Introduction
The major impact of hepatocyte growth factor (HGF)
on the development and metastasis of prostate cancer may be via its
action on prostate stem cells or their progeny. Understanding the
sequence of events in the above process would have important
implications in understanding the biology of prostate cancer and in
the development of new therapies. Stem cells constitute a small
population of relatively undifferentiated cells that express no
differentiation markers of tissue and exhibit diverse expression of
tight junction (TJ) proteins.
TJs are crucial to the process of metastasis and
have been previously shown to be regulated by HGF. TJs are the
apical cellular structure of both epithelial and endothelial cells
and play a key role in the control of paracellular passage and cell
adhesion. Previous studies have demonstrated a correlation between
a reduction in TJs and tumour differentiation, and experimental
evidence has emerged that places TJs as the frontline structures
that tumour cells must overcome in order to successfully
metastasize (1–5). Although a considerable body of
research exists on TJs and their role in a number of diseases, it
is only in the last few years that their possible role in
tumourigenesis and tumour progression has been studied. Yet, to
date, most of the research has focused on cell lines and to a
limited degree on colorectal and pancreatic cancers, with a few
studies carried out on breast cancer concentrating on claudin-1
(SEMP-1), claudin-7, ZO-1 and ZO-2 expression (5–10).
Changes in the expression of TJ proteins may be due
to regulatory mechanisms or promoter methylation. Regulatory
mechanisms may be via the suggested pathway of
epithelial-to-mesenchymal transition (EMT) as the process of
acquisition of an invasive phenotype by tumours of epithelial
origin can be regarded as a pathological version of EMT (11,12).
TJs determine epithelial cell polarity and disappear during EMT.
Snail and Slug are factors thought to be responsible for this loss
(13). Regulation also occurs via
the Rho GTPase family, which is able to regulate TJ assembly
(14). Thus, TJs can be regulated
in response to physiological and tissue-specific requirements
(15). TJs are able to rapidly
change their permeability and functional properties in response to
stimuli, permitting dynamic fluxes of ions and solutes in addition
to the passage of whole cells. HGF, a cytokine secreted by stromal
cells, was found to modulate the expression and function of TJ
molecules in human breast cancer cell lines (10). HGF decreased the transepithelial
resistance and increased the paracellular permeability of human
breast cancer cell lines, MDA-MB-231 and MCF-7. qPCR revealed that
HGF modulated the levels of several TJ molecule (occludin,
claudin-1, claudin-5, JAM-1 and JAM-2) mRNA transcripts in
MDA-MB-231 and MCF-7 cells. Western blotting and
immunohistochemistry also showed modulation of expression of the TJ
molecule, occludin. It was suggested that HGF disrupts TJ function
in human breast cancer cells by affecting changes in the expression
of TJ molecules at both the mRNA and protein levels. It has also
been shown that HGF treatment results in disassociation of MCF-7
and T47D cells in culture, and a loss of claudin-7 expression
within 24 h (6).
Few studies have reported on the role of TJs in
prostate cancer. Those studies that were carried out focused on
clinical samples and investigated the expression of claudins
(16–19) even though there was an interest in
the role of TJs in the prostate early on (20–23)
which has not been further researched.
Our present study, therefore, aimed to determine
whether HGF plays a similar role in the regulation of TJs in human
prostate cells. Cells were subjected to HGF treatment and the
distribution and expression of ZO-1, ZO-2, ZO-3, occludin,
claudin-1 and claudin-5 were determined. Any changes in junctional
integrity were also assessed. The data obtained showed that HGF was
able to regulate the function of TJs in human prostate cells by
modulating the expression of a number of TJ molecules at the
molecular and protein levels. It is anticipated that further
studies will reveal an important role of TJs in prostate cancer
cell behaviour.
Materials and methods
Reagents and antibodies
Anti-occludin (N-19 epitope, sc-8145 and H-279
epitope, sc-5562), anti-ZO-1, anti-ZO-2, anti-ZO-3, anti-claudin-1,
anti-claudin-5, anti-AF6 and anti-β-actin (sc-8432) antibodies were
purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA) and
Pharmingen International (San Diego, CA, USA). The anti-occludin
(c-terminal epitope, 71–1500) antibody was purchased from Zymed
(Invitrogen Ltd., Paisley, UK). Peroxidase-conjugated anti-mouse
and anti-rabbit IgG for western blotting were from Sigma-Aldrich
(Poole, UK). FITC- or TRITC-conjugated anti-mouse, anti-goat and
anti-rabbit IgG were from Sigma-Aldrich. FITC-conjugated dextran
(40 kDa) was obtained from Molecular Probes (Eugene, OR, USA).
Carbonate filter inserts with a pore size of 0.4 μm (for 24-well
plates) were from Greiner Bio-One Ltd. (Stonehouse, Glos, UK). Gold
electrode arrays (8W10E) were purchased from Applied Biophysics
Inc. (Troy, NY, USA).
Cell lines
Human prostate cancer cell lines PC-3, DU-145,
CAHPV-10, PZHPV-7 and LNCap [European Collection of Animal Cell
Cultures (ECACC), Wiltshire, UK] were routinely maintained in
Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich)
supplemented with 10% foetal calf serum (FCS), penicillin and
streptomycin (Sigma-Aldrich). RWPE-1, a normal adult prostate
parental epithelial cell and WPESTEM, a stem cell-like derivative
of RWPE-1 (ATCC, LGC Standards, Teddington, UK) were routinely
cultured using the keratinocyte serum-free medium (K-SFM) kit
[Invitrogen (Gibco); cat. no. 17005-042] with bovine pituitary
extract (BPE) and human recombinant epidermal growth factor
(EGF).
Transepithelial resistance (TER)
TER was measured using an EVOM Volt-Ohm meter (EVOL,
World Precision Instruments, Aston, Herts, UK), equipped with a
pair of STX-2 chopstick electrodes (WPI, Sarasota, FL, USA), as we
previously reported (10,24). Briefly, cells were seeded into the
0.4-μm pore size insert (Greiner Bio-One Ltd., Stonehouse, Glos,
UK) and allowed to reach full confluency, after which fresh medium
was replaced for further experiments. Inserts without cells,
inserts with cells in medium and inserts with cells with HGF (at
varying concentrations) were tested for a period of 2 h. Electrodes
were placed at the upper and lower chambers, and resistance was
measured with the Volt-Ohm meter.
Immunofluorescence staining of human
prostate cancer cells
For immunofluorescence staining, cells (30,000
cells/well) were grown in 16-well chamber slides (LAB-TEK
International, Sussex, UK) and incubated at 37°C in a 5% incubator
for a set period of time (0–24 h). After incubation, the culture
medium was aspirated, the wells were rinsed with balanced salt
solution (BSS) buffer and the cells were fixed in methanol for 20
min at −20°C. After fixation, the cells were washed twice using BSS
buffer and permeabilised by the addition of 200 μl of 0.1% Triton
X-100 (Sigma-Aldrich) detergent in phosphate-buffered solution
(PBS) for 5 min at room temperature. Cells were rinsed twice with
PBS buffer, and 200 μl of blocking buffer (10% horse serum in PBS)
was added to each well and the chamber slide was incubated for 40
min at room temperature on a bench rocker. The wells were washed
once with wash buffer (3% horse serum in TBS buffer containing 0.1%
Tween-20), and 100 μl of the primary antibodies prepared in wash
buffer was added to the appropriate wells. The chamber slide was
incubated on the rocker for a further 60 min at room temperature.
Wells were washed twice with TBS buffer (with 0.1% Tween-20), and
the cells were incubated in 100 μl of the secondary antibodies
[fluorescein isothiocyanate (FITC) or tetramethylrhodamine
isothiocyanate (TRITC) conjugates; Sigma-Aldrich] (diluted in the
same manner as the primary antibodies) for 50 min. The chamber
slide was wrapped in foil to prevent light from reaching the
conjugate. Finally, the wells were rinsed twice with wash buffer,
and once in BSS buffer mounted with FluorSave™
(Calbiochem-Novabiochem Ltd., Nottingham, UK) reagent and
visualised using an Olympus BX51 microscope with a Hamamatsu
(Welwyn Garden City, Herts, UK) Orca ER digital camera at a
magnification of ×100 using an oil immersion lens.
SDS-PAGE and western blotting
Total cell lysates and tissues were prepared as
follows. Cells were pelleted and lysed in HCMF buffer plus 0.5%
SDS, 0.5% Triton X-100, 2 mM CaCl2, 100 μg/ml
phenylmethylsulfonyl fluoride, 1 mg/ml leupeptin, 1 mg/ml aprotinin
and 10 mM sodium orthovanadate for 40 min. Sample buffer was added,
and the proteins were boiled at 100°C for 5 min before
clarification at 13,000 × g for 10 min. Equal amounts of protein
from each cell sample were added onto an 8% polyacrylamide gel.
Following electrophoresis, proteins were blotted onto
nitrocellulose sheets and blocked in 10% horse serum for 60 min
before probing with the specific primary antibodies, followed with
the peroxidase-conjugated secondary antibody (1:2,000). Protein
bands were visualised with the Supersignal West Dura Extended
Duration substrate chemiluminescent system (Perbio Science UK Ltd.,
Cramlington, UK) and detected using a CCD UVIprochemi system
(UVItec Ltd., Cambridge, UK).
Electric cell-substrate impedance sensing
measurements
The electric cell-substrate impedance sensing (ECIS,
Applied Biophysics, Troy, NY, USA) system was used to measure
transepithelial impedance in order to ascertain differences in
barrier function in the presence or absence of HGF as a potential
regulator of TJ function. For ECIS measurements, 104
cells were seeded into each well of an electrode array (96W10E or
8W10E). Medium (0.4 ml/well) was added, and after 24 h in culture,
confluence and viability of the cell monolayer were confirmed by
light microscopy and electrically. Impedance data were obtained
from cell-covered electrode arrays using a 1-V AC signal at 4,000
Hz.
Statistical analysis
Statistical analysis was performed by Minitab
version 13.32 (Minitab Inc., State College, PA, USA) using a
two-sample Student’s t-test.
Results
Redistribution of TJ proteins during HGF
co-culture
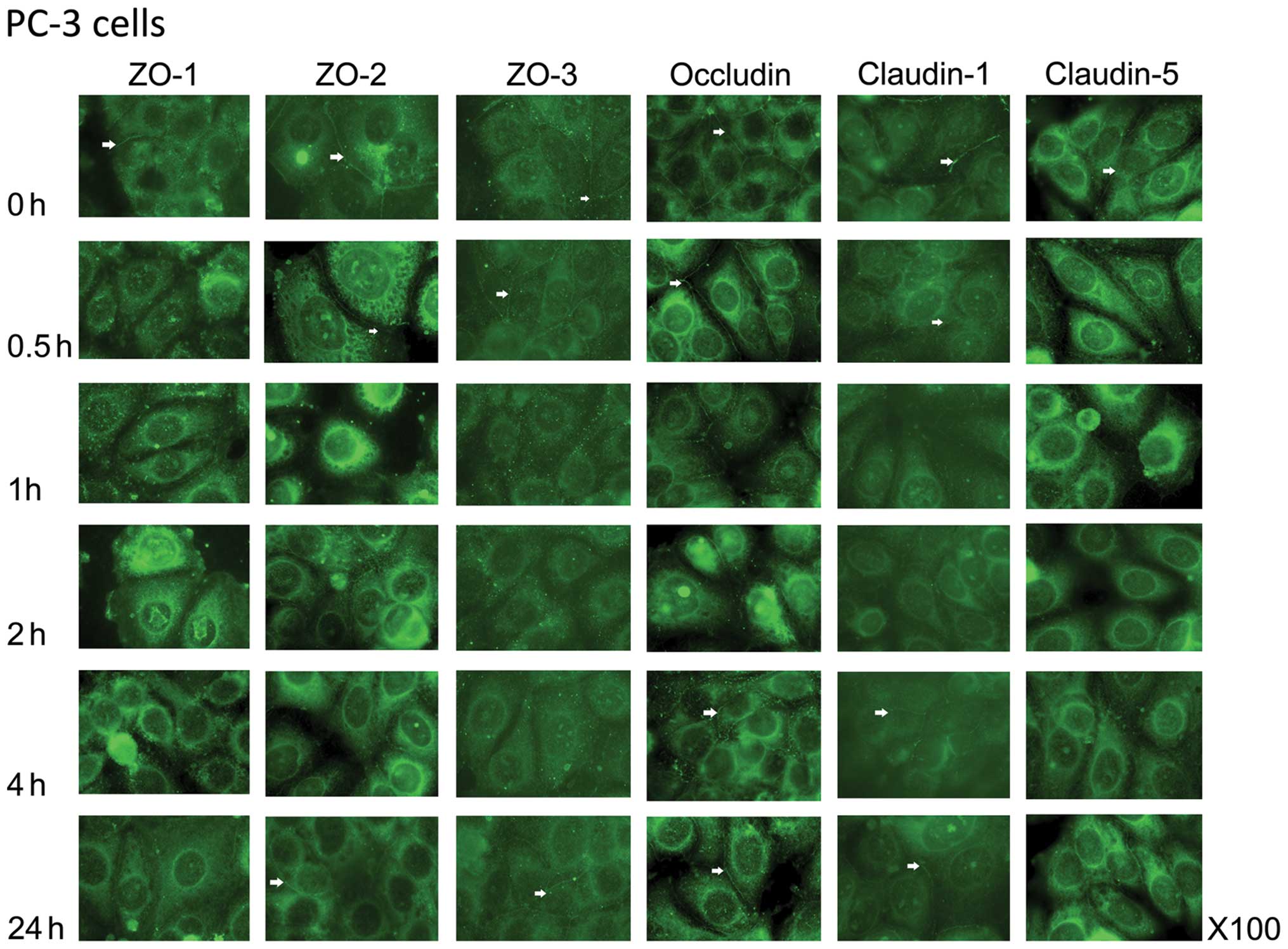
Confluent cultures of all 4 prostate cell lines
exhibited normal TJ location as visualised using
immunofluorescence. ZO-1, ZO-2 and ZO-3 were observed at the
cell-cell junctions of the cells, as were occludin and claudin-1.
For the most part, claudin-5 was found less obviously at the cell
membrane, but quite extensively in the peri-nuclear region
(Fig 1).
In the PC-3 cells, after a 0.5-h co-culture with HGF
(25 ng/ml) a reduction in the cell junction location of ZO-1 was
noted, which by 1 h had resulted in a loss of ZO-1 at the cell
periphery and an increased signal at the peri-nuclear region
(Fig. 1). This was more pronounced
at 2 and 4 h of co-culture. After a 24-h treatment, there was some
re-establishment of ZO-1 at the cell-cell junctions. There was a
similar re-distribution of ZO-2, albeit a slower reduction at the
cell-cell junction (at 1 h not 0.5 h as with ZO-1), although after
a 24-h co-culture, re-location to the cell periphery was not
complete. Concomitant with this was the change in location of ZO-3
(Fig. 1). In contrast, the location
of occludin did not show such changes after treatment with HGF.
Although the staining pattern became ‘spotty’ by 1 h of co-culture,
there was altogether, no loss of this protein from the cell
periphery. However, claudin-1 expression was lost by 0.5 h of
co-culture and did not recover after 24 h. Claudin-5 distribution
was lost by a 1-h treatment and also did not recovered by 24 h. It
was observed that claudin-5 expression was overall more intense
than claudin-1 expression.
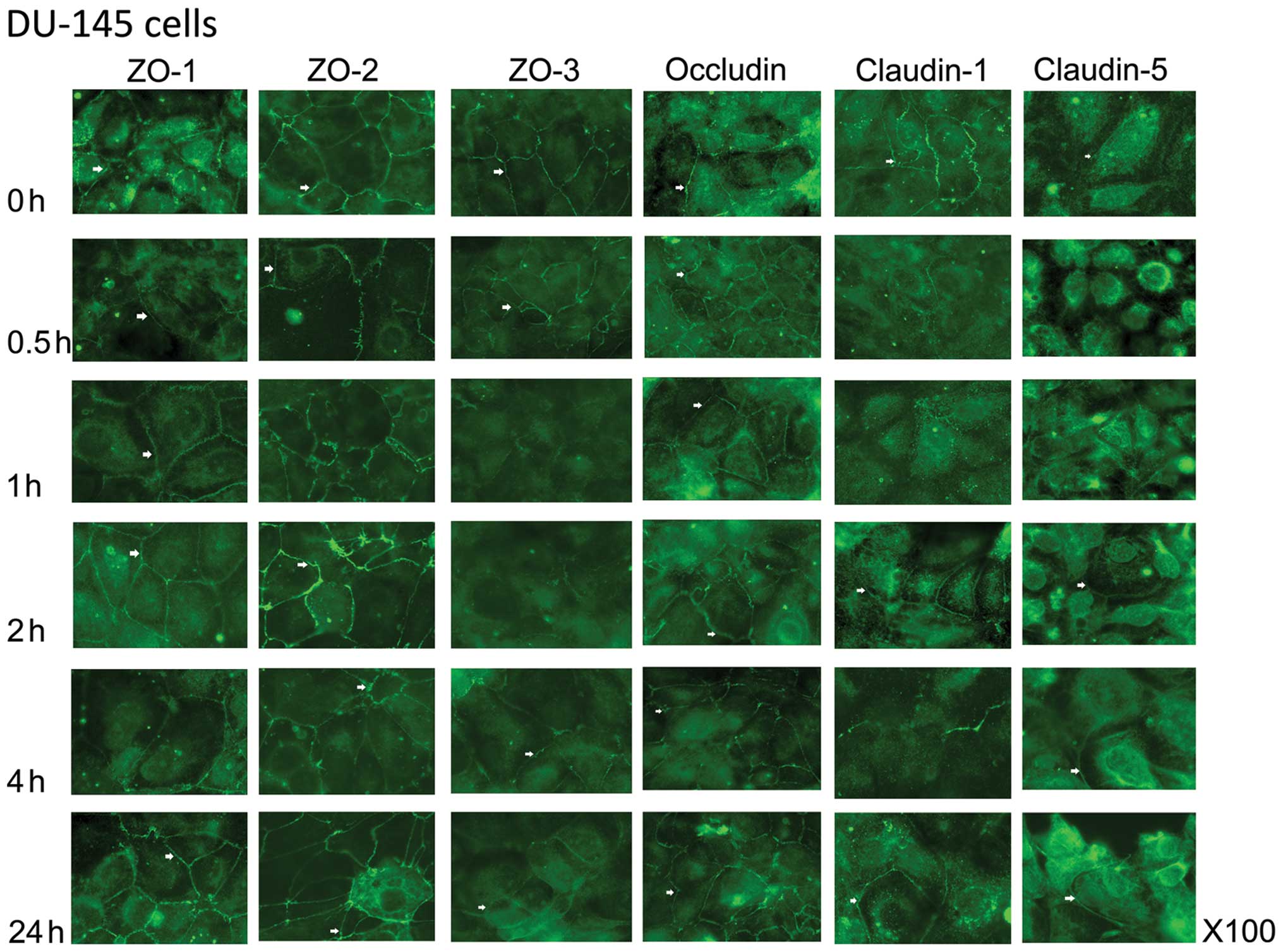
In the DU-145 cells, expression of ZO-1 was not
noticeably reduced until 4 h of co-culture with HGF and recovered
fully by 24 h (Fig. 2). A similar
pattern was also observed with ZO-2. ZO-3 expression, however, was
reduced by 1 h of co-culture with HGF and recovery to the cell
membrane was visible after 4 h of co-culture. In comparison to PC-3
cells, occludin was strongly visible at the cell periphery in
DU-145 cells and co-culture with HGF did not greatly reduce the
expression of this protein. Some spotting was noticeable by 4 h of
co-culture with HGF, but the TJ staining pattern was clearly
visible. Claudin-1 was lost from the cell junctions after only 30
min of co-culture with HGF and became spotty after 1 h in DU-145
cells. Recovery to the TJs was almost complete by 2 h of
co-culture. Claudin-5 was weakly stained in the control DU-145
cells, together with strong peri-nuclear staining. This was more
concentrated around the nucleus after just 30 min of co-culture
with HGF, but re-location to the cell periphery was apparent after
2 h.
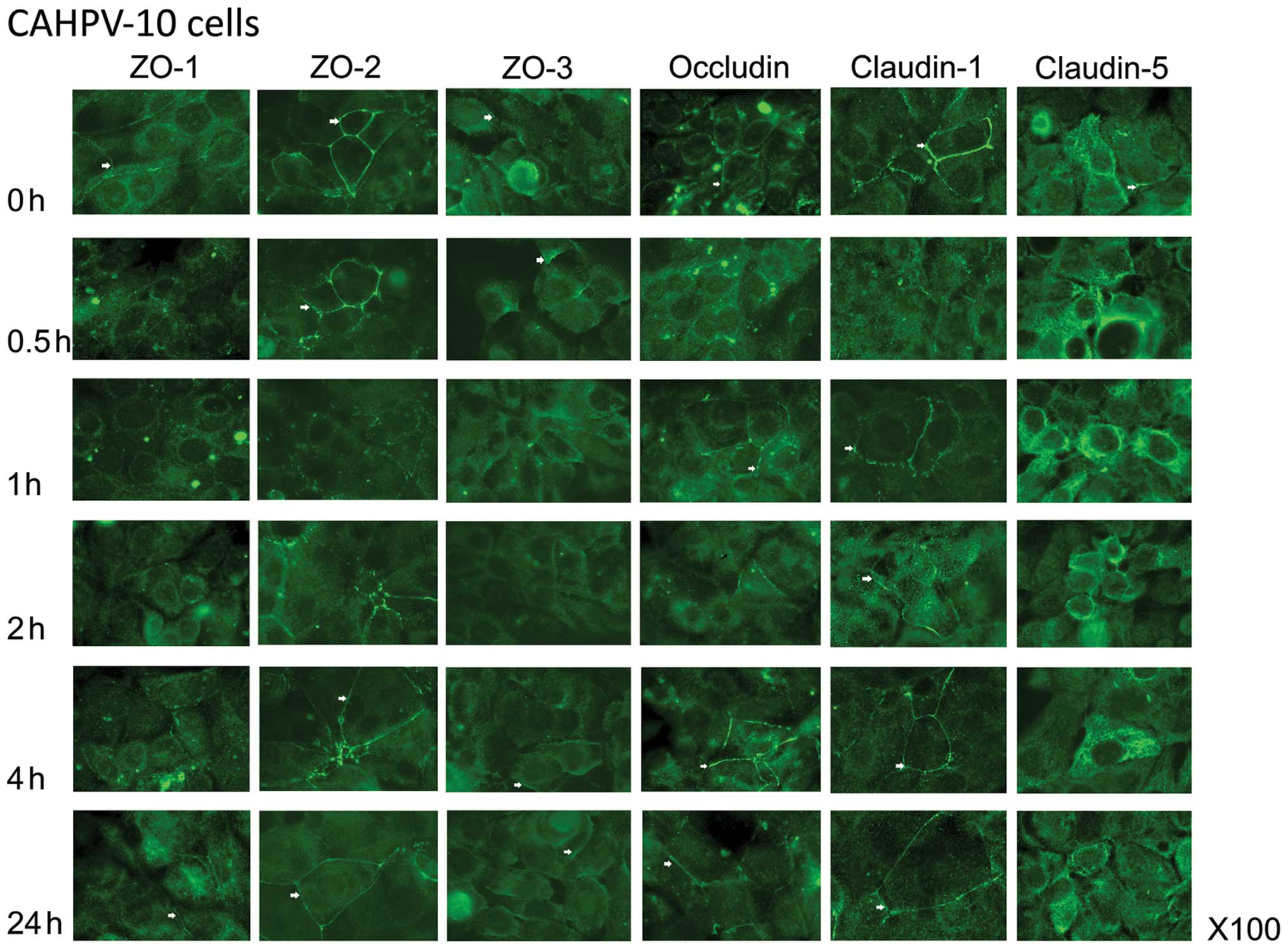
CAHPV-10 cells exhibited clear junctional staining
for ZO-1 that was lost completely by 1 h of co-culture with HGF
(Fig. 3). There was relocation
towards the nucleus which had only just dissipated by 24 h of
co-culture with HGF. ZO-2 expression was very strong in the
CAHPV-10 cells and became dotty and almost completely lost from the
cell periphery after 1 h of HGF treatment. Location of ZO-2 at the
TJ area was almost reconstituted after 4 h. In contrast, ZO-3 was
weakly located at the cell periphery in the CAHPV-10 cells and
appeared to increase at the cell periphery after HGF treatment.
Occludin was shown to be located at both the cell membrane and
nuclear membrane in the CAHPV-10 cells, both of which were reduced
by 2 h of co-culture with HGF. By 24 h of co-culture, cell membrane
integrity appeared to be intact, but there was no return of
occludin to the nuclear region. Claudin-1 was lost from the cell
membrane after 30 min of treatment with HGF, and re-location was
apparent after only 1 h. The location of claudin-5 was again
visible in the peri-nuclear region of CAHPV-10 cells and there
appeared to be an increase in intensity after treatment with
HGF.
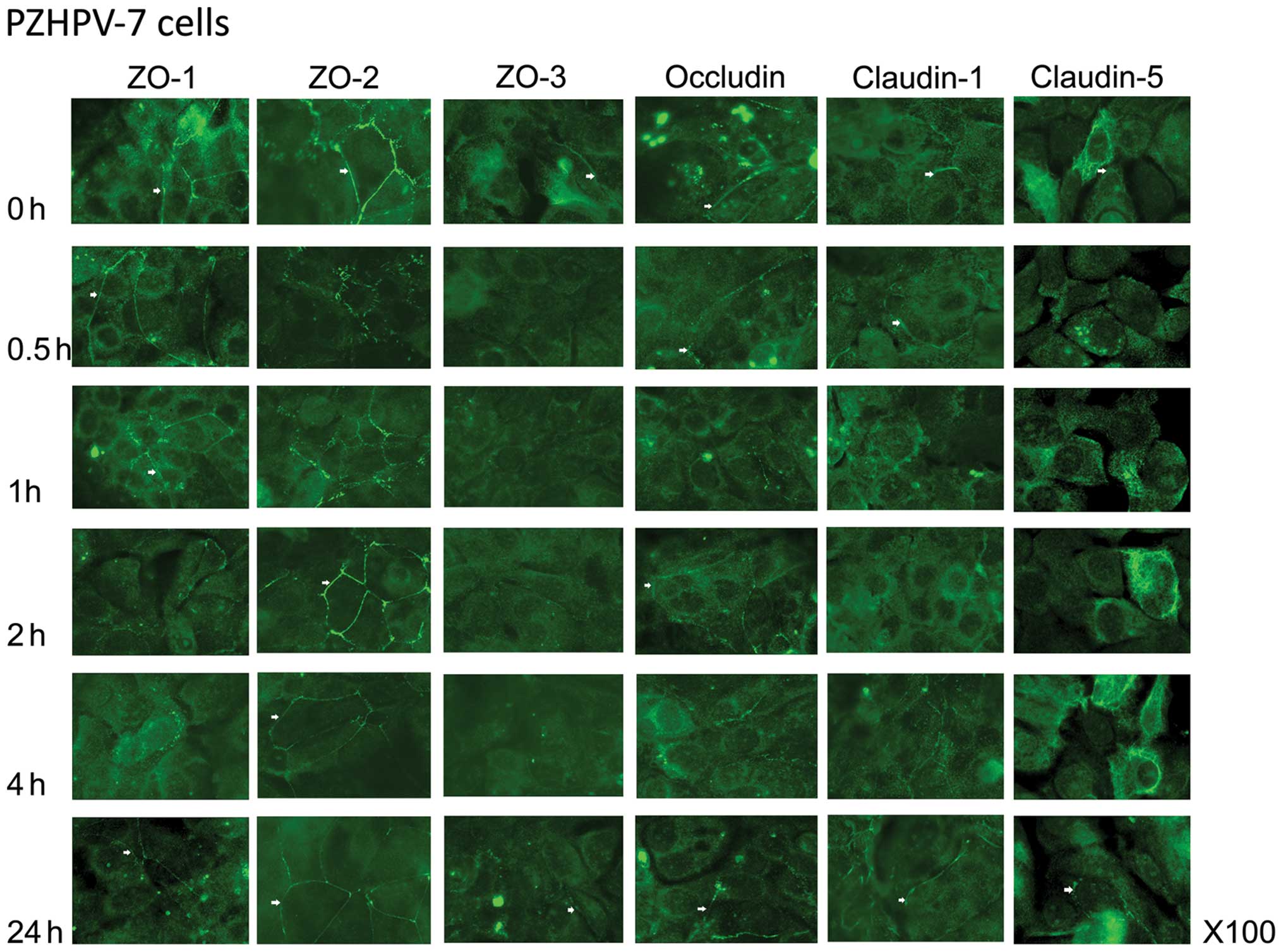
ZO-1 was lost from the cell membrane in the PZHPV-7
cells after a 2-h treatment with HGF (Fig. 4). There appeared to be a stronger
staining towards the leading edge of the cell. Recovery to the cell
membrane was slow and not complete after a 24-h culture. ZO-2
staining became spotty at the cell membrane after 30 min of
co-culture with HGF but recovered by 2 h. The pattern of staining
of ZO-3 in the PZHPV-7 cells was very weak, although visible at the
cell membrane before HGF treatment and was completely lost by a
30-min co-culture. Levels had recovered by 24 h. Occludin was not
completely lost from the cell membrane in the PZHPV-7 cells but was
dotty after a 30-min co-culture with HGF. Claudin-1 was reduced at
the cell periphery and showed increased cytoplasmic staining in the
PZHPV-7 cells by 2 h of co-culture with HGF before recovery by 4 h.
Claudin-5 was strongly stained in the peri-nuclear region of
PZHPV-7 cells, weakly stained at the cell membrane and remained so
until 24 h of treatment with HGF, when there was some relocation to
the cell periphery.
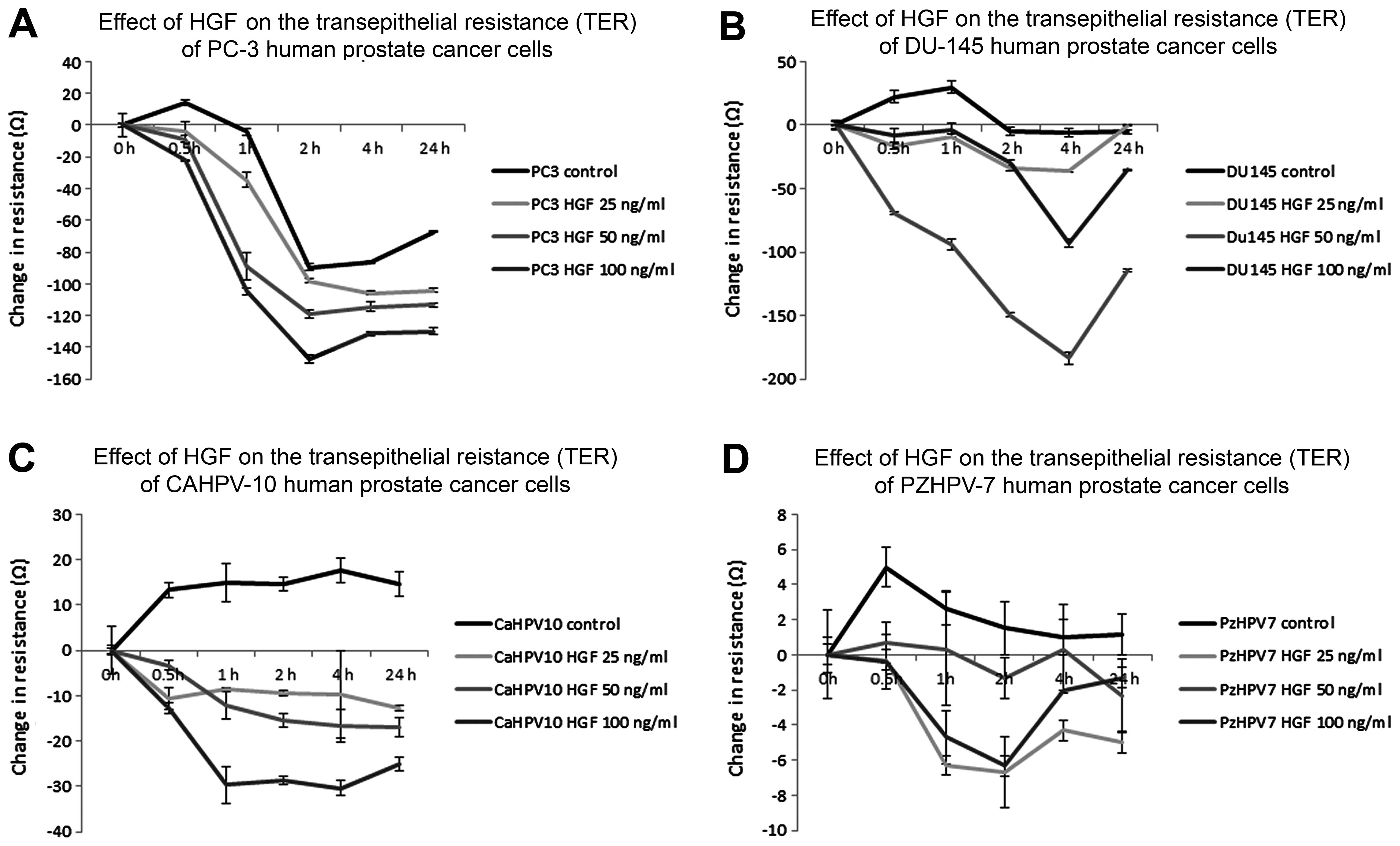
Changes in transepithelial resistance
(TER) of cells during HGF co-culture
Co-culture of confluent PC-3 cells with HGF at
increasing concentrations resulted in a marked reduction in TER
over 24 h as compared to the control cells (Fig. 5A). This occurred in a
concentration-dependent manner, with the highest concentration of
HGF (100 ng/ml) causing the greatest reduction in resistance.
Maximum reduction was achieved after 4 h of treatment with HGF
(control vs. HGF 25 ng/ml, P<0.0001; 50 ng/ml, P=0.0004; 100
ng/ml P<0.0001). A similar effect was observed in the DU-145
cells treated with HGF (Fig. 5B);
however, the greatest reduction in TER was achieved by HGF at 50
ng/ml. Maximum effect of HGF at all concentrations occurred
following 4 h of treatment (control vs. HGF 25 ng/ml, P=0.002; 50
ng/ml, P=0.0002; 100 ng/ml, P=0.0005). CAHPV-10 cells reacted in
the same way to HGF as the PC-3 cells, in that TER was reduced with
increasing HGF concentration (Fig.
5C). A maximum effect was observed by a 1 h co-culture with HGF
(control vs. HGF 25ng/ml, P=0.007; 50 ng/ml, P=0.006; 100ng/ml,
P=0.007). In contrast to the other three prostate cancer cell
lines, PZHPV-7 cells showed maximum reduced resistance with 25
ng/ml HGF at 1 h (Fig. 5D); the
higher concentrations of HGF also reduced TER in these cells, but
not to such a great extent (control vs. HGF 25 ng/ml, P=0.003; 50
ng/ml, P=0.03; 100 ng/ml, P=0.007).
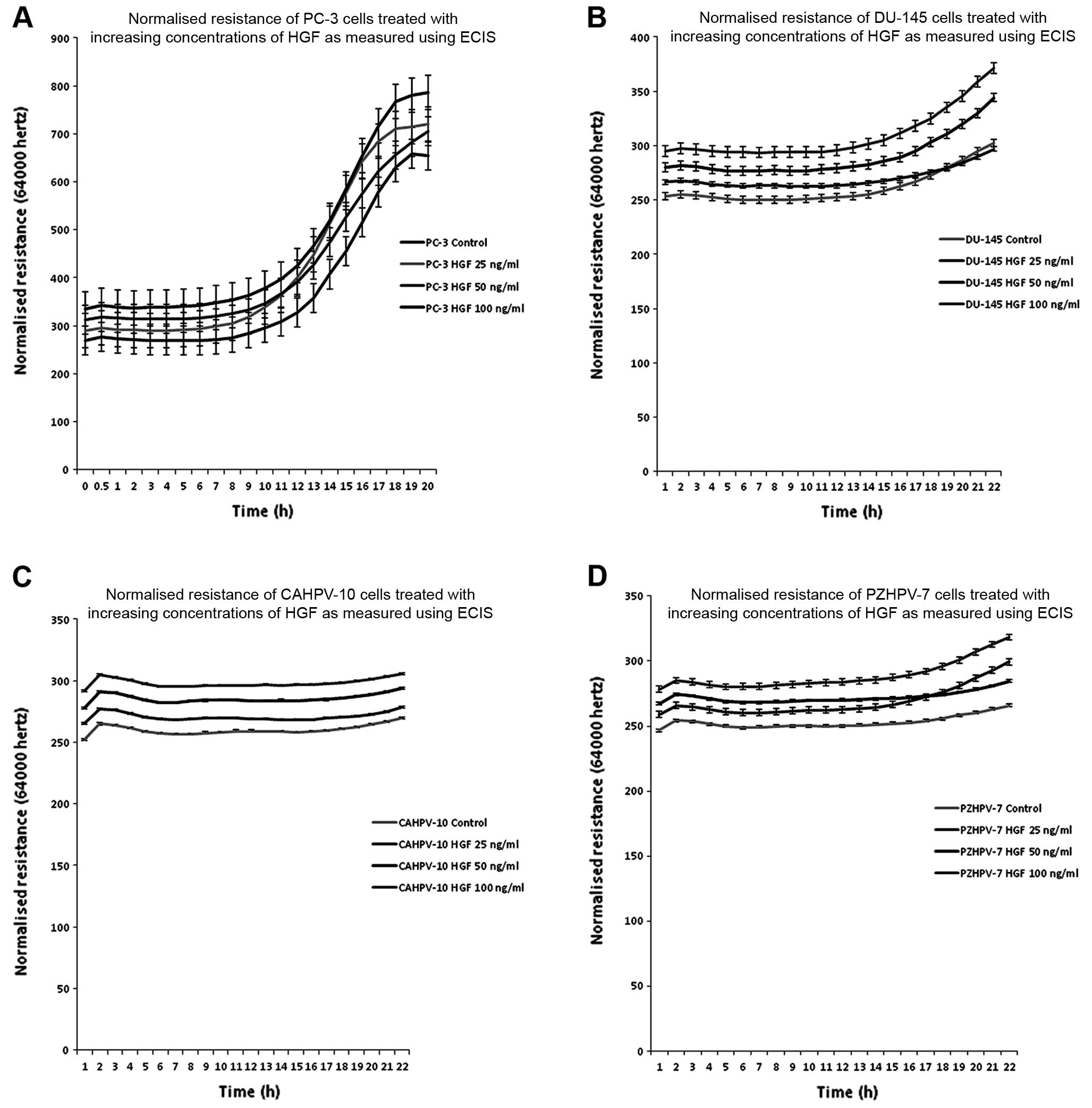
Changes in cell behaviour using ECIS
during HGF co-culture
All four prostate cancer cell lines were treated
with three concentrations of HGF (25, 50 and 100 ng/ml) in order to
determine the effect that HGF has on cell behaviour using ECIS. As
with TER, HGF caused a concentration-dependent increase in cell
attachment (Fig. 6A), with a
maximum response at 20 h of co-culture (control vs. HGF 100 ng/ml,
P<0.0001). However, HGF at 25 ng/ml produced the greatest
response in the DU-145 cells (control vs. HGF 25 ng/ml,
P<0.0001) (Fig. 6B). This also
occurred in the CAHPCV-10 cells (control vs. HGF 25 ng/ml,
P=0.0003) (Fig. 6C). HGF (100
ng/ml) caused the most response in the PZHPV-7 cells (control vs.
HGF 100 ng/ml, P<0.0001) (Fig.
6D).
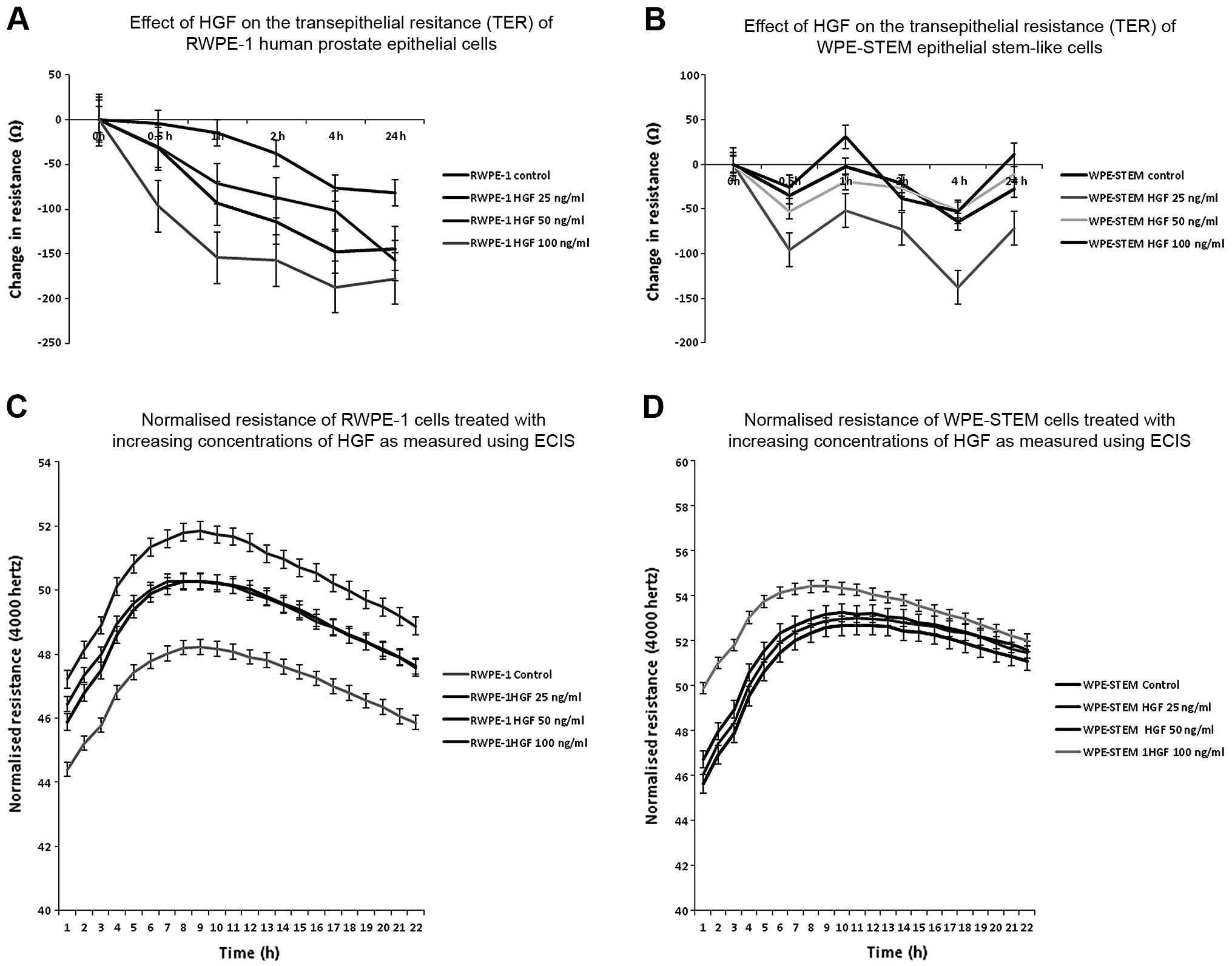
Effect of HGF co-culture on the behaviour
of normal and stem cell-like prostate cells
We then decided to investigate whether HGF had a
similar effect on the TJ function of normal prostate epithelial
cells. All three concentrations produced a reduced resistance in
the RWPE-1 cells, as expected (Fig.
7A). The greatest reduction was caused by 100 ng/ml HGF and no
further effect was noted after 4 h of co-culture (at 4 h
co-culture: control cells vs. HGF 25 ng/ml, P=0.002; HGF 50 ng/ml,
P=0.0008; HGF 100 ng/ml, P=0.0002). When we carried out the same
experiment on WPE-STEM prostate stem-like cells, we found that HGF
caused a biphasic modulation of TJ function as assessed using TER
(Fig. 7B). An initial decrease in
TER at 30 min of co-culture was followed by an increase at 1 h,
then a maximum decrease again by 4 h of co-culture before a return
to almost initial levels by 24 h. The most marked effect was
observed at an HGF concentration of 25 ng/ml, followed by 50 ng/ml.
HGF at 100 ng/ml caused increased resistance after 1 h of
co-culture which was higher than the control cells (at 4 h
co-culture: control cells vs. HGF 25 ng/ml, P=0.0002; HGF 50 ng/ml,
P=0.003; HGF 100 ng/ml, P=0.04). It is not unknown for high
concentrations of HGF to elicit an inhibitory effect, and so it
appears here that this may regulate the TJ function in these
stem-like prostate cells.
When we used ECIS to assess the cell behaviour of
RWPE-1 cells, HGF caused a concentration-dependent increase in cell
attachment (control vs. HGF 100 ng/ml, P<0.005) (Fig. 7C). This was also noted in the
WPE-STEM cells, although to a lesser extent (control vs. HGF 100
ng/ml, P>0.05) (Fig. 7D).
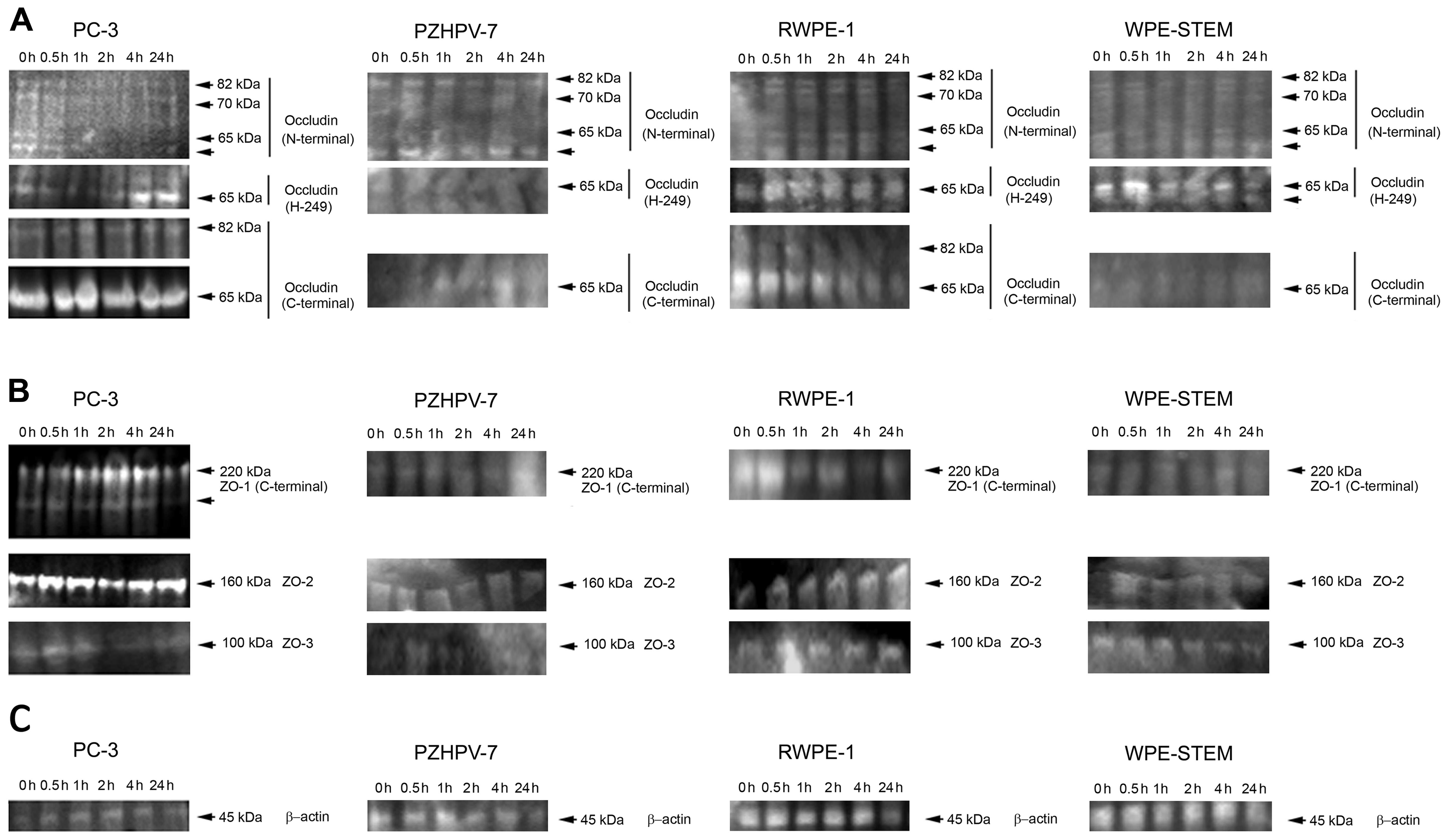
Changes in protein expression levels of
TJ molecules during HGF co-culture
Three different antibodies to occludin were used,
each produced to different epitopes N-terminal, H-249 and
C-terminal regions. In all the cells probed (PC-3, PZHPV-7, RWPE-1
and WPE-STEM), 4 different sizes of occludin protein were
identified by the N-terminal antibody of 82 kDa, as expected: one
at ~70 kDa, one at 65 kDa and one unknown protein of smaller size
(Fig. 8A). The H-249 antibody
identified only the 65-kDa protein in all 4 cell lines, with an
additional smaller band visible in the WPE-STEM cells. The
C-terminal antibody identified two proteins in the PC-3 cells,
again at 82 and 65 kDa (shown separately with reduced exposure for
clarity). Probing with the N-terminal antibody revealed an apparent
time-related response to HGF with occludin reduced from 0.5 h of
treatment for all four occludin proteins in PC-3 cells. In
contrast, there was an increase in the 65-kDa protein by a 4-h
treatment with HGF when probing with the H-249 antibody. No change
was observed with the C-terminal antibody in PC-3 cells. In the
PZHPV-7 cells, there was a decrease after probing with the
N-terminal antibody by 2 h of co-culture with HGF which returned to
initial levels by 4 h. There was no other change in occludin levels
in the PZHPV-7 cells. There was some reduction in occludin levels
after HGF treatment in the RWPE-1 cells (assessed by probing with
the C-terminal antibody). Although there was no change noted in
levels of occludin expression in the WPE-STEM cells after probing
with the N-terminal antibody, a biphasic response was observed when
probing with the H-249 antibody (Fig.
8A).
Western blotting for ZO-1 in the PC-3 cells revealed
two protein bands (Fig. 8B). There
was an increase in protein expression for the expected 220-kDa
protein by 1 h, which continued to increase in intensity after 4 h
of treatment with HGF, before returning to initial levels by 24 h.
The smaller protein was increased in intensity between 2 and 4 h of
treatment with HGF, before being reduced at 24 h. In the PZHPV-7
cells, ZO-1 was increased after 24 h of treatment with HGF
(Fig. 8B). Levels of ZO-1 were
decreased in the RWPE-1 cells after HGF treatment for 1 h. Again, a
small biphasic change in ZO-1 expression was noted in the WPE-STEM
cells (Fig. 5B). Protein expression
of ZO-2 was decreased after a 2 h treatment with HGF before
returning to initial levels in PC-3 cells. No change was observed
in the PZHPV-7 and WPE-STEM cells. Levels of ZO-2 were increased in
the RWPE-1 cells by 2 h of treatment with HGF (Fig. 8B). In the PC-3 cells, levels for
ZO-3, although very weak in intensity overall showed a drop at 2 h
(Fig. 8B). This was also observed
in the PZHPV-7 and WPE-STEM cells. β-actin was used as a loading
control (Fig. 8C).
In vivo growth with HGF
RWPE-1 and WPE-STEM cells were used to assess
tumourigenicity in an athymic nude mouse model. After 4 weeks of
growth, no tumours were noted; after dissection, no evidence was
observed of any growth. We can only conclude that neither cell line
possessed tumourigenic properties, even in the presence of HGF.
Discussion
The present study showed that HGF, a cytokine that
is strongly indicated as a regulator of tumour progression and
metastasis (25,26), decreased transepithelial resistance
in human prostate cancer cells and cancer stem cell derivatives by
modulating the expression of TJ molecules at both the transcript
and protein levels and by redistribution of TJ proteins. HGF may
regulate TJ structure formation by modulating the expression of
ZO-1 and ZO-2, two TJ molecules thought to be key proteins in the
regulation of TJ structure. Increasing evidence suggests that TJs
may play a key role in cancer metastasis (27). We previously demonstrated that HGF
promotes both migration and invasion of PC-3 prostate cancer cell
(28–30).
Our previous research showed that HGF decreases
trans-endothelial resistance, and increases paracellular
permeability in both human endothelial and breast cancer cells
(2,10). HGF is able to cause loss of TJs,
increased permeability and decreased TER in several cell types.
This loss of function leads to increased chemotactic migration in
retinal pigment epithelial monolayers (30). HGF regulates Sertoli-Sertoli TJs in
mammalian testes, inducing a reduction in expression and a
redistribution of occludin (31).
HGF has also been shown to promote the relocalisation of ZO-1 from
TJs to the cytoplasm in MDCK cells (32) and to decrease TER in a non-tumoural
gastric epithelial cell line (33);
a similar result in T84 human intestinal cells (34), but not in tracheal epithelial cells
(35).
Conversely, a number of studies have shown that in
certain cases, HGF provides a protective effect against TJ
disruption. This was demonstrated in cerebrovascular endothelial
cells, where treatment with recombinant HGF attenuated the decrease
in expression of occludin and ZO-1 after sustained cerebral
ischemia (36). In addition, HGF
increases TER in uterine epithelial cells (37). In human pulmonary artery endothelial
cells, HGF rapidly and dose-dependently increased transendothelial
resistance (38), although there
was no examination of the expression of any TJ molecules. This
present work is the first study to examine the effect of HGF on TJs
in prostate cancer cells. PC-3 cells had reduced transepithelial
resistance and increased paracellular permeability resulting in
reduced barrier function upon addition of HGF.
RT-PCR revealed that HGF modulated the expression of
a number of TJ molecules, a response that was consistent over
numerous experiments (n=5). The loss of expression of ZO-1, ZO-2
and ZO-3 was an expected response to HGF. However, the increase in
expression of occludin was not foreseen, as was the lack of
response to HGF by claudin-1 and claudin-5. At the protein level,
there was an apparent initial increase in expression of ZO-1,
followed by a reduction. There was also a signal for two proteins
after probing with ZO-1, a phenomenon we had not previously
observed. There was some loss of ZO-2 and ZO-3 at the protein
level, but this did not follow the observations of changes in
expression at the RNA level. The increased message level of
occludin was not reflected by the protein levels of occludin, as
probed using an antibody with an epitope to the N-terminal region
of the protein. However, after probing with a second antibody with
an epitope mapping to a more central region of occludin, there was
a concurrent increase in occludin signal of the 65-kDa size.
A possible explanation for the inconsistency of
reduced expression in message to protein following HGF stimulation
may be that the possible reduction in protein was reflected, but
that protein degradation occurs much more slowly than that of mRNA.
Consequently, as the mRNA levels of ZO-1 and ZO-2 began to decrease
after 1 h of incubation with HGF, we did not see an equivalent
reduction in protein. Occludin at the high molecular weight level
was reduced after a 0.5-h incubation with HGF. An extra band was
also observed at ~70 kDa. We previously demonstrated this band in
human breast cancer cells (10).
Previous reports have shown that phosphorylation of serine residues
on occludin increases the formation of TJs (25) and that occludin confers adhesiveness
in proportion to the level of protein expressed (39). Phosphorylation of occludin is
probably an important step in regulating TJ formation and
permeability. Farshori and Kachar (40) have shown that, in MDCK epithelial
cell monolayers, the 62–65 kDa occludin bands are phosphorylated on
serine and threonine, whereas the 71-kDa band is phosphorylated on
serine only. Wong (41) described
the appearance of two clusters of occludin in MDCK cells: one group
of low molecular weight (LMW) of ~65–68 kDa, and one group of high
molecular weight (HMW) of ~70–75 kDa. LMW occludins are found in
cells that have no cell-cell contact, i.e. no intact TJs. The HMW
occludins (hyperphosphorylated LMWs) are the functional forms of
occludin that participate in the formation of the TJ barrier. Such
results suggest that the phosphorylation of occludin may be a
mechanism by which localisation and function are regulated. We have
seen that a decrease in HMW occludin in breast cancer cell lines,
upon addition of HGF, corresponds to loss of TJ integrity, and so
to a loss of TJ function (10).
Based on our data here, there was an increase in
levels of the 65-kDa occludin band suggesting, that in these cells,
HGF caused an increase in serine and threonine phosphorylation,
with a concurrent reduction in serine-only phosphorylated occludin.
This could thus lead to a reduction in TJ integrity, as serine-only
phosphorylated occludin is required for TJ formation in cell types
that express occludin. The apparent disparity in expression pattern
between occludin antibodies of various epitopes might reflect the
sensitivities of these antibodies to changes in phosphorylation
status.
It has been shown that phosphorylation of the
tyrosine residues on ZO-1 may result in the dismantling of TJs and
a decrease in TER (25), which we
also showed to contribute to changes in TJ formation in human
breast cancer cells (10). ZO
proteins are involved in the organisation of epithelial and
endothelial intercellular junctions, binding to the cytoplasmic
carboxyl termini of junctional transmembrane proteins and so
linking them to the actin cytoskeleton. HGF-induced ZO-1
phosphorylation may contribute to the disassembly of the TJ
complex, resulting in transient disruption of TJ function. The two
ZO-1 proteins observed expressed by PC-3 cells may reflect a
difference in phosphorylation status of ZO-1 in these cells.
Early research on claudins suggested that tissue
expression patterns varied between different claudin species
(42). It is evident from our study
here, that HGF has no effect on the expression of either claudin-1
or claudin-5 in these human prostate cells. Immunostaining with
ZO-1, ZO-2, ZO-3, occludin, claudin-1 and claudin-5 showed that HGF
affected the distribution of these TJ molecules. After co-culture
with HGF, there was a more densely stained cytoplasm and relocation
of ZO-1 to ruffled membrane areas of the cells. Both ZO-2 and ZO-3
showed a loss of signal at the TJ region with increased cytoplasmic
staining in the case of ZO-2. ZO-3 expression remained less strong
throughout the experiment overall. Occludin staining was well
defined and following treatment with HGF became dotty and
discontinuous with increased cytoplasmic staining. There was also a
concurrent loss in TJ location of both claudins.
As stated in the introduction, there have been few
studies investigating the role of TJs in prostate cancer cells and
those that have been carried out were focused on the claudin family
of proteins. Studies using clinical samples have shown that
claudin-1 is lost in prostate adenocarcinoma (16) as discovered using
immunohistochemistry. In LNcaP prostate cells, two isoforms of
claudin-7 were found to regulate the expression of PSA, with the
truncated form predominantly expressed in healthy prostate tissues
(18). In addition, occludin was
observed to be lost in unpolarized cells of Gleason grades 4 and 5
(n=64) with general downregulation associated with loss of cell
polarity (43). There have been no
reports of the modulation of TJ function and formation in human
prostate cancer cells.
Claudin proteins have been targeted as a means of
modulating TJ function. Tight junctions in human prostate cells can
be modulated successfully, as shown by a study suggesting that
flavonoids extracted from Orostachys japonicus A. Berger
suppressed cancer migration and invasion by tightening TJs through
the suppression of claudin expression, and by suppressing MMPs in
LNCaP human prostate cancer cells, which at least in part resulted
from the suppression of the PI3K/Akt signaling pathway (44).
Moreover, in mice, low serum testosterone has been
associated with reduced transcript and protein levels of claudin-4
and claudin-8, resulting in defective TJ ultrastructure in benign
prostate glands. Expression of claudin-4 and claudin-8 was
negatively correlated with the mononuclear inflammatory infiltrate
caused by testosterone deprivation. Testosterone suppression also
induced an autoimmune humoral response directed toward prostatic
proteins. Testosterone supplementation in castrated mice resulted
in re-expression of TJ components in prostate epithelium and
significantly reduced prostate inflammatory cell numbers. These
data demonstrate that TJ architecture in the prostate is related to
changes in serum testosterone levels, and identify an
androgen-regulated mechanism that potentially contributes to the
development of prostate inflammation and consequent pathology
(45). In targeting TJs,
CPE-mediated cytotoxicity was observed in human prostate cancer
cell lines, but was barely detected in normal human PrECs. The
cytotoxic effect depended not only on the expression level of
Claudin-4 protein but also on its subcellular localization. These
results suggest that Claudin-4-targeted therapy using CPE may be a
new treatment for prostate cancer (46).
The WPE-STEM cells demonstrated a difference in
response to treatment with HGF, which may have a direct bearing on
their stem cell-like phenotype. It has been shown that HGF secreted
by prostate cancer stem-like cells/cancer-initiating cells and
prostate myofibroblasts has a role in the maintenance of prostate
cancer stem-like cells/cancer-initiating cells in an autocrine and
paracrine manner (47). In mice,
immunohistochemical analysis in prostatectomies revealed
significant enrichment of c-MET-positive cells at the invasive
front, and demonstrated co-expression of c-MET with stem-like
markers CD49b and CD49f. In conclusion, activation of c-MET in
prostate cancer cells induced a stem-like phenotype, indicating a
dynamic relation between differentiated and stem-like cells in this
malignancy. Its mediation of efficient tumour formation in
vivo and predominant receptor expression at the invasive front
indicate that c-MET regulates tumour infiltration in surrounding
tissues putatively by acquisition of a stem-like phenotype
(48).
The rapidly increasing studies linking TJ function
and regulation with cancer cell dissociation, motility and
invasion, all events involved in metastasis, strongly suggest that
TJs are a key element in the prevention of cancer cell
dissemination.
In conclusion, from the data presented here, we can
conclude that HGF, a cytokine known to be involved in metastatic
progression, disrupts the TJs of human prostate cancer cells. HGF
affects this disruption by: i) modulating TJ molecule transcripts
and ii) modulating the protein expression of ZO-1, ZO-2 and
occludin. These changes were concurrent with relocation of ZO-1,
ZO-2, ZO-3, occludin and claudin-1 and claudin-5 from the TJ
locality. Both occludin and ZO-1 showed some relocation to the
ruffled membrane areas of migrating cells. The disruption of the TJ
structure by HGF could thus be an initial event in the metastasis
of human prostate cancer cells and as such requires further
investigation to evaluate its usefulness as a target for
anti-metastasis intervention.
Acknowledgements
The authors would like to thank Cancer Research
Wales for supporting their work and Dr Jane Lane for proof-reading
the manuscript.
References
|
1
|
Martin TA, Watkins G, Mansel RE and Jiang
WG: Loss of tight junction plaque molecules in breast cancer
tissues is associated with a poor prognosis in patients with breast
cancer. Eur J Cancer. 40:2717–2725. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Martin TA, Mansel RE and Jiang WG:
Antagonistic effect of NK4 on HGF/SF induced changes in the
transendothelial resistance (TER) and paracellular permeability of
human vascular endothelial cells. J Cell Physiol. 192:268–275.
2002. View Article : Google Scholar
|
|
3
|
Ren J, Hamada J, Takeichi N, Fujikawa S
and Kobayashi H: Ultrastructural differences in junctional
intercellular communication between highly and weakly metastatic
clones derived from rat mammary carcinoma. Cancer Res. 50:358–362.
1990.
|
|
4
|
Satoh H, Zhong Y, Isomura H, et al:
Localization of 7H6 tight junction-associated antigen along the
cell border of vascular endothelial cells correlates with
paracellular barrier function against ions, large molecules, and
cancer cells. Exp Cell Res. 222:269–274. 1996. View Article : Google Scholar
|
|
5
|
Hoevel T, Macek R, Mundigl O, Swisshelm K
and Kubbies M: Expression and targeting of the tight junction
protein CLDN1 in CLDN1-negative human breast tumor cells. J Cell
Physiol. 191:60–68. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kominsky SL, Argani P, Korz D, et al: Loss
of the tight junction protein claudin-7 correlates with
histological grade in both ductal carcinoma in situ and invasive
ductal carcinoma of the breast. Oncogene. 22:2021–2033. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Krämer F, White K, Kubbies M, Swisshelm K
and Weber BH: Genomic organization of claudin-1 and its assessment
in hereditary and sporadic breast cancer. Hum Genet. 7:249–256.
2002.PubMed/NCBI
|
|
8
|
Chlenski A, Ketels KV, Korovaitseva GI,
Talamonti MS, Oyasu R and Scarpelli DG: Organization and expression
of the human zo-2 gene (tjp-2) in normal and neoplastic tissues.
Biochim Biophys Acta. 1493:319–324. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bell J, Walsh S, Nusrat A and Cohen C:
Zonula occludens-1 and Her-2/neu expression in invasive breast
carcinoma. Appl Immunohistochem Mol Morphol. 11:125–129. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Martin TA, Watkins G, Mansel RE and Jiang
WG: Hepatocyte growth factor disrupts tight junctions in human
breast cancer cells. Cell Biol Int. 28:361–371. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ikenouchi J, Matsuda M, Furuse M and
Tsukita S: Regulation of tight junctions during the
epithelium-mesenchyme transition: direct repression of the gene
expression of claudins/occludin by Snail. J Cell Sci.
116:1959–1967. 2003. View Article : Google Scholar
|
|
12
|
Martin TA, Goyal A, Watkins G and Jiang
WG: Expression of the transcription factors snail, slug, and twist
and their clinical significance in human breast cancer. Ann Surg
Oncol. 12:488–496. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ohkubo T and Ozawa M: The transcription
factor Snail downregulates the tight junction components
independently of E-cadherin downregulation. J Cell Sci.
117:1675–1685. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gopalakrishnan S, Raman N, Atkinson SJ and
Marrs JA: Rho GTPase signaling regulates tight junction assembly
and protects tight junctions during ATP depletion. Am J Physiol.
275:C798–C809. 1998.PubMed/NCBI
|
|
15
|
Wong V and Gumbiner BM: A synthetic
peptide corresponding to the extracellular domain of occludin
perturbs the tight junction permeability barrier. J Cell Biol.
136:399–409. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Krajewska M, Olson AH, Mercola D, Reed JC
and Krajewski S: Claudin-1 immunohistochemistry for distinguishing
malignant from benign epithelial lesions of prostate. Prostate.
67:907–910. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hewitt KJ, Agarwal R and Morin PJ: The
claudin gene family: expression in normal and neoplastic tissues.
BMC Cancer. 6:1862006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zheng JY, Yu D, Foroohar M, Ko E, Chan J,
Kim N, Chiu R and Pang S: Regulation of the expression of the
prostate-specific antigen by claudin-7. J Membr Biol. 194:187–197.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Long H, Crean CD, Lee WH, Cummings OW and
Gabig TG: Expression of Clostridium perfringens enterotoxin
receptors claudin-3 and claudin-4 in prostate cancer epithelium.
Cancer Res. 61:7878–7881. 2001.
|
|
20
|
Meyer HW, Freytag C, Freytag T and Richter
W: Effect of proteases and other treatments on the proliferative
assembly of tight junction strands in the rat prostate tissue. Exp
Pathol. 34:237–244. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kachar B and Reese TS: Evidence for the
lipidic nature of tight junction strands. Nature. 296:464–466.
1982. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kachar B and Reese TS: Formation of
misplaced and reflexive tight junction strands in prostate
epithelial cells. J Ultrastruct Res. 82:90–95. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kachar B and Pinto da Silva P: Rapid
massive assembly of tight junction strands. Science. 213:541–544.
1981. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Martin TA, Das T, Mansel RE and Jiang WG:
Enhanced tight junction function in human breast cancer cells by
antioxidant, selenium and polyunsaturated lipid. J Cell Biochem.
101:155–166. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiang WG, Martin TA, Matsumoto K, Nakamura
T and Mansel RE: Hepatocyte growth factor/scatter factor decreases
the expression of occludin and transendothelial resistance (TER)
and increases paracellular permeability in human vascular
endothelial cells. J Cell Physiol. 181:319–329. 1999. View Article : Google Scholar
|
|
26
|
Rosen EM, Zitnik RJ, Elias JA, Bhargava
MM, Wines J and Goldberg ID: The interaction of HGF-SF with other
cytokines in tumor invasion and angiogenesis. EXS. 65:301–310.
1993.PubMed/NCBI
|
|
27
|
Martin TA and Jiang WG: Tight junctions
and their role in cancer metastasis. Histol Histopathol.
16:1183–1195. 2001.PubMed/NCBI
|
|
28
|
Hurle RA, Davies G, Parr C, Mason MD,
Jenkins SA, Kynaston HG and Jiang WG: Hepatocyte growth
factor/scatter factor and prostate cancer: a review. Histol
Histopathol. 20:1339–1349. 2005.PubMed/NCBI
|
|
29
|
Davies G, Jiang WG and Mason MD:
Matrilysin mediates extracellular cleavage of E-cadherin from
prostate cancer cells: a key mechanism in hepatocyte growth
factor/scatter factor-induced cell-cell dissociation and in vitro
invasion. Clin Cancer Res. 7:3289–3297. 2001.
|
|
30
|
Jin M, Barron E, He S, Ryan SJ and Hinton
DR: Regulation of RPE intercellular junction integrity and function
by hepatocyte growth factor. Invest Ophthalmol Vis Sci.
43:2782–2790. 2002.PubMed/NCBI
|
|
31
|
Catizone A, Ricci G and Galdieri M:
Hepatocyte growth factor modulates Sertoli-Sertoli tight junction
dynamics. J Cell Physiol. 216:253–260. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Grisendi S, Arpin M and Crepaldi T: Effect
of hepatocyte growth factor on assembly of zonula occludens-1
protein at the plasma membrane. J Cell Physiol. 176:465–471. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hollande F, Blanc EM, Bali JP, Whitehead
RH, Pelegrin A, Baldwin GS and Choquet A: HGF regulates tight
junctions in new nontumorigenic gastric epithelial cell line. Am J
Physiol Gastrointest Liver Physiol. 280:G910–G921. 2001.PubMed/NCBI
|
|
34
|
Nusrat A, Parkos CA, Bacarra AE, Godowski
PJ, Delp-Archer C, Rosen EM and Madara JL: Hepatocyte growth
factor/scatter factor effects on epithelia. Regulation of
intercellular junctions in transformed and nontransformed cell
lines, basolateral polarization of c-met receptor in transformed
and natural intestinal epithelia, and induction of rapid wound
repair in a transformed model epithelium. J Clin Invest.
93:2056–2065. 1994.
|
|
35
|
Shen BQ, Panos RJ, Hansen-Guzmán K,
Widdicombe JH and Mrsny RJ: Hepatocyte growth factor stimulates the
differentiation of human tracheal epithelia in vitro. Am J Physiol.
272:L1115–L1120. 1997.PubMed/NCBI
|
|
36
|
Date I, Takagi N, Takagi K, Tanonaka K,
Funakoshi H, Matsumoto K, Nakamura T and Takeo S: Hepatocyte growth
factor attenuates cerebral ischemia-induced increase in
permeability of the blood-brain barrier and decreases in expression
of tight junctional proteins in cerebral vessels. Neurosci Lett.
407:141–145. 2006. View Article : Google Scholar
|
|
37
|
Grant-Tschudy KS and Wira CR: Hepatocyte
growth factor regulation of uterine epithelial cell transepithelial
resistance and tumor necrosis factor alpha release in culture. Biol
Reprod. 72:814–821. 2005. View Article : Google Scholar
|
|
38
|
Liu F, Schaphorst KL, Verin AD, Jacobs K,
Birukova A, Day RM, Bogatcheva N, Bottaro DP and Garcia JG:
Hepatocyte growth factor enhances endothelial cell barrier function
and cortical cytoskeletal rearrangement: potential role of glycogen
synthase kinase-3beta. FASEB J. 16:950–962. 2002. View Article : Google Scholar
|
|
39
|
Fanning AS, Mitic LL and Anderson JM:
Transmembrane proteins in the tight junction barrier. J Am Soc
Nephrol. 10:1337–1345. 1999.PubMed/NCBI
|
|
40
|
Farshori P and Kachar B: Redistribution
and phosphorylation of occludin during opening and resealing of
tight junctions in cultured epithelial cells. J Membr Biol.
170:147–156. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wong V: Phosphorylation of occludin
correlates with occludin localization and function at the tight
junction. Am J Physiol. 273:C1859–C1867. 1997.PubMed/NCBI
|
|
42
|
Morita K, Furuse M, Fujimoto K and Tsukita
S: Claudin multigene family encoding four-transmembrane domain
protein components of tight junction strands. Proc Natl Acad Sci
USA. 96:511–516. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Busch C, Hanssen TA, Wagener C and OBrink
B: Down-regulation of CEACAM1 in human prostate cancer: correlation
with loss of cell polarity, increased proliferation rate, and
Gleason grade 3 to 4 transition. Hum Pathol. 33:290–298. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Shin DY, Lee WS, Jung JH, et al:
Flavonoids from Orostachys japonicus A. Berger inhibit the
invasion of LNCaP prostate carcinoma cells by inactivating Akt and
modulating tight junctions. Int J Mol Sci. 14:18407–18420.
2013.PubMed/NCBI
|
|
45
|
Meng J, Mostaghel EA, Vakar-Lopez F,
Montgomery B, True L and Nelson PS: Testosterone regulates tight
junction proteins and influences prostatic autoimmune responses.
Horm Cancer. 2:145–156. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Maeda T, Murata M, Chiba H, Takasawa A,
Tanaka S, Kojima T, Masumori N, Tsukamoto T and Sawada N:
Claudin-4-targeted therapy using Clostridium perfringens
enterotoxin for prostate cancer. Prostate. 72:351–360. 2012.
View Article : Google Scholar
|
|
47
|
Nishida S, Hirohashi Y, Torigoe T, Inoue
R, Kitamura H, Tanaka T, Takahashi A, Asanuma H, Masumori N,
Tsukamoto T and Sato N: Prostate cancer stem-like
cells/cancer-initiating cells have an autocrine system of
hepatocyte growth factor. Cancer Sci. 104:431–436. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
van Leenders GJ, Sookhlall R, Teubel WJ,
de Ridder CM, Reneman S, Sacchetti A, Vissers KJ, van Weerden W and
Jenster G: Activation of c-MET induces a stem-like phenotype in
human prostate cancer. PLoS One. 6:e267532001.PubMed/NCBI
|