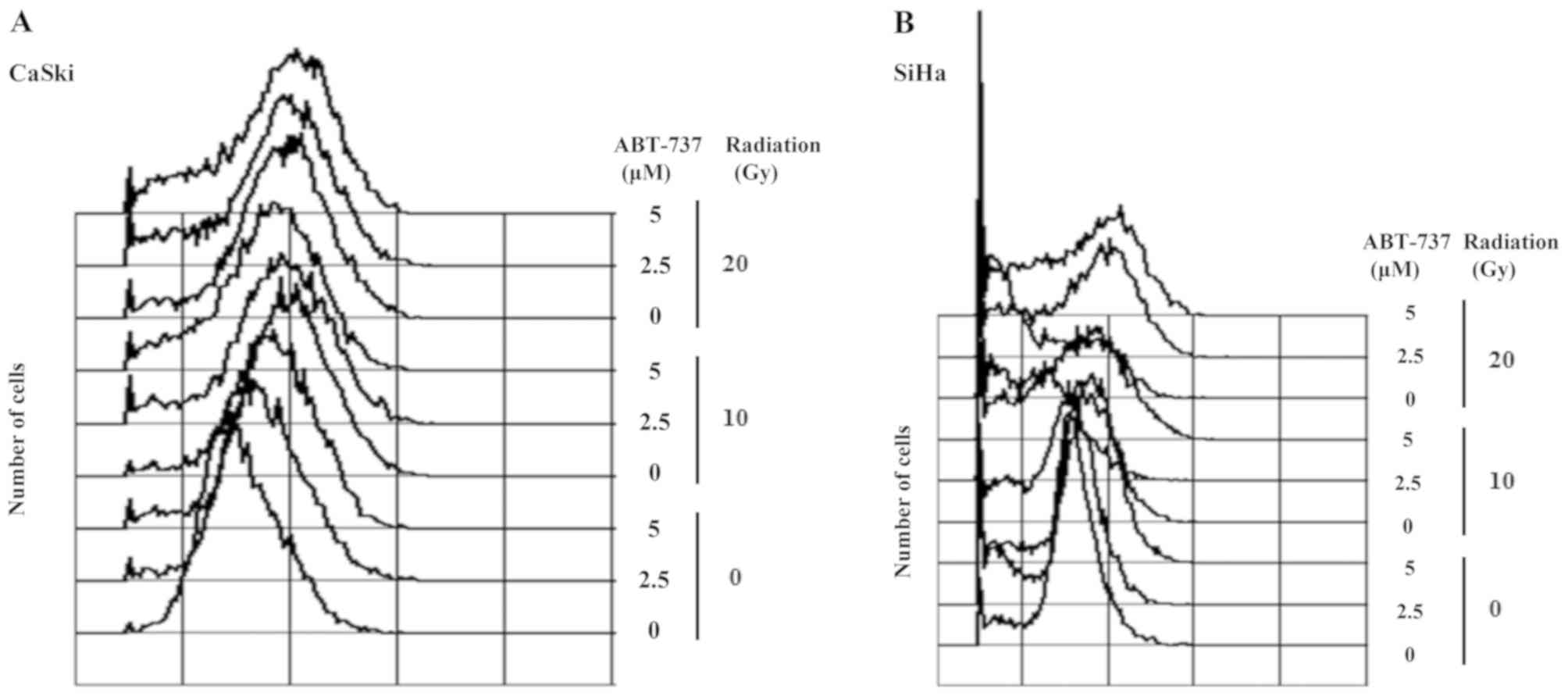
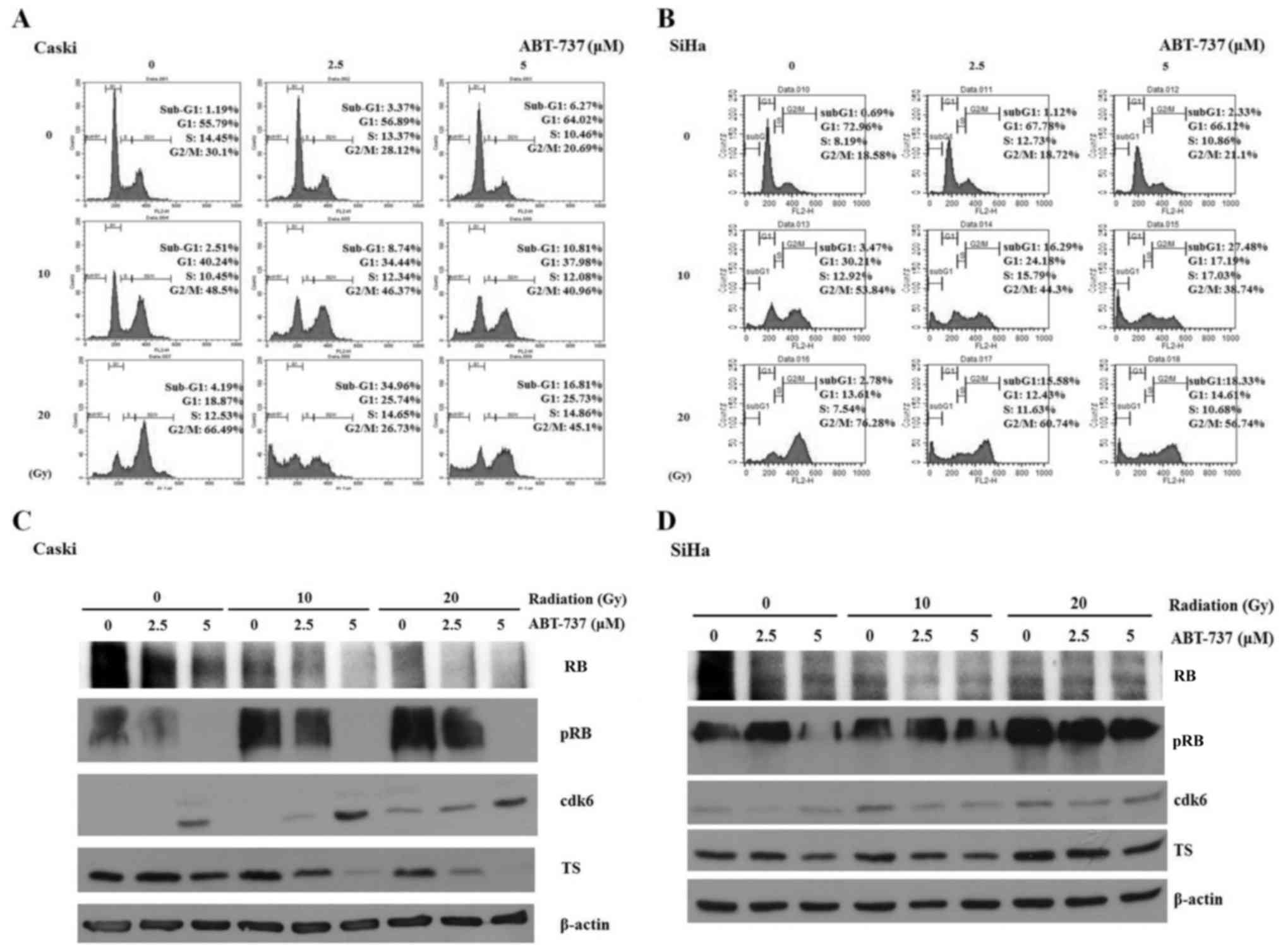
The cell cycle was arrested in the G2/M
phase when CaSki and SiHa cells were treated with irradiation only;
however, the percentage of cells in the sub-G1 phase was
amplified in CaSki and SiHa cells co-treated with ABT-737 and
irradiation (Fig. 6A and B). Western
blotting results demonstrated that pRB protein expression was
upregulated in CaSki cells treated with irradiation alone but was
downregulated in the cells treated with ABT-737. The protein
expression levels of CDK6 were increased, but those of TS were
decreased in CaSki cells co-treated with ABT-737 and irradiation
(Fig. 6C). The protein expression
levels of pRB, CDK6 and TS were not affected in SiHa cells treated
with ABT-737 and irradiation (Fig.
6D).
Discussion
Although there was no significant association
between Bcl-2 mRNA level and the stage of cervical cancer, the
Bcl-2 protein expression level in cervical cancer tissues was
significantly higher compared with in the normal cervix, chronic
cervicitis or cervical intraepithelial neoplasia (23). Bcl-2 level was positively correlated
with clinical stage (23). In
addition, the number of malignant lesions expressing Bcl-2 protein
was higher compared with premalignant lesions (24). In cervical intraepithelial neoplasia
lesions, Bcl-2 expression increased as the severity of the cancer,
measured by the grade, increased (24). Based on these studies, the
mechanistic effects of inhibiting Bcl-2 and irradiating cervical
cancer cells were investigated. To the best of our knowledge, there
are no reports demonstrating the effects of combined Bcl-2
inhibition and irradiation on cervical cancer. In the present
study, a target strategy was proposed to improve patient prognosis
via Bcl-2 inhibition, and the effect of combined Bcl-2 inhibition
and irradiation on cervical cancer cells was evaluated. As an
inhibitor of Bcl-2, ABT-737 is one of the best-characterized BH3
mimetics that activates pro-apoptotic Bcl-2-like protein 11 (BIM)
(8,25). Activation of BIM is associated with
markers associated with a favorable overall survival prognosis for
patients with cervical cancer (26).
Therefore, the possible mechanisms of Bcl-2 inhibition and the
enhanced effect of combined Bcl-2 inhibitor and irradiation
treatment in reducing cancer cell progression were
investigated.
The activity of the Bcl-2 family members is mediated
through its interaction with the mitochondria, especially with
VDAC1, the outer mitochondrial membrane (OMM) transporter, to
regulate mitochondria-mediated apoptosis, frequently referred to as
the intrinsic pathway, by regulating OMM permeability (6,27).
ABT-737 enhances the effect of apoptosis by activating
pro-apoptotic proteins such as BIM, and therefore induces cell
death primarily through mitochondria-mediated apoptosis (4,7–9). Sugiyama et al (28) demonstrated that BIM interacts with
VDAC, and this interaction was enhanced during apoptosis. The
present study demonstrated that ABT-737 and irradiation induced
apoptosis in cervical cancer CaSki and SiHa cells. The MMP
decreased following treatment with increased doses of irradiation
and ABT-737, particularly in the CaSki cells. Upon stimulation of
apoptosis, mitochondrial permeability transition occurred via the
mitochondrial permeability transition pore, which is composed of
VDAC at the OMM, adenine nucleotide at the inner mitochondrial
membrane and cyclophilin D in the mitochondrial matrix, after
which, the MMP collapsed (29,30). The
proteins in the mitochondrial intermembrane space, such as
cytochrome c, are released when a cell undergoes apoptosis.
The data presented in the present study suggest that ABT-737 and
radiation may exert a synergistic apoptotic effect on cervical
cancer cell lines. Bcl-2 protects against the apoptosis of cervical
cancer cells via its interaction with the N-terminal part of VDAC1
(4). There is potential for ABT-737
to bind to Bcl-2 with a high affinity and disrupt its interaction
with VDAC1, resulting in loss of the MMP and enhanced apoptosis.
Therefore, ABT-737 and irradiation may induce apoptosis via the
loss of MMP caused by the interaction of VDAC1 with pro-apoptotic
proteins mediated by a ROS-dependent intrinsic pathway in CaSki and
SiHa cells. In previous studies, ABT-737 also improved the
efficiency of radiotherapy and chemotherapy in breast cancer
(31) and non-small cell lung cancer
cell lines (32), and it may induce
the autophagy of prostate cancer via releasing Beclin-1 (33).
In the present study, ABT-737 increased ROS
production and enhanced the apoptotic effect of irradiation in
CaSki cells. Increased levels of mitochondrial ROS initiate
intrinsic apoptosis, leading to the release of mitochondrial
apoptogenic factors such as cytochrome c, an
apoptosis-inducing factor, into the cytosol (34). ABT-737 and irradiation may induce
apoptosis via the ROS-dependent mitochondria-mediated apoptosis
pathway. A limitation of the present study was that Bcl-2 and
cytochrome c expression were not measured. ABT-737 has been
demonstrated to bind to Bcl-2 with a high affinity and disrupts the
interaction with the pro-apoptotic proteins, Bax/Bak, thus
enhancing apoptosis. Bax and Bak homodimers promote apoptosis via
pore formation within the mitochondria, leading to permeabilization
of the mitochondrial outer membrane, release of cytochrome c
and activation of the caspase cascade (35). Hepatocellular carcinoma cells with
increased levels of Bcl-2 may resist the effect of ABT-737 by
activation of the ROS-Janus kinase-autophagy pathway (36). In the present study, ABT-737 enhanced
the apoptotic effect of irradiation on the loss of the MMP in
cervical cancer CaSki and SiHa cells. The cancer cells may protect
themselves from cell apoptosis via autophagy to remove the damaged
mitochondria (37,38). However, experiments to determine
whether autophagy was induced when cells were treated with ABT-737
and irradiation were inconclusive (data not shown).
RB functions to prevent uncontrolled cell growth by
arresting cell cycle progression until a cell is ready to divide.
At this point, RB is phosphorylated and becomes inactive, thus
allowing the cell cycle to progress. Based on the results of the
present study, ABT-737 may potentiate the irradiation effect to
decrease cell viability by promoting or inducing apoptosis. ABT-737
and radiation treatment decreased pRB expression in the CaSki
cells. TS serves a vital role in early DNA biosynthesis (39). Healthy DNA synthesis and insertion of
required for the normal functions of the body and in order to avoid
cancerous activity. Furthermore, the synthesis of important
nucleotides must be inhibited for cell growth. Therefore, TS has
become an important target for cancer treatment through
chemotherapy or radiotherapy. In the present study, the protein
expression levels of TS were decreased in CaSki cells co-treated
with ABT-737 and radiation. The sensitivity of TS to ABT-737 and
irradiation may provide another application for treating patients
with cervical cancer. CDK6 in conjunction with CDK4 acts as a
switch that initially appears in G1 and subsequently
directs the cell toward the S phase of the cell cycle (40). By studying a DNA damage-induced
senescence model of human fibroblasts, Brookes et al
(41) demonstrated an unexpected
role for CDK4 in contributing to a G2/M cell cycle
arrest and the senescent phenotype. The findings of the present
study may explain that ABT-737, especially when combined with
irradiation, increases the expression of CDK6 and consequently
induces a G2/M cell cycle arrest.
The apoptotic effects were conflicting when CaSki
and SiHa cells were treated with ABT-737 and irradiation. It is
possible that different methods of detecting apoptosis may result
in different sensitivities. A cell cycle study may not be
sufficient to demonstrate apoptosis. Cell-cycle analysis would
reveal the fragmentation of DNA (sub-G1) that occurs in
the late stage of apoptosis, and which may occur during necrosis as
well. Therefore, the number of cells in the sub-G1 phase
is usually lower than that reported by other assays that detect
both early and late stages of apoptosis.
Radiotherapy is the cornerstone of cervical cancer
treatment, especially for the locally advanced stages (16). ABT-737 can improve the sensitivity of
HeLa cervical cancer cells to irradiation and thus, induce cell
apoptosis (17). However, the HeLa
cancer cells are of an adenocarcinoma histological type. CaSki and
SiHa cells are derived from SCC of the uterine cervix. When CaSki
and SiHa cells were subjected to ABT-737 and irradiation treatment,
the CaSki cells became more sensitive than SiHa cells based on the
results of the in vitro assays, which may have been due to
the levels of antioxidant enzymes or HPV 16 copy number in each
cell type. Filippova et al (42) reported that SiHa cells are more
resistant to doxorubicin and cisplatin treatment, and express
higher levels of antioxidant enzymes than CaSki cells. These
different profiles may have contributed to their different
responses to treatments with chemotherapy. SiHa cells contain 1 to
2 copies of the HPV 16 genome per cell, whereas CaSki cells contain
~600 copies per cell (43). Upon
treatment of these two cell lines with TNF at varying
concentrations, a viability analysis revealed that the CaSki cells
with a higher HPV 16 level were sensitive to the cytokine, whereas
SiHa cells with only 1 to 2 copies of the genome were relatively
resistant (44). This indicated that
certain aspects of the high number of HPV 16 copies in the CaSki
genome, such as the increased amount of one or more viral proteins,
may contribute to its TNF sensitivity (44).
In conclusion, the present study highlighted the
potential implication of the combinatorial approach for treating
cervical cancer cells with both a Bcl-2 inhibitor and radiation
therapy. The percentages of cells in the sub-G1 phase
were amplified in CaSki and SiHa cells co-treated with ABT-737 and
irradiation. ABT-737 may enhance the induction of apoptosis in
human cervical SCC via loss of the MMP and an ROS-dependent
intrinsic apoptosis pathway in CaSki and SiHa cells. Although the
CDK6 expression was increased, the reduced expression levels of pRB
and TS indicate the subsequent G1/S checkpoint
regulation of cell progression in CaSki cells was abrogated. Based
on these findings, therapeutic strategies should use ABT-737 as a
target to induce cell apoptosis, reduce cell growth and
progression, and enhance the therapeutic efficacy of radiation
therapy. Therefore, ABT-737 may be a potential irradiation adjuvant
for treating patients with cervical cancer.
Acknowledgements
Not applicable.
Funding
The present study was funded by The Taiwan Ministry
of Science and Technology (grant no. MOST 105-2314-B-040-016-MY2)
and Chung Shan Medical University Hospital (grant no.
CSH-2017-D-002).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the author on reasonable request.
Authors' contributions
PW and HS conceived and designed the study. WW and
JK performed experiments and analyzed the data. TW, SY, CW and CY
conducted data analysis and interpretation. PW wrote the
manuscript. WW edited and revised the manuscript. All authors
discussed results/discussion and collaborated in drafting the
manuscript.
Ethics approval and consent to
participate
The present study was approved by The Institutional
Review Board of Chung Shan Medical University Hospital (Taichung,
Taiwan).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ABT-737
|
4-[4-[(4′-chloro[1,1′-biphenyl]-2-yl)
methyl]-1-piperazinyl]-N-[[4-[[(1R)-3-(dimethylamino)-1-[(phenylthio)methyl]propyl]amino]-3-nitrophenyl]
sulfonyl]-benzamide
|
|
Bcl-2
|
B-cell lymphoma 2
|
|
BH3
|
Bcl-2 homology domain 3
|
|
MMP
|
mitochondrial membrane potential
|
|
JC-1
|
5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimi-dazolylcarbocyanine
iodide
|
|
PARP
|
poly ADP ribose polymerase
|
|
ROS
|
reactive oxygen species
|
|
TS
|
thymidylate synthase
|
|
VDAC1
|
voltage-dependent anion channel 1
|
References
|
1
|
Wu CH, Lin YW, Wu TF, Ko JL and Wang PH:
Clinical implication of voltage-dependent anion channel 1 in
uterine cervical cancer and its action on cervical cancer cells.
Oncotarget. 7:4210–4225. 2016.PubMed/NCBI
|
|
2
|
Abu-Hamad S, Arbel N, Calo D, Arzoine L,
Israelson A, Keinan N, Ben-Romano R, Friedman O and Shoshan-Barmatz
V: The VDAC1 N-terminus is essential both for apoptosis and the
protective effect of anti-apoptotic proteins. J Cell Sci.
122:1906–1916. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Arbel N and Shoshan-Barmatz V:
Voltage-dependent anion channel 1-based peptides interact with
Bcl-2 to prevent antiapoptotic activity. J Biol Chem.
285:6053–6062. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shoshan-Barmatz V, De Pinto V,
Zweckstetter M, Raviv Z, Keinan N and Arbel N: VDAC, a
multi-functional mitochondrial protein regulating cell life and
death. Mol Aspects Med. 31:227–285. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Adams JM and Cory S: The Bcl-2 apoptotic
switch in cancer development and therapy. Oncogene. 26:1324–1337.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Youle RJ and Strasser A: The BCL-2 protein
family: Opposing activities that mediate cell death. Nat Rev Mol
Cell Biol. 9:47–59. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Del Gaizo Moore V, Brown JR, Certo M, Love
TM, Novina CD and Letai A: Chronic lymphocytic leukemia requires
BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2
antagonist ABT-737. J Clin Invest. 117:112–121. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Oltersdorf T, Elmore SW, Shoemaker AR,
Armstrong RC, Augeri DJ, Belli BA, Bruncko M, Deckwerth TL, Dinges
J, Hajduk PJ, et al: An inhibitor of Bcl-2 family proteins induces
regression of solid tumours. Nature. 435:677–681. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen S, Dai Y, Harada H, Dent P and Grant
S: Mcl-1 down-regulation potentiates ABT-737 lethality by
cooperatively inducing Bak activation and Bax translocation. Cancer
Res. 67:782–791. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mason KD, Vandenberg CJ, Scott CL, Wei AH,
Cory S, Huang DC and Roberts AW: In vivo efficacy of the Bcl-2
antagonist ABT-737 against aggressive Myc-driven lymphomas. Proc
Natl Acad Sci USA. 105:17961–17966. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hann CL, Daniel VC, Sugar EA,
Dobromilskaya I, Murphy SC, Cope L, Lin X, Hierman JS, Wilburn DL,
Watkins DN and Rudin CM: Therapeutic efficacy of ABT-737, a
selective inhibitor of BCL-2, in small cell lung cancer. Cancer
Res. 68:2321–2328. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kutuk O and Letai A: Alteration of the
mitochondrial apoptotic pathway is key to acquired paclitaxel
resistance and can be reversed by ABT-737. Cancer Res.
68:7985–7994. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tagscherer KE, Fassl A, Campos B, Farhadi
M, Kraemer A, Böck BC, Macher-Goeppinger S, Radlwimmer B, Wiestler
OD, Herold-Mende C and Roth W: Apoptosis-based treatment of
glioblastomas with ABT-737, a novel small molecule inhibitor of
Bcl-2 family proteins. Oncogene. 27:6646–6656. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gilormini M, Malesys C, Armandy E, Manas
P, Guy JB, Magné N, Rodriguez-Lafrasse C and Ardail D: Prefer ntial
targeting of cancer stem cells in the radiosensitizing effect of
ABT-737 on HNSCC. Oncotarget. 7:16731–16744. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Taiwan Cancer Registry Annual Report, .
Taiwan: Health promotion administration, ministry of health and
welfare. https://www.hpa.gov.tw/File/Attach/5191/File_6166.pdf
|
|
16
|
Marth C, Landoni F, Mahner S, McCormack M,
Gonzalez-Martin A and Colombo N; ESMO Guidelines Committee, :
Cervical cancer: ESMO clinical practice guidelines for diagnosis,
treatment and follow-up. Ann Oncol. 28 (Suppl 4):iv72–iv83. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang H, Yang YB, Shen HM, Gu J, Li T and
Li XM: ABT-737 induces Bim expression via JNK signaling pathway and
its effect on the radiation sensitivity of HeLa cells. PLoS One.
7:e524832012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tomczak K, Czerwińska P and Wiznerowicz M:
The cancer genome atlas (TCGA): An immeasurable source of
knowledge. Contemp Oncol (Pozn). 19:A68–A77. 2015.PubMed/NCBI
|
|
19
|
Pecorelli S: Revised FIGO staging for
carcinoma of the vulva, cervix, and endometrium. Int J Gynecol
Obstet. 105:103–104. 2009. View Article : Google Scholar
|
|
20
|
Reers M, Smiley ST, Mottola-Hartshorn C,
Chen A, Lin M and Chen LB: Mitochondrial membrane potential
monitored by JC-1 dye. Methods Enzymol. 260:406–417. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Elefantova K, Lakatos B, Kubickova J,
Sulova Z and Breier A: Detection of the mitochondrial membrane
potential by the cationic dye JC-1 in L1210 cells with massive
overexpression of the plasma membrane ABCB1 drug transporter. Int J
Mol Sci. 19(pii): E19852018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shah BP, Pasquale N, De G, Tan T, Ma J and
Lee KB: Core-shell nanoparticle-based peptide therapeutics and
combined hyperthermia for enhanced cancer cell apoptosis. ACS Nano.
8:9379–9387. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou XL and Wang M: Expression levels of
survivin, Bcl-2, and KAI1 proteins in cervical cancer and their
correlation with metastasis. Genet Mol Res. 14:17059–17067. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kamaraddi S, Nayak A, Honnappa S and
Swarup A: Expression of Bcl-2 marker in premalignant lesions of
cervical cancer. Int J Reprod Contracept Obstet Gyneco. 15:965–969.
2016. View Article : Google Scholar
|
|
25
|
Fulda S, Galluzzi L and Kroemer G:
Targeting mitochondria for cancer therapy. Nat Rev Drug Discov.
9:447–464. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim BW, Cho H, Ylaya K, Kitano H, Chung
JY, Hewitt SM and Kim JH: Bcl-2-like Protein 11 (BIM) expression is
associated with favorable prognosis for patients with cervical
cancer. Anticancer Res. 37:4873–4879. 2017.PubMed/NCBI
|
|
27
|
Shoshan-Barmatz V, Zakar M, Rosenthal K
and Abu-Hamad S: Key regions of VDAC1 functioning in apoptosis
induction and regulation by hexokinase. Biochim Biophys Acta.
1787:421–430. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sugiyama T, Shimizu S, Matsuoka Y, Yoneda
Y and Tsujimoto Y: Activation of mitochondrial voltage-dependent
anion channel by apro-apoptotic BH3-only protein Bim. Oncogene.
21:4944–4956. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kroemer G, Galluzzi L and Brenner C:
Mitochondrial membrane permeabilization in cell death. Physiol Rev.
87:99–163. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Halestrap AP and Richardson AP: The
mitochondrial permeability transition: A current perspective on its
identity and role in ischaemia/reperfusion injury. J Mol Cell
Cardiol. 78:129–141. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li JY, Li YY, Jin W, Yang Q, Shao ZM and
Tian XS: ABT-737 reverses the acquired radioresistance of breast
cancer cells by targeting Bcl-2 and Bcl-xL. J Exp Clin Cancer Res.
31:1022012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kim KW, Moretti L, Mitchell LR, Jung DK
and Lu B: Combined Bcl-2/mammalian target of rapamycin inhibition
leads to enhanced radiosensitization via induction of apoptosis and
autophagy in non-small cell lung tumor xenograft model. Clin Cancer
Res. 15:6096–6105. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lian J, Wu X, He F, Karnak D, Tang W, Meng
Y, Xiang D, Ji M, Lawrence TS and Xu L: A natural BH3 mimetic
induces autophagy in apoptosis-resistant prostate cancer via
modulating Bcl-2-Beclin1 interaction at endoplasmic reticulum. Cell
Death Differ. 18:60–71. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Redza-Dutordoir M and Averill-Bate DA:
Activation of apoptosis signalling pathways by reactive oxygen
species. Biochim Biophys Acta. 1863:2977–2992. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chipuk JE, Moldoveanu T, Lambi F, Parsons
MJ and Green DR: The BCL-2 family reunion. Mol Cell. 37:299–310.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ni Z, Wang B, Dai X, Ding W, Yang T, Li X,
Lewin S, Xu L, Lian J and He F: HCC cells with high levels of Bcl-2
are resistant to ABT-737 via activation of the ROS-JNK-autophagy
pathway. Free Radic Biol Med. 70:194–203. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Das DN, Naik PP, Mukhopadhyay S, Panda PK,
Sinha N, Meher BR and Bhutia SK: Elimination of dysfunctional
mitochondria through mitophagy suppresses benzo[a]pyrene-induced
apoptosis. Free Radic Biol Med. 112:452–463. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Swiader A, Nahapetyan H, Faccini J,
D'Angelo R, Mucher E, Elbaz M, Boya P and Vindis C: Mitophagy acts
as a safeguard mechanism against human vascular smooth muscle cell
apoptosis induced by atherogenic lipids. Oncotarget. 7:28821–28835.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Peters GJ, Backus HH, Freemantle S, van
Triest B, Codacci-Pisanelli G, van der Wilt CL, Smid K, Lunec J,
Calvert AH, Marsh S, et al: Induction of thymidylate synthase as a
5-fluorouracil resistance mechanism. Biochim Biophys Acta.
1587:194–205. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Bertoli C, Skotheim JM and de Bruin RA:
Control of cell cycle transcription during G1 and S phases. Nat Rev
Mol Cell Biol. 14:518–528. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Brookes S, Gagrica S, Sanij E, Rowe J,
Gregory FJ, Hara E and Peters G: Evidence for a CDK4-dependent
checkpoint in a conditional model of cellular senescence. Cell
Cycle. 14:1164–1173. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Filippova M, Filippov V, Williams VM,
Zhang K, Kokoza A, Bashkirova S and Duerksen-Hughes P: Cellular
levels of oxidative stress affect the response of cervical cancer
cells to chemotherapeutic agents. Biomed Res Int. 2014:5746592014.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
ATCC: CRL-1550, HTB-35, . 2005, http://www.atcc.org
|
|
44
|
Filippova M, Brown-Bryan TA, Casiano CA
and Duerksen-Hughes PJ: The human papillomavirus 16 E6 protein can
either protect or further sensitize cells to TNF: Effect of dose.
Cell Death Differ. 12:1622–1635. 2005. View Article : Google Scholar : PubMed/NCBI
|