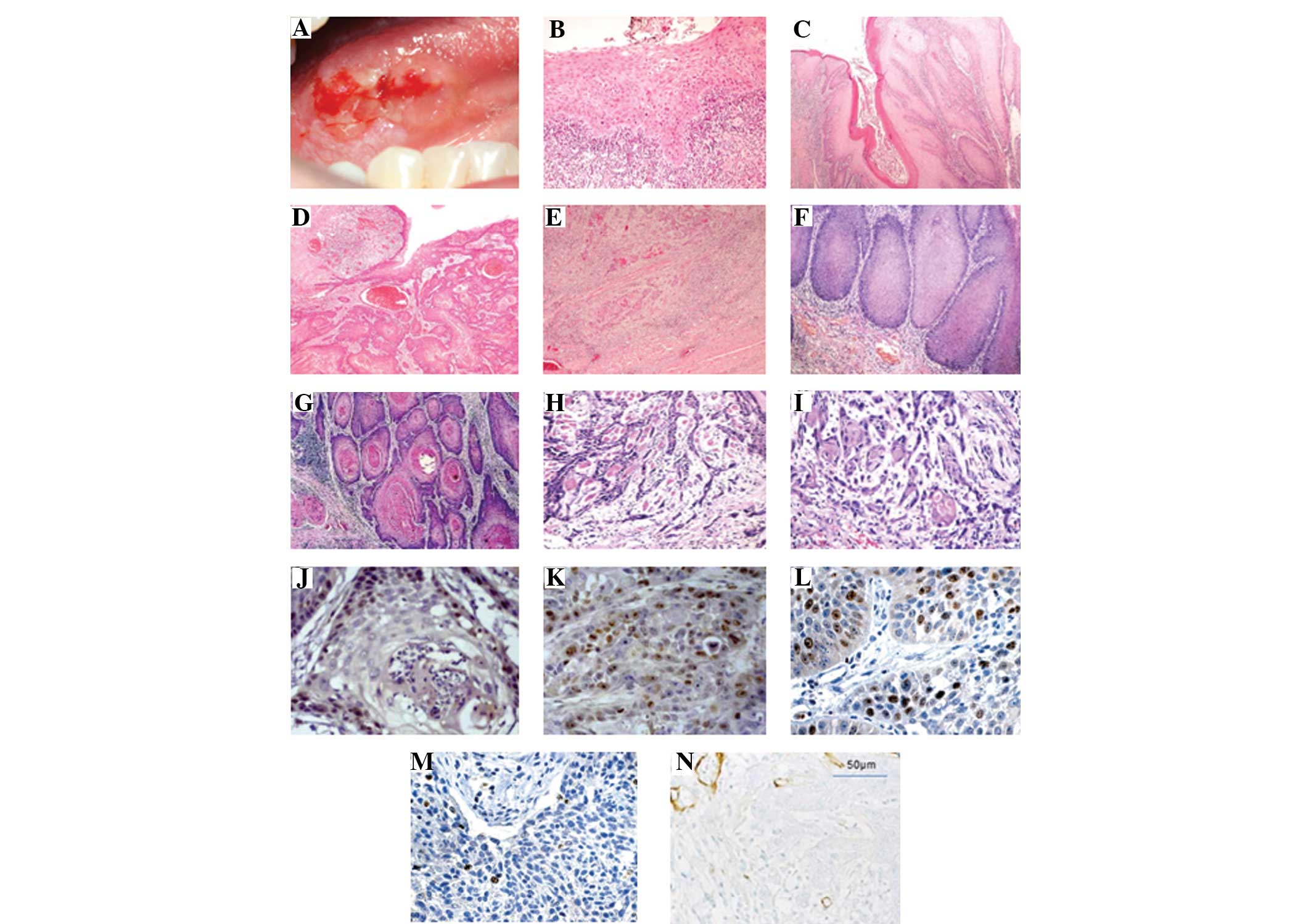
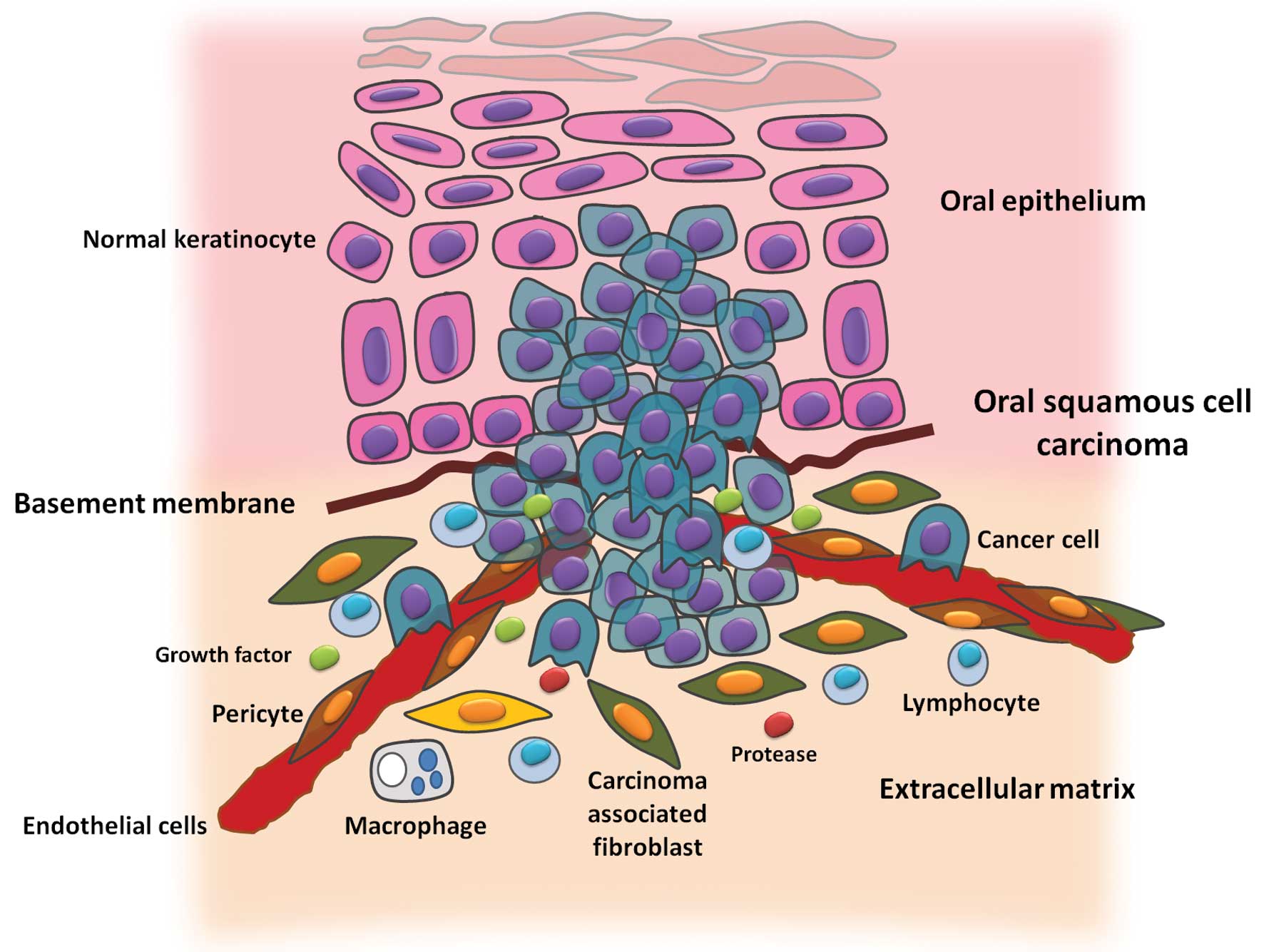
|
1
|
Dissanayaka WL, Pitiyage G, Kumarasiri PV,
Liyanage RL, Dias KD and Tilakaratne WM: Clinical and
histopathologic parameters in survival of oral squamous cell
carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol. 113:518–525.
2012.
|
|
2
|
Bray F, Sankila R, Ferlay J and Parkin DM:
Estimates of cancer incidence and mortality in Europe in 1995. Eur
J Cancer. 38:99–166. 2002.
|
|
3
|
Parkin D, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108.
2005.
|
|
4
|
Koontongkaew S: The tumor microenvironment
contribution to development, growth, invasion and metastasis of
head and necksquamous cell carcinomas. J Cancer. 4:66–83. 2013.
|
|
5
|
Wangsa D, Ryott M, Avall-Lundqvist E, et
al: Ki-67 expression predicts locoregional recurrence in stage I
oral tongue carcinoma. Br J Cancer. 99:1121–1128. 2008.
|
|
6
|
Jung DW, Che ZM, Kim J, Kim K, Kim KY and
Williams D: Tumor-stromal crosstalk in invasion of oral squamous
cell carcinoma: a pivotal role of CCL7. Int J Cancer. 127:332–344.
2010.
|
|
7
|
Centelles PV, Seoane-Romero JM, Gómez I,
Diz-Dios P, de Melo NS and Seoane J: Timing of oral cancer
diagnosis: Implications for prognosis and survival. Oral Cancer.
Ogbureke KUE: InTech; pp. 173–188. 2012
|
|
8
|
Bettendorf O, Piffkò J and Bànkfalvi A:
Prognostic and predictive factors in oral squamous cell cancer:
important tools for planning individual therapy? Oral Oncol.
40:110–119. 2004.
|
|
9
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000.
|
|
10
|
Tumuluri V, Thomas GA and Fraser IS:
Analysis of the Ki-67 antigen at the invasive tumour front of human
oral squamous cell carcinoma. J Oral Pathol Med. 31:598–604.
2002.
|
|
11
|
Wilkey JF, Buchberger G, Saucier K, et al:
Cyclin D1 overexpression increases susceptibility to
4-nitroquinoline-1 -oxide-induced dysplasia and neoplasia in murine
squamous oral epithelium. Mol Carcinog. 48:853–861. 2009.
|
|
12
|
Neville B, Damm D, Allen C and Bouquot J:
Oral and Maxillofacial Pathology. 3rd edition. Saunders Elsevier;
Philadelphia, PA: pp. 356–367. 2009
|
|
13
|
Jerjes W, Upile T, Petrie A, et al:
Clinicopathological parameters, recurrence, locoregional and
distant metastasis in 115 T1–T2 oral squamous cell carcinoma
patients. Head Neck Oncol. 2:92010.
|
|
14
|
Sapp JP, Eversole LR and Wysocki GP:
Contemporary Oral and Maxillofacial Pathology Chapter 6: Epithelial
Disorders. 2nd edition. Mosby Year Book Inc; Maryland Heights, MO:
pp. 184–193. 2004
|
|
15
|
Neville BW and Day TA: Oral cancer and
precancerous lesions. CA Cancer J Clin. 52:195–215. 2002.
|
|
16
|
Wang Z, Zhang B, Jiang L, et al: RACK1, an
excellent predictor for poor clinical outcome in oral squamous
carcinoma, similar to Ki67. Eur J Cancer. 45:490–496. 2009.
|
|
17
|
Rivera MCA: 4NQO carcinogenesis: A model
of oral squamous cell carcinoma. Int J Morphol. 30:309–314.
2012.
|
|
18
|
Fuentes B, Duaso J, Droguett D, et al:
Progressive extracellular matrix disorganization in chemically
induced murine oral squamous cell carcinoma. ISRN Pathology.
2012.
|
|
19
|
Rivera CA, Droguett DA, Kemmerling U and
Venegas BA: Chronic restraint stress in oral squamous cell
carcinoma. J Dent Res. 90:799–803. 2011.
|
|
20
|
Pindborg JJ, Reichart PA, Smith CJ and Van
der Waal I: WHO International Histological Classification of
Tumours Histological typing of cancer and precancer of the oral
mucosa. Springer-Verlag; New York: 1997
|
|
21
|
Wang X, Zhang J, Fan M, et al: The
expression of E-cadherin at the invasive tumor front of oral
squamous cell carcinoma: immunohistochemical and RT-PCR analysis
with clinicopathological correlation. Oral Surg Oral Med Oral
Pathol Oral Radiol Endod. 107:547–554. 2009.
|
|
22
|
Bànkfalvi A and Piffkò J: Prognostic and
predictive factors in oral cancer: the role of the invasive tumour
front. J Oral Pathol Med. 29:291–298. 2000.
|
|
23
|
Kurokawa H, Zhang M, Matsumoto S, et al:
The high prognostic value of the histologic grade at the deep
invasive front of tongue squamous cell carcinoma. J Oral Pathol
Med. 34:329–333. 2005.
|
|
24
|
Bryne M, Koppang HS, Lilleng R and
Kjaerheim A: Malignancy grading of the deep invasive margins of
oral squamous cell carcinomas has high prognostic value. J Pathol.
166:375–381. 2005.
|
|
25
|
Oliveira L, Ribeiro-Silva A, Costa J,
Simões A, Matteo M and Zucoloto S: Prognostic factors and survival
analysis in a sample of oral squamous cell carcinoma patients. Oral
Surg Oral Med Oral Pathol Oral Radiol Endod. 106:685–695. 2008.
|
|
26
|
Leemans CR, Braakhuis BJ and Brakenhoff
RH: The molecular biology of head and neck cancer. Nat Rev Cancer.
11:9–22. 2010.
|
|
27
|
Choi S and Myers J: Molecular pathogenesis
of oral squamous cell carcinoma: implications for therapy. J Dent
Res. 87:14–32. 2008.
|
|
28
|
Massano J, Regateiro F, Januário G and
Ferreira A: Oral squamous cell carcinoma: review of prognostic and
predictive factors. Oral Surg Oral Med Oral Pathol Oral Radiol
Endod. 102:67–76. 2006.
|
|
29
|
van Houten VM, Tabor MP, van den Brekel
MW, et al: Mutated p53 as a molecular marker for the diagnosis of
head and neck cancer. J Pathol. 198:476–486. 2002.
|
|
30
|
Maddocks OD and Vousden KH: Metabolic
regulation by p53. J Mol Med (Berl). 89:237–245. 2011.
|
|
31
|
Yerushalmi R, Woods R, Ravdin PM, Hayes M
and Gelmon KA: Ki-67 in breast cancer: prognostic and predictive
potential. Lancet Oncol. 11:1742010.
|
|
32
|
Bitu CC, Carrera M, Lopes MA, Kowalski LP,
Soares FA and Coletta RD: HOXB7 expression is a prognostic factor
for oral squamous cell carcinoma. Histopathology. 60:662–665.
2012.
|
|
33
|
Shah N and Sukumar S: The Hox genes and
their roles in oncogenesis. Nat Rev Cancer. 10:361–371. 2010.
|
|
34
|
Tucci R, Campos MS, Matizonkas-Antonio LF,
Durazzo M, dos Pinto Junior DS and Nunes FD: HOXB5 expression in
oral squamous cell carcinoma. J Appl Oral Sci. 19:125–129.
2011.
|
|
35
|
Liao WT, Jiang D, Yuan J, et al: HOXB7 as
a prognostic factor and mediator of colorectal cancer progression.
Clin Cancer Res. 17:3569–3578. 2011.
|
|
36
|
Fan HX, Li HX, Chen D, Gao ZX and Zheng
JH: Changes in the expression of MMP2, MMP9, and ColIV in stromal
cells in oral squamous tongue cell carcinoma: relationships and
prognostic implications. J Exp Clin Cancer Res. 31:902012.
|
|
37
|
Kumar V, Abbas AK and Aster JC: Robbins
Basic Pathology Chapter 7: Neoplasia. 8th ed. Saunders;
Philadelphia, PA: pp. 298–299. 2012
|
|
38
|
Tamamura R, Nagatsuka H, Siar CH, et al:
Comparative analysis of basal lamina type IV collagen alpha chains,
matrix metalloproteinases-2 and −9 expressions in oral dysplasia
and invasive carcinoma. Acta Histochem. 115:113–119. 2013.
|
|
39
|
de Vicente JC, Fresno MF, Villalain L,
Vega JA and Hernández Vallejo G: Expression and clinical
significance of matrix metalloproteinase-2 and matrix
metalloproteinase-9 in oral squamous cell carcinoma. Oral Oncol.
41:283–293. 2005.
|
|
40
|
Baba Y, Iyama K, Ikeda K, et al: The
Expression of type IV collagen α6 chain is related to the prognosis
in patients with esophageal squamous cell carcinoma. Ann Surg
Oncol. 15:555–565. 2008.
|
|
41
|
Kenny PA, Lee GY and Bissell MJ: Targeting
the tumor microenvironment. Front Biosci. 12:3468–3474. 2007.
|
|
42
|
Zhang J and Liu J: Tumor stroma as targets
for cancer therapy. Pharmacol Ther. 137:200–215. 2013.
|
|
43
|
Liotta LA and Kohn EC: The
microenvironment of the tumour-host interface. Nature. 411:375–379.
2001.
|
|
44
|
Shimoda M, Mellody KT and Orimo A:
Carcinoma-associated fibroblasts are a rate-limiting determinant
for tumour progression. Semin Cell Dev Biol. 21:19–25. 2010.
|
|
45
|
Räsänen K and Vaheri A: Activation of
fibroblasts in cancer stroma. Exp Cell Res. 316:2713–2722.
2010.
|
|
46
|
Xouri G and Christian S: Origin and
function of tumor stroma fibroblasts. Semin Cell Dev Biol.
21:40–46. 2010.
|
|
47
|
de-Assis EM, Pimenta LG, Costa-e-Silva E,
Souza PE and Horta MC: Stromal myofibroblasts in oral leukoplakia
and oral squamous cell carcinoma. Med Oral Patol Oral Cir Bucal.
17:e733–e738. 2012.
|
|
48
|
Thode C, Jørgensen TG, Dabelsteen E,
Mackenzie I and Dabelsteen S: Significance of myofibroblasts in
oral squamous cell carcinoma. J Oral Pathol Med. 40:201–207.
2011.
|