Introduction
The Sleeping Beauty (SB) system is a genetically
engineered insertional mutagenesis system that consists of two
components: a transposon, which is a series of DNA mobile elements
flanked by indirect repeat sequences, and SB transposase, which
catalyzes the mobilization and reintegration of transposon into
mouse genomic DNA (1,2). Integration into the host chromosome
provides prolonged expression of the transgene. In addition, the SB
system has the advantages of both viral and non-viral vector
systems (3). For this reason, the
SB system has attracted much attention as a promising delivery
system, as well as a discovery tool for cancer-associated genes.
Its ability to achieve long-term in vivo expression suggests
that the somatic integration of oncogenes using SB is a feasible
approach to the development of molecularly defined tumorigenic
mouse models (4). Although
xenograft models and genetically engineered mice are able to mimic
human cancer progression (5), the
present mouse model systems do not correspond to humans with regard
to genetic backgrounds (i.e., different genetic variants, genetic
mutations and subsequent protein expression) (6–8). A
hallmark of human cancer is genetic complexity, meaning a number of
different mutations are commonly involved (9). The complex genetic alterations in
different types of cancer cause various histological subtypes and
can explain the heterogeneous nature of a given neoplasm (9). Carlson et al(4) succeeded in eliciting tumor formation
in mouse hepatocytes by hydrodynamic injection of a transposon
containing an activated NRAS oncogene. Although those investigators
studied the simple feature of the tumors induced by insertional
mutagenesis, the exact origins and tumor subtypes have yet to be
determined.
In the present study, the c-Myc, HRAS and shp53
oncogenes were delivered with SB into C57BL/6 mice. A malignant
undifferentiated tumor was subsequently identified at the injection
site on the subcutis of the right side of the lower ventral
abdominal area. The nature of the tumor was identified by
histological and immunohistochemical methods (Table I).
 | Table IThe immunohistochemical panel. |
Table I
The immunohistochemical panel.
| Immunostains | Type | Manufacturer,
location | Dilution |
|---|
| 1 | CD45 | M | Abcam, Cambridge,
UK | 1:2,000 |
| 2 | CD163 | M | Abcam, Cambridge,
UK | 1:2,000 |
| 3 | CD68 | M | Abcam, Cambridge,
UK | 1:4,000 |
| 4 | Desmin | M | Abcam, Cambridge,
UK | 1:1,000 |
| 5 | Myogenin | M | Abcam, Cambridge,
UK | 1:100 |
| 6 | Melanoma | M | Abcam, Cambridge,
UK | 1:1,000 |
| 7 | S100 | M | Abcam, Cambridge,
UK | 1:400 |
| 8 | α-SMA | M | Abcam, Cambridge,
UK | 1:400 |
| 9 | Pan-cytokeratin | M | Abcam, Cambridge,
UK | 1:3,000 |
| 10 | Cytokeratin 7 | P | Abcam, Cambridge,
UK | 1:4,000 |
| 11 | Cytokeratin 20 | M | Abcam, Cambridge,
UK | 1:2,000 |
| 12 | MDM2 | M | Abcam, Cambridge,
UK | 1:1,000 |
| 13 | CDK4 | M | Abcam, Cambridge,
UK | 1:1,000 |
Materials and methods
Animals
Female 5-week-old C57BL/6 mice were purchased from
Orient Bio (Yongin, Korea). The mice were housed at the laboratory
animal facility at Asan Institute for Life Sciences under specific
pathogen-free conditions and used according to the guidelines of
the Institutional Animal Care and Use Committee of Asan Institute
for Life Sciences.
Plasmid construction
Plasmids encoding the SB transposase (pPGK/SB13) and
transposon vectors (PT2/BH) with multiple cloning sites between two
indirect repeat sequences (IR/DRs) were used for this study.
Plasmids pPGK/SB13 and PT2/BH were kind gifts from Drs David
Largaespada and Perry Hackett at the University of Minnesota. The
cDNA encoding either c-Myc or HRAS was inserted into the pCXEGFP
plasmid (kindly provided by Dr Masaru Okabe at the Osaka
University, Japan) and the transcriptional cassettes were cloned
into PT2/BH. PT2/shp53/GFP4, transposon plasmids encoding a short
hairpin RNA against tumor suppressor P53, was a generous gift from
Dr John Ohlfest at the University of Minnesota. DNA used for
injection was prepared using an EndoFree Plasmid Maxi kit (cat. no.
12362, Qiagen) according to the manufacturer’s instructions.
DNA plasmid injections
Animals received a mixture of three types of
transposon and the plasmid encoding the transposase, as detailed
above. The molar ratio of transposase-encoding plasmids to
transposon plasmids was 1:2. First, three types of transposon were
mixed in equal amounts in total 50 μg and then the transposase
encoding plasmids was added to the transposon mixture with 50 μl of
phosphate-buffered saline (PBS). The DNA mixture was collected with
an insulin syringe (31 G) and injected subcutaneously near the
right inferior mammary gland. Transposon genes and transposase were
regarded to be 7,000 and 5,000 kb in size, respectively, rounded to
the nearest kb.
Animal PET imaging
Radiopharmaceutical preparation
Decay-corrected radiochemical yields ranged from 60
to 70% and after high-performance liquid chromatography (HPLC)
purification, the radiochemical purity was 98±1.2% (mean ± SD). The
specific activity of the [18F]Flu-deoxy-glucose (FDG)
obtained was >100 TBq/mmol. PET scans were performed using a
microPET Focus 120 system (microPET, Concorde Microsystem, Inc.)
with resolutions of 1.18 (radial), 1.13 (tangential) and 1.44 mm
(axial) at the center of the field of view. Each mouse was injected
with 7.4 (0.2 mCi) and 37 MBq (1 mCi) [18F]FDG into the
tail vein and 10-min static PET scans were obtained. Each mouse was
maintained under isoflurane anesthesia during the uptake and
scanning periods. A heating pad and heat lamp were used to maintain
body temperature at ~37°C. PET images were reconstructed by OSEM2D
with a cut-off frequency of 0.5 cycles per pixel. No attenuation
correction was applied.
Tumor monitoring and necropsy
Mice were carefully examined three times each week
to detect tumors. Tumors were measured using a digital caliper. The
tumor volume (v) in mm3 was calculated using the formula
v = L × W2/2, where L is the longest diameter and W is
the tumor length that is perpendicular to L. When the tumor reached
100 mm3 in volume, the mice were humanely euthanized and
subjected to a necropsy in which the tumor was excised with the
circumferential tissue for histopathological examination.
Histology and
immunohistochemistry
After macroscopic examination, the excised tissues
were fixed in 10% neutral-buffered formalin. The specimens were
embedded in paraffin and 3-μm sections were cut and stained using a
hematoxylin and eosin (H&E) stain. In addition,
immunohistochemical markers [all from Abcam, (Cambridge, UK)] were
used to subtype the tumor as follows: CD45 (1:2,000), CD163
(1:2,000) and CD68 (1:4,000) for histiocytic sarcoma; desmin
(1:1,000) and myogenin (1:100) for pleomorphic rhabdomyosarcoma;
HMB45 (1:1,000) and S100 (1:400) for malignant melanoma, α-smooth
muscle actin (α-SMA; 1:400) for leiomyosarcoma; pan-cytokeratin
(1:3,000), cytokeratin 7 (1:4,000) and cytokeratin 20 (1:2,000) for
undifferentiated carcinoma; and murine double minute 2 (MDM2;
1:1,000) and cyclin-dependent kinase 4 (CDK4; 1:1,000) for
pleomorphic liposarcoma (Table
I).
Detection of the expression of
genes
Expression of the c-Myc and HRAS genes was
identified by immunohistochemistry with anti-c-Myc (1:1,000, Abcam)
and HRAS (1:1,000, Abcam) antibodies, respectively. For the
detection of the shp53 gene, an anti-GFP antibody (1:4,000, Abcam)
was used as a GFP gene was contained in the shp53 DNA plasmid as a
reporter gene.
Results
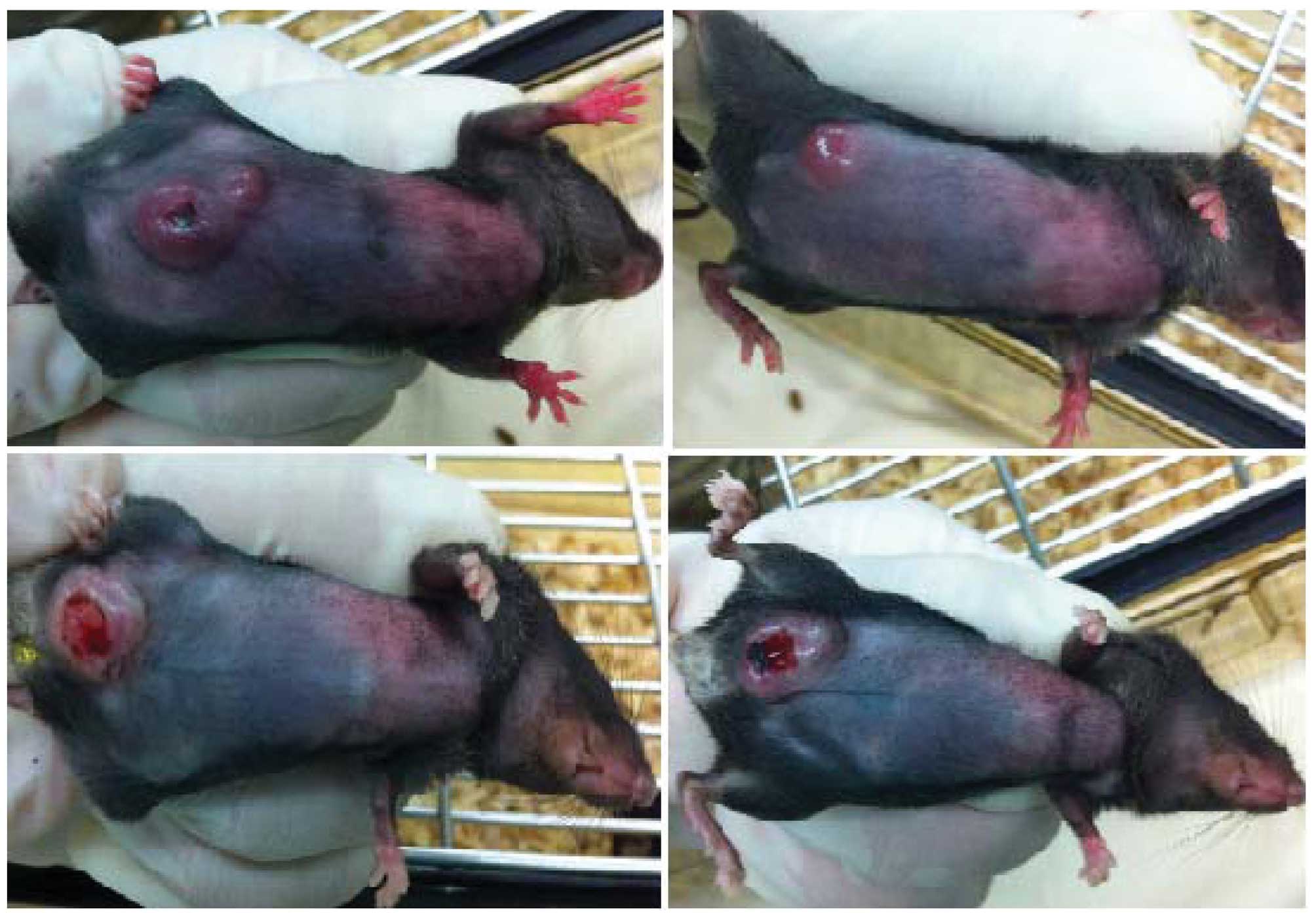
Tumor observation
Recipients (12/12) of the three transposons and the
SB transposase developed single or coupled, nodular, subcutaneous
neoplasms ~30 days after injection (Fig. 2). Each tumor grew rapidly from
initial detection to eventually reach a large volume.
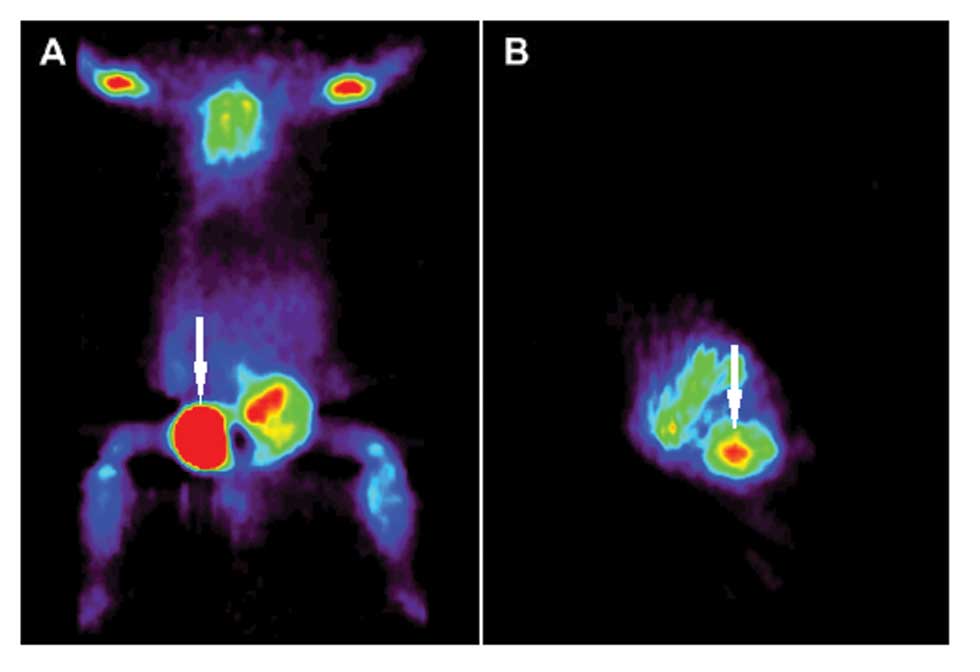
Animal PET imaging
Different dimensions, both transverse and
longitudinal, of PET-CT images for subcutaneous neoplasms were
detected on the injection sites. The tumors are observed as a
red-pinkish-colored small ball (Fig.
3).
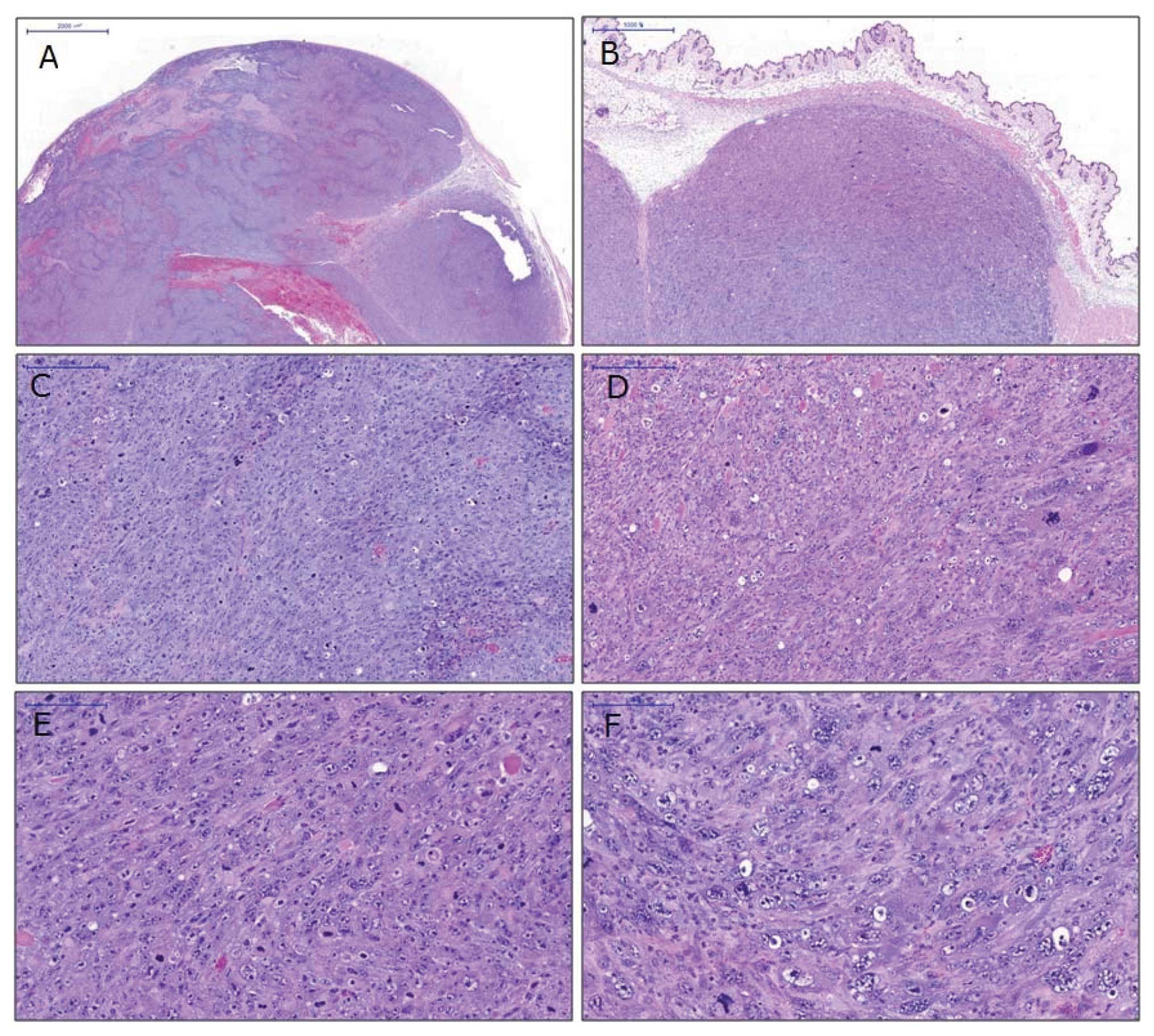
Gross and microscopic findings
A well-demarcated, ovoid nodule was located in the
subcutaneous soft tissue where the DNA plasmids were injected.
Metastatic foci in non-injected sites were not observed. The tumor
showed high cellularity with abundant mitoses and apoptosis. Both
epithelial and mesenchymal components were evident. The tumor cells
were undifferentiated with pleomorphic features characterized by
round to oval cells with pale basophilic cytoplasm and
hyperchromatic nuclei with prominent nucleoli (Fig. 4). Multinucleated giant cells were
occasionally identified. Areas of necrosis were also evident in the
central part of the tumor. However, the overlying skin and its
associated adnexa, including hair follicles, sebaceous glands and
mammary glands, did not exhibit dysplastic changes. All the tumors
had the same morphological features.
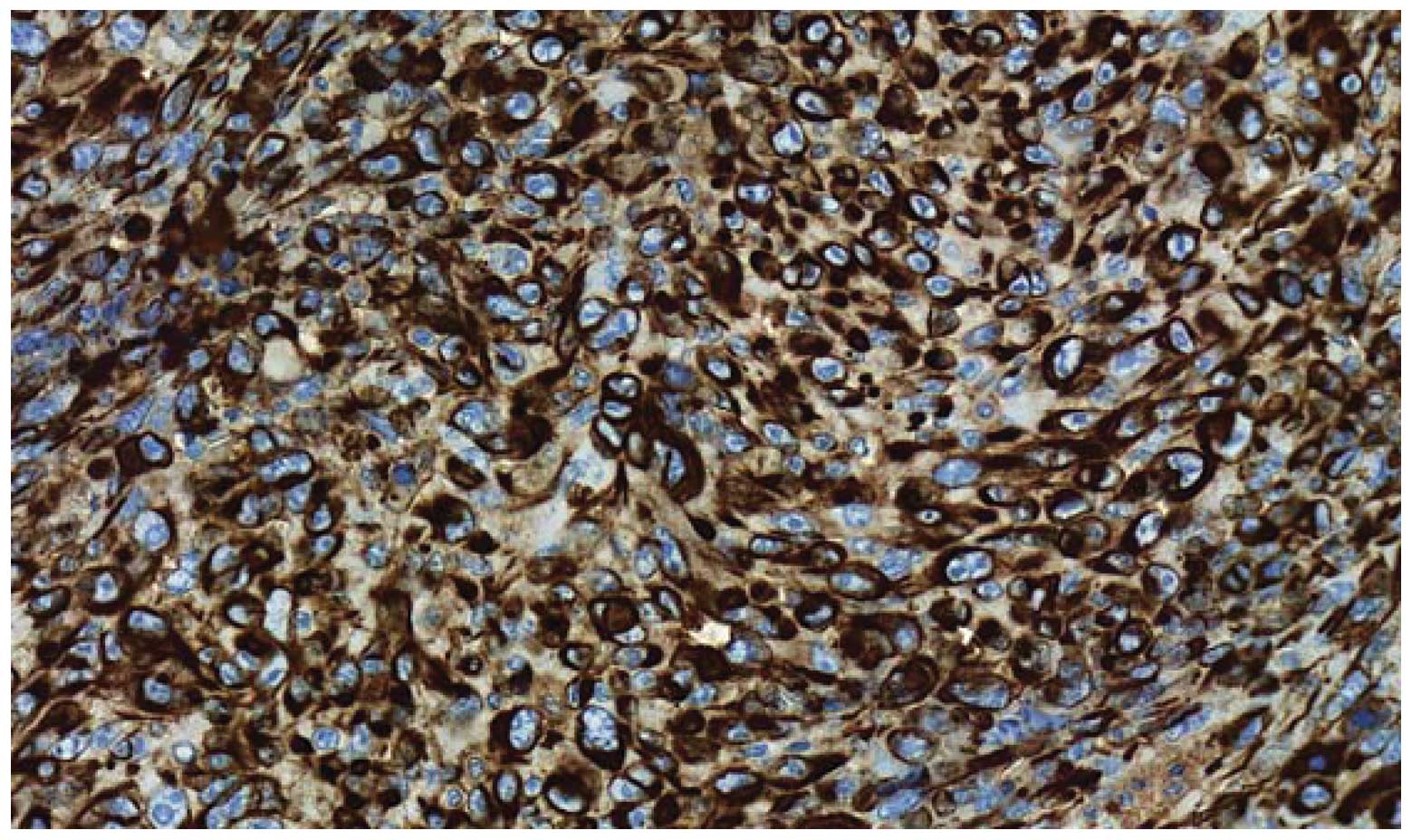
Immunohistochemical findings
As summarized in Table
II, only pan-cytokeratin was found to be positively expressed
by the tumor cells (Fig. 5),
suggesting the epithelial origin of the tumor. The other markers
were completely negative, except in the tumor-associated stroma.
Consequently, we diagnosed this tumor as a sarcomatoid
carcinoma.
 | Table IIImmunohistochemical profile. |
Table II
Immunohistochemical profile.
| Tissue of marker | IHC antibody | Reactivity |
|---|
| Epithelial tissue
marker | Pan-cytokeratin | + |
| Cytokeratin 7 | − |
| Cytokeratin 20 | − |
| Muscular tissue
marker | Myogenin | − |
| Desmin | − |
| α-SMA | − |
| Hematopoietic cell
marker | CD45 | − |
| CD163 | − |
| CD68 | − |
| Melnoma marker | Melanoma | − |
| S100 | − |
| Adipose tiusse
marker | CDK4 | − |
| MDM2 | − |
| Adenocarinoma
marker | CEA | − |
Identification of gene expression in the
tumor
Expression of the c-Myc and HRAS oncogenes was
demonstrated by immunohistochemical positivity for the c-Myc and
HRAS antibodies, respectively. A positive signal was detected with
an anti-GFP antibody, indicating expression of the shp53 DNA.
Discussion
In the present study, we induced tumors in mice via
the injection of transposons encoding three oncogenes and a
plasmid-expressing transposase. The histological features of the
induced tumor were poorly differentiated, thus we used
immunohistochemical markers to characterize this lesion.
Multinucleated giant cells were occasionally identified. Areas of
necrosis were also identified in the central part of the tumor.
Notably, the tumor expressed only pan-cytokeratin and all other
markers used in this study were completely negative. As a result,
we diagnosed the tumor as a sarcomatoid carcinoma.
To the best of our knowledge, there are no
spontaneously occurring subcutis malignant epithelial tumors that
morphologically resemble the tumor induced in this mouse study. The
origin of this tumor remains unknown as the overlying epidermis and
associated adnexa were not histologically dysplastic. Mammary
tissues were also intact and normal mammary tissues were embedded
within the tumor. This finding is consistent with the fact that
mammary tissues lack response to the SB transposon system. Although
this system has recently been shown to be able to induce various
types of SB transposon-induced tumors, certain types of tumors such
as those of the lung, mammary gland, prostate and pancreas, have
not yet been generated with the SB transposon system in the mouse
(10).
As the oncogenes were injected into the subcutis
area, the origin of the tumor is assumed to be the skin adnexa,
although this could not be precisely defined. c-Myc overexpression
is associated with an undifferentiated phenotype in cultured
astrocytes (11) and gastric
carcinomas (12). In addition,
c-Myc is downregulated during myogenic differentiation (13,14).
HRAS overexpression is also associated with an anaplastic phenotype
in mammary adenocarcinomas (15).
Therefore, we hypothesize that injection of these oncogenes induced
the growth of tumors with an undifferentiated phenotype.
The tumors induced in this study appeared to be of
mesenchymal origin morphologically. Features of the tumors
including undifferentiated pleomorphic cells characterized by
hyperchromatic nuclei with prominent nucleoli and basophilic
spindle-shaped cytoplasm resembled sarcomatous tumor. However,
results of immunohistochemical staining showed this tumor to be
carcinoma. p53 gene knockout is involved in presenting sarcomatoid
features, as recent studies showed that tumor-suppressor gene
mutations within the stroma allowed mesenchymal proliferation
(16).
Insertional mutagenesis is commonly used in gene
discovery studies in the field of oncology. This method is easy and
more rapid compared to conventional genetic engineering (e.g., gene
knockout). As a result, previous studies have focused on the use of
the SB transposon system for insertional mutagenesis (17,18).
However, detailed histological characterization of the induced
tumors has not previously been performed. Thus, this study is, to
the best of our knowledge, the first report of the histological and
immunohistochemical identification of subcutaneous tumors induced
by transposable elements.
Acknowledgements
The authors gratefully acknowledge the Institute for
Innovative Cancer Research (IICR) for providing research funds.
References
|
1
|
Belur LR, Podetz-Pedersen KM, Sorenson BS,
Hsu AH, Parker JB, Carlson CS, Saltzman DA, Ramakrishnan S and
McIvor RS: Inhibition of angiogenesis and suppression of colorectal
cancer metastatic to the liver using the Sleeping Beauty Transposon
System. Mol Cancer. 10:142011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Howell VM: Sleeping beauty-a mouse model
for all cancers? Cancer Lett. 317:1–8. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Song JS, Kim HP and Rubin E: Development
of a Sleeping Beauty-based telomerase gene delivery system for
hepatocytes. Biosci Biotechnol Biochem. 75:227–231. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Carlson CM, Frandsen JL, Kirchhof N,
McIvor RS and Largaespada DA: Somatic integration of an
oncogene-harboring Sleeping Beauty transposon models liver tumor
development in the mouse. Proc Natl Acad Sci USA. 102:17059–17064.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kim IS and Baek SH: Mouse models for
breast cancer metastasis. Biochem Biophys Res Commun. 394:443–447.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thyagarajan T, Totey S, Danton MJ and
Kulkarni AB: Genetically altered mouse models: the good, the bad
and the ugly. Crit Rev Oral Biol Med. 14:154–174. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rivera J and Tessarollo L: Genetic
background and the dilemma of translating mouse studies to humans.
Immunity. 28:1–4. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Radiloff DR, Rinella ES and Threadgill DW:
Modeling cancer patient populations in mice: complex genetic and
environmental factors. Drug Discov Today Dis Models. 4:83–88. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Andrechek ER and Nevins JR: Mouse models
of cancers: opportunities to address heterogeneity of human cancer
and evaluate therapeutic strategies. J Mol Med. 88:1095–1100. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dupuy AJ: Transposon-based screens for
cancer gene discovery in mouse models. Semin Cancer Biol.
20:261–268. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lassman AB, Dai C, Fuller GN, Vickers AJ
and Holland EC: Overexpression of c-Myc promotes an
undifferentiated phenotype in cultured astrocytes and allows
elevated Ras and Akt signaling to induce gliomas from
GFAP-expressing cells in mice. Neuron Glia Biol. 1:157–163. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ishii HH, Gobe GC, Pan W, Yoneyama J and
Ebihara Y: Apoptosis and cell proliferation in the development of
gastric carcinomas: associations with c-myc and p53 protein
expression. J Gastroenterol Hepatol. 17:966–972. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yeilding NM, Procopio WN, Rehman MT and
Lee WM: c-myc mRNA is down-regulated during myogenic
differentiation by accelerated decay that depends on translation of
regulatory coding elements. J Biol Chem. 273:15749–15757. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Schneider MD, Perryman MB, Payne PA, Spizz
G, Roberts R and Olson EN: Autonomous expression of c-myc in BC3H1
cells partially inhibits but does not prevent myogenic
differentiation. Mol Cell Biol. 7:1973–1977. 1987.PubMed/NCBI
|
|
15
|
Aldaz CM, Gollahon LS and Chen A:
Systematic HRAS amplification in ovary-independent mammary tumors:
correlation with progressively anaplastic phenotypes. Cancer Res.
53:5339–5344. 1993.PubMed/NCBI
|
|
16
|
Hill R, Song Y, Cardiff RD and Van Dyke T:
Selective evolution of stromal mesenchyme with p53 loss in response
to epithelial tumorigenesis. Cell. 123:1001–1011. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Keng VW, Villanueva A, Chiang DY, et al: A
conditional transposon-based insertional mutagenesis screen for
genes associated with mouse hepatocellular carcinoma. Nat
Biotechnol. 27:264–274. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Goryshin IY, Jendrisak J, Hoffman LM, Meis
R and Reznikoff WS: Insertional transposon mutagenesis by
electroporation of released Tn5 transposition complexes. Nat
Biotechnol. 18:97–100. 2000. View
Article : Google Scholar : PubMed/NCBI
|