Introduction
Neuroblastoma is the most common pediatric
extracranial solid tumor of neural crest origin.
Characteristically, it can exhibit either a favorable or an
unfavorable phenotype. Favorable neuroblastomas are treatable with
minimal interventions, whereas unfavorable neuroblastomas are
aggressive and require extensive treatments, including autologous
stem cell rescue (1). Long-term
survival of children with unfavorable neuroblastoma is hence the
lowest among childhood cancers. Approximately half of unfavorable
neuroblastoma cases have MYCN amplification, which is
associated with high MYCN expression, older age of onset, advanced
stage disease, rapid tumor progression, and the worst prognosis
(2,3). A recent study also showed that
non-MYCN amplified neuroblastomas of the unfavorable subset
express high levels of MYC instead of MYCN, which appears to be the
determining factor of their aggressiveness (4).
OSU-0312 is a celecoxib-derived PDK1 inhibitor that
has a growth suppressive effect on various cancer cell lines
(5,6). When RAS is activated through external
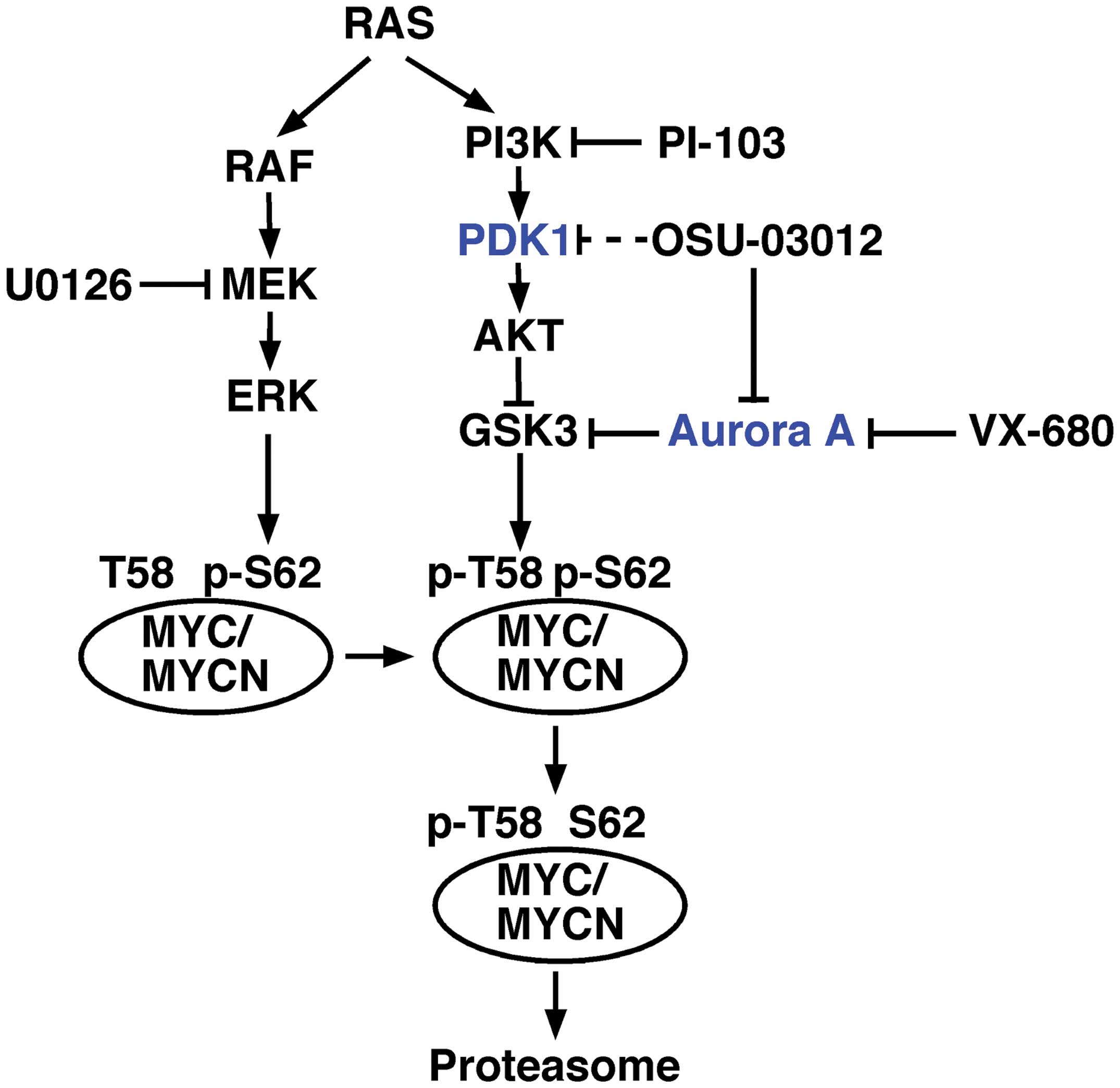
stimuli or mutation, two distinct cellular pathways are activated:
the RAF/MEK/ERK and PI3/PDK1/AKT/GSK3 pathways. These pathways in
turn affect the stability of MYC family proteins (7–9)
(Fig. 1). In various cancer cell
lines, when MEK is inhibited by U0126, there is a marked reduction
in MYC protein expression levels with concomitant growth
suppression (10). Based on these
observations, it is plausible that inhibition of any point in the
RAF/MEK/ERK and/or PI3K/PDK1/AKT pathways can destabilize MYC and
MYCN. Hence, OSU-0312 was predicted to destabilize MYC and MYCN
protein in neuroblastoma cells.
However, since PDK1 phosphorylates AKT to activate
it, and OSU-03012 is an inhibitor of PDK1, it was puzzling that
under normal cell culture conditions, AKT was barely phosphorylated
and OSU-03012 did not affect the p-AKT status in neuroblastoma
cells (as confirmed below). This observation suggests that PDK1 is
not the main target of OSU-03012 in neuroblastoma cells. In
addition, it has been reported that GSK3β is phosphorylated by
Aurora kinase A, and that Aurora A knockdown results in reduced MYC
levels (11). These observations
collectively suggest that OSU-03012 inhibits Aurora kinase A, which
in turn destabilizes MYC and MYCN in neuroblastoma cells (Fig. 1). In the present study, we
investigated this possibility.
Materials and methods
Neuroblastoma cell lines
The neuroblastoma cell lines were grown in RPMI-1640
supplemented with 5% (v/v) fetal bovine serum and OPI (1 mM
oxaloacetate, 0.45 mM pyruvate, 0.2 unit/ml insulin, at final
concentrations). These cell lines tested negative for mycoplasma,
and their identity was validated by the original source or by
microsatellite analysis (P.S. White, Children’s Hospital of
Philadelphia, Philadelphia, PA, USA; unpublished data). The IMR5 (a
clone of IMR32) and CHP134 cell lines were obtained from Dr Roger
H. Kennett (Wheaton College, Wheaton, IL, USA). The SKNBE(2)C cell line was from Robert Ross (Fordham
University, New York, NY, USA). The SKNAS cell line was from Dr C.
Patrick Reynolds (The Texas Tech University Health Sciences Center,
Lubbock, TX, USA). CHP134, IMR5 and SKBBE(2)C are MYCN-amplified cell lines,
whereas SKNAS is a non-MYCN-amplified cell line.
MTS assay
An MTS
[3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium,
inner salt] assay (a water-soluble form of the MTT assay) was
performed as described in our previous study (12). OSU-03012 was purchased from Cayman
Chemical Company (Ann Arbor, MI, USA). VX-680 was purchased from LC
Laboratories (Woburn, MA, USA). The stock solutions of the
inhibitors were prepared at 10 mM in DMSO, and stored at −20°C.
Western blot analysis
Western blotting was performed as previously
described (13,14). Light emission signals were captured
by an LAS-3000 digital image analyzer (Fujifilm). Cell extracts
were made in 2D gel sample buffer (9 M urea, 2% Nonidet-P40, 2%
2-mercaptoethanol and 0.32% pH 3.0 to 10.0 2D Pharmalyte), and the
protein content of the samples was determined by the Bio-Rad
protein assay kit using bovine serum albumin as a standard and the
sample buffer as the blank. Antibodies used to detect proteins of
interest are described in the figure legends.
Molecular docking analysis in silico
The 3D structure of AURKA (PDB ID: 3DAJ) was
obtained from the Brookhaven Protein Databank. The structures of
FXG and OSU-03012 were constructed using MOE (version 2007;
Chemical Computing Group, Montreal, Canada). The docking
simulations and interaction energy calculations were performed
using MOE Dock of MOE. The most stable docking structures of FXG
and OSU-03012 with AURKA were displayed by MOE.
Cell-free Aurora kinase A assay
The HTScan® Aurora A kinase assay kit
(Cell Signaling Technology Inc., Danvers, MA, USA) was used to
examine the inhibitory activity of OSU-03012 and VX-680 against
recombinant Aurora kinase A, according to the manufacturer’s
instructions.
Results
OSU-03012 and VX-680 suppress the growth
of neuroblastoma cells
First, we examined the dose response of OSU-03012
and VX-680, a potent Aurora kinase inhibitor (16), on the growth of neuroblastoma cell
lines (with and without MYCN amplification). As shown in
Fig. 2A, both compounds were
significantly growth inhibitory against the neuroblastoma cells in
dose-dependent manners.
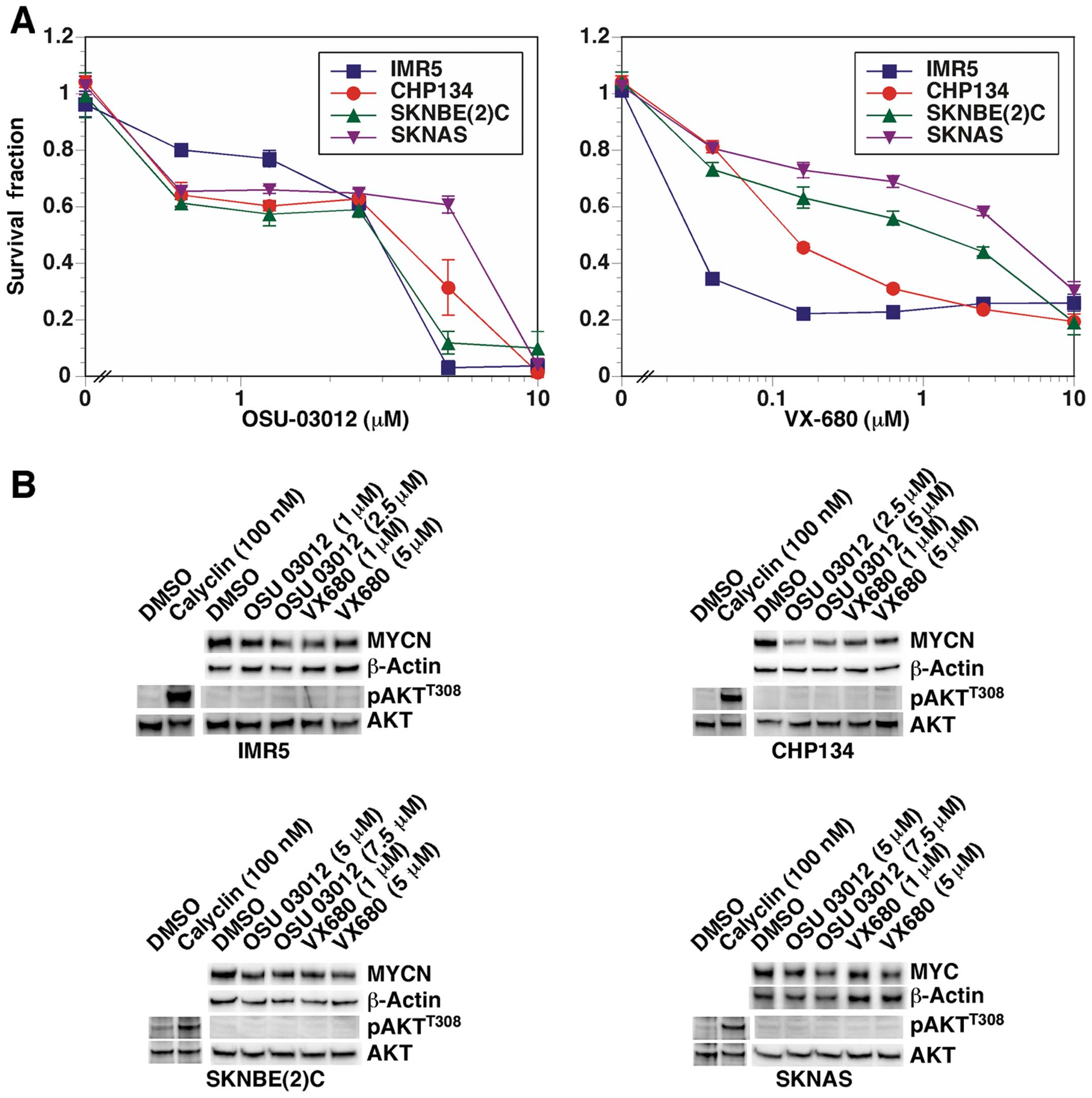 | Figure 2(A) OSU-03012 and VX-680 suppress the
growth of neuroblastoma cells. CHP134, IMR5, SKNBE(2)C and SKNAS
neuroblastoma cells were treated with OSU-03012 and VX-680 for 2
days and then subjected to MTS assay. (B) OSU-03012 and VX-680
destabilized MYC and MYCN, while AKT phosphorylation was not
affected. CHP134, IMR5, SKNBE(2)C and SKNAS neuroblastoma cells
were treated with OSU-03012 and VX-680 for 1 day and subjected to
western blot assay. Anti-pan MYC monoclonal antibody, NCM II 143,
was used to detect MYC and MYCN proteins. The rabbit anti-AKT
(polyclonal) and anti-pAKTT308 (monoclonal, clone D25E6)
antibodies were from Cell Signaling Technology Inc. (Danvers, MA,
USA). Calyculin-treated (for 30 min) CHP134, IMR5, SKNBE(2)C and
SKNAS cells were used to show that the anti-pAKTT308
could detect endogenous levels of pAKTT308. |
OSU-03012 and VX-680 destabilize MYC and
MYCN
We next assessed the effect of OSU-03012 and VX-680
on the stability of MYC and MYCN in the neuroblastoma cell lines.
As shown in Fig 2B, both compounds
destabilized MYC and MYCN at low μM concentrations following one
day of drug treatment.
OSU-03012 and VX-680 do not affect the
phosphorylation status of AKT
The status of AKT phosphorylation and the effect of
OSU-03012 and VX-680 on it were examined next. As shown in Fig. 2B, under the normal culture
condition, AKT was not phosphorylated at detectable levels nor was
its phosphorylation status affected following drug treatment. The
anti-pAKTT308 antibody used was functional, as it
detected pAKTT308 upon treatment of the neuroblastoma
cell lines with a protein phosphatase inhibitor, calyculin. This
observation suggests that PDK1 is not the main target of OSU-03012
in neuroblastoma cells.
In silico analysis reveals that OSU-03012
inhibits Aurora kinase A
FXG is a derivative of compound 6, an Aurora kinase
inhibitor, discovered through site-specific dynamic combinatorial
chemistry by Cancilla et al (15). Furthermore, co-crystallization of
FXG and Aurora A has been performed and the coordinates have been
deposited in the Brookhaven Protein Database (PDB ID: 3DAJ).
Therefore, we used this complex (FXG docking into Aurora A) as the
positive control to examine whether OSU-03012 has any likelihood of
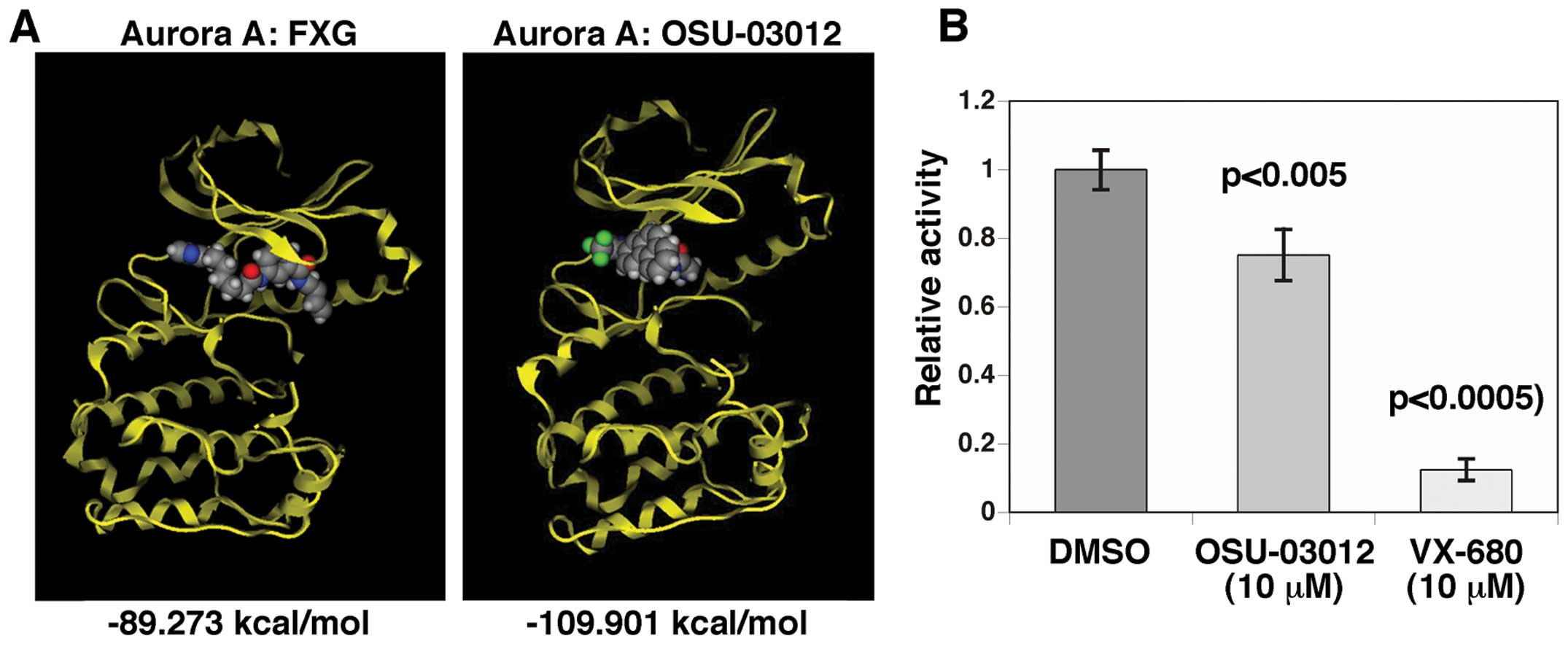
binding to Aurora kinase A. We performed in silico docking
analysis of OSU-03012 and Aurora kinase A. As shown in Fig. 3A, OSU-03012 exhibited a lower
calculated binding energy in comparison to a positive control
compound (FXG) against Aurora kinase A. This result suggests that
OSU-03012 has enough potential to bind to and exhibit an inhibitory
effect on Aurora kinase A.
OSU-03012 inhibits Aurora kinase A in an
in vitro assay
To further assess if, in fact, OSU-03012 can inhibit
Aurora kinase A, we performed an in vitro Aurora kinase A
inhibition assay. As shown in Fig.
3B, OSU-03012 inhibited Aurora kinase A, although its efficacy
was less than that of the potent Aurora kinase A inhibitor, VX-680
(16).
Discussion
It has been well documented that the stability of
MYC family proteins are in part regulated through their
phosphorylation status. ERK-mediated serine 62 (S62)
phosphorylation appears to be the first signal regulating the MYC
protein stability, which stabilizes MYC proteins. However, S62
phosphorylation triggers phosphorylation of MYC family proteins at
threonine 58 (T58) via GKS3. This leads to dephosphorylation of
S62. MYC proteins phosphorylated at T58 are then degraded through
the proteasome (Fig. 1) (7,9).
As shown in Fig. 1,
inhibition at any point in the RAF/MEK/ERK and PI3K/PDK1/AKT/GSK3
pathways by small-molecule inhibitors would therefore destabilize
MYC family proteins. We initially became interested in OSU-03012 as
this small-molecule inhibitor was reported to inhibit PDK1.
OSU-03012 is a derivative of celecoxib, which has a weak PDK1
inhibitory activity (6). Based on
this observation, Zhu et al developed OSU-03012 as a more
potent PDK1 inhibitor through structural optimization of
celecoxib.
In fact, treatment of neuroblastoma cells with
OSU-03012 resulted in destabilization of MYC family proteins
(Fig. 2). However, AKT, the
downstream effector of PDK1, was barely phosphorylated under normal
cell culture conditions, and OSU-03012 did not affect the status of
AKT phosphorylation. Thus, PDK1 is not a likely target of
OSU-03012, and this compound may affect another point of the
signaling cascades that regulate the stability of MYC family
proteins. Yacoub et al (17)
reported a similar observation that OSU-03012 activity was not
closely correlated with inhibition of PDK-1 and the phosphorylation
status of AKT, and argued that OSU-03012 must have additional
targets apart from PDK-1 in its cytotoxic actions in lung cancer
cells. Thus, the potential target of OSU-03012, apart from PDK1,
that could affect the stability of MYC family proteins remained
unknown.
Notably, it was reported that knockdown of Aurora
kinase A by RNAi considerably reduced the expression of MYC
(11). This observation and the
findings mentioned above collectively indicated that OSU-03012
could inhibit Aurora kinase A, hence reducing the stability of MYC
family proteins. In the present study, we investigated this
possibility. In fact, in silico docking analysis and an
in vitro Aurora kinase A inhibition assay demonstrated that
OSU-03012 can bind and inhibit Aurora kinase A.
To date, many small-molecule inhibitors of protein
kinases have been reported (18).
These inhibitors often have potencies of nM IC50 values
against cognate protein kinases in cell-free assay (19), and they are considered highly
selective. For example, VX-608 has an IC50 value of 36
nM against Aurora kinase A in cell-free assay (20). However, Bain et al (19) demonstrated that all of the
small-molecule protein kinase inhibitors that they tested had
substantial off-target effects on protein kinases other than their
cognate kinases. Collectively, our results suggest that OSU-03012
affects multiple cellular targets, including Aurora kinase A, to
exhibit its growth suppressive and MYC and MYCN-destabilizing
effects in neuroblastoma and other cancer cells.
Acknowledgements
The present study was supported by the NIH grant
CA127571 and a grant from the St. Baldrick’s Foundation.
References
|
1
|
Cohn SL, Pearson AD, London WB, et al: The
International Neuroblastoma Risk Group (INRG) classification
system: an INRG Task Force report. J Clin Oncol. 27:289–297. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brodeur GM, Seeger RC, Schwab M, Varmus HE
and Bishop JM: Amplification of N-myc in untreated human
neuroblastomas correlates with advanced disease stage. Science.
224:1121–1124. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Seeger RC, Brodeur GM, Sather H, et al:
Association of multiple copies of the N-myc oncogene with
rapid progression of neuroblastomas. New Engl J Med. 313:1111–1116.
1985.PubMed/NCBI
|
|
4
|
Wang LL, Suganuma R, Ikegaki N, et al:
Neuroblastoma of undifferentiated subtype, prognostic significance
of prominent nucleolar formation, and MYC/MYCN protein expression:
a report from the Children’s Oncology Group. Cancer. 119:3718–3726.
2013.PubMed/NCBI
|
|
5
|
Kucab JE, Lee C, Chen CS, et al: Celecoxib
analogues disrupt Akt signaling, which is commonly activated in
primary breast tumours. Breast Cancer Res. 7:R796–R807. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhu J, Huang JW, Tseng PH, et al: From the
cyclooxygenase-2 inhibitor celecoxib to a novel class of
3-phosphoinositide-dependent protein kinase-1 inhibitors. Cancer
Res. 64:4309–4318. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sears R, Nuckolls F, Haura E, Taya Y,
Tamai K and Nevins JR: Multiple Ras-dependent phosphorylation
pathways regulate Myc protein stability. Genes Dev. 14:2501–2514.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Adhikary S and Eilers M: Transcriptional
regulation and transformation by Myc proteins. Nat Rev Mol Cell
Biol. 6:635–645. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schulein C and Eilers M: An unsteady
scaffold for Myc. EMBO J. 28:453–454. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Marampon F, Ciccarelli C and Zani BM:
Down-regulation of c-Myc following MEK/ERK inhibition halts the
expression of malignant phenotype in rhabdomyosarcoma and in non
muscle-derived human tumors. Mol Cancer. 5:312006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang H, Liu Q, Cheng GZ, He L, Nicosia SV
and Cheng JQ: Crosstalk between Aurora-A and GSK3β/β-catenin
pathways induces c-Myc and cyclin D1. Proc Amer Assoc Cancer Res.
46:55072005.
|
|
12
|
Tang XX, Zhao H, Robinson ME, et al:
Implications of EPHB6, EFNB2, and EFNB3 expressions
in human neuroblastoma. Proc Natl Acad Sci USA. 97:10936–10941.
2000.
|
|
13
|
Regan PL, Jacobs J, Wang G, et al: Hsp90
inhibition increases p53 expression and destabilizes MYCN and MYC
in neuroblastoma. Int J Oncol. 38:105–112. 2011.PubMed/NCBI
|
|
14
|
Torres J, Regan PL, Edo R, et al:
Biological effects of induced MYCN hyper-expression in
MYCN-amplified neuroblastomas. Int J Oncol. 37:983–991.
2010.PubMed/NCBI
|
|
15
|
Cancilla MT, He MM, Viswanathan N, et al:
Discovery of an Aurora kinase inhibitor through site-specific
dynamic combinatorial chemistry. Bioorg Med Chem Lett.
18:3978–3981. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Harrington EA, Bebbington D, Moore J, et
al: VX-680, a potent and selective small-molecule inhibitor of the
Aurora kinases, suppresses tumor growth in vivo. Nat Med.
10:262–267. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yacoub A, Park MA, Hanna D, et al:
OSU-03012 promotes caspase-independent but PERK-, cathepsin B-,
BID-, and AIF-dependent killing of transformed cells. Mol
Pharmacol. 70:589–603. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sawyer TK, Wu JC, Sawyer JR and English
JM: Protein kinase inhibitors: breakthrough medicines and the next
generation. Expert Opin Investig Drugs. 22:675–678. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bain J, Plater L, Elliott M, et al: The
selectivity of protein kinase inhibitors: a further update. Biochem
J. 408:297–315. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tyler RK, Shpiro N, Marquez R and Eyers
PA: VX-680 inhibits Aurora A and Aurora B kinase activity in human
cells. Cell Cycle. 6:2846–2854. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Segerstrom L, Baryawno N, Sveinbjornsson
B, et al: Effects of small molecule inhibitors of PI3K/Akt/mTOR
signaling on neuroblastoma growth in vitro and in
vivo. Int J Cancer. 129:2958–2965. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Otto T, Horn S, Brockmann M, et al:
Stabilization of N-Myc is a critical function of Aurora A in human
neuroblastoma. Cancer Cell. 15:67–78. 2009. View Article : Google Scholar : PubMed/NCBI
|