Introduction
Breast cancer is the most common malignant tumor in
women. The risk of breast cancer is influenced by multiple factors,
including family and childbearing histories, hormone levels,
obesity and diabetes mellitus. Cancer stem cells, first isolated
from acute leukemia patients in 1997 (1), may lead to treatment resistance and
the high metastatic potential of tumors. Al-Hajj et al
(2) reported that as few as 100
cells with the expression profile
CD44+CD24−/low/Lin−, now known to
be indicative of the stem cell phenotype, were able to form tumors
in severely immunodeficient nude mice and to give rise to
tumorigenic breast cells. Stem cells are characterized by high
chemotherapy and radiotherapy resistance, tolerance to hypoxia, and
high oncogenicity, invasiveness and metastatic potential. These
characteristics greatly enhance the risk for local recurrence and
distant metastasis. Therefore, the destruction of cancer stem cells
is critical for effective long-term cancer treatment.
Triptolide is extracted from the root of
Tripterygium wilfordii. It has been reported to have various
pharmacological effects such as anti-inflammation, anti-oxidant and
anti-angiogenesis (3). In
particular, the antitumor activity of triptolide has attracted the
most interest (4). It was found to
effectively induce prostate and bile duct cancer cell apoptosis,
and to inhibit the growth and metastasis of these tumor cells
(5). It is also the most effective
chemotherapeutic drug for monocytic and myelocytic leukemia,
increasing the remission rates to 71 and 87%, respectively
(6). This antitumor effect was
found to be associated with the downregulation of cyclin and
anti-apoptosis genes and with the upregulation of pro-apoptosis
genes (7). In addition, triptolide
was found to downregulate expression of the transcription factor
NF-κB in multiple myeloma (8).
However, the observations were mainly carried out in tumor cell
lines and primary tumor cells, while the effects of triptolide on
tumor stem cells remain largely unknown. In this study, we
investigated the cytotoxicity of triptolide in both human primary
breast cancer cells (BCCs) and breast cancer stem cells (BCSCs) in
order to explore the clinical potential of triptolide for breast
cancer treatment.
Materials and methods
Major reagents and devices
The following reagents and devices were used: fetal
bovine serum (FBS), DMEM-F12, DMEM, B27, trypsin, collagenase I,
streptomycin + benzylpenicillin (all from Gibco-BRL, Gaithersburg,
MD, USA); oct4 and sox2 antibodies (Santa Cruz Biotechnology, Santa
Cruz, CA, USA); CD24-PE and CD44-FITC monoclonal antibodies
(Beckman Coulter, Brea, CA, USA); reverse transcription kits
(Toyobo, Osaka, Japan); alkaline fibroblastic growth factor (bFGF)
and epidermal growth factor (EGF) (Peprotech, Rocky Hill, NJ, USA);
human and bovine insulin as well as hyaluronidase (all from Sigma,
St. Louis, MO, USA); 99.7% triptolide (Pharmaceutical Sciences, Sun
Yat-sen University, Guangzhou, China); apoptosis detection kits
(BD, USA), CCK-8 cell viability kits (Dojindo Laboratories,
Kumamoto, Japan); a flow cytometer (BD, USA); a constant
CO2 cell culture incubator (Thermo Fisher Scientific,
USA); an inverted fluorescence microscope (Nikon TE 2000-U; Nikon,
Tokyo, Japan) and a Coulter counting plate.
Isolation and cultivation of tumorigenic
breast cancer stem cells
Tumorigenic breast cancer cells were isolated from
the invasive ductal carcinoma samples, which have been described in
the literature (9). Briefly, cell
suspensions were cultured at a density of 1×105/ml in
stem cell culture medium DMEM-F12 containing 2×10−5 g/l
EGF, 5×10−3 g/l insulin, 1×10−5 g/l bFGF and
B27 at 37°C under a 5% CO2 atmosphere with saturated
humidity. The medium was exchanged every 2 to 3 days. After 7 to 9
days, mammospheres of ~100 μm in diameter were observed. The
mammospheres were digested with trypsin solution containing 0.05%
EDTA for 5 to 10 min and filtered through a 400-mesh screen. The
resulting cell suspension was cultured in stem cell culture medium,
and the cell phenotype was characterized as described below.
Cultivation of primary breast cancer
cells
Mammospheres were digested as described and the
suspension was cultured in DMEM with 5% FBS at 37°C under a 5%
CO2 atmosphere with saturated humidity. The culture
medium was exchanged every 2 to 3 days. Cells in the logarithmic
growth phase were digested with 0.25% trypsin and collected for
experiments.
Identification of breast cancer stem
cells
Detection of CD44 and CD24
expression
Mammospheres (cultured for 7 to 9 days) were
digested to a single-cell suspension and then resuspended in PBS
containing 2.5% FBS. Both CD44-FITC and CD24-PE antibodies were
added at 20 μl per 1×106 cells, and the suspension was
incubated for 30 min on ice under darkness. The labeled cells were
rinsed with PBS containing 2.5% FBS and fixed in PBS containing 10
mg/ml paraformaldehyde. Fixed and stained cells were examined for
CD44/24 expression by flow cytometry. Control cultures were
processed identically except for the addition of the
antibodies.
Detection of sox2, oct4 and nanog
expression
Levels of nanog, sox2 and oct4 mRNA and proteins in
the mammospheres were measured by immunofluorescence and
semi-quantitative RT-PCR. The primer sequences of nanog, sox2, oct4
and β-actin are as follows: nanog forward, 5′-GTC
TTCTGCTGAGATGC-3′ and reverse, 5′-AGTTGTTTT TCTGCCACC-3′;
sox2 forward, 5′-TGCAGTACAACTCCA TGACCA-3′ and reverse,
5′-GTGCTGGGACATGTGAAG TCT-3′; oct4 forward,
5′-CTCCTGAAGCAGAAGAGGAT CAC-3′ and reverse,
5′-CTTCTGGCGCCGGTTACAGAA CCA-3′; β-actin forward,
5′-CATGTACGTTGCTATCCA GGC-3′ and reverse,
5′-CTCCTTAATGTCACGCACGAT-3′.
Cytotoxicity assays
CCK-8 cell viability assay
Single-cell suspensions from the digested
mammospheres were cultured for one day on 96-well plates at 37°C
under 5% CO2 and saturated humidity. The culture medium
was replaced with the same medium containing 0.1, 0.5 or 1 μM
triptolide and treated for 24, 48 and 72 h. Colorimetric assays
were performed 3 h after CCK-8 was added. Control cells were
treated with DMSO (vehicle) instead of triptolide. The OD value was
measured, and the percentage of dead cells was calculated using the
following formula: 100% × (OD of the control group - OD of the
experimental group)/OD of the control group. The experiment was
repeated 3 times using three independent cultures.
Apoptosis assay
Cells cultured on 96-well plates were treated with
0.1, 0.5 or 1 μM triptolide (or vehicle) and assayed after 24, 48
and 72 h. Adherent cells were washed twice with PBS prior to and
following digestion with 0.25% trypsin. Then, 100 μl binding buffer
and 5 μl FITC-labeled Annexin V were added at room temperature
under darkness. Thirty minutes later, 5 μl PI was added (50 μg/ml).
After 5 min, 400 μl binding buffer was added. The proportion of
apoptotic cells was assayed by flow cytometry. For cells in
suspension, the suspended microsphere cells digested to cell
suspension were labeled as described above; except that 10 μl
FITC-labeled Annexin V was added.
Animal studies
Mammosphere cells (1×105) were implanted
in the left flank of 5-week-old female BALB/c nude mice. Cells
treated with triptolide (concentration 1 μM) before implantation
did not form tumors (n=10; followed up until day 70). When the
tumors reached ~5 mm in maximal diameter, triptolide (1 μM) or PBS
(mock) was intravenously injected into the nude mice (day 0),
followed by repeated injections on days 3, 7, 10 and 14. Tumor
volumes were measured twice a week using an external caliper, and
the tumor volume (V) was calculated as follows: V = length × width
× height.
All animal procedures were approved by the Animal
Care and Use Committee of Sun Yat-Sen University.
Statistical analyses
The statistical program SPSS 13.0 was used for all
analyses. The proportion of apoptotic cells was compared between
the vehicle- and triptolide-treated groups using the Chi-square
test. A P<0.05 was considered to indicate a statistically
significant difference.
Results
Cultivation and isolation of primary
breast cancer cells and breast cancer stem cells
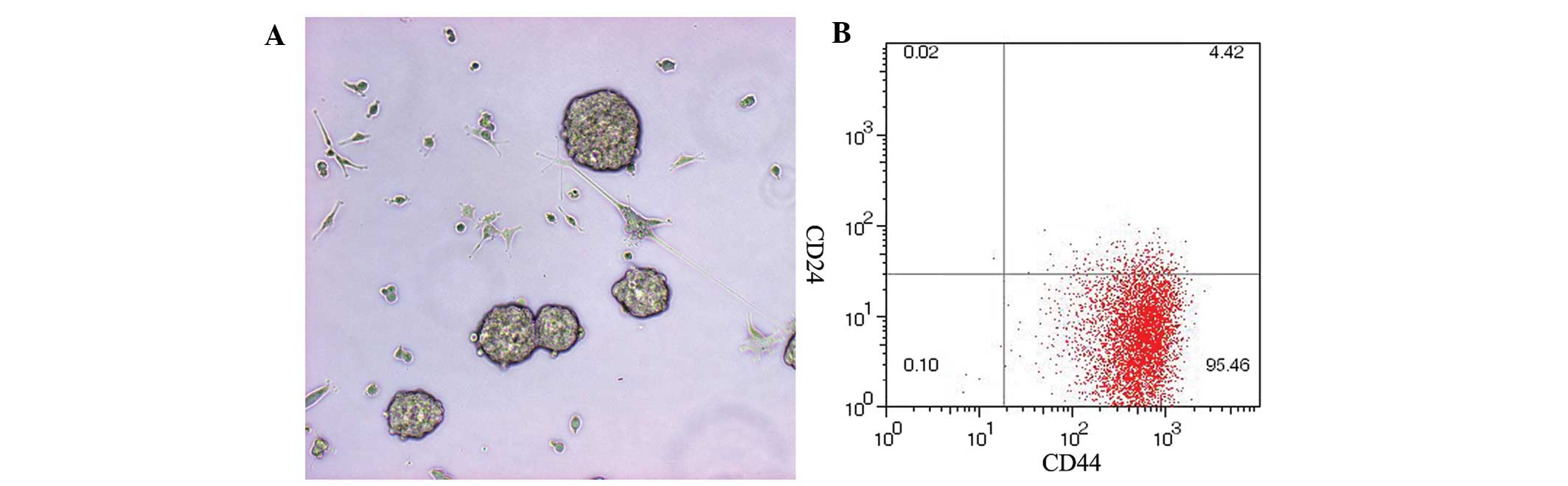
BCCs isolated from invasive ductal carcinoma samples
formed mammospheres after 7 to 9 days in a serum-free stem cell
selective medium (Fig. 1A).
Mammospheres were digested with 2.5% trypsin to obtain a
single-cell suspension for phenotype analysis. Flow cytometry
revealed that the majority of mammosphere cells (95±0.5%) were
CD44+CD24−/low (Fig. 1B), consistent with a stem cell
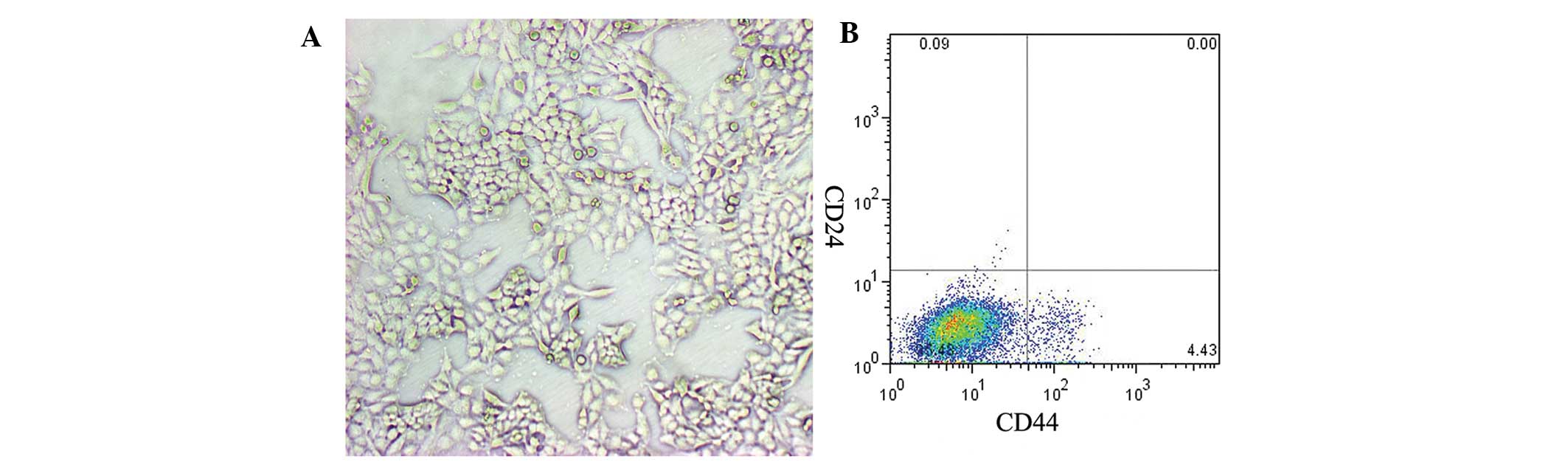
phenotype. After mammospheres were cultured in DMEM + 5% FBS
(Fig. 2A); however, surviving cells
adhered to the substrate and the proportion of
CD44+CD24−/low cells decreased from 98 to 5%
(Fig. 2B), suggesting
differentiation of BCSCs into primary BCCs. Mammospheres also
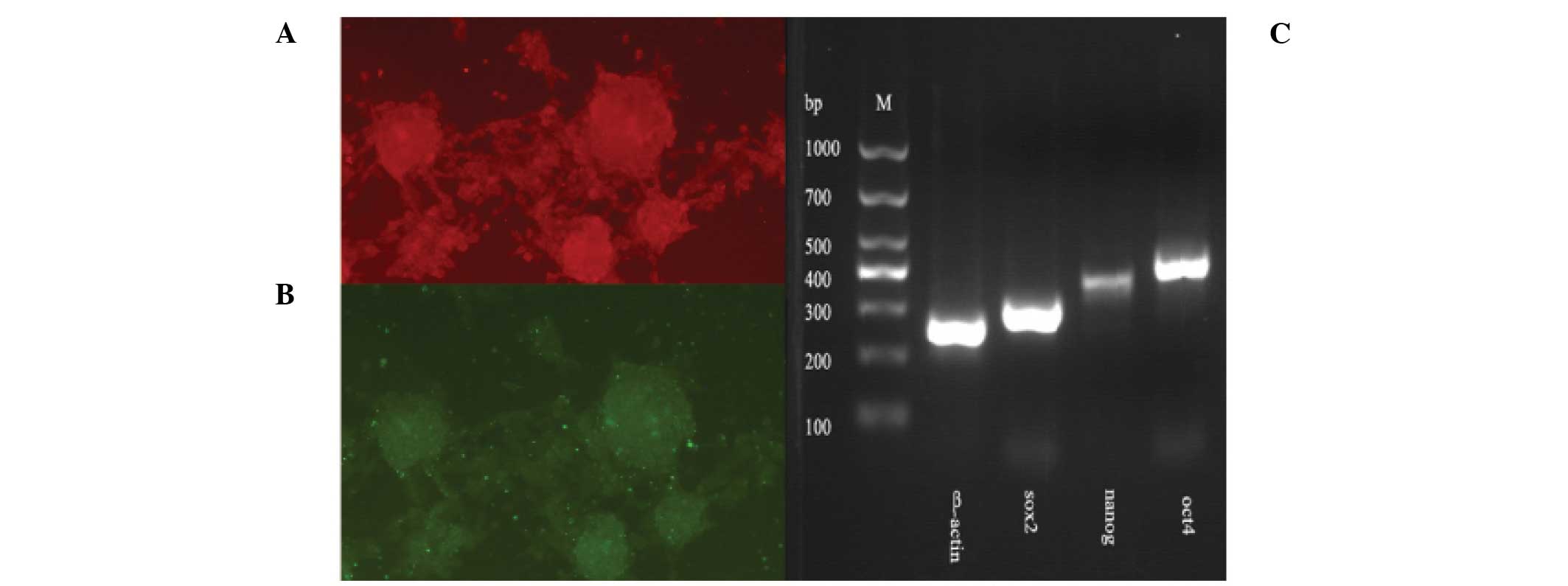
expressed the stem cell markers sox2, oct4 and nanog at both the
protein and mRNA levels (Fig. 3),
confirming that the mammospheres contained BCSCs.
Cytotoxic effect of triptolide on human
primary breast cancer cells and breast cancer stem cells
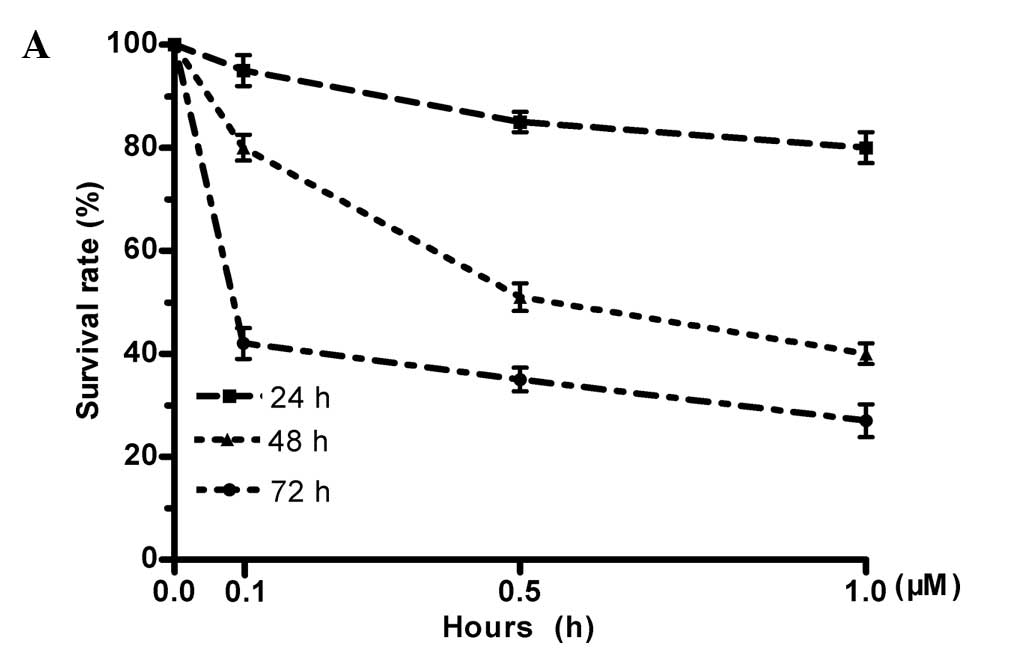
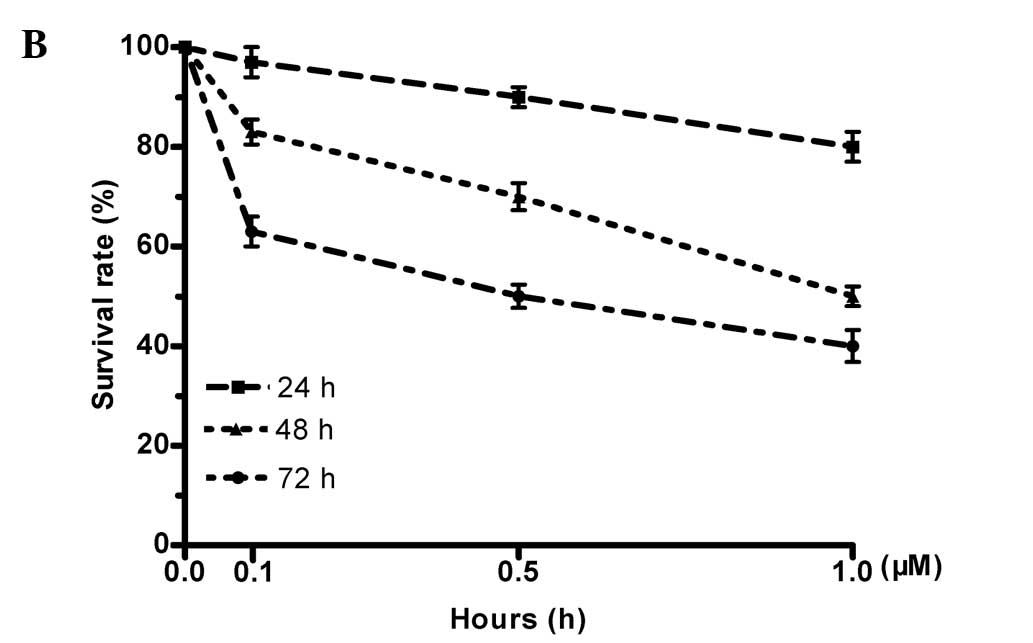
Triptolide was cytotoxic to both primary BCCs and
BCSCs at 0.1, 0.5 and 1 μM. Cell death increased with incubation
time at all concentrations tested in both cancer cell types
(Figs. 4 and 5). After 72 h, the death rate of the BCCs
relative to vehicle-treated controls was 33.1±2.7% at 0.1 μM,
49.3±3.5% at 0.5 μM and 65.6±5.2% at 1 μM of triptolide. Triptolide
was significantly less toxic against BCSCs; however, the percentage
of death relative to vehicle-treated BCSCs was 15.5±2.3% at 0.1 μM,
22.8±3.7% at 0.5 μM and 37.9±3.9% at 1 μM triptolide after 72 h
(Fig. 5).
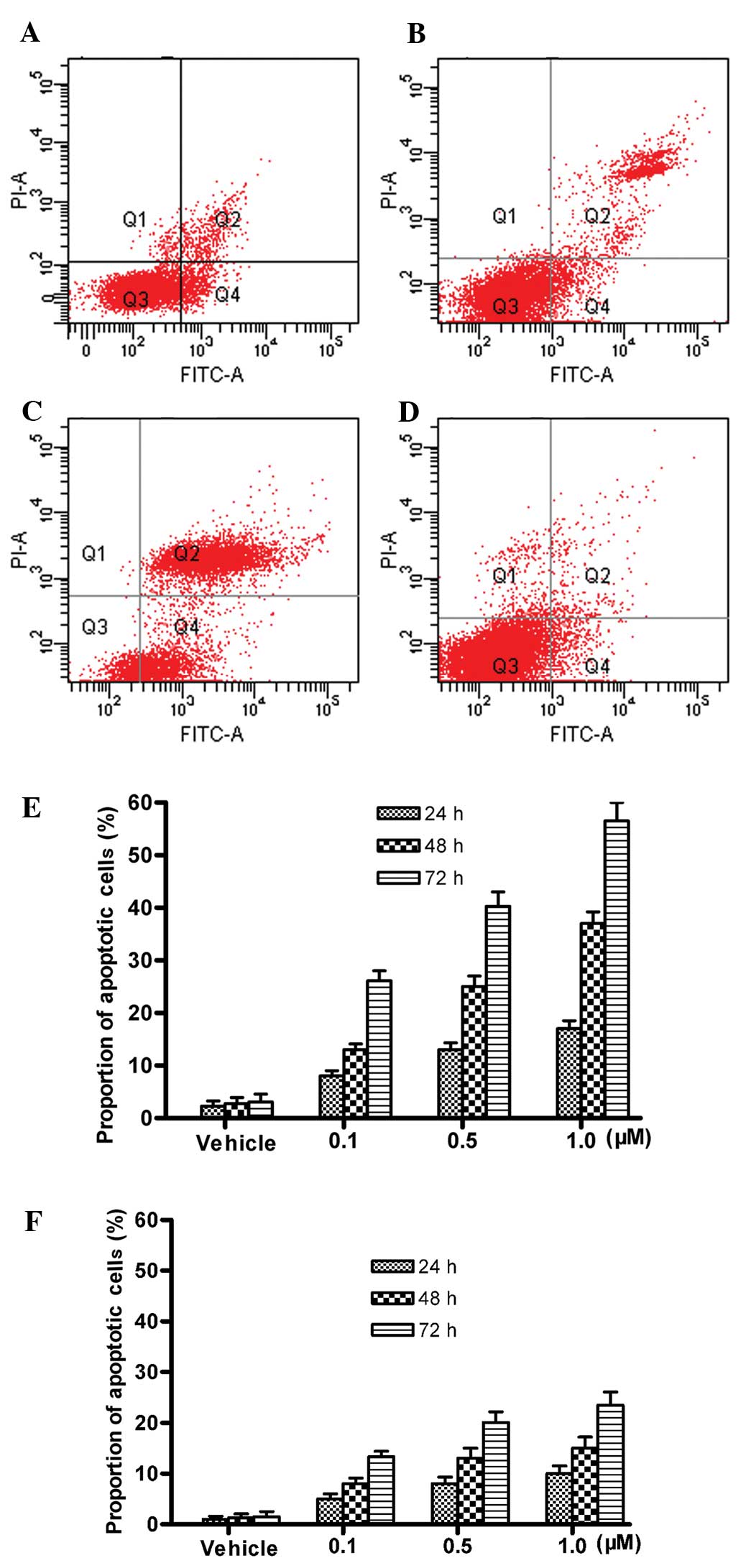
Induction of apoptosis by triptolide
Consistent with results from the CCK-8 measurement,
the proportion of apoptotic cells gradually increased over time and
with increasing concentrations of triptolide in both primary BCCs
and BCSCs. After 72 h, 26.1±1.9% of BCCs were apoptotic at 0.1 μM,
40.3±2.7% at 0.5 μM and 56.5±4.6% at 1 μM triptolide compared to
only 3.1%±1.5% of BCCs treated with vehicle (P<0.05) (Fig. 6). The proportion of early apoptotic
BCCs was 13.3±1.1% at 0.1 μM, 20.1±2.1% at 0.5 μM and 23.5±2.6% at
1 μM triptolide compared to only 1.5±1.0% of BCCS treated with
vehicle (P<0.05) (Fig. 6F). Also
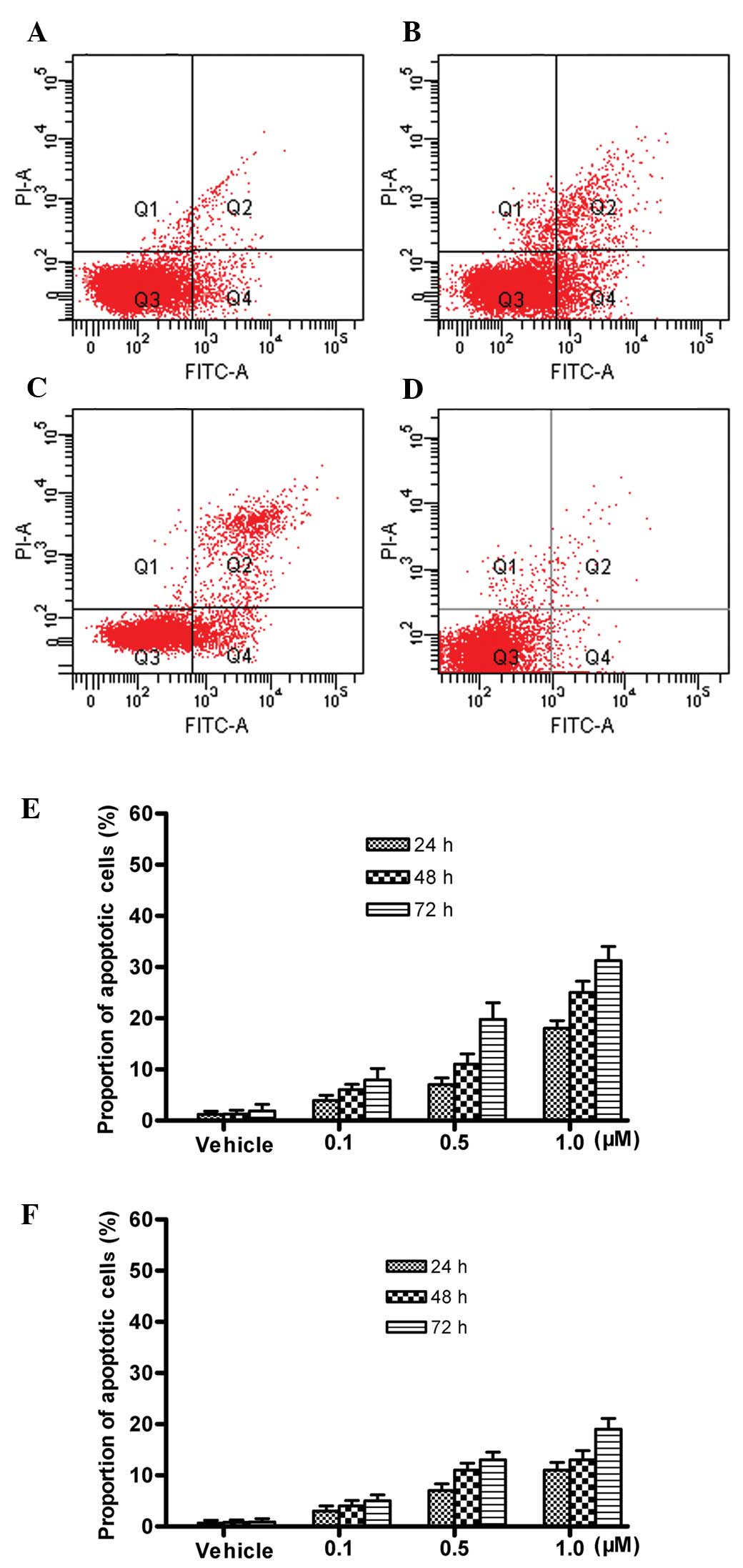
consistent with the lower cytotoxicity of triptolide against BCSCs,
the percentage of apoptosis was 7.9±2.3% at 0.1 μM, 19.8±3.2% at
0.5 μM and 31.3±2.7% at 1 μM triptolide, significantly lower than
that in the BCCs (P<0.05), but still significantly higher than
that in the vehicle-treated BCSCs (1.9±1.3%) (P<0.05) (Fig. 7); the proportion of early apoptotic
BCCs was 5±1.2% at 0.1 μM, 13±1.5% at 0.5 μM and 19±2.1% at 1 μM
triptolide compared to only 0.9±0.6% of BCSCs treated with vehicle
(P<0.05) (Fig. 7F)
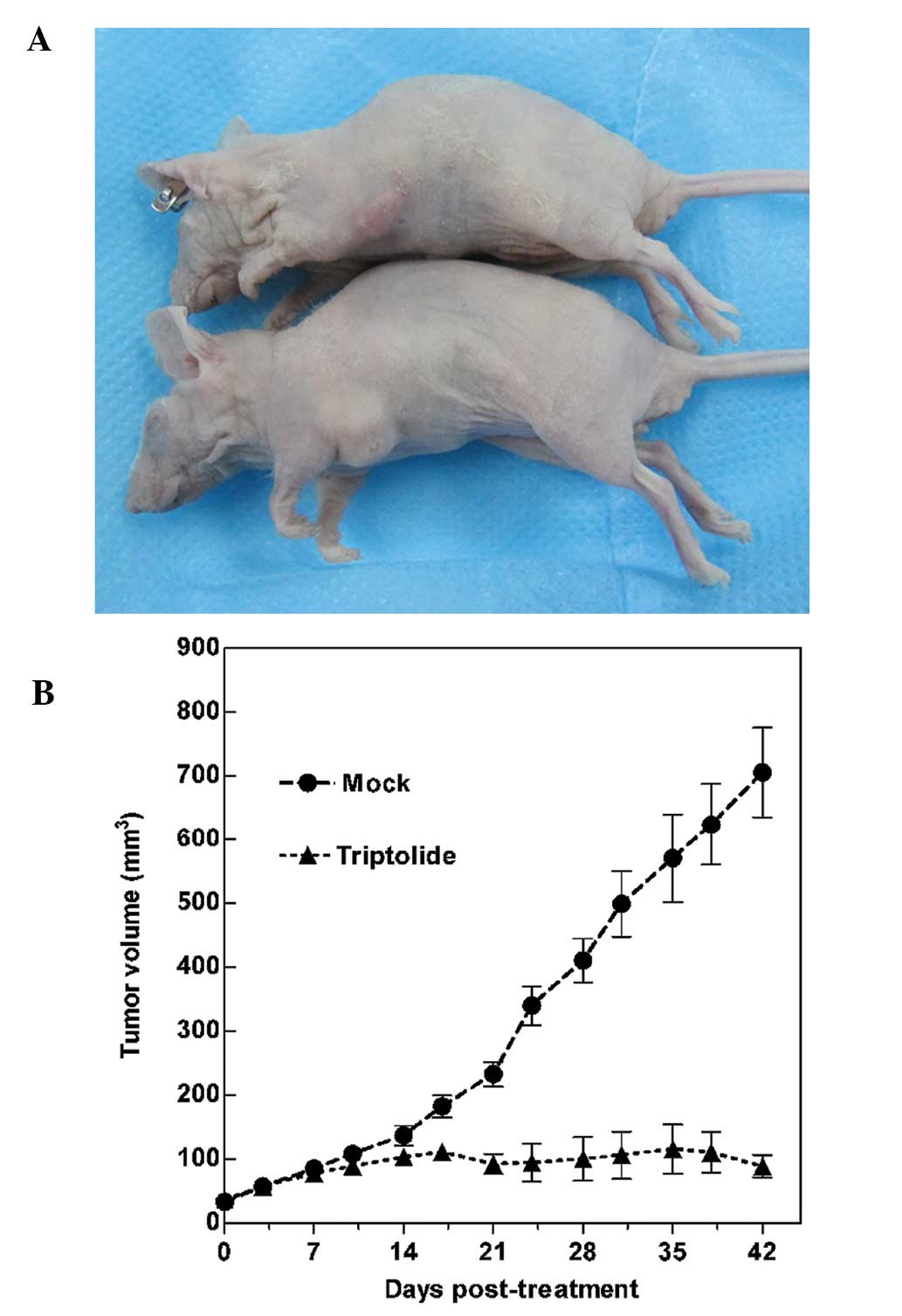
Treatment of tumors in vivo
Mammospheres were used to form tumors when injected
into the mammary fat pad of 5-week-old female nude BALB/c mice.
Cells incubated with triptolide at 1 μM before implantation did not
form tumors (n=15; observation until day 70). To assess the
antitumor efficacy of triptolide, when the tumors were ~5 mm in
maximal diameter, triptolide (1 μM) or PBS (mock) was intravenously
injected into nude mice on days 0, 3, 7, 10 and 14. After 15 days,
the tumors began to have a trend of reduced volume. Triptolide
treatment significantly inhibited tumor growth (P<0.05; Fig. 8).
Discussion
There is still no standard therapeutic regimen to
prevent breast cancer metastasis, the major cause of death in
breast cancer patients (10–14).
It is generally believed that tumor stem cells promote tumor
metastasis in addition to increasing the recurrence risk and
treatment resistance. There is now substantial evidence for the
presence of stem cells in many tumor types, although definitive
identification of these cells is still difficult. Current studies
on tumor origins suggest that tumor stem cells are derived from
native tissue stem cells or progenitor cells. If this cancer stem
cell population does indeed facilitate metastasis, then targeted
therapies may show substantial antitumor effects.
Only a minority of breast cancer cells have
metastatic potency. In 2003, Al-Hajj et al (2) demonstrated that as few as 100
CD44+CD24−/low/Lin− cells were
able to form tumors in immunodeficient nude mice. A subsequent
series of studies indicated that tumorigenic breast cancer cells
are CD44+CD24−/low (15,16),
consistent with other stem cells. At present, mammosphere
cultivation, cell separation and selection culture and the aldehyde
dehydrogenase 1 (ALDH1) assay are the most commonly used methods to
identify stem cells. In this study, a special serum-free stem cell
culture medium was used for suspension cultivation of mammospheres
to select stem cells. The stem cell phenotype was then confirmed by
surface antigen expression (CD44+CD24−/low).
In addition, these mammospheres expressed three transcription
factors, sox2, nanog and oct4, characteristic of many pluripotent
cells. After stem cells were cultured in DMEM with serum, the
proportion of CD44+CD24−/low cells decreased
significantly and cells took on the morphological appearance of
primary tumor cells, illustrating that stem cells can differentiate
into cancer cells under appropriate conditions.
There is no effective treatment for preventing the
genesis, development, recurrence, or metastasis of tumors (17,18).
To explore new treatment options, we examined the cytotoxicity of
triptolide, a diterpenoid triepoxide extracted from the Chinese
herb “common Threewingnut root” known to have anti-inflammatory,
immunosuppressant and antitumor effects (19). Triptolide can inhibit tumor growth
and metastasis by inducing apoptosis and inhibiting
neovascularization (20). Recently,
triptolide was demonstrated to be a wide-spectrum tumor inhibitor
with significant toxic effects on approximately 60 tumor types,
including breast, gastric, epidermoid, bile duct, lung, pancreatic
and bladder cancers, as well as granulocytic leukemia and melanoma.
Triptolide induces apoptosis by activating the caspase pathway,
inducing p53 expression and activating MAPK signaling (21,22).
Liu et al (3) found that
triptolide upregulates wild-type p53 expression and inhibits mutant
p53 expression. Triptolide does have side-effects, including
reduced appetite, anorexia, vomiting, abdominal pain, diarrhea,
esophageal irritation and leucopenia. However, these side-effects
are generally reversible and dose-dependent.
In the present study, breast cancer stem cells
extracted from human breast cancer tissue exhibited a significant
dose- and time-dependent sensitivity to the cytotoxic actions of
triptolide. After 72 h, 1 μM triptolide destroyed 65.6±5.2% of
primary BCCs and 37.9±3.9% of BCSCs, while 56.5±4.6% of BCCs and
31.3±2.7% of BCSCs were undergoing apoptosis as indicated by
Annexin V and PI staining. The proportion of dead cells was very
similar to the proportion of apoptotic cells for both cancer cell
types, suggesting that triptolide destroys tumor cells by inducing
apoptosis. However, the cytotoxicity of triptolide was
significantly weaker against BCSCs, consistent with the generally
greater chemoresistance of stem cells compared to primary tumor
cells. Nonetheless, this study confirms the substantial
cytotoxicity of triptolide on breast cancer stem cells. Similarly,
we demonstrated that in vivo triptolide was effective in
controlling tumor growth.
In conclusion, triptolide can effectively kill
primary breast cancer cells and breast cancer stem cells, at least
in part by inducing apoptosis. This study provides a basis for
further animal experiments investigating the therapeutic potential
of triptolide and its derivatives against breast and other forms of
cancer.
References
|
1
|
Bonnet D and Dick JE: Human acute myeloid
leukemia is organized as a hierarchy that originates from a
primitive hematopoietic cell. Nat Med. 3:730–737. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Al-Hajj M, Wicha MS, Benito-Hernandez A,
Morrison SJ and Clarke MF: Prospective identification of
tumorigenic breast cancer cells. Proc Natl Acad Sci USA.
100:3983–3988. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu J, Jiang Z, Xiao J, et al: Effects of
triptolide from Tripterygium wilfordii on ERalpha and p53
expression in two human breast cancer cell lines. Phytomedicine.
16:1006–1013. 2009.
|
|
4
|
Lu L, Kanwar J, Schmitt S, et al:
Inhibition of tumor cellular proteasome activity by triptolide
extracted from the Chinese medicinal plant ‘thunder god vine’.
Anticancer Res. 31:1–10. 2011.PubMed/NCBI
|
|
5
|
Yang S, Chen J, Guo Z, et al: Triptolide
inhibits the growth and metastasis of solid tumors. Mol Cancer
Ther. 2:65–72. 2003.PubMed/NCBI
|
|
6
|
Pigneux A, Mahon FX, Uhalde M, et al:
Triptolide cooperates with chemotherapy to induce apoptosis in
acute myeloid leukemia cells. Exp Hematol. 36:1648–1659. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhou Y, Xu Q and Huang Y: An in vitro
study of Triptolide inhibiting proliferation of vascular
endothelial cells. Practical J China. 6:564–566. 2004.
|
|
8
|
Lou YJ, Jin J and Wang YG: Triptolide
inhibits transcription factor NF-kappaB and induces apoptosis of
multiple myeloma cells. Leuk Res. 29:99–105. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Alvi AJ, Clayton H, Joshi C, et al:
Functional and molecular characterisation of mammary side
population cells. Breast Cancer Res. 5:R1–R8. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen Y and Rittling SR: Novel murine
mammary epithelial cell lines that form osteolytic bone metastases:
effect of strain background on tumor homing. Clin Exp Metastasis.
20:111–120. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Moltzahn FR, Volkmer JP, Rottke D and
Ackermann R: ‘Cancer stem cells’ - lessons from Hercules to fight
the Hydra. Urol Oncol. 26:581–589. 2008.
|
|
12
|
Liu S, Dontu G and Wicha MS: Mammary stem
cells, self-renewal pathways and carcinogenesis. Breast Cancer Res.
7:86–95. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Abraham BK, Fritz P, McClellan M,
Hauptvogel P, Athelogou M and Brauch H: Prevalence of
CD44+/CD24−/low cells in breast cancer may
not be associated with clinical outcome but may favor distant
metastasis. Clin Cancer Res. 11:1154–1159. 2005.PubMed/NCBI
|
|
14
|
Ling LJ, Wang S, Liu XA, et al: A novel
mouse model of human breast cancer stem-like cells with high
CD44+CD24−/lower phenotype metastasis to
human bone. Chin Med J. 121:1980–1986. 2008.PubMed/NCBI
|
|
15
|
Ponti D, Costa A, Zaffaroni N, et al:
Isolation and in vitro propagation of tumorigenic breast cancer
cells with stem/progenitor cell properties. Cancer Res.
65:5506–5511. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ginestier C, Hur MH, Charafe-Jauffret E,
et al: ALDH1 is a marker of normal and malignant human mammary stem
cells and a predictor of poor clinical outcome. Cell Stem Cell.
1:555–567. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fan M, Yan PS, Hartman-Frey C, et al:
Diverse gene expression and DNA methylation profiles correlate with
differential adaptation of breast cancer cells to the antiestrogens
tamoxifen and fulvestrant. Cancer Res. 66:11954–11966. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Eriksson M, Guse K, Bauerschmitz G, et al:
Oncolytic adenoviruses kill breast cancer initiating
CD44+CD24−/low cells. Mol Ther. 15:2088–2093.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qiu D and Kao PN: Immunosuppressive and
anti-inflammatory mechanisms of triptolide, the principal active
diterpenoid from the Chinese medicinal herb Tripterygium
wilfordii Hook. f. Drugs R D. 4:1–18. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang S, Chen J, Guo Z, et al: Triptolide
inhibits the growth and metastasis of solid tumors. Mol Cancer
Ther. 2:65–72. 2003.PubMed/NCBI
|
|
21
|
Frese S, Pirnia F, Miescher D, et al:
PG490-mediated sensitization of lung cancer cells to
Apo2L/TRAIL-induced apoptosis requires activation of ERK2.
Oncogene. 22:5427–5435. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang P, Zhang R and Wang Y: Research
progress on the antitumor molecular mechanism of triptolide. Acta
Chin Med Pharmacol. 3:79–82. 2009.(In Chinese).
|