Introduction
Anaplastic thyroid carcinoma (ATC), which accounts
for 2–5% of thyroid carcinomas, is one of the most aggressive and
resistant human malignancies (1,2). The
disease is usually advanced by the time of diagnosis and 75% of the
patients develop distant metastasis during later progression. The
multimodal treatment of ATC includes surgical extirpation,
radiotherapy and chemotherapy (usually doxorubicin or paclitaxel)
(3). Despite combined therapy, the
prognosis of the disease is poor with an average survival time of
only 6–8 months (3,4).
Sorafenib, a multikinase inhibitor, targets kinases
of different receptors such as VEGFR-2, VEGFR-3, PDGFR, RET and
BRAF and thus exhibits antitumor and anti-angiogenic activities.
Sorafenib has been approved for the treatment of advanced renal
cell carcinoma, unresectable hepatocellular carcinoma and tested in
preclinical and ongoing clinical studies in breast carcinoma, colon
cancer and melanoma (5). Different
studies in patients with undifferentiated and radioiodine
refractory-differentiated thyroid carcinoma (DTC) have demonstrated
superiority of sorafenib over the standard care with doxorubicin
(6–9). Therefore, the FDA has recently
approved this chemotherapeutic drug for treatment of differentiated
thyroid cancer that does not respond to radioiodine therapy
(http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm376443.htm).
However, patients treated with sorafenib may develop
toxic side effects including hand-foot syndrome, rash, fatigue,
diarrhea, and hypertension (10).
Since these side-effects are, at least in part, dose-dependent, a
reduction of sorafenib dose without reducing the therapeutic effect
may be of high clinical value.
Metformin, a commonly prescribed, well-tolerated
anti-diabetic agent, may be a candidate for such a combined
therapy. Its anti-proliferative effect on thyroid carcinoma cells
was clearly demonstrated (11).
Moreover, metformin amplifies the anti-mitogenic effect of
chemotherapeutic agents such as doxorubicin and cisplatin whose
dose may be reduced in a combined therapy (11).
In the present study, a synergistic effect of
sorafenib and metformin on the growth inhibition of anaplastic
thyroid cancer was demonstrated. Sorafenib inhibited anaplastic
thyroid cell growth by blocking cell cycle progression and inducing
apoptosis. On the molecular level, sorafenib decreased the cell
growth rate by inhibiting MAPK signaling pathway. Furthermore,
sorafenib inhibited clonal cell growth and thyroid cancer sphere
formation, a characteristic of cancer stem cells. Of note,
metformin amplified the anti-mitogenic effect of sorafenib and
synergistically decreased clonal cell growth and sphere formation
in ATC cells. In conclusion, the results showed that combined
chemotherapy of metformin with sorafenib reduced the dose-dependent
side-effects of this chemotherapeutic drug.
Materials and methods
Cell cultures
The HTh74 anaplastic thyroid cancer cell line was
kindly provided by Dr Heldin, Uppsala, Sweden. Cells were cultured
in F12 medium supplemented with 10% fetal calf serum (FCS, v/v), 1%
MEM (v/v), 100 U/ml penicillin and 100 μg/ml streptomycin.
The stable doxorubicin-resistant thyroid carcinoma
cell line HTh74Rdox was established as described below (12). Briefly, the HTh74Rdox cell line was
derived by continuous exposure of this cell line to 0.5 μg/ml
doxorubicin for >6 months. The IC50 value for
doxorubicin was 153.53±16.43 μg/ml, which corresponds to an 85-fold
increase compared to doxorubicin-sensitive parental HTh74 cells.
Flow cytometric analysis after Hoechst 33342 dye staining
demonstrated that ~80% of the doxorubicin-resistant cells were
detectable as a side population cell fraction, enriched with cancer
stem cells that expressed transporters of the ATP-binding cassette
(ABC) gene family (12).
Monolayer cultures of human thyrocytes isolated from
nodular goiters of 6 patients undergoing thyroidectomy were
established and cultured as described previously (13,14).
This study was approved by the Local Ethics Committee. In all cases
informed consent was obtained.
Cell viability assay
Cell viability was assessed using the 3-(4,
5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT)
assay as described by the manufacturer (Sigma-Aldrich, St. Louis,
MO, USA). All the experiments were repeated at least three times in
quadruplicate.
Cell cycle and apoptotic analysis
Analysis of cell cycle arrest was performed in
low-density cultures (1×105 cells/6 cm dish, 40–50%
confluence), whereas apoptosis was investigated in high-density
cultures (4×105 cells/6 cm dish, confluent >90%)
(15). The cell cycle was analysed
by measuring the amount of propidium iodide (PI) incorporation into
cellular DNA in ethanol-fixed cells. Apoptotic and necrotic cell
death was analyzed by double staining with fluorescein
isothiocyanate (FITC)-conjugated Annexin V and PI according to the
manufacturer’s instructions (BD Biosciences, Heidelberg, Germany).
The green and red fluorescence of Annexin V/PI-stained viable cells
and PI-stained fixed cells were analyzed with a FACSVerse flow
cytometer (BD Biosciences) using a peak fluorescence gate to
exclude cell aggregates during cell cycle analysis. The number of
viable (Annexin V−/PI−), apoptotic (Annexin
V+/PI−) and necrotic (Annexin
V+/PI+) cells and the proportion of cells in
different cell cycle phases were calculated with the FACSDiva
software (BD Biosciences).
Colorimetric assay of caspase-3
activity
Caspase-3 activity (2×106 cells seeded in
10 cm dishes) was determined using a colorimetric assay kit
(Sigma-Aldrich) according to the manufacturer’s instructions. The
intra- and inter-assay CV of caspase-3 assay was 4.49 and 6.46%,
respectively.
In vitro clonal analysis
HTh74 and HTh74Rdox cells were plated at clonal
density (200 cells/well) in triplicates in 6-well plates and
treated with the indicated concentrations (1–10 μM) of sorafenib
with or without metformin. Formed colonies were stained with Giemsa
and the percentage of cells that initiated a clone was determined
as cloning efficiency.
Sphere formation assay
Tumor spheres that consist of stem cells and their
progenitor cells were generated by placing HTh74 cells
(1×104 cells/ml) into serum-free DMEM/F12 medium
containing B27 (1:50 dilution), bFGF (20 ng/ml) and EGF (20 ng/ml),
as described previously (11).
Sphere-forming efficiency (SFE) was calculated as the number of
sphere-like structures (large diameter >50 μm) formed in 7 days
divided by the original number of cells seeded and expressed as a
percentage mean (± SD).
Western blot analysis
Cells following different treatments were lysed with
RIPA buffer. The proteins were treated with 4X sample buffer
containing dithiothreitol and boiled for 10 min. An equal amount of
protein (30 μg) was subjected to 12.5% SDS polyacrylamide gel and
separated proteins were transferred to NC membranes. The membranes
were blocked in 5% skim milk for 1 h at room temperature. The
immunoblots were incubated overnight at 4°C with anti-cyclin D1,
anti-ERK, and anti-phosphorylated ERK1/2 (Thr202 and
Tyr204) antibodies (all from Santa Cruz Biotechnology,
Inc., Santa Cruz, CA, USA) in 5% BSA/TBST at a dilution of 1:1,000,
1:1,0000, and 1:2,000, respectively. The following day, the
membranes were incubated with a horseradish peroxidase-conjugated
secondary antibody (Santa Cruz Biotechnology, Inc.) for 1 h at room
temperature. The immunoreactive bands were detected with a
chemiluminescence substrate kit (ProteinSimple, Santa Clara, CA,
USA) under the FluorChem FC2 system.
Flow cytometry for side population
cells
To isolate the thyroid cancer side population
fraction, FACS was performed using the Hoechst 33342 dye staining
method as described previously (16). Briefly, cancer cells were labeled
with 5 μg/ml Hoechst 33342 dye (Sigma-Aldrich) either alone or in
combination with 50 μM verapamil (Sigma-Aldrich), which is an
inhibitor of ABCG2 transporter. The cells were counterstained with
1 μg/ml PI to exclude dead cells. A 350-nm UV laser was used to
excite Hoechst 33342 dye and PI. Analysis was performed on a
fluorescence-activated cell sorter (BD Biosciences) using a
dual-wavelength analysis (blue, 424–444 nm and red, 675 nm).
Statistical analysis
Statistical analysis was performed with SPSS13.0
software. Numerical data are expressed as mean ± SD. P<0.05 was
considered to indicate a statistically significant difference.
Results
Inhibition of cell growth by sorafenib
with or without metformin
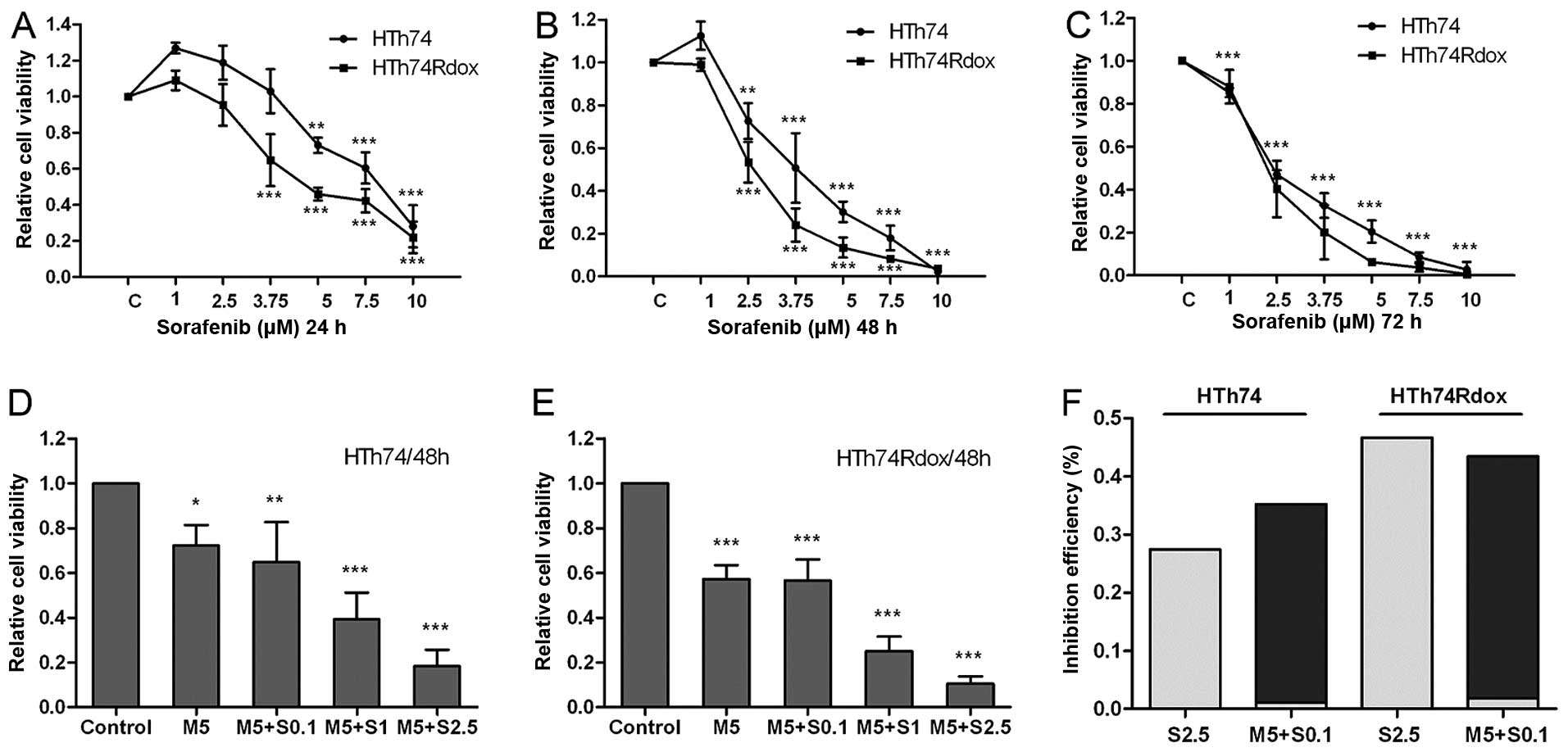
Human HTh74 and HTh74Rdox ATC cells were treated
with various concentrations of sorafenib for 24–72 h. As shown in
Fig. 1A–C, sorafeninb significantly
inhibited growth of the HTh74 and HTh74Rdox cells in a dose- and
time-dependent manner. The mean EC50 values in the 48-h
cell viability assay were ~3.28 μM for HTh74 cells, and 2.47 μM for
HTh74Rdox cells, indicating that HTh74Rdox cells, which are
enriched with cancer stem cells, were more sensitive to sorafenib
than their parental HTh74 cells.
To determine whether metformin influenced the
anti-mitogenic effect of sorafenib, viability of HTh74 and
HTh74Rdox cells was analysed after combined treatment with the two
drugs. As shown in Fig. 1D–E, there
was additional growth inhibition in the combination group.
Metformin (5 μM) combined with 0.1 μM sorafenib exhibited the
equivalent growth inhibitory effect as 2.5 μM as monotherapy with
sorafenib in HTh74 and HTh74Rdox cells (~35–45% inhibition
efficiency, Fig. 1D–F).
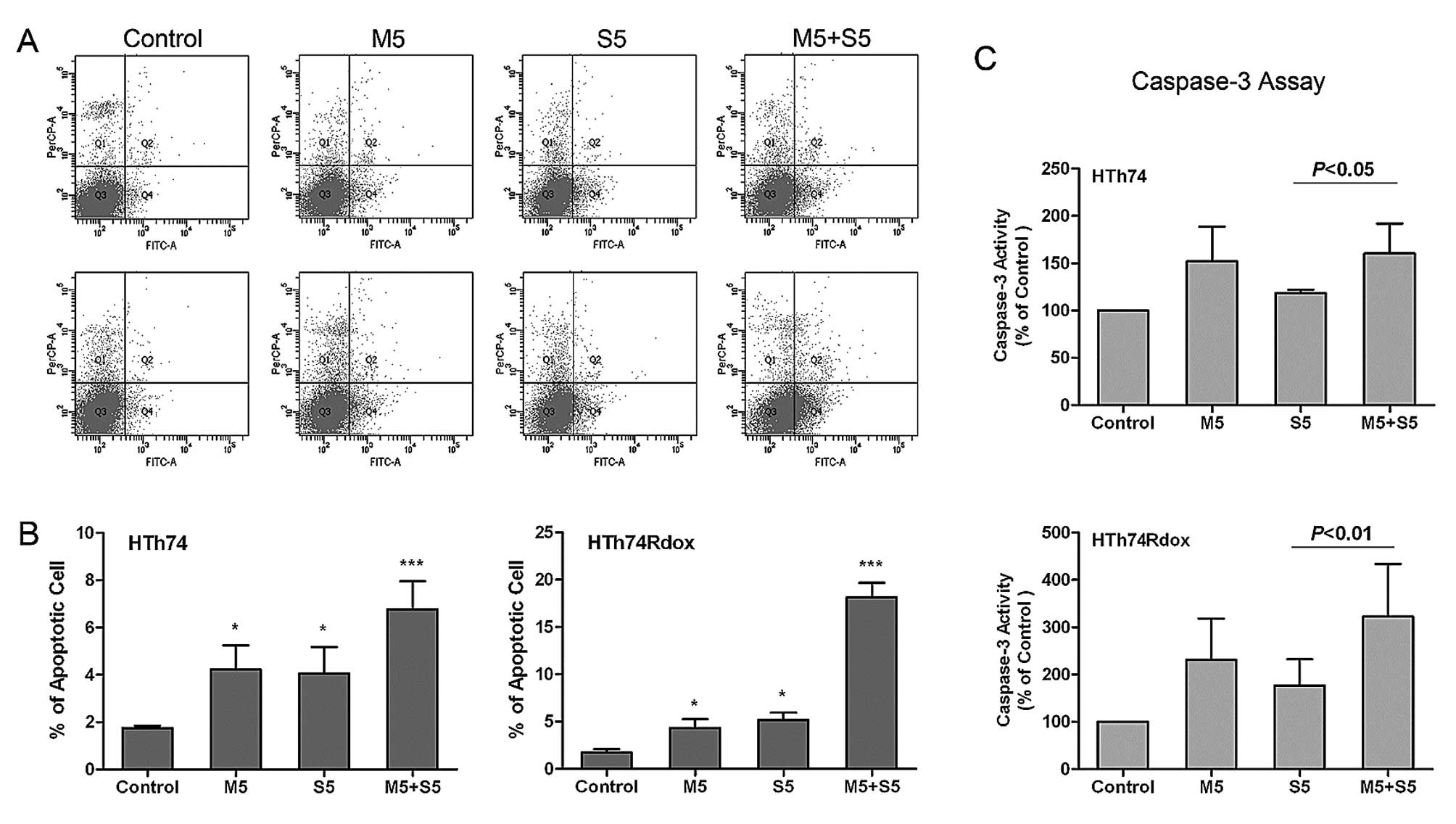
Induction of apoptosis by sorafenib and
metformin
To investigate the mechanism of growth inhibition by
sorafenib and to evaluate whether sorafenib and metformin act
synergistically, the induction of apoptosis was analyzed after
different treatments. As shown in Fig.
2, sorafenib and metformin as monotherapy enhanced apoptosis of
HTh74 and HTh74Rdox cells. The addition of 5 μM sorafenib or 5 μM
metformin increased the percentage of apoptotic cells from 1.7 to
4.1% and 3.1%, respectively, in HTh74 cells and from 1.7 to 4.3%
and 4.9%, respectively, in HTh74Rdox cells (Fig. 2A and B). In the sorafenib/metformin
group, the induction of apoptosis was much more pronounced (5.4% in
HTh74 cells and 17.9% in HTh74Rdox cells) compared to sorafenib or
metformin as monotherapy.
In addition, agent-induced apoptosis was assayed by
detection of caspase-3 activity in HTh74 and HTh74Rdox cells. In
response to sorafenib or metformin treatment, caspase-3 activity
markedly increased by 18.4 and 52.0%, respectively, in HTh74 cells
and 77.3 and 131.0%, respectively, in HTh74Rdox cells (Fig. 2C). Compared to the results of
Annexin V-FITC and PI staining, the combination of sorafenib with
metformin was more effective in inducing caspase-3 activity than as
monotherapy (caspase-3 activity was increased by 60.2 and 222.2% in
HTh74 and HTh74Rdox cells, respectively). Thus, in HTh74Rdox the
pro-apoptotic effect of sorafenib and metformin was more pronounced
than that in HTh74 cells.
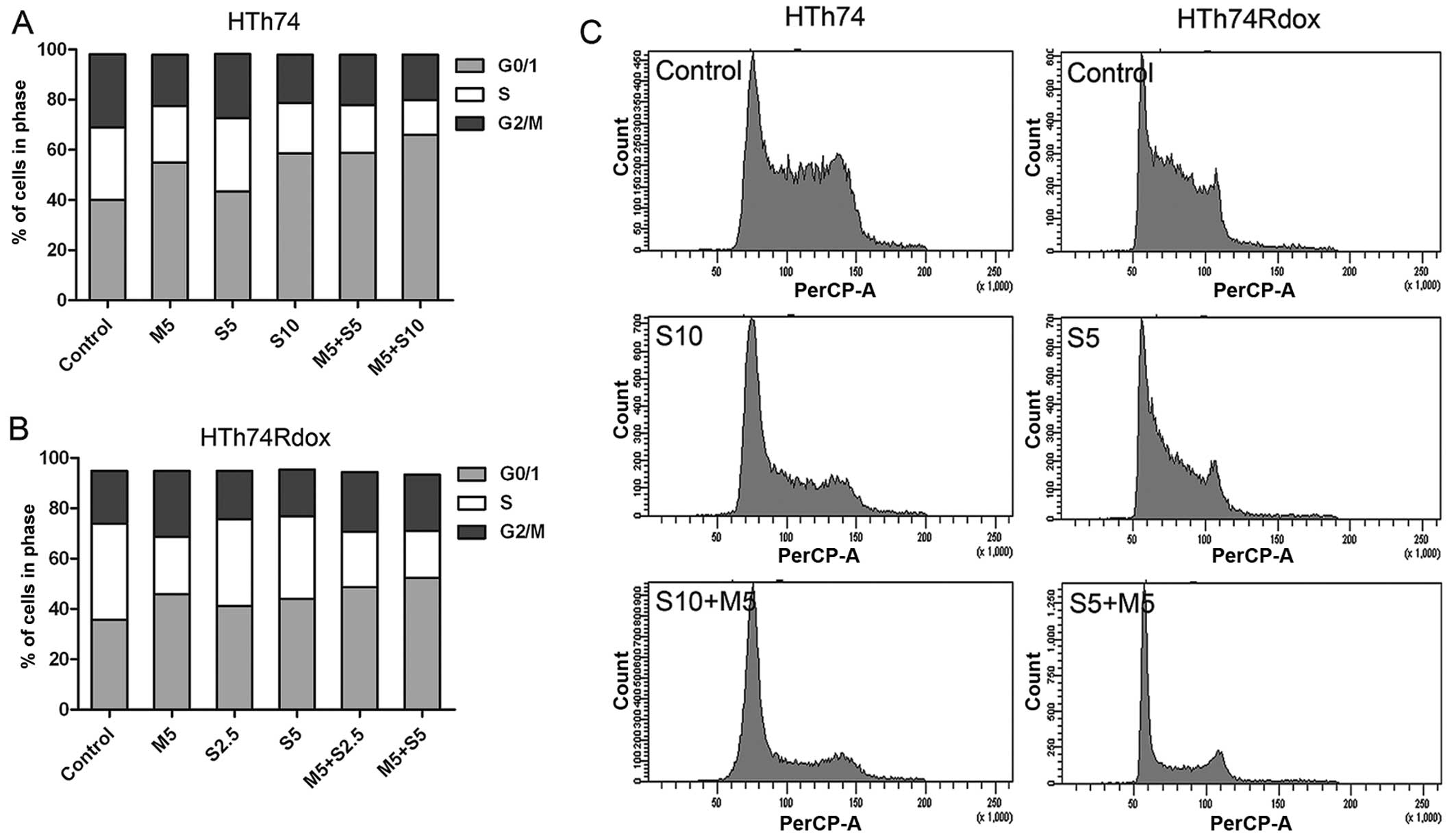
Sorafenib and metformin cause cell cycle
arrest
The effect of sorafenib and metformin on the cell
cycle progression was analyzed by flow cytometry after 24-h
treatment.
Sorafenib and metformin treatment led to the
accumulation of cells in G1 phase with a consecutive decrease in
the percentage of cells in S phase in HTh74 and HTh74Rdox cells
(Fig. 3). The effect on cell cycle
arrest induced by sorafenib was weaker than that by metformin. In
the combination group, similar data were obtained although with a
more pronounced decrease of cells in S phase than by sorafenib
alone.
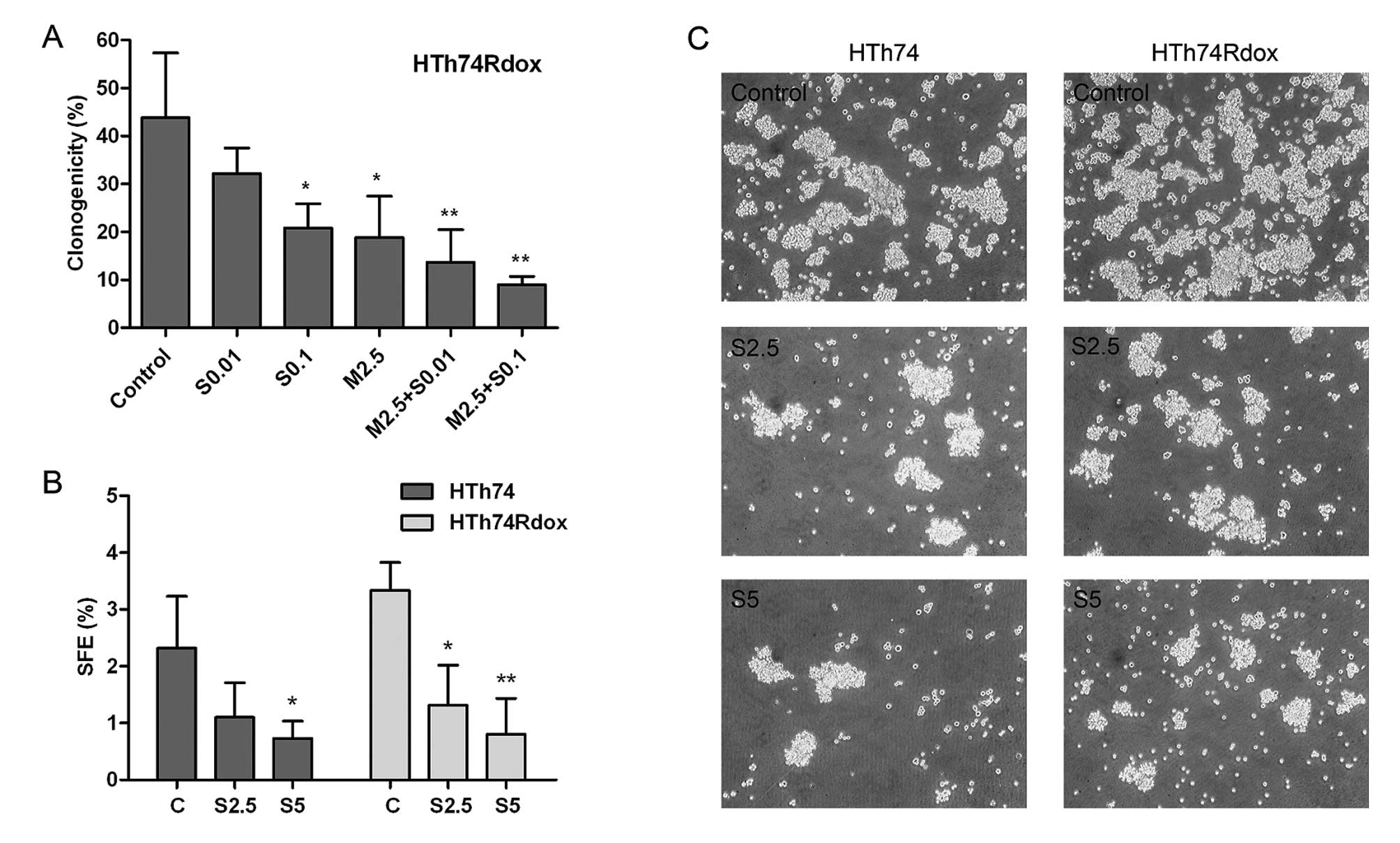
Inhibition of colony and tumor sphere
formation by sorafenib
The self-renewal capacity of anaplastic thyroid
cancer cells was analyzed by clonal formation and sphere formation
assays. Colony formation assay was performed only in HTh74Rdox
cells which are enriched with cancer stem cells and are more
clonogenic than HTh74 cells (11,12).
As observed in Fig. 4A, after
treatment with 0.1 μM sorafenib or 2.5 μM metformin, the number of
HTh74Rdox colonies formed was significantly reduced. Combination of
2.5 μM metformin with 0.01 or 0.1 μM sorafenib resulted in the
further decrease of clonogenicity.
The effect of sorafenib on HTh74 and HTh74Rdox
cells, in the presence or absence of metformin, was investigated by
sphere formation assay. In response to increasing doses of
sorafenib, SFE was significantly lower in the two cell lines with
~52 and 61% reduction at the concentration of 2.5 μM sorafenib and
69 and 76% reduction at the concentration of 5 μM sorafenib in
HTh74 and HTh74Rdox cells, respectively (Fig. 4B and C). In the presence of 5 μM
metformin, the sphere formation efficiency was almost completely
suppressed by 2.5 μM sorafenib (data not shown).
Sorafenib and metformin inhibit cell
growth via MAPK pathway
To evaluate the effect of sorafenib, and sorafenib
plus metformin on a pathway involved in growth inhibition and
targeted by the two drugs, phosphorylation of ERK, a key protein of
MAPK pathway, was analyzed in HTh74 and HTh74Rdox cells. Western
blotting revealed that sorafenib markedly decreased the
phosphorylation of ERK in a dose-dependent manner (Fig. 5A). Metformin also decreased the
phosphorylation of ERK. However, a synergistically inhibitory
effect of sorafenib plus metformin was not observed. The expression
of cyclin D1 was reduced following sorafenib treatment with or
without metformin.
Discussion
Removal of the tumor mass and subsequent ablation of
the remaining thyroid cancer tissue by radioiodine therapy is a
prerequisite for cure of thyroid cancer. Therefore, anaplastic
thyroid carcinomas that lack radioiodine uptake have a very poor
prognosis (17). Furthermore, life
expectancy of patients with progressive local and metastatic
well-differentiated thyroid cancer that secondarily became
refractory to radioiodine treatment is also very limited (6).
As an evolving new strategy to improve prognosis of
these thyroid carcinomas, multi-targeted tyrosine kinase inhibitors
have been investigated in clinical studies (18). Recently, the FDA has approved
sorafenib as the first drug of this group (http://www.fda.gov). Sorafenib, an orally ingested
drug, exerts its anti-angiogenic and anti-mitogenic effect by
targeting BRAF, VEGFR1 and 2, RET and thereby important
growth-regulating signaling pathways of thyroid cancer (19). In differentiated thyroid cancer the
role of genetic aberrations in the RET-RAS-RAF-MAPK signaling
pathway in tumor pathogenesis and progression is well established
(20). Rearrangements in the RET
proto-oncogene which is involved in the initiation of tumor
formation are detectable in up to 25% of PTCs (21). Twenty nine to 69% of PTCs harbor a
BRAF V600E mutation, which is associated with recurrent and
persistent disease (21). RAS
mutations and downstream signaling PIK3CA mutations occur in almost
50% of FTCs and >10% of Hurthle cell carcinomas (21).
In a xenograft model it was demonstrated that
sorafenib obstructs RAF kinases and thereby inhibits growth of ATC
cells (20). Furthermore, sorafenib
inhibited the growth and angiogenesis of orthotopic ATC xenografts
in nude mice (22). These carcinoma
cells lacked any known molecular aberrations which argues against
the hypothesis that the effect of sorafenib is limited to tumors
with these mutations. Subsequent findings demonstrated that PTC
cells carrying the RET/PTC1 rearrangement were more sensitive to
sorafenib than those carrying a BRAF mutation (23).
Clinical studies confirmed the efficacy of sorafenib
for patients with radioiodine refractory-differentiated thyroid
carcinoma (DTC). A recent meta-analysis which included seven
studies on radioiodine-refractory DTC, showed a partial response in
22% and stable disease in 52% of the patients (24). Median progression-free survival was
12.4 month (95% CI: 10.4–14.7). Sorafenib has also been utilized
for patients with advanced ATC (9).
Two out of 20 patients had a partial remission and 5 had stable
disease with a duration of 4 months (range, 3–11 months). At least
in patients with radioiodine-refractory thyroid carcinomas with a
poor prognosis multi-kinase inhibitors such as sorafenib are
considered promising.
However, toxic side effects such as hand-foot
syndrome, diarrhea, fatigue, rash and weight loss that occur in up
to 80% of sorafenib-treated patients are a concern (24). Due to the side effects dose
reduction was necessary in >60% of the patients and in 6–25% of
patients treatment was even discontinued (24). Therefore, adjuvant therapy that
allows dose reduction of sorafenib without decreasing its efficacy
may be an option to overcome this problem.
Metformin, a widely used, well-tolerated
antidiabetic drug, has recently been demonstrated to potentiate the
anti-mitogenic effects of doxorubicin and cisplatin in ATC cells
(11). The combined therapy with
metformin enabled a significant reduction of these chemotherapeutic
drugs without reducing their anti-proliferative capacity (11).
In the present study, we have demonstrated that the
anti-mitogenic effect of sorafenib was potentiated by the addition
of metformin. The addition of metformin allowed a dose reduction of
sorafenib by up to 25% without a decrease of the growth-inhibitory
effect.
Sorafenib reduced viability of ATC cells by blocking
cell cycle progression via G0/1 phase arrest and S phase inhibition
and by inducing cell apoptosis. The effects were more pronounced
when metformin was added to the culture.
Tumor sphere formation and clonal growth which
reflect self-renewal and characteristic proliferation pattern, are
a hallmark of cancer stem cells (12,25).
In the present results, sorafenib significantly reduced clonal
growth of HTh74Rdox cells. In addition, the drug inhibited tumor
sphere formation by decreasing tumor sphere number and size. Again,
metformin amplified the effect of sorafenib in the two experiments.
These data suggest that sorafenib and metformin target cancer cells
and their derived stem cells. A potentiating effect of metformin as
adjuvant to sorafenib has recently been reported in
cholangiocarcinoma cells (26).
Sorafenib and metformin inhibited MAP kinase
signaling as demonstrated by reduced ERK phosphorylation. A
synergistic effect was, however, not detected. Therefore, the
synergistic inhibition of cell growth may be explained by the
different targets of the two drugs. Metformin additionally
decreases thyroid carcinoma cell growth by inhibition of the
AMPK-mTOR pathway and, as mentioned earlier, sorafenib by targeting
other receptor-dependent kinases (11).
In conclusion, the multikinase inhibitor sorafenib
and metformin synergistically decreased the growth rate of ATC
cells. These drugs share a common target in cancer therapy, the MAP
kinase pathway. Additionally, each drug inhibits other
growth-regulatory signaling pathways and exert their anti-mitogenic
effect on the derived cancer stem cells. The synergistic effect of
metformin suggests this drug as an adjuvant to sorafenib treatment
to reduce dose-dependent side-effects. Clinical studies are
necessary to evaluate whether a combined therapy of sorafenib and
metformin are useful for the treatment of radioiodine-refractory
DTC and anaplastic thyroid cancer in diabetics and other patients
with hyper-insulinemia.
Acknowledgements
We are grateful to Martina Kleinhardt for her
excellent technical support. This study was supported by a grant
from Biomedic EV and by IKFE services and supported by Deutsche
Forschungsgemeinschaft funded-Graduate College 1208 (Charite,
Berlin).
References
|
1
|
Ordonez N, Baloch Z, Matias-Guiu X, et al:
Undifferentiated (anaplastic) carcinoma. World Health Organization
Classification of Tumours of Endocrine Organs. DeLellis RA, Lloyd
RV, Heitz PU and Eng C: IARC Press; Lyons: pp. 77–80. 2004
|
|
2
|
Smallridge RC, Marlow LA and Copland JA:
Anaplastic thyroid cancer: molecular pathogenesis and emerging
therapies. Endocr Relat Cancer. 16:17–44. 2009. View Article : Google Scholar
|
|
3
|
Pasieka JL: Anaplastic thyroid cancer.
Curr Opin Oncol. 15:78–83. 2003. View Article : Google Scholar
|
|
4
|
Granata R, Locati L and Licitra L:
Therapeutic strategies in the management of patients with
metastatic anaplastic thyroid cancer: review of the current
literature. Curr Opin Oncol. 25:224–228. 2013.PubMed/NCBI
|
|
5
|
Wilhelm SM, Adnane L, Newell P, Villanueva
A, Llovet JM and Lynch M: Preclinical overview of sorafenib, a
multikinase inhibitor that targets both Raf and VEGF and PDGF
receptor tyrosine kinase signaling. Mol Cancer Ther. 7:3129–3140.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brose MS, Nutting CM, Jarzab B, et al:
Sorafenib in radioactive iodine-refractory, locally advanced or
metastatic differentiated thyroid cancer: a randomised,
double-blind, phase 3 trial. Lancet. 384:319–328. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gupta-Abramson V, Troxel AB, Nellore A, et
al: Phase II trial of sorafenib in advanced thyroid cancer. J Clin
Oncol. 26:4714–4719. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Haugen BR and Kane MA: Approach to the
thyroid cancer patient with extracervical metastases. J Clin
Endocrinol Metab. 95:987–993. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Savvides P, Nagaiah G, Lavertu P, et al:
Phase II trial of sorafenib in patients with advanced anaplastic
carcinoma of the thyroid. Thyroid. 23:600–604. 2013. View Article : Google Scholar :
|
|
10
|
Duntas LH and Bernardini R: Sorafenib:
rays of hope in thyroid cancer. Thyroid. 20:1351–1358. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen G, Xu S, Renko K and Derwahl M:
Metformin inhibits growth of thyroid carcinoma cells, suppresses
self-renewal of derived cancer stem cells, and potentiates the
effect of chemotherapeutic agents. J Clin Endocrinol Metab.
97:E510–E520. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zheng X, Cui D, Xu S, Brabant G and
Derwahl M: Doxorubicin fails to eradicate cancer stem cells derived
from anaplastic thyroid carcinoma cells: Characterization of
resistant cells. Int J Oncol. 37:307–315. 2010.PubMed/NCBI
|
|
13
|
Broecker M, Hammer J and Derwahl M:
Excessive activation of tyrosine kinases leads to inhibition of
proliferation in a thyroid carcinoma cell line. Life Sci.
63:2373–2386. 1998. View Article : Google Scholar
|
|
14
|
Manole D, Schildknecht B, Gosnell B, Adams
E and Derwahl M: Estrogen promotes growth of human thyroid tumor
cells by different molecular mechanisms. J Clin Endocrinol Metab.
86:1072–1077. 2001.PubMed/NCBI
|
|
15
|
Isakovic A, Harhaji L, Stevanovic D, et
al: Dual antiglioma action of metformin: cell cycle arrest and
mitochondria-dependent apoptosis. Cell Mol Life Sci. 64:1290–1302.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lan L, Cui D, Nowka K and Derwahl M: Stem
cells derived from goiters in adults form spheres in response to
intense growth stimulation and require thyrotropin for
differentiation into thyrocytes. J Clin Endocrinol Metab.
92:3681–3688. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Reddi HV, Madde P, McDonough SJ, et al:
Preclinical efficacy of the oncolytic measles virus expressing the
sodium iodide symporter in iodine non-avid anaplastic thyroid
cancer: a novel therapeutic agent allowing noninvasive imaging and
radioiodine therapy. Cancer Gene Ther. 19:659–665. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Thomas L, Lai SY, Dong W, et al: Sorafenib
in metastatic thyroid cancer: a systematic review. Oncologist.
19:251–258. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wilhelm SM, Carter C, Tang L, et al: BAY
43-9006 exhibits broad spectrum oral antitumor activity and targets
the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in
tumor progression and angiogenesis. Cancer Res. 64:7099–7109. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Salvatore G, De Falco V, Salerno P, et al:
BRAF is a therapeutic target in aggressive thyroid carcinoma. Clin
Cancer Res. 12:1623–1629. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Schneider TC, Abdulrahman RM, Corssmit EP,
Morreau H, Smit JW and Kapiteijn E: Long-term analysis of the
efficacy and tolerability of sorafenib in advanced radioiodine
refractory differentiated thyroid carcinoma: final results of a
phase II trial. Eur J Endocrinol. 167:643–650. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim S, Yazici YD, Calzada G, et al:
Sorafenib inhibits the angiogenesis and growth of orthotopic
anaplastic thyroid carcinoma xenografts in nude mice. Mol Cancer
Ther. 6:1785–1792. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Henderson YC, Ahn SH, Kang Y and Clayman
GL: Sorafenib potently inhibits papillary thyroid carcinomas
harboring RET/PTC1 rearrangement. Clin Cancer Res. 14:4908–4914.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shen CT, Qiu ZL and Luo QY: Sorafenib in
the treatment of radioiodine-refractory differentiated thyroid
cancer: a meta-analysis. Endocr Relat Cancer. 21:253–261. 2014.
View Article : Google Scholar
|
|
25
|
Mitsutake N, Iwao A, Nagai K, et al:
Characterization of side population in thyroid cancer cell lines:
cancer stem-like cells are enriched partly but not exclusively.
Endocrinology. 148:1797–1803. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ling S, Feng T, Ke Q, et al: Metformin
inhibits proliferation and enhances chemosensitivity of
intrahepatic cholangiocarcinoma cell lines. Oncol Rep.
31:2611–2618. 2014.PubMed/NCBI
|