Introduction
Gastric cancer (GC) is a malignant tumor originating
from the gastric mucosa. It ranks the first in tumor incidence of
Chinese population. Regional difference is observed in the
incidence of GC. The northwest and eastern coastal areas of China
are high incidence regions of GC (1). GC mainly affects people over 50 years,
with the male-female ratio of 2:1. Recently, the incidence of GC
becomes younger owing to changes in environmental and dietary
habits, overloaded working and increased infection rate of
Helicobacter pylori (2). GC
occurs in any part of the stomach, and more than half of GC cases
structurally involve the gastric antrum (3). Based on the pathological
classification, the majority of GC belongs to adenocarcinoma.
Early-stage symptoms of GC are atypical, manifesting as similar
symptoms of gastritis and gastric ulcer, such as upper abdominal
discomfort and hernia (4).
Unfortunately, detective rate of early-stage GC is low, leading to
a poor prognosis of GC patients (5).
The overall survival of GC remains at 20%, which is a global health
problem (6). Advanced GC has an
extreme poor prognosis, with <15% of 5-year survival (7). Development of effective and sensitive
hallmarks for GC contributes to improving the clinical outcome of
the affected (8).
Long non-coding RNAs (lncRNAs) are transcribed by
RNA polymerase II with >200 nucleotides in length. They are a
research focus on tumor-targeted therapy (9). lncRNAs are dysregulated in tumors, and
mediate oncogenes or tumor-suppressor genes to further influence
tumor progression (10). They exert
diverse functions in regulating cellular behavior (11). In tumor biology, lncRNAs have been
widely explored since they are capable of regulating
drug-resistance and malignant phenotypes of tumor cells (12,13).
Tumor-related lncRNAs may be promising targets applied in tumor
detection (14).
EZH2 encodes a histone lysine N-methyltransferase
that is involved in DNA methylation to inhibit transcription of
other genes. EZH2 also methylates H3K27me3 (15). The methylation activity of EZH2
promotes heterochromatinization and thus silences downstream genes
(16). Mutation or overexpression of
EZH2 is associated with multiple types of cancers (17–21).
Abnormally activated EZH2 can inhibit expression of
tumor-suppressor genes. Therefore, inhibition of EZH2 activity is
able to alleviate tumor growth (22).
This study explored the biological function of
lncRNA ST7-AS1 in the malignant progression of GC. The potential
interaction between ST7-AS1 and EZH2 was investigated, which may
provide new directions for developing therapeutic strategies for
GC.
Patients and methods
Subjects
GC tissues and matched adjacent normal tissues were
surgically harvested from GC patients in The Fourth Affiliated
Hospital of China Medical University (Shenyang, China) from April
2016 to October 2018. Resected samples were placed into liquid
nitrogen until analyses. Enrolled GC patients were pathologically
diagnosed and had no medical history of other malignancies. This
study was approved by the Ethics Committee of The Fourth Affiliated
Hospital of China Medical University and informed consent was
received from each subject.
Cell culture and transfection
Epithelial cells of gastric mucosa (GES-1) and GC
cell lines (AGS, MG803 and SGC-7901) provided by American Type
Culture Collection (ATCC) were cultured in Roswell Park Memorial
Institute-1640 (RPMI-1640) containing 10% fetal bovine serum (FBS)
(both from HyClone) and 1% penicillin-streptomycin in a 5%
CO2 incubator at 37°C. Prior to transfection, cells were
seeded in a 6-well plate with 1×104 cells/well.
Serum-free medium (1.5 ml) and 0.5 ml of Lipofectamine™ 2000
(Invitrogen; Thermo Fisher Scientific, Inc.) containing
transfection vectors were mixed. At 75–85% confluence, 2.0 ml of
transfection mixture was applied in each well. Complete medium was
replaced 4–6 h later.
Western blot analysis
Total protein was extracted from cells or tissues
using radioimmunoprecipitation assay (RIPA) and loaded for
electrophoresis (Beyotime). After transferring on a polyvinylidene
fluoride (PVDF) membranes (Millipore), it was blocked in 5% skim
milk for 2 h, incubated with primary antibodies at 4°C overnight
and secondary antibodies for 2 h. Bands were exposed by
electrochemiluminescence (ECL) and analyzed by Image Software
(NIH).
RNA extraction and quantitative
real-time polymerase chain reaction (qRT-PCR)
RNA extraction was performed using TRIzol method
(Invitrogen; Thermo Fisher Scientific, Inc.). The extracted RNA was
quantified and reverse transcribed into complementary deoxyribose
nucleic acid (cDNA), followed by PCR using SYBR-Green method
(Takara). QRT-PCR was performed at 94°C for 5 min, and 40 cycles at
94°C for 30 sec, 55°C for 30 sec and 72°C for 90 sec.
Cell counting kit-8 (CCK-8) assay
Cells were seeded in a 96-well plate with
2×103 cells/well. Absorbance (A) at 450 nm was recorded
at the appointed time points using the CCK-8 kit (Dojindo
Laboratories) for depicting the viability curve.
Apoptosis determination
Apoptotic rate in GC cells was determined through
calculating caspase-3 activity using a relevant commercial kit
(Beyotime).
Flow cytometry
Cells were fixed in 75% ethanol at 4°C overnight,
and washed with phosphate-buffered saline (PBS) twice. After
incubation with RNase A at 37°C for 30 min, cells were dyed with
propidium iodide (PI). Cell cycle distribution was finally analyzed
by FACSCalibur flow cytometry (BD Biosciences).
RNA immunoprecipitation (RIP)
Cells were treated according to the procedures of
Millipore Magna RIP™ RNA-Binding Protein Immunoprecipitation Kit
(Millipore). Cell lysate was incubated with anti-EZH2 or IgG
antibody at 4°C for 6 h. A protein-RNA complex was captured and
digested with 0.5 mg/ml proteinase K containing 0.1% sodium dodecyl
sulphate (SDS) to extract RNA. The magnetic beads were repeatedly
washed with RIP washing buffer to remove non-specific adsorption as
much as possible. Finally, the extracted RNA was subjected to mRNA
level determination using qRT-PCR.
Transwell assay
Fifty microliters of FN (100 µg/ml) was coated in
the bottom of Transwell chambers. Cell density was adjusted to
1×106/ml. One hundred microliters of suspension was
applied to the upper chamber of Transwell chambers (Millipore)
pre-coated with 100 µl of diluted Matrigel (BD Biosciences). Into
the lower chamber, 600 µl of medium containing 10% FBS was applied.
After 48 h of incubation, invasive cells were fixed in methanol for
30 min and dyed with 0.1% crystal violet for 10 min. Invasive cells
were captured and counted in 6 randomly selected fields per sample.
Migration assay was similarly performed except for Matrigel
pre-coating.
Statistical analysis
Statistical Product and Service Solutions (SPSS)
19.0 software (IBM Corp.) was used for data analyses. Data were
expressed as mean ± standard deviation. Intergroup differences were
analyzed by the t-test. P<0.05 was considered statistically
significant.
Results
Upregulation of ST7-AS1 in GC
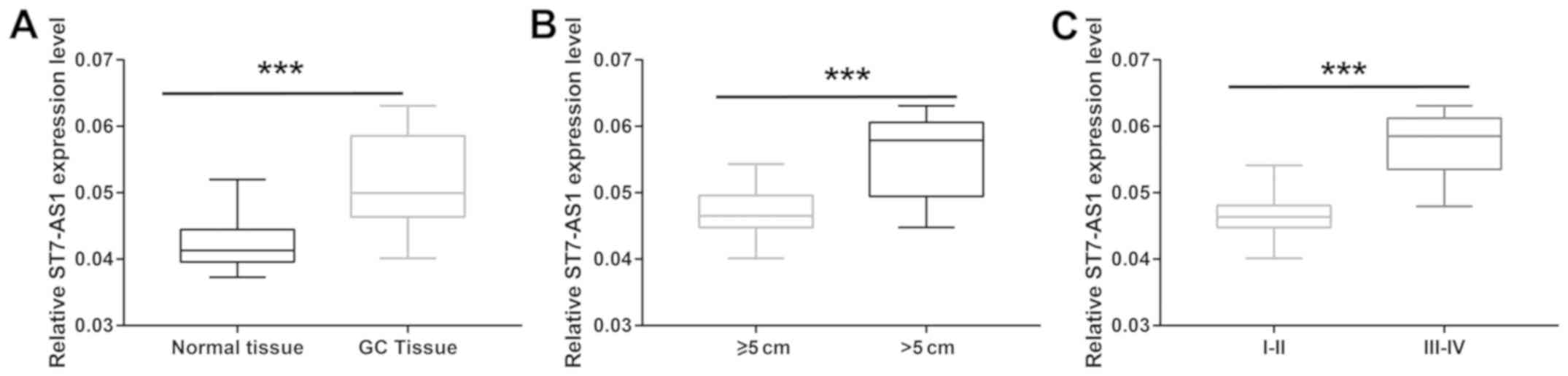
Expression pattern of ST7-AS1 in GC tissues was
examined. As qRT-PCR revealed, ST7-AS1 was upregulated in GC
tissues relative to normal ones (Fig.
1A). Moreover, ST7-AS1 level was higher in GC tissues of >5
cm in size than those ≤5 cm (Fig.
1B). Higher level of ST7-AS1 was observed in GC patients with
stage III–IV compared with those with stage I–II (Fig. 1C). It indicated that ST7-AS1 was
involved in the progression of GC.
Knockdown of ST7-AS1 suppresses
viability, arrests cell cycle and induces apoptosis of GC
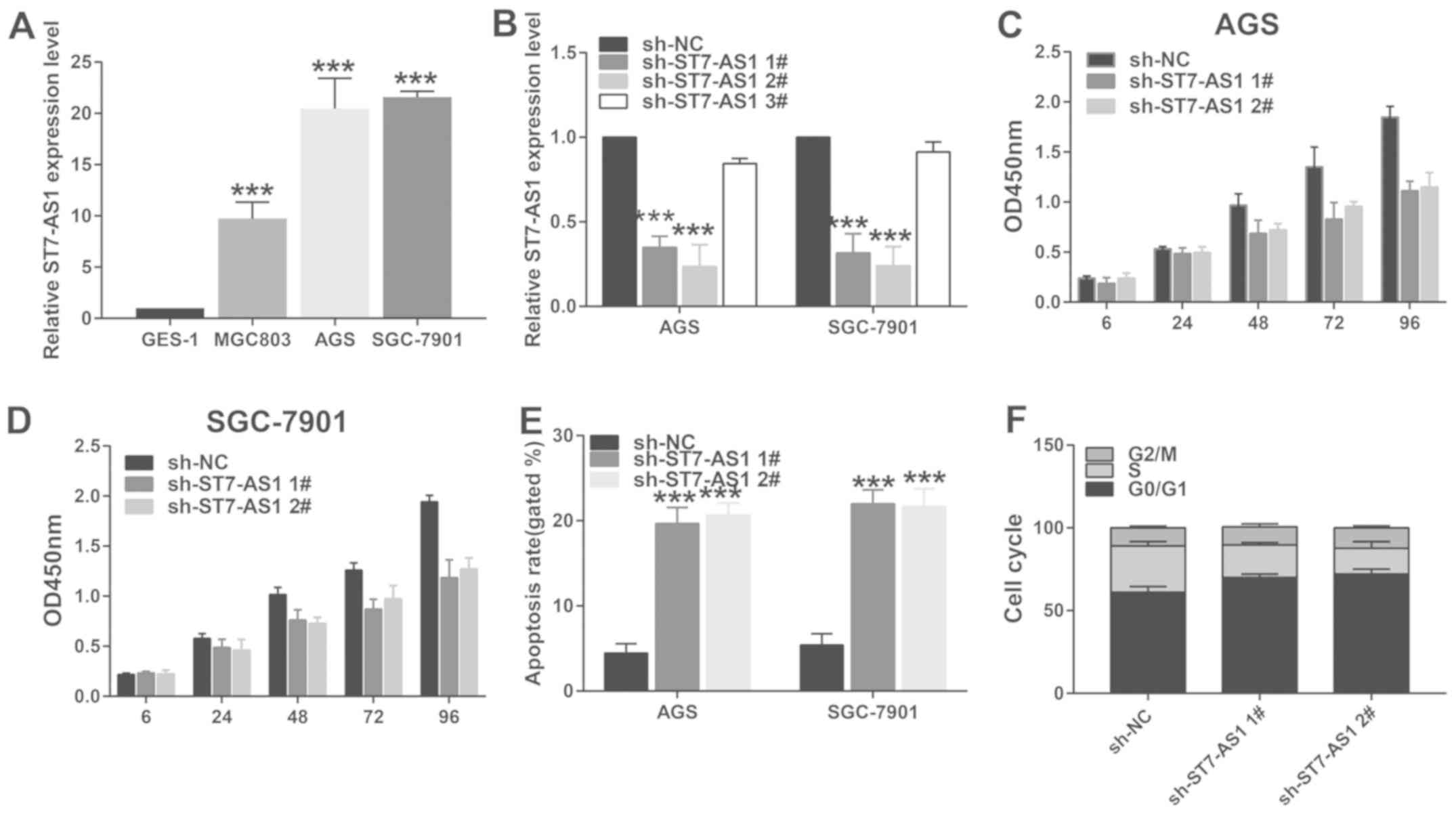
Compared with epithelial cells of gastric mucosa,
ST7-AS1 was upregulated in GC cells, especially in AGS and SGC-7901
cells (Fig. 2A). Three transfection
vectors of sh-ST7-AS1 (sh-ST7-AS1 1#, sh-ST7-AS1 2# and sh-ST7-AS1
3#) were tested for their transfection efficacy. QRT-PCR data
revealed pronounced transfection efficacy in the former two vectors
(Fig. 2B). In AGS and SGC-7901 cells
transfected with sh-ST7-AS1 1# or sh-ST7-AS1 2#, the viability
greatly decreased compared with controls (Fig. 2C and D). Apoptotic rate was elevated
by transfection of sh-ST7-AS1 1# or sh-ST7-AS1 2# (Fig. 2E). Moreover, cell ratio in G0/G1
phase was enhanced after transfection of sh-ST7-AS1 1# or
sh-ST7-AS1 2# in GC cells, indicating arrested cell cycle
progression (Fig. 2F).
Knockdown of ST7-AS1 suppressed
migratory and invasive abilities of GC
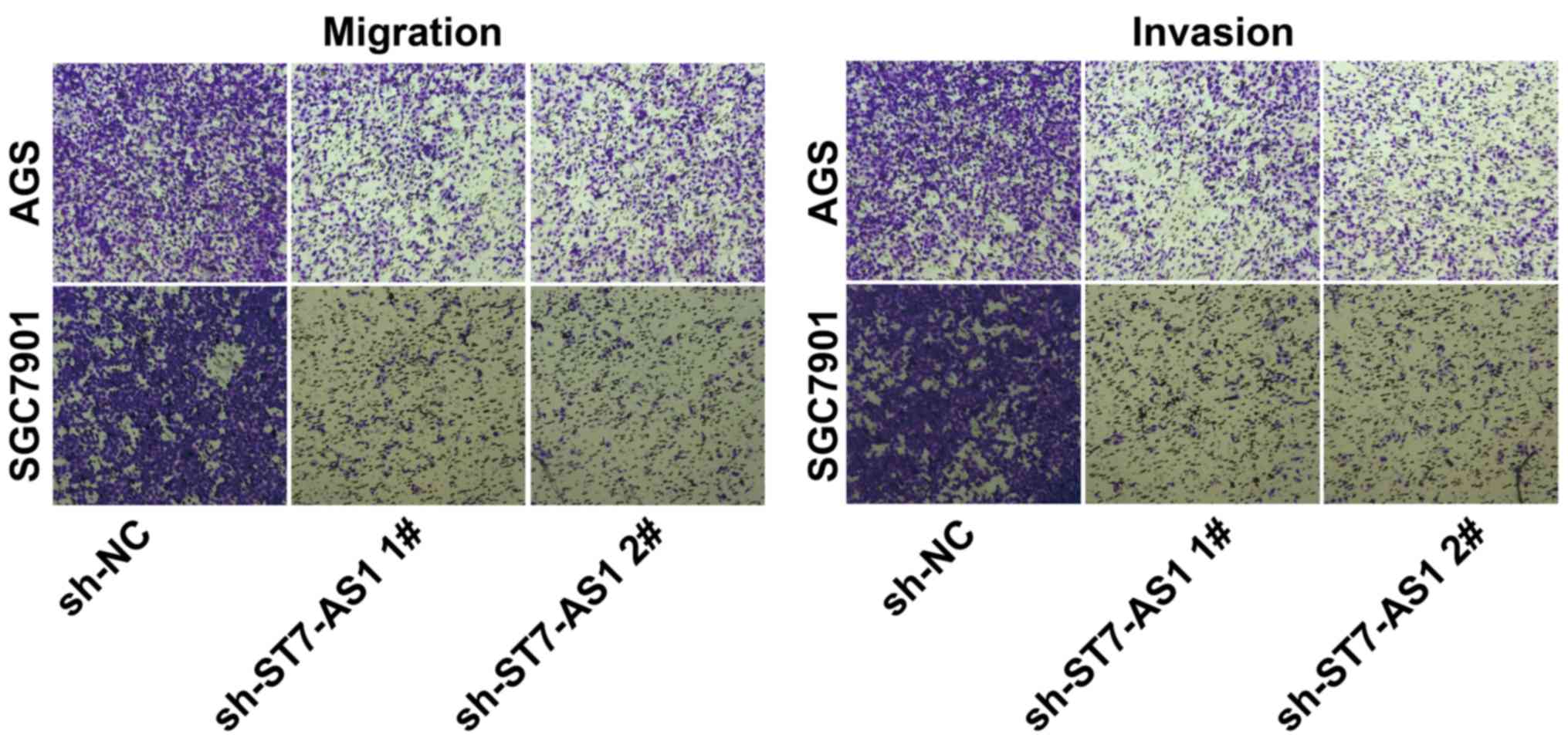
After transfection of sh-ST7-AS1 1# or sh-ST7-AS1 2#
in AGS and SGC-7901 cells, Transwell assay illustrated attenuated
migratory and invasive abilities relative to controls (Fig. 3).
ST7-AS1 mediats cellular behavior of
GC via interacting with EZH2
To uncover the molecular mechanism of ST7-AS1 in
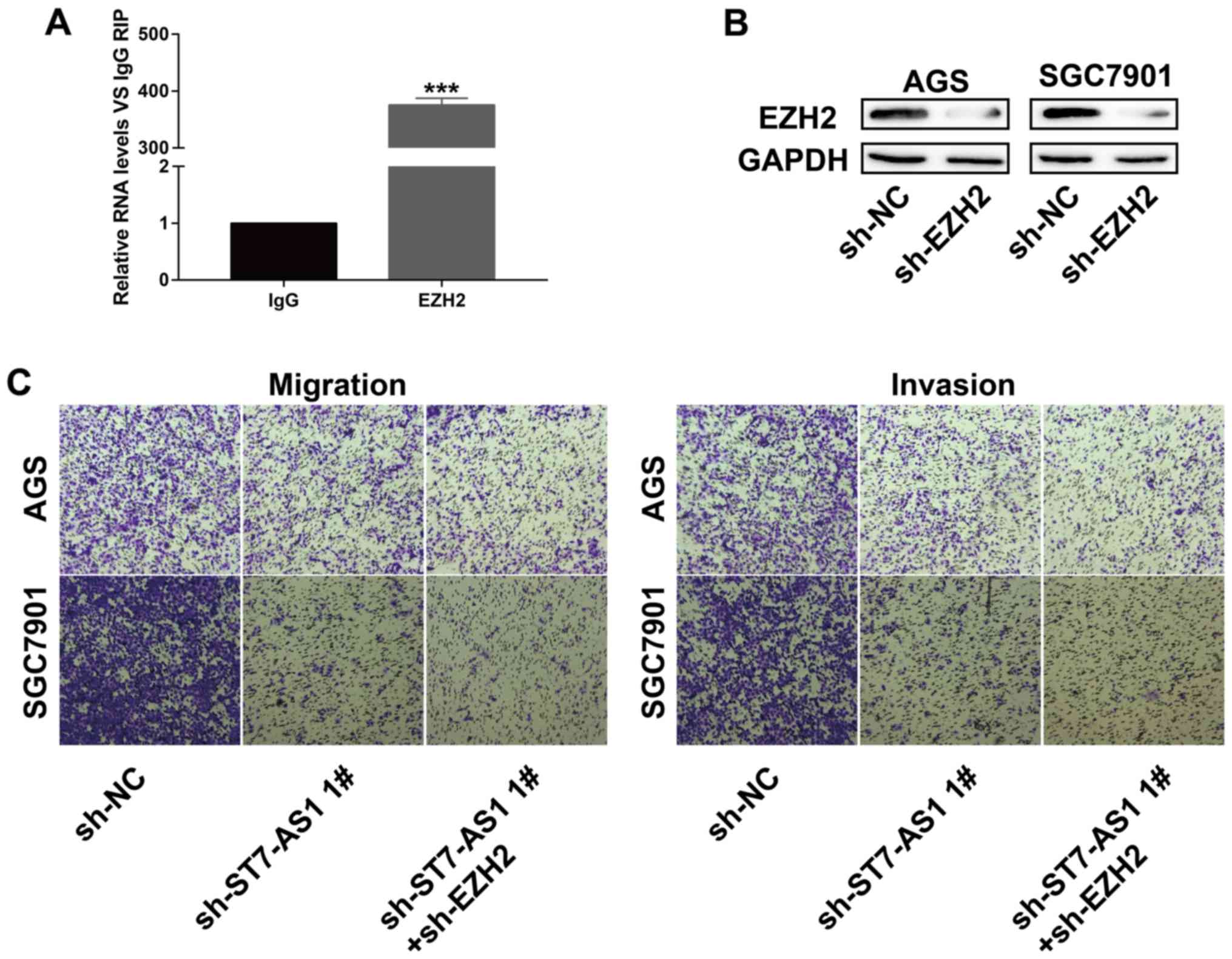
regulating cellular behavior of GC, RIP assay was conducted to
assess the potential interaction between ST7-AS1 and EZH2. ST7-AS1
was greatly enriched in anti-EZH2 antibody relative to control IgG,
verifying the interaction between ST7-AS1 and EZH2 (Fig. 4A). Transfection of sh-EZH2 markedly
downregulated protein level of EZH2, presenting an effective
transfection efficacy in GC cells (Fig.
4B). Interestingly, the attenuated migratory and invasive
abilities of GC cells transfected with sh-ST7-AS1 1# were further
inhibited by co-transfection of sh-EZH2 (Fig. 4C). It is indicated that ST7-AS1
accelerated GC cells to migrate and invade via interacting with
EZH2.
Discussion
GC is a common malignancy worldwide, especially in
China (23). Recently,
identification of novel therapeutic hallmarks of GC have been
widely conducted (24). GC is
characterized by infinitely excessive proliferation and growth of
tumor cells (25). Oncogene
activation and tumor-suppressor gene inactivation are the main
causes of tumorigenesis (26).
Traditional treatments for GC include surgery, chemotherapy and
radiotherapy. These therapeutic strategies destroy normal tissues
and cells while destroying tumor tissues (27). In addition, the development of
chemotherapy-resistance markedly limits the therapeutic efficacy of
GC (28). Hence, it is urgent to
search for effective targets for GC treatment.
lncRNAs barely encode proteins, but mediate gene
expression at multiple levels (29,30).
Increasing evidence has proven the role of lncRNAs in tumor
progression (31). They are capable
of mediating epigenetic regulation and cellular behavior (32). lncRNA ST7-AS1 is a newly discovered
one located on 7q31.2 (33). In this
study, ST7-AS1 was upregulated in GC tissues and cell lines.
ST7-AS1 level was higher in GC patients with worse tumor stage and
larger tumor size, indicating its carcinogenic role in GC.
Moreover, knockdown of ST7-AS1 attenuated proliferative, migratory
and invasive abilities, arrested cell cycle and induced apoptosis
of GC cells.
Epigenetic modifications are involved in gene
expressions of tumor-related molecules. Polycomb repressive complex
2 (PRC2) regulates transcription of target genes mainly by
trimethylation of H3K27me3 (34).
Multiple studies have shown that EZH2 is the methylation enzyme
subunit of PRC2. Overexpression or mutation of EZH2 can induce
tumorigenesis and promote tumor progression (35). EZH2 is able to mediate multiple
pathological processes in cells, such as cell cycle, cell
senescence, and cell differentiation (36).
The present study confirmed the interaction between
ST7-AS1 and EZH2 through RIP assay. Subsequently, we speculated
whether EZH2 was involved in the malignant progression of GC
regulated by ST7-AS1. Notably, the decreased migratory and invasive
abilities in GC cells with ST7-AS1 knockdown were further
attenuated by EZH2 knockdown. Collectively, ST7-AS1 mediated
malignant phenotypes of GC cells via interacting with EZH2.
In conclusion, upregulated ST7-AS1 in GC accelerated
proliferative, migratory and invasive abilities, and inhibited
apoptosis, thus aggravating the progression of GC. lncRNA ST7-AS1
could be utilized as a promising target for improving clinical
outcomes of GC patients.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
SC and FM designed the study and performed the
experiments, SC and YW collected the data, FM and PL analyzed the
data, SC and FM prepared the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
The Fourth Affiliated Hospital of China Medical University
(Shenyang, China). Signed informed consents were obtained from the
patients and/or guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Karimi P, Islami F, Anandasabapathy S,
Freedman ND and Kamangar F: Gastric cancer: Descriptive
epidemiology, risk factors, screening, and prevention. Cancer
Epidemiol Biomarkers Prev. 23:700–713. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ding Z, Jiang L, Zhang K and Huang R:
Short- and long-term outcomes of conversion in laparoscopic
gastrectomy for gastric cancer. J BUON. 23:1004–1012.
2018.PubMed/NCBI
|
|
3
|
Xu C, Cui H, Li H, Wu Y, An H and Guo C:
Long non-coding RNA ZEB2-AS1 expression is associated with disease
progression and predicts outcome in gastric cancer patients. J
BUON. 24:663–671. 2019.PubMed/NCBI
|
|
4
|
de Boer WB, Ee H and Kumarasinghe MP:
Neoplastic lesions of gastric adenocarcinoma and poximal polyposis
syndrome (GAPPS) are gastric phenotype. Am J Surg Pathol. 42:1–8.
2018.PubMed/NCBI
|
|
5
|
Markowski AR, Markowska A and
Guzinska-Ustymowicz K: Pathophysiological and clinical aspects of
gastric hyperplastic polyps. World J Gastroenterol. 22:8883–8891.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nomura E and Okajima K:
Function-preserving gastrectomy for gastric cancer in Japan. World
J Gastroenterol. 22:5888–5895. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Akimoto T, Muto S, Kutsuwada T, Kutsuwada
K and Nagata D: Peritoneal dialysis and malignancy: An experience
with a patient complicated by gastric carcinoma. Clin Med Insights
Case Rep. 12:11795476198351762019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Çolak Ş, Gürbulak B, Çakar E and Bektaş H:
Resection of mucosal and submucosal gastrointestinal lesions and a
double endoscope experience. JSLS. 23:e2018.00096. 2019. View Article : Google Scholar
|
|
9
|
Huarte M: The emerging role of lncRNAs in
cancer. Nat Med. 21:1253–1261. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Peng WX, Koirala P and Mo YY:
LncRNA-mediated regulation of cell signaling in cancer. Oncogene.
36:5661–5667. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Botti G, Marra L, Malzone MG, Anniciello
A, Botti C, Franco R and Cantile M: LncRNA HOTAIR as prognostic
circulating marker and potential therapeutic target in patients
with tumor diseases. Curr Drug Targets. 18:27–34. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu Y, Guo G, Zhong Z, Sun L, Liao L, Wang
X, Cao Q and Chen H: Long non-coding RNA FLVCR1-AS1 sponges miR-155
to promote the tumorigenesis of gastric cancer by targeting c-Myc.
Am J Transl Res. 11:793–805. 2019.PubMed/NCBI
|
|
13
|
Li J, Xu Q, Wang W and Sun S: MIR100HG: A
credible prognostic biomarker and an oncogenic lncRNA in gastric
cancer. Biosci Rep. 39:BSR201901712019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Marín-Béjar O, Mas AM, González J,
Martinez D, Athie A, Morales X, Galduroz M, Raimondi I, Grossi E,
Guo S, et al: The human lncRNA LINC-PINT inhibits tumor cell
invasion through a highly conserved sequence element. Genome Biol.
18:2022017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yamagishi M and Uchimaru K: Targeting EZH2
in cancer therapy. Curr Opin Oncol. 29:375–381. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yan KS, Lin CY, Liao TW, Peng CM, Lee SC,
Liu YJ, Chan WP and Chou RH: EZH2 in cancer progression and
potential application in cancer therapy: A Friend or Foe? Int J Mol
Sci. 18:11722017. View Article : Google Scholar
|
|
17
|
Wassef M, Luscan A, Aflaki S, Zielinski D,
Jansen PW, Baymaz HI, Battistella A, Kersouani C, Servant N,
Wallace MR, et al: EZH1/2 function mostly within canonical PRC2 and
exhibit proliferation-dependent redundancy that shapes mutational
signatures in cancer. Proc Natl Acad Sci USA. 116:6075–6080. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sun C, Ban Y, Wang K, Sun Y and Zhao Z:
SOX5 promotes breast cancer proliferation and invasion by
transactivation of EZH2. Oncol Lett. 17:2754–2762. 2019.PubMed/NCBI
|
|
19
|
Labbé DP, Sweeney CJ, Brown M, Galbo P,
Rosario S, Wadosky KM, Ku SY, Sjöström M, Alshalalfa M, Erho N, et
al: TOP2A and EZH2 provide early detection of an aggressive
prostate cancer subgroup. Clin Cancer Res. 23:7072–7083. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Emran AA, Chatterjee A, Rodger EJ, Tiffen
JC, Gallagher SJ, Eccles MR and Hersey P: Targeting DNA methylation
and EZH2 activity to overcome melanoma resistance to immunotherapy.
Trends Immunol. 40:328–344. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ramakrishnan S, Granger V, Rak M, Hu Q,
Attwood K, Aquila L, Krishnan N, Osiecki R, Azabdaftari G, Guru K,
et al: Inhibition of EZH2 induces NK cell-mediated differentiation
and death in muscle-invasive bladder cancer. Cell Death Differ.
26:2100–2114. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yu L, Despotovic N, Kovacs MS, Pin CL and
Luyt LG: 18F-labeled PET probe targeting enhancer of Zeste
homologue 2 (EZH2) for cancer imaging. ACS Med Chem Lett.
10:334–340. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li Z, Liu ZM and Xu BH: A meta-analysis of
the effect of microRNA-34a on the progression and prognosis of
gastric cancer. Eur Rev Med Pharmacol Sci. 22:8281–8287.
2018.PubMed/NCBI
|
|
24
|
Lin Y, Zhang CS, Li SJ, Li Z and Sun FB:
LncRNA LOC554202 promotes proliferation and migration of gastric
cancer cells through regulating p21 and E-cadherin. Eur Rev Med
Pharmacol Sci. 22:8690–8697. 2018.PubMed/NCBI
|
|
25
|
Hernanz N, Rodríguez de Santiago E, Marcos
Prieto HM, Jorge Turrión MÁ, Barreiro Alonso E, Rodríguez Escaja C,
Jiménez Jurado A, Sierra M, Pérez Valle I, Volpato N, et al:
Characteristics and consequences of missed gastric cancer: A
multicentric cohort study. Dig Liver Dis. 51:894–900. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chen C, Tang X, Liu Y, Zhu J and Liu J:
Induction/reversal of drug resistance in gastric cancer by
non-coding RNAs (Review). Int J Oncol. 54:1511–1524.
2019.PubMed/NCBI
|
|
27
|
Yamagata Y, Saito K, Ban S, Fujii A and
Oya M: The origin of p40-negative and CDX2-positive primary
squamous cell carcinoma of the stomach: Case report. World J Surg
Oncol. 17:532019.PubMed/NCBI
|
|
28
|
Jain VS, Kawale D, Jain SM, Waghmare C and
Pemmaraju G: Various addiction patterns, dietary habits, associated
medical problems, and socioeconomic status in gastrointestinal
malignancies: A prospective study in rural area of Maharashtra,
India. J Cancer Res Ther. 15:104–107. 2019.PubMed/NCBI
|
|
29
|
Jarroux J, Morillon A and Pinskaya M:
History, discovery, and classification of lncRNAs. Adv Exp Med
Biol. 1008:1–46. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Andersen RE and Lim DA: Forging our
understanding of lncRNAs in the brain. Cell Tissue Res. 371:55–71.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Léveillé N and Baglio SR:
Exosome-transferred lncRNAs at the core of cancer bone lesions.
Crit Rev Oncol Hematol. 139:125–127. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mao Z, Wu Y, Zhou J and Xing C:
Salinomycin reduces epithelial-mesenchymal transition-mediated
multidrug resistance by modifying long noncoding RNA HOTTIP
expression in gastric cancer cells. Anticancer Drugs. 30:892–899.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Qin H, Xu J, Gong L, Jiang B and Zhao W:
The long noncoding RNA ST7-AS1 promotes laryngeal squamous cell
carcinoma by stabilizing CARM1. Biochem Biophys Res Commun.
512:34–40. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Huang M, Hou J, Wang Y, Xie M, Wei C, Nie
F, Wang Z and Sun M: Long noncoding RNA LINC00673 is activated by
SP1 and exerts oncogenic properties by interacting with LSD1 and
EZH2 in gastric cancer. Mol Ther. 25:1014–1026. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen DL, Ju HQ, Lu YX, Chen LZ, Zeng ZL,
Zhang DS, Luo HY, Wang F, Qiu MZ, Wang DS, et al: Long non-coding
RNA XIST regulates gastric cancer progression by acting as a
molecular sponge of miR-101 to modulate EZH2 expression. J Exp Clin
Cancer Res. 35:1422016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yang Y, Zhu F, Wang Q, Ding Y, Ying R and
Zeng L: Inhibition of EZH2 and EGFR produces a synergistic effect
on cell apoptosis by increasing autophagy in gastric cancer cells.
Onco Targets Ther. 11:8455–8463. 2018. View Article : Google Scholar : PubMed/NCBI
|