Introduction
In spite of the recent advances in cytoreductive
surgery and chemotherapeutic agents, epithelial ovarian cancer
(EOC) remains the leading cause of cancer-related death in women
diagnosed with gynecologic malignancies (1). Cell cycle dysregulation is closely
related to the development of many malignant tumors, including
ovarian cancer.
Polo-like protein kinase 1 (PLK1) is a highly
conserved cyclin-dependent serine/threonine kinase, which is
closely related to several events in eukaryotic cells during
mitosis, such as centrosome replication, spindle formation,
chromosome segregation, cytokinesis, and DNA damage repair
(2,3). The activities of PLKl are elevated in
a variety of human cancer cells and is implicated in the poor
prognosis of lung cancer, colorectal cancer, breast cancer,
prostate cancer, malignant melanoma, lymphoma and bladder cancer
(4–10). Tumor-suppressor gene P53 is
implicated in the regulation of cell cycle and in tumorigenesis,
and also plays an important role in the cell cycle G2/M DNA damage
checkpoint regulation and replication arrest (11). More than 50% of human cancers
contain P53 gene mutations and mutant P53 protein expression
(12). P21WAF1 promotes
cell cycle arrest in response to many stimuli, such as DNA damage,
and acts as an important trancriptional target of P53, and
regulates cyclin-dependent kinase (CDK)1 and cyclinB1 complexes in
the G2/M DNA damage checkpoint and cell cycle transition (13). P21WAF1 is a tumor
suppressor, but it also can behave as an oncogene in certain
cellular contexts in human tumors (14).
In the present study, we inhibited the expression of
PLK1 and P53 in ovarian cancer SK-OV-3 cells by RNA interference
and detected the expression levels of PLK1, P53 and
P21WAF1 in different ovarian tissues. We evaluated the
associations between their expression levels and the clinical
pathological factors of the EOC cases. We also determined the
survival time of EOC patients by Kaplan-Meier analysis, and aimed
to explore the role of concomitant PLK1, P53 and P21WAF1
expression in the prognosis of ovarian cancer.
Materials and methods
Cell transfection
Human ovarian carcinoma SK-OV-3 cells were purchased
from the Chinese Academy of Sciences Cell Bank (Shanghai, China),
and cultured in completed RPMI-1640 medium (HyClone, Logan, Utah,
USA), at 37°C with 5% CO2. Cells were harvested in a
logarithmic phase of growth for all experiments as described below.
PLK1 and P53 shRNA lentiviral plasmids (Santa Cruz Biotechnology,
Santa Cruz, CA, USA) were used for cell transfection, respectively,
which was performed following the protocol of the shRNA Plasmid
Transfection reagent (Santa Cruz Biotechnology). Stably transfected
SK-OV-3 cells were isolated by puromycin (Clontech, Mountain View,
CA, USA) selection after transfection for 48 h. Three cell groups
were used for the next step study: SK-OV-3, PLK1 shRNA SK-OV-3 and
P53 shRNA SK-OV-3 cells.
sqRT-PCR
Cell total RNA was isolated using TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA), and first-strand
cDNA was synthesized from 1 μg total RNA according to the protocol
of the RevertAid First Strand cDNA Synthesis kit (Fermentas, EU).
Primers used in sqRT-PCR were PLK1, P53 and P21WAF1
(Santa Cruz Biotechnology), and β-actin sense,
5′-ACGCACCCCAACTACAACTC-3′ and anti-sense,
5′-TCTCCTTAATGTCACGCACGA-3′. PCR cycling parameters included:
denaturation (94°C, 30 sec), annealing (56°C, 30 sec) and extension
(72°C, 30 sec). Equal amounts of PCR products were electrophoresed
on 1.2% agarose gels and visualized by ethidium bromide staining.
The specific bands of the PCR products were analyzed by Image-Pro
Plus 6.0 system, β-actin was used as a control for normalization.
RT-PCR was performed 3 times independently.
Western blot analysis
The antibodies used in the western blot analysis,
following the manufacturer’s protocols, were mouse anti-human
monoclonal PLK1, rabbit anti-human polyclonal growth arrest and DNA
damage-inducible gene 45 (GADD45)a, goat anti-human polyclonal
14-3-3σ (Santa Cruz Biotechnology), mouse anti-human monoclonal
P53, rabbit anti-human polyclonal phospho-P53 (Ser15), mouse
anti-human monoclonal P21WAF1, and mouse anti-human
monoclonal β-actin (Beyotime Biotechnology, Haimen, Jiangsu,
China). Total protein was extracted using RIPA lysis buffer for
western blot analysis and IP (Beyotime Biotechnology), and the
protein concentration was determined using the BCA assay. Equal
amounts of protein (30 μg) were separated by 10% SDS-PAGE and
transferred onto PVDF membranes. The detection of hybridized
protein was performed using an enhanced chemiluminescence kit
(Zhongshan Golden Bridge Biotechnology, Peking, China), β-actin was
used as a control for normalization. The relative values of
specific bands were analyzed by Image-Pro Plus 6.0 system.
MTT assay
Cells (1×104 cells/well) were planted
into 96-well plates, and 100 μl medium containing 10% FBS was added
into each well. Five duplicate wells were set up for each group.
Cells were cultured continuously for 7 days, and 20 μl MTT reagent
(5 μg/ml; Sigma-Aldrich, St. Louis, MO, USA) was added into each
well, and incubated for another 4 h. The initial medium was
aspirated and 150 μl DMSO was added. The absorbance of the samples
was measured by a microplate spectrophotometer (Thermo Spectronic,
Madison, WI, USA) at 492 nm. All experiments were conducted in
triplicate. A cell growth curve was plotted vs. time by Origin 8
software.
Flow cytometric analysis
Approximately 1×106 cells were put into a
single-cell suspension and treated with PBS solution, and were
prepared following the manufacturer’s protocol of the Annexin
V-FITC Apoptosis Detection kit (Beyotime Biotechnology). Then, the
rates of apoptosis were analyzed with the FACScan system (BD
Biosciences, San Jose, CA, USA).
Matrigel invasion assay
A Transwell chamber (8-μm pore size; Millipore,
Bedford, MA, USA) covered with 100 μl of 1 μg/ml Matrigel (BD
Biosciences, Franklin Lakes, NJ, USA) was used to measure cell
invasive ability. Cells (1×105) were seeded into each
upper chamber with 200 μl fresh medium without FBS, and 500 μl
medium with 20% FBS was added into each lower chamber. Three
duplicate wells were set up for each group. After 12 h, the cells
were fixed with methanol for 5 min, and the cells were stained by
hematoxylin for 30 min. The upper chamber was cleaned and inverted,
and cell numbers were counted on the lower membrane under a high
power lens (x400) in 5 random visual fields.
Animal model
The experimental protocol was approved by the
Zhengzhou University Ethics Committee for Animal Experimentation.
Female BALB/c nude mice (4–5 weeks old, 13–17 g) were purchased
from Vital River Laboratory Animal Technology Co., Ltd. (Peking,
China), and were randomly assigned into 3 groups with 10 mice per
group. Approximately 1×1012 cells were suspended in 2 μl
PBS and intraperitoneally injected into each mouse, respectively.
The survival time was recorded for each mouse.
Patients and tissue samples
Ninety-one ovarian specimens were obtained from
patients during surgery at the Department of Gynaecology, The First
Affiliated Hospital of Zhengzhou University (from May 2008 to
August 2010). The samples consisted of 20 specimens of normal
ovarian tissues (obtained from patients who underwent hysterectomy
and oophorectomy for multiple uterine myoma other than ovarian
tumors), 19 specimens of benign ovarian tumor tissues (10 serous
and 9 mucinous cystadenomas), and 52 specimens of EOC tissues (30
serous and 22 mucinous cystadenocarcinomas). The median age of the
ovarian cancer patients was 53 years (range from 21 to 75). All of
the ovarian cancer patients did not receive preoperative
radiochemotherapy. The enrolled ovarian cancer patients all
received comprehensive surgical staging and standardized
postoperative chemotherapy (paclitaxel combined with platinum), and
relapsed patients received second-line chemotherapy drugs for
combination chemotherapy. The tissue samples were collected after
surgical resection immediately and promptly saved in liquid
nitrogen. The consent of all enrolled patients was obtained for
sample collection before surgery, and the present study was
approved by the Local Ethics Committee of Zhengzhou University. All
tissue samples were verified independently by 2 pathologists before
IHC by H&E staining.
Immunohistochemistry
Immunohistochemical staining was performed following
the protocol of the Universal SP kit (Zhongshan Golden Bridge
Biotechnology). For PLK1, P53 and P21WAF1 protein,
staining localized in the nucleus was considered positive.
Immunoreactive scoring was performed by 2 pathologists
independently using Image J; semi-quantitative counting method was
used to determine positive staining.
Statistical analysis
Values are expressed as mean ± standard deviation
(SD). Count data were analyzed by χ2 test and Fisher’s
exact test. Measurement data were analyzed by one-way ANOVA and
Bonferroni’s test using SPSS 17.0 software package. A difference
was considered statistically significant when P-value was
<0.05.
Results
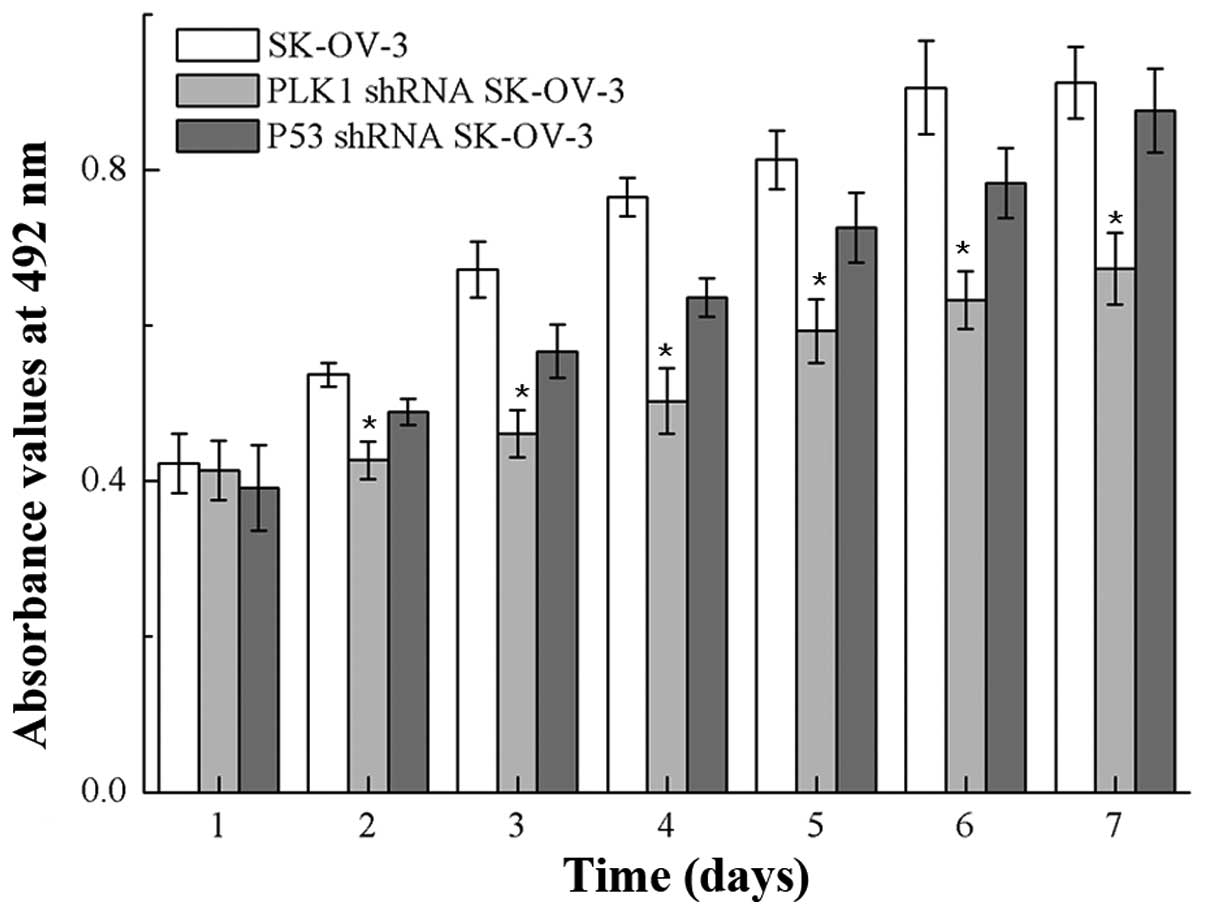
Suppression of cell proliferation after
RNA interference
After knockdown of PLK1 or P53, cell proliferation
was obviously suppressed from day 2 when compared with the control
cells, and a difference in proliferation was also detected between
the PLK1-null and P53-null SK-OV-3 cells (P<0.05) (Fig. 1).
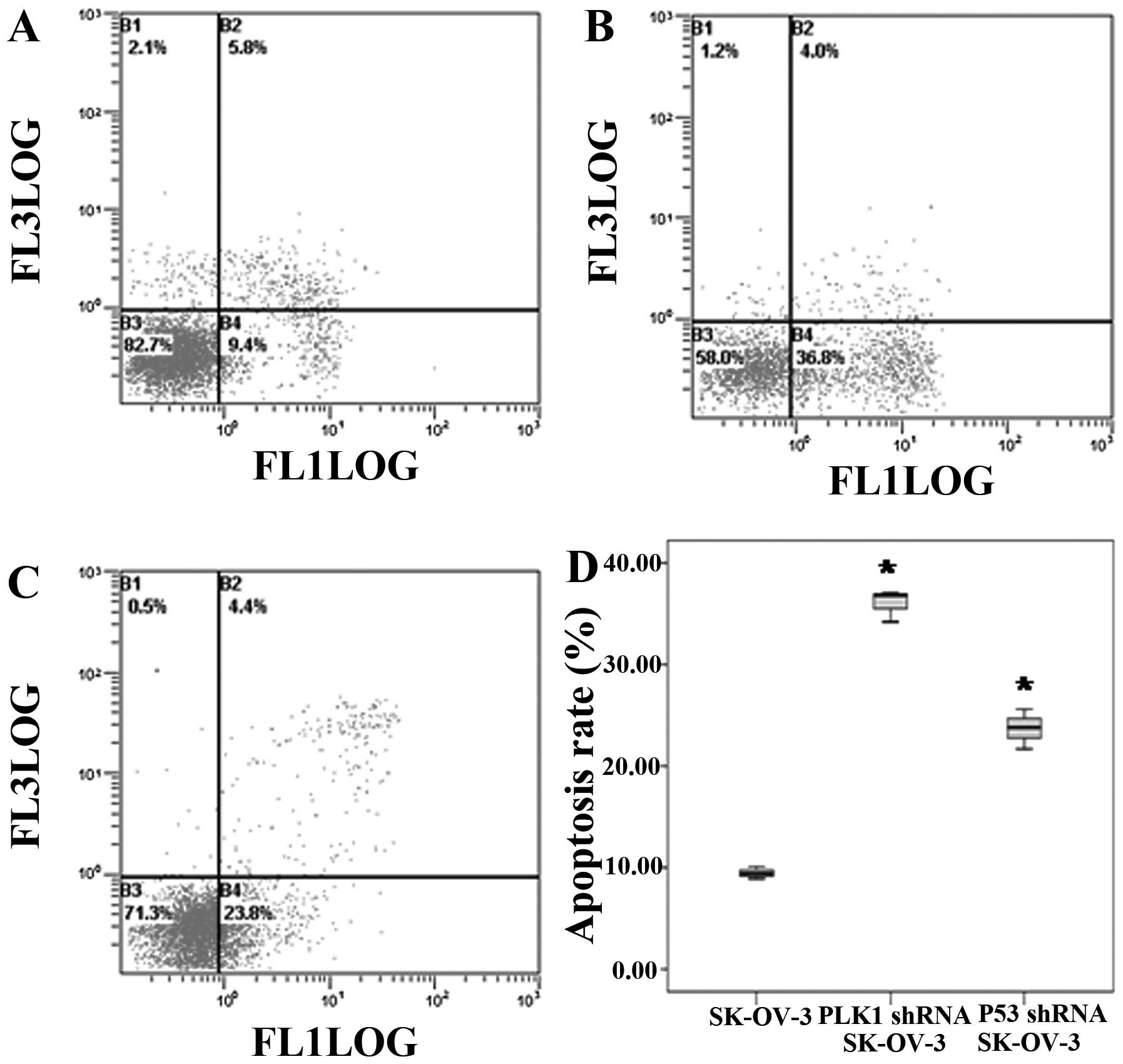
Cell apoptosis is induced by RNA
interference
After inhibition of PLK1 or P53, the cell apoptosis
rates were markedly increased (F=236.833, P<0.05), and a
difference in apoptosis was also detected between the PLK1-null and
P53-null SK-OV-3 cells (P<0.05) (Fig. 2).
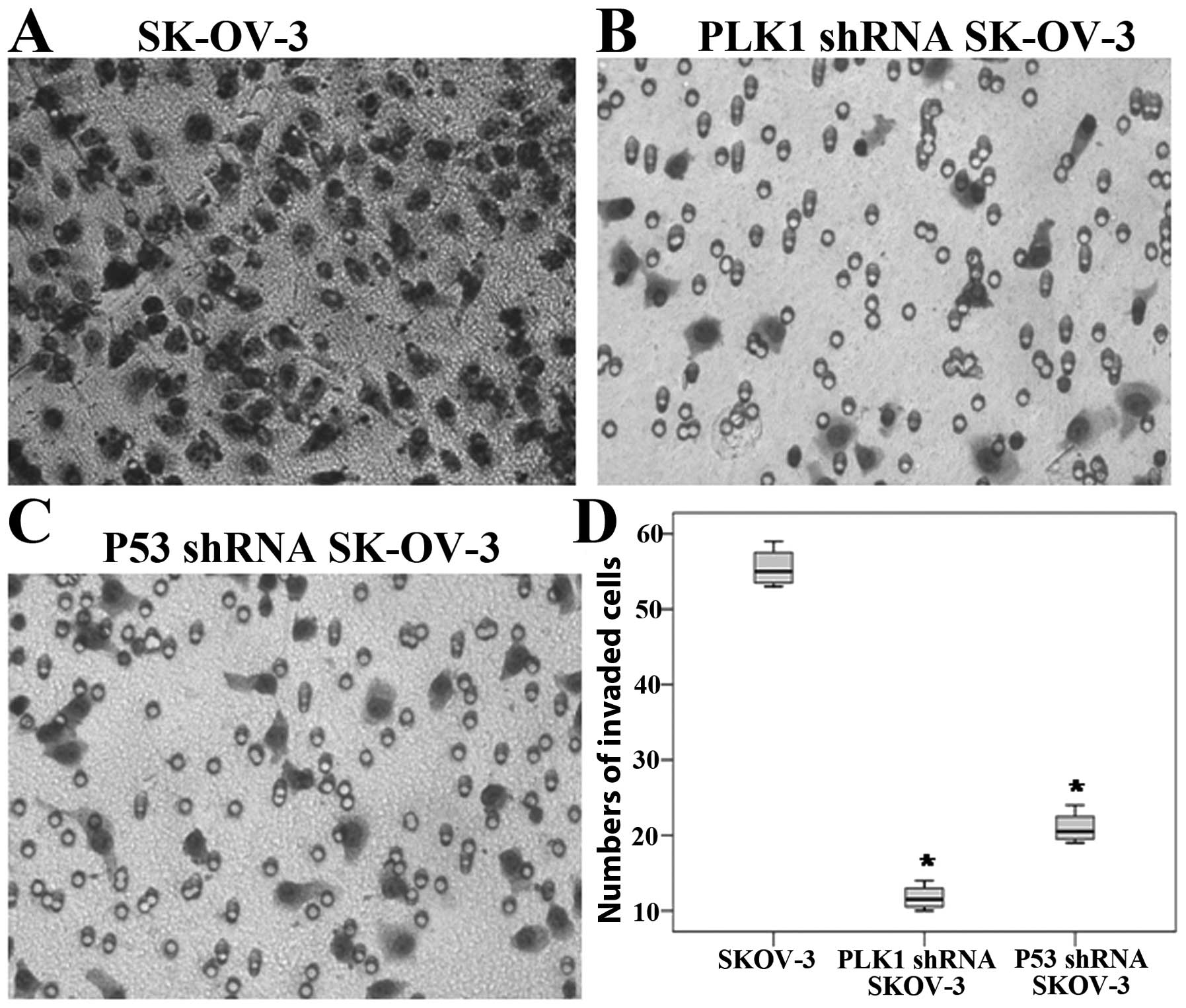
Cell invasion is inhibited by RNA
interference
In the Matrigel invasion assay, the number of
invaded cells on the lower membrane was markedly decreased in the
PLK1-null and P53-null SK-OV-3 cells compared to the control
(F=437.469, P<0.05), and a difference was also detected between
the PLK1-null and P53-null SK-OV-3 cells (P<0.05) (Fig. 3).
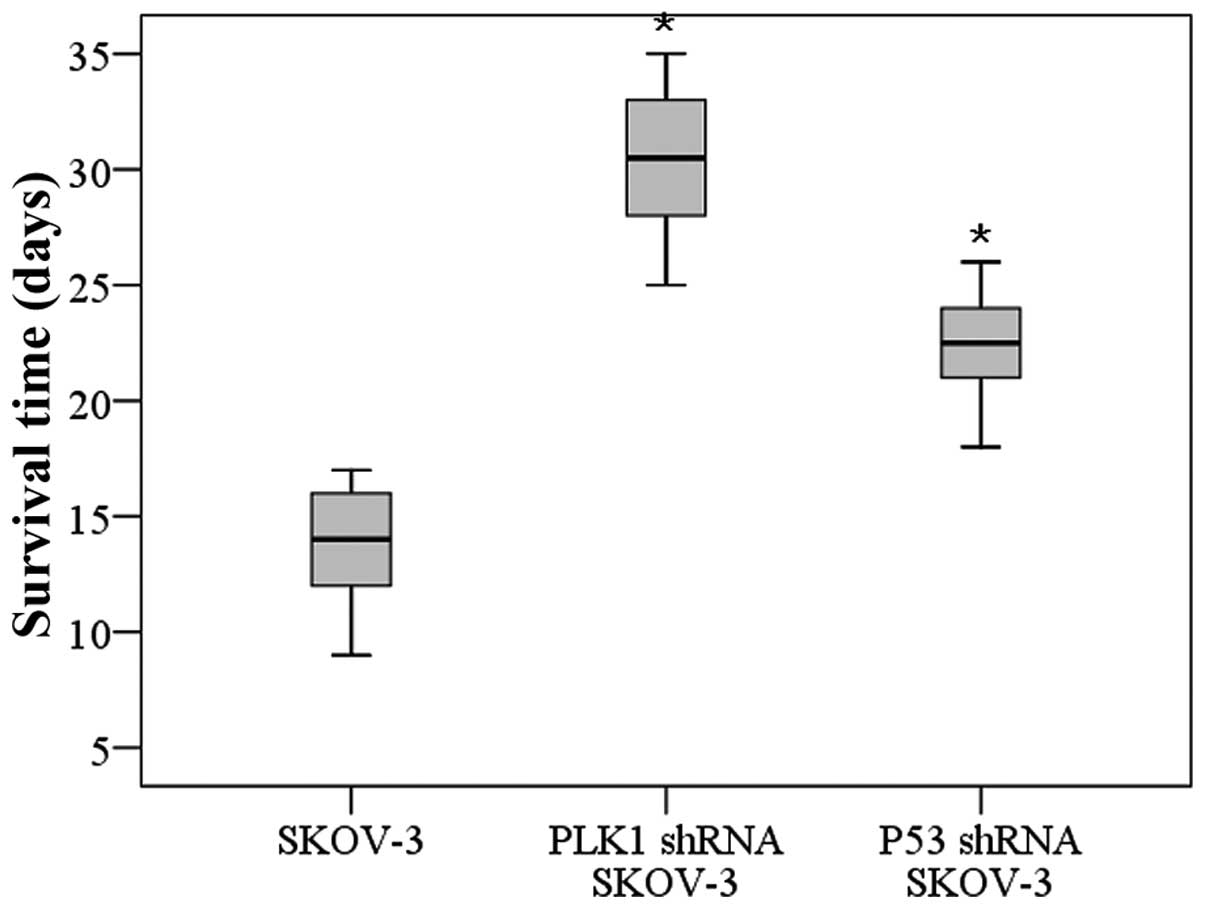
Survival time of animals is prolonged
after RNA interference
The survival time of the animals injected with
PLK1-null and P53-null SK-OV-3 cells was obviously prolonged when
compared to the survival of mice injected with the SK-OV-3 cells
(F=95.703, P<0.05), and a difference was also detected between
the PLK1-null and P53-null SK-OV-3 cell groups (P<0.05)
(Fig. 4).
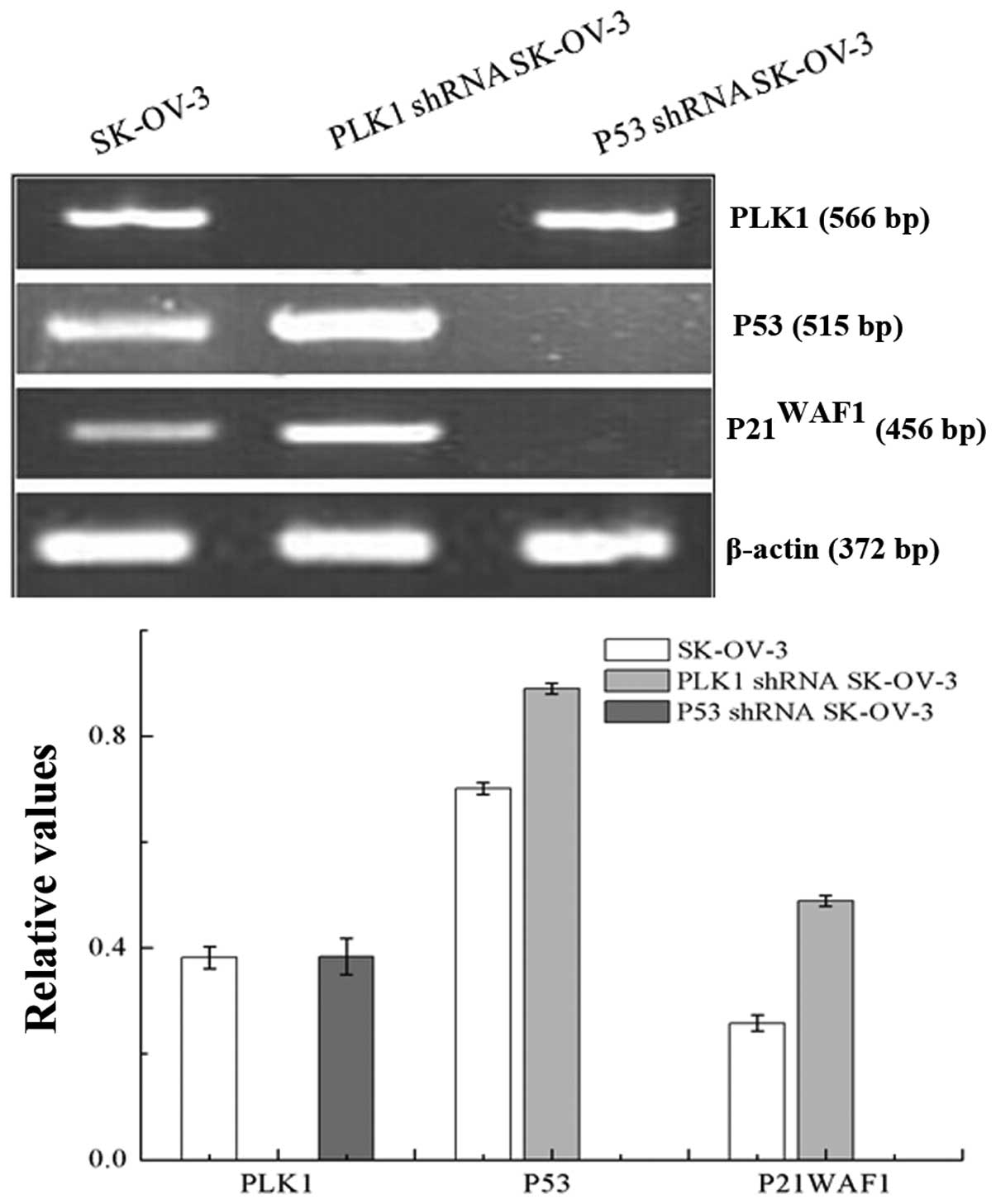
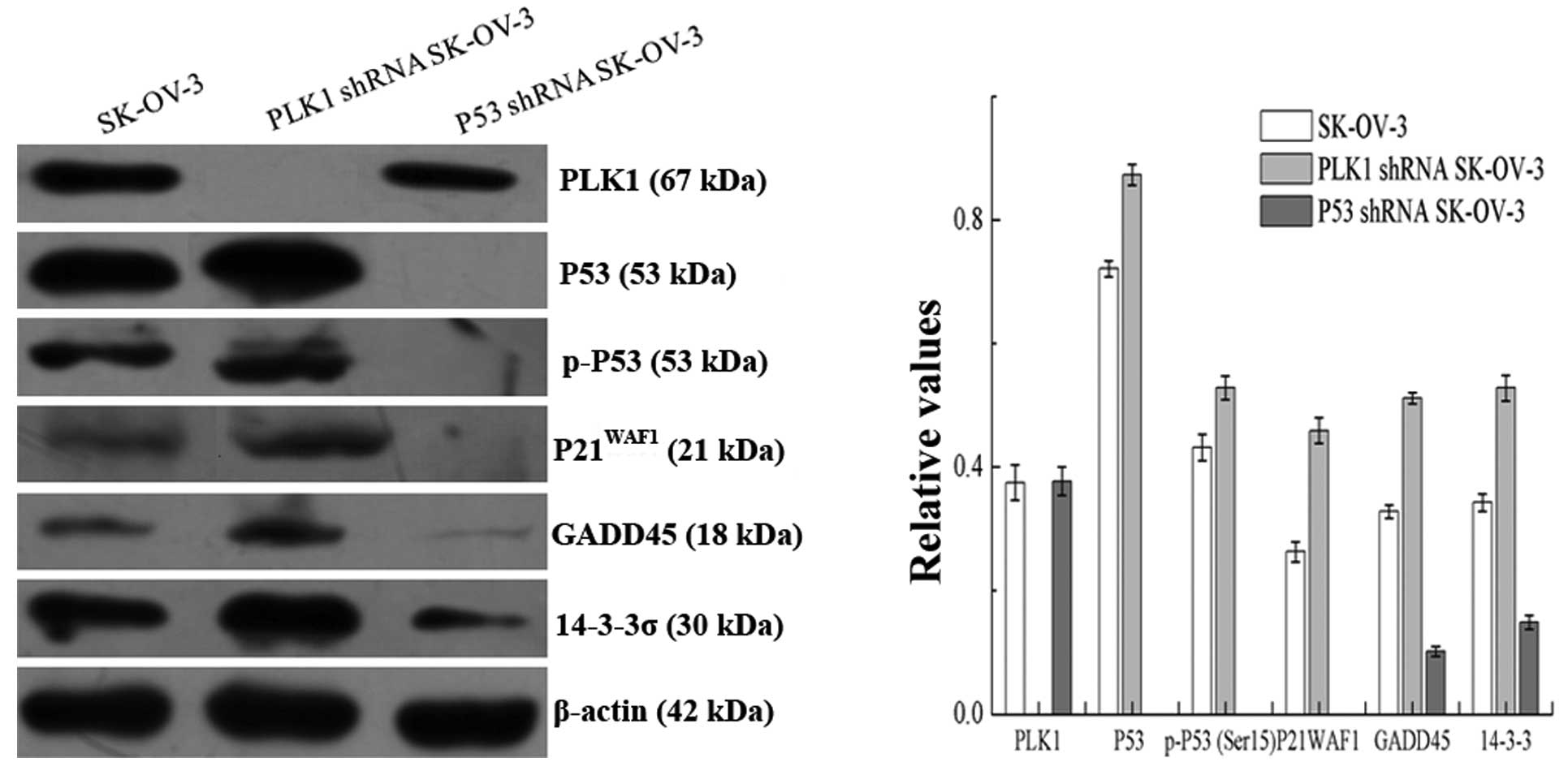
Protein and mRNA expression changes in
the SK-OV-3 cells after RNA interference
As a result of PLK1 knockdown, a significant
upregulation in the levels of P53, p-P53 (Ser15),
P21WAF1, GADD45 and 14-3-3σ was observed in the PLK1
shRNA SK-OV-3 cells. After P53 knockdown, levels of P53, p-P53
(Ser15), P21WAF1, GADD45 and 14-3-3σ were markedly
decreased in the P53 shRNA SK-OV-3 cells, without obvious changes
in PLK1. However, differences in the fold-change were noted in the
PLK1-null and P53-null SK-OV-3 groups (Figs. 5 and 6).
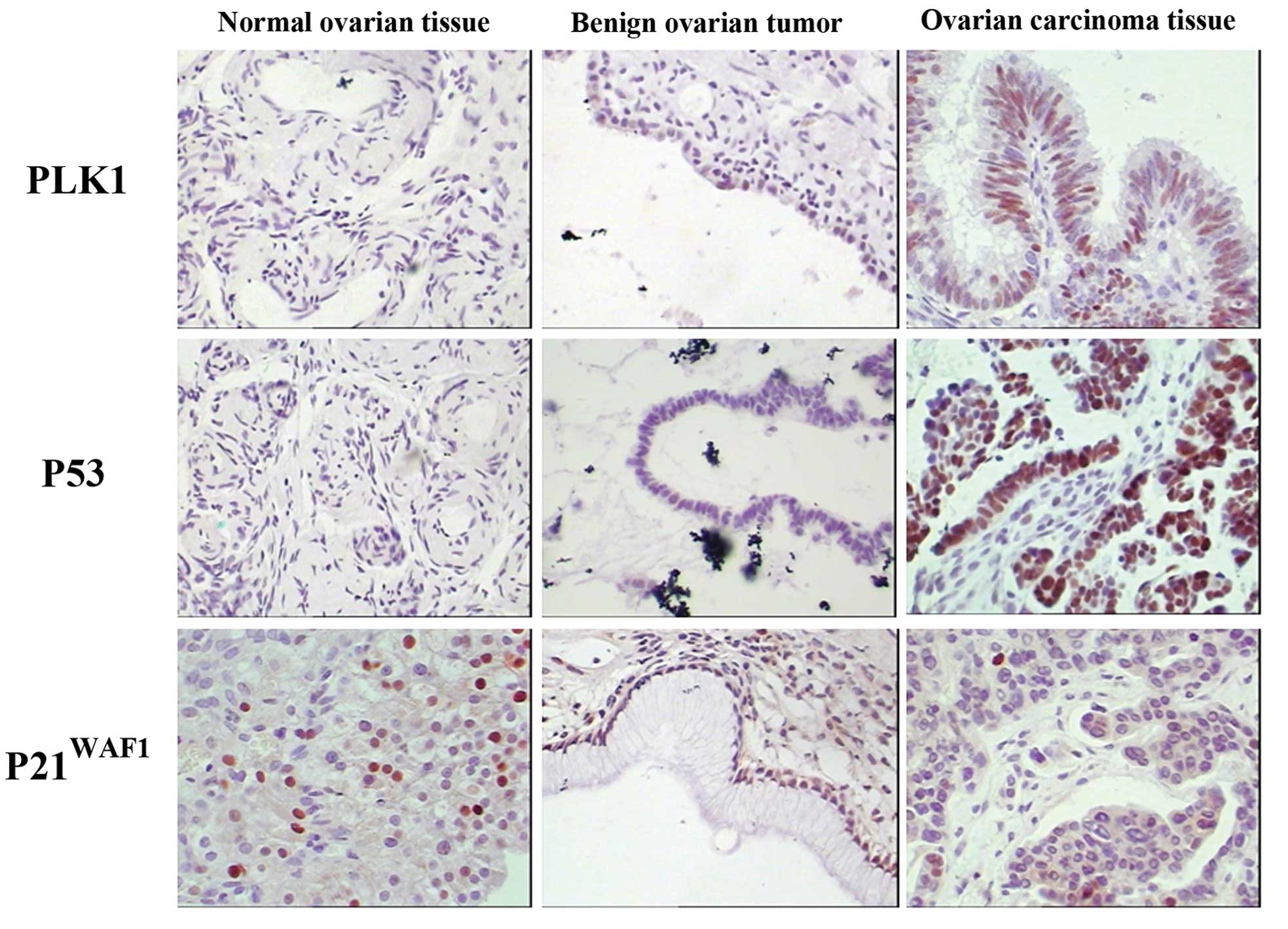
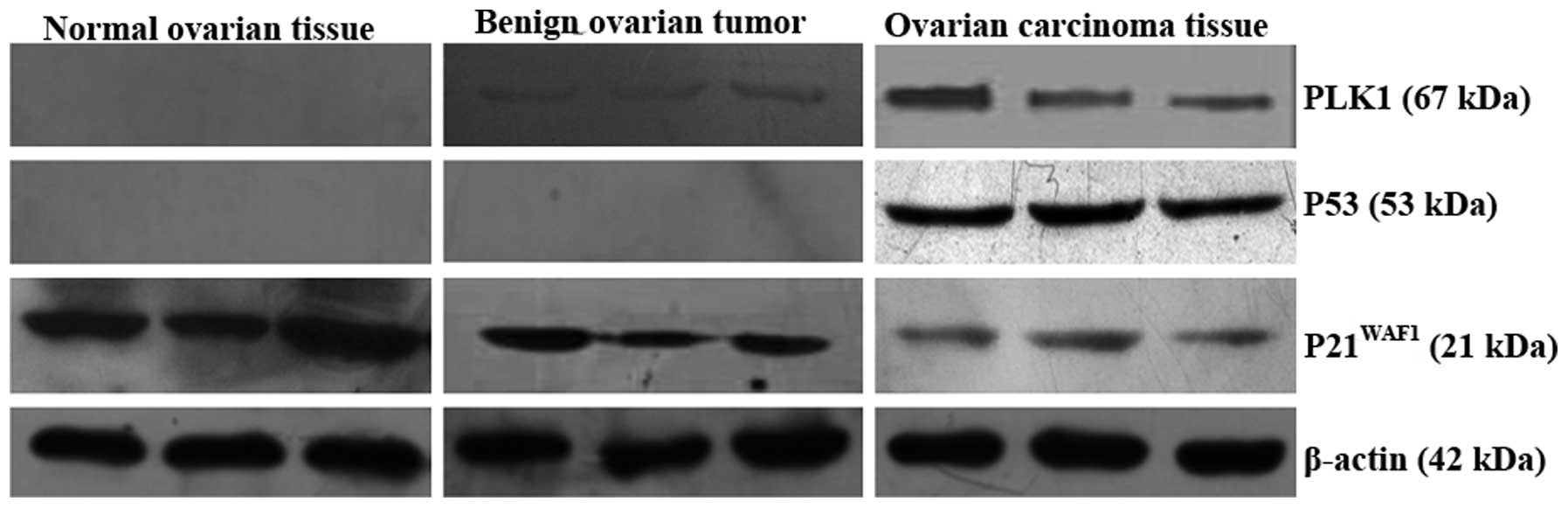
Expression of PLK1, P53 and
P21WAF1 in the different ovarian tissues
In the EOC tissues, positive staining for PLK1 and
P53 was obviously higher than that in the benign ovarian tumor and
normal ovarian tissues (χ2=17.266, P<0.05;
χ2=42.656, P<0.05). P21WAF1 was markedly
lower in the ovarian cancer tissues (χ2=20.270,
P<0.05) (Table I and Fig. 7). Consistent results were observed
in the western blot analysis (Fig.
8).
 | Table IExpression of PLK1, P53 and
P21WAF1 in the different ovarian tissues as detected by
immunohistochemistry. |
Table I
Expression of PLK1, P53 and
P21WAF1 in the different ovarian tissues as detected by
immunohistochemistry.
| | PLK1 | P53 |
P21WAF1 |
|---|
| |
|
|
|
|---|
| Tissue types | N | − | + | ++ | Positive rate
(%) | − | + | ++ | Positive rate
(%) | − | + | ++ | Positive rate
(%) |
|---|
| Normal ovarian
tissues | 20 | 20 | 0 | 0 | 0.0 | 20 | 0 | 0 | 0.0 | 2 | 18 | 0 | 90.0 |
| Benign ovarian
tumors | 19 | 18 | 1 | 0 | 5.6 | 19 | 0 | 0 | 0.0 | 7 | 12 | 0 | 63.2 |
| Ovarian carcinoma
tissues | 52 | 32 | 11 | 9 | 38.5a,b | 17 | 13 | 22 | 67.3a | 35 | 11 | 6 | 32.7a |
Association of PLK1, P53 and
P21WAF1 expression with the clinicopathological
characteristics of the ovarian cancer case
As shown in Table
II, the expression levels of PLK1 and P53 were significantly
associated with FIGO stage and histological differentiation, and
P21WAF1 was obviously associated with FIGO stage in the
ovarian carcinoma cases (P<0.05).
 | Table IIRelationship between expression of
PLK1, P53 and P21WAF1 in ovarian cancer and clinical
characteristics of the epithelial ovarian cancer cases. |
Table II
Relationship between expression of
PLK1, P53 and P21WAF1 in ovarian cancer and clinical
characteristics of the epithelial ovarian cancer cases.
| | PLK1 | P53 |
P21WAF1 |
|---|
| |
|
|
|
|---|
| Groups | N | + | % | P-value | + | % | P-value | + | % | P-value |
|---|
| Age (years) |
| <50 | 12 | 4 | 33.3 | 0.747a | 6 | 50.0 | 0.173a | 5 | 41.7 | 0.496a |
| ≥50 | 40 | 16 | 40.0 | | 29 | 72.5 | | 12 | 30.0 | |
| FIGO stage |
| I–II | 16 | 2 | 12.5 | 0.014 | 6 | 37.5 | 0.004 | 10 | 62.5 | 0.002 |
| III–IV | 36 | 18 | 50.0 | | 29 | 80.6 | | 7 | 11.8 | |
| Histological
differentiation |
| G1 | 10 | 0 | 0.0 | 0.008a | 3 | 30.0 | 0.009a | 4 | 40.0 | 0.711a |
| G2–G3 | 42 | 20 | 16.2 | | 32 | 76.2 | | 13 | 31.0 | |
| Lymph node
metastasis |
| Yes | 20 | 10 | 50.0 | 0.176 | 16 | 80.0 | 0.123 | 8 | 40.0 | 0.374 |
| No | 32 | 10 | 31.3 | | 19 | 59.4 | | 9 | 28.1 | |
| Ascites |
| Yes | 28 | 11 | 39.3 | 0.895 | 20 | 71.4 | 0.494 | 10 | 35.7 | 0.616 |
| No | 24 | 9 | 37.5 | | 15 | 62.5 | | 7 | 29.2 | |
Correlations between PLK1, P53 and
P21WAF1 in 52 EOC cases
Firstly, 14 subjects were PLK1-positive and
P53-negative, and 29 were PLK1-negative and P53-positive, which
suggested a negative correlation between the expression of PLK1 and
P53 (r=−0.629, P<0.05). Secondly, 29 subjects were P53-positive
and P21WAF1-negative, and 11 patients were P53-negative
and P21WAF1-positive, which indicated a negative
correlation between P53 and P21WAF1 (r=−0.476,
P<0.05). Thirdly, no correlation was observed between expression
of PLK1 and P21WAF1 (P>0.05).
Univariate and multivariate logistic
regression analysis
Based on the present data, PLK1 (OR=3.333, P=0.040,
95% CI: 0.102–1.092), P53 (OR=15.080, P<0.05, 95% CI:
3.713–65.252), P21WAF1 (OR=21.459, P=0.017, 95% CI:
3.192–68.521), FIGO stage (OR=26.867, P<0.05, 95% CI:
5.584–129.274), histological differentiation (OR=18.238, P=0.035,
95% CI: 1.267–48.121) and lymph metastasis (OR=5.000, P=0.025, 95%
CI: 1.221–20.483) were the prognosis factors of ovarian cancer
shown by univariate logistic regression analysis. However, only
PLK1 (OR=2.288, P=0.025, 95% CI: 0.105–50.050) was an independent
prognostic factor for ovarian cancer, which was analyzed by
multivariate logistic regression analysis.
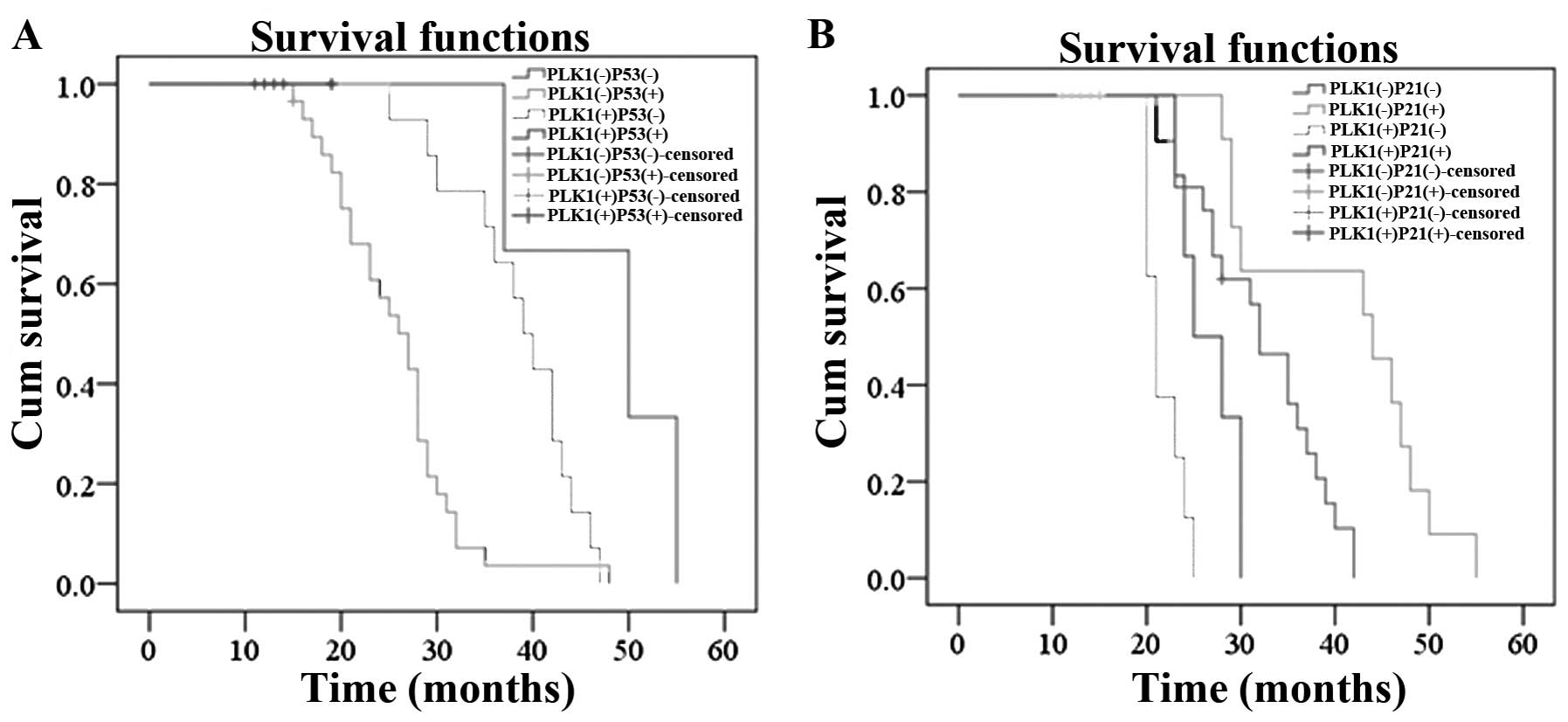
Kaplan-Meier analysis
Up to December 2012, the median follow-up was 29
months for 52 ovarian cancer patients (range from 11 to 55 months);
45 patients were alive at the end of the follow-up, 34 subjects had
relapsed or succumbed to the disease (Table III). When PLK1 and P53 were
combined to evaluate the overall survival time, we found that the
overall survival of PLK1-positive/P53-positive subjects was
obviously shorter than the survival of the other expression groups
in the 52 ovarian cancer patients (χ2=17.246, P=0.037).
When assessing PLK1 combined with P21WAF1, the overall
survival time of the subjects with
PLK1-positive/P21WAF1-negative expression was markedly
shorter than patients in the other expression groups
(χ2=48.428, P<0.05) (Fig.
9).
 | Table IIIFollow-up of 52 ovarian cancer
patients after surgery. |
Table III
Follow-up of 52 ovarian cancer
patients after surgery.
| | | Relapse | |
|---|
| | |
| |
|---|
| FIGO stage | N | Stable | ≤6 months | >6 months | Death |
|---|
| I | 5 | 5 | 0 | 0 | 0 |
| II | 11 | 8 | 1 | 2 | 0 |
| III | 24 | 4 | 12 | 7 | 1 |
| IV | 12 | 1 | 5 | 0 | 6 |
Discussion
PLK1 exhibits high activity in tissues and cells
with a high mitotic index, and plays an important role in cell
cycle progression. Inhibition of the activity of PLK1 enables cell
cycle arrest in the G2/M phase (15,16).
Wild-type P53 is a tumor-suppressor gene, which plays an important
role in the regulation of the cell cycle and tumor outcome. P53
upregulates cell growth arrest and apoptosis-related genes in
response to stress signals, thereby influencing programmed cell
death, cell differentiation, and cell cycle control mechanisms. P53
is required for DNA damage checkpoint and functions to upregulate
expression of its 3 trancriptional targets, GADD45, 14-3-3σ and
P21WAF1 (17,18). In response to DNA damage and other
stimuli, P21WAF1 can bind to and inhibit the kinase
activity of CDKs leading to growth arrest at specific stages in the
cell cycle (14). GADD45, a protein
involved in DNA replication and repair, binds to both CDKs and
PCNA, and serves as a link between P53-dependent cell cycle
checkpoint and DNA repair (18). In
response to tumor growth, 14-3-3σ increases the rate of
P53-regulated inhibition of G2/M cell cycle progression (19).
It has been proven that P53 is a critical target of
PLK1, and its function is abrogated through the physical
interaction with PLK1. PLK1 binds to P53 at the P53-binding domain,
amino acid sequence comprising residues from 99 to 329 of PLK1, and
inhibits its transactivation activity as well as its pro-apoptotic
function (20). The present in
vitro results showed that the cell proliferation was inhibited,
the apoptosis rate was increased, and cell invasion was suppressed
after PLK1 or P53 knockdown in SK-OV-3 cells. The survival time was
prolonged after PLK1 or P53 knockdown in the animals. Furthermore,
a notable phenomenon shown in the present study, was that the
inhibitory effects of PLK1 knockdown were more obvious than P53
knockdown in the SKOV-3 cells. Inhibition of PLK1 in SKOV-3 cells
increased the expression of P53, p-P53 (Ser15), P21WAF1,
GADD45 and 14-3-3σ. In contrast, knockdown of P53 decreased the
expression of P53, p-P53 (Ser15), P21WAF1, GADD45 and
14-3-3σ, without obvious changes in PLK1. These data suggest that
abnormal expression of PLK1 implicated in ovarian carcinogenesis
may owe its ability to inhibition of the activity of P53.
Increasing evidence suggests that the level of PLK1
expression has prognostic value for predicting outcomes in patients
with various types of cancers such as lung cancer, squamous cell
carcinomas of the head and neck, melanomas, and ovarian carcinomas
(21,22). Mutation and overexpression of P53
are common in EOC; positive staining for P53 was detected in almost
all different subtypes of ovarian cancer, and the group of patients
with P53-positive tumors showed worse survival (23,24).
Overexpression of P53 is not a feature of benign epithelial ovarian
tumors or early-stage borderline ovarian tumors (25). P21WAF1 is often
misregulated in human cancers including ovarian cancer, and
patients with downregulation of P21WAF1 were found to
show worse survival time in ovarian serous carcinoma (26,27).
Our previous study also showed that P21WAF1 was
downregulated in EOC tissues, and was related with a later FIGO
stage (28).
In the present study, abnormal expression levels of
PLK1, P53 and P21WAF1 protein in EOC tissues were
detected by immunohistochemistry and western blot analysis. The
expression levels of PLK1 and P53 were significantly higher in the
EOC tissues, while P21WAF1 was obviously lower. Higher
expression levels of PLK1 and P53 were detected in patients with
later FIGO stage and worse histological differentiation, but lower
P21WAF1 in later FIGO stage. These results suggest that
misregulation of PLK1, P53 and P21WAF1 are implicated in
the progression of ovarian cancer.
P53 plays key roles in cell cycle signal cross-talk,
can receive various upstream cell stimuli, and can be directly or
indirectly mediated by different signal transduction proteins, such
as PLK1, ATM and ATR (20,29,30).
The present study showed that PLK1, P53 and P21WAF1
could be used to assess the prognosis respectively, but
multivariate logistic regression analysis verified that only PLK1
was an independent prognostic factor for ovarian cancer. The
overall survival time assessed by Kaplan-Meier curves analysis
showed that the overall survival time of the patients with
PLK1-positive/P53-positive or
PLK1-positive/P21WAF1-negative was obviously shorter
than that of other patients at the end of the follow-up. These
results indicate that misregulation of P53 and P21WAF1
may be a common event in ovarian cancer, and may be used to assess
the prognosis, but only PLK1 could serve as an independent
prognostic factor for ovarian cancer. These results indicate that
misregulation of PLK1 coinciding with P53 and P21WAF1 in
EOC highly suggests poor prognosis.
In conclusion, according to the above in
vitro data and due to the limited number of enrolled patients
with follow-up, the present study showed that abnormal expression
of PLK1 is implicated in ovarian carcinogenesis and may owe its
ability to inhibition of the activity of P53, and misregulation of
PLK1 coincident with P53 and P21WAF1 in EOC suggests
poor prognosis.
Acknowledgements
We thank Huixiang Li and Kuisheng Chen for their
technical support in regards to immunohistochemistry, and the
Clinical Medicine Key Disciplines Laboratory of Henan Province for
assistance with the experiments. Our study was supported by the
Hospital Youth Fund Project of the First Affiliated Hospital of
Zhengzhou University.
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Strebhardt K and Ullrich A: Targeting
polo-like kinase 1 for cancer therapy. Nat Rev Cancer. 6:321–330.
2006. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kishi K, van Vugt MA, Okamoto K, Hayashi Y
and Yaffe MB: Functional dynamics of Polo-like kinase 1 at the
centrosome. Mol Cell Biol. 29:3134–3150. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wolf G, Elez R, Doermer A, Holtrich U,
Ackermann H, Stutte HJ, Altmannsberger HM, Rübsamen-Waigmann H and
Strebhardt K: Prognostic significance of polo-like kinase (PLK)
expression in non-small cell lung cancer. Oncogene. 14:543–549.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Takahashi T, Sano B, Nagata T, Kato H,
Sugiyama Y, Kunieda K, Kimura M, Okano Y and Saji S: Polo-like
kinase 1 (PLKI) is overexpressed in primary colorectal cancers.
Cancer Sci. 94:148–152. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Weichert W, Kristiansen G, Winzer KJ,
Schmidt M, Gekeler V, Noske A, Müller BM, Niesporek S, Dietel M and
Denkert C: Polo-like kinase isoforms in breast cancer: expression
patterns and prognostic implications. Virchows Arch. 446:442–450.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Weichert W, Schmidt M, Gekeler V, Denkert
C, Stephan C, Jung K, Loening S, Dietel M and Kristiansen G:
Polo-like kinase 1 is overexpressed in prostate cancer and linked
to higher tumor grades. Prostate. 60:240–245. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Strebhardt K, Kneisel L, Linhart C, Bernd
A and Kaufmann R: Prognostic value of pololike kinase expression in
melanomas. JAMA. 283:479–480. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mito K, Kashima K, Kikuchi H, Daa T,
Nakayama I and Yokoyama S: Expression of polo-like kinase (PLK1) in
non-Hodgkin’s lymphomas. Leuk Lymphoma. 46:225–231. 2005.
View Article : Google Scholar
|
|
10
|
Yamamoto Y, Matsuyama H, Kawauchi S,
Matsumoto H, Nagao K, Ohmi C, Sakano S, Furuya T, Oga A, Naito K
and Sasaki K: Overexpression of polo-like kinase 1 (PLKl) and
chromosomal instability in bladder cancer. Oncology. 70:231–237.
2006. View Article : Google Scholar
|
|
11
|
Stark GR and Taylor WR: Control of the
G2/M transition. Mol Biotechnol. 32:227–248. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Levine AJ: p53, the cellular gatekeeper
for growth and division. Cell. 88:323–331. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Charrier-Savournin FB, Château MT, Gire V,
Sedivy J, Piette J and Dulic V: p21-mediated nuclear retention of
cyclin B1-Cdk1 in response to genotoxic stress. Mol Biol Cell.
15:3965–3976. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abbas T and Dutta A: p21 in cancer:
intricate networks and multiple activities. Nat Rev Cancer.
9:400–414. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Song B, Liu XS and Liu X: Polo-like kinase
1 (Plk1): an unexpected player in DNA replication. Cell Div.
7:32012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yim H and Erikson RL: Polo-like kinase l
depletion induces DNA damage in early S prior to caspase
activation. Mol Cell Biol. 29:2609–2621. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hayashi MT and Karlseder J: DNA damage
associated with mitosis and cytokinesis failure. Oncogene.
32:4593–4601. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Murray AW: Creative blocks: cell-cycle
checkpoints and feedback controls. Nature. 359:599–604. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jeong JH, Kang SS, Park KK, Chang HW,
Magae J and Chang YC: p53-independent induction of G1 arrest and
p21WAF1/CIP1 expression by ascofuranone, an isoprenoid
antibiotic, through downregulation of c-Myc. Mol Cancer Ther.
9:2102–2113. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ando K, Ozaki T, Yamamoto H, Furuya K,
Hosoda M, Hayashi S, Fukuzawa M and Nakagawara A: Polo-like kinase
1 (Plkl) inhibits p53 function by physical interaction and
phosphorylation. J Biol Chem. 279:25549–25561. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ahmad N: Polo-like kinase (Plk) 1: a novel
target for the treatment of prostate cancer. FASEB J. 18:5–7. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Weichert W, Denkert C, Schmidt M, Gekeler
V, Wolf G, Köbel M, Dietel M and Hauptmann S: Polo-like kinase
isoform expression is a prognostic factor in ovarian carcinoma. Br
J Cancer. 90:815–821. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Marks JR, Davidoff AM, Kerns BJ, Humphrey
PA, Pence JC, Dodge RK, Clarke-Pearson DL, Iglehart JD, Bast RC Jr
and Berchuck A: Overexpression and mutation of p53 in epithelial
ovarian cancer. Cancer Res. 51:2979–2984. 1991.PubMed/NCBI
|
|
24
|
Skírnisdóttir I and Seidal T: Prognostic
impact of concomitant p53 and PTEN on outcome in early stage (FIGO
I–II) epithelial ovarian cancer. Int J Gynecol Cancer.
21:1024–1031. 2011. View Article : Google Scholar
|
|
25
|
Berchuck A, Kohler MF, Hopkins MP,
Humphrey PA, Robboy SJ, Rodriguez GC, Soper JT, Clarke-Pearson DL
and Bast RC: Overexpression of p53 is not a feature of benign and
early-stage borderline epithelial ovarian tumors. Gynecol Oncol.
52:232–236. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Buchynska LG, Nesina IP, Yurchenko NP,
Bilyk OO, Grinkevych VN and Svintitsky VS: Expression of p53,
p21WAF1/CIP1, p16INK4A and Ki-67 proteins in serous ovarian tumors.
Exp Oncol. 29:49–53. 2007.PubMed/NCBI
|
|
27
|
Terauchi F, Okamoto A, Nagashima T,
Kobayashi Y, Moritake T, Yamamoto Y, Takakura S, Iwaki S and Ogura
H: Clinical significance of p21WAF1/CIP1 and p53
expression in serous cystadenocarcinoma of the ovary. Oncol Rep.
14:363–368. 2005.PubMed/NCBI
|
|
28
|
Shi HR and Zhang RT: Expression and
significance of P53, P21WAF1 and CDK1 proteins in epithelial
ovarian cancer. Ai Zheng. 28:882–885. 2009.(In Chinese). PubMed/NCBI
|
|
29
|
Banin S, Moyal L, Shieh S, Taya Y,
Anderson CW, Chessa L, Smorodinsky NI, Prives C, Reiss Y, Shiloh Y
and Ziv Y: Enhanced phosphorylation of p53 by ATM in response to
DNA damage. Science. 281:1674–1677. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Canman CE, Lim DS, Cimprich KA, Taya Y,
Tamai K, Sakaguchi K, Appella E, Kastan MB and Siliciano JD:
Activation of the ATM kinase by ionizing radiation and
phosphorylation of p53. Science. 281:1677–1679. 1998. View Article : Google Scholar : PubMed/NCBI
|