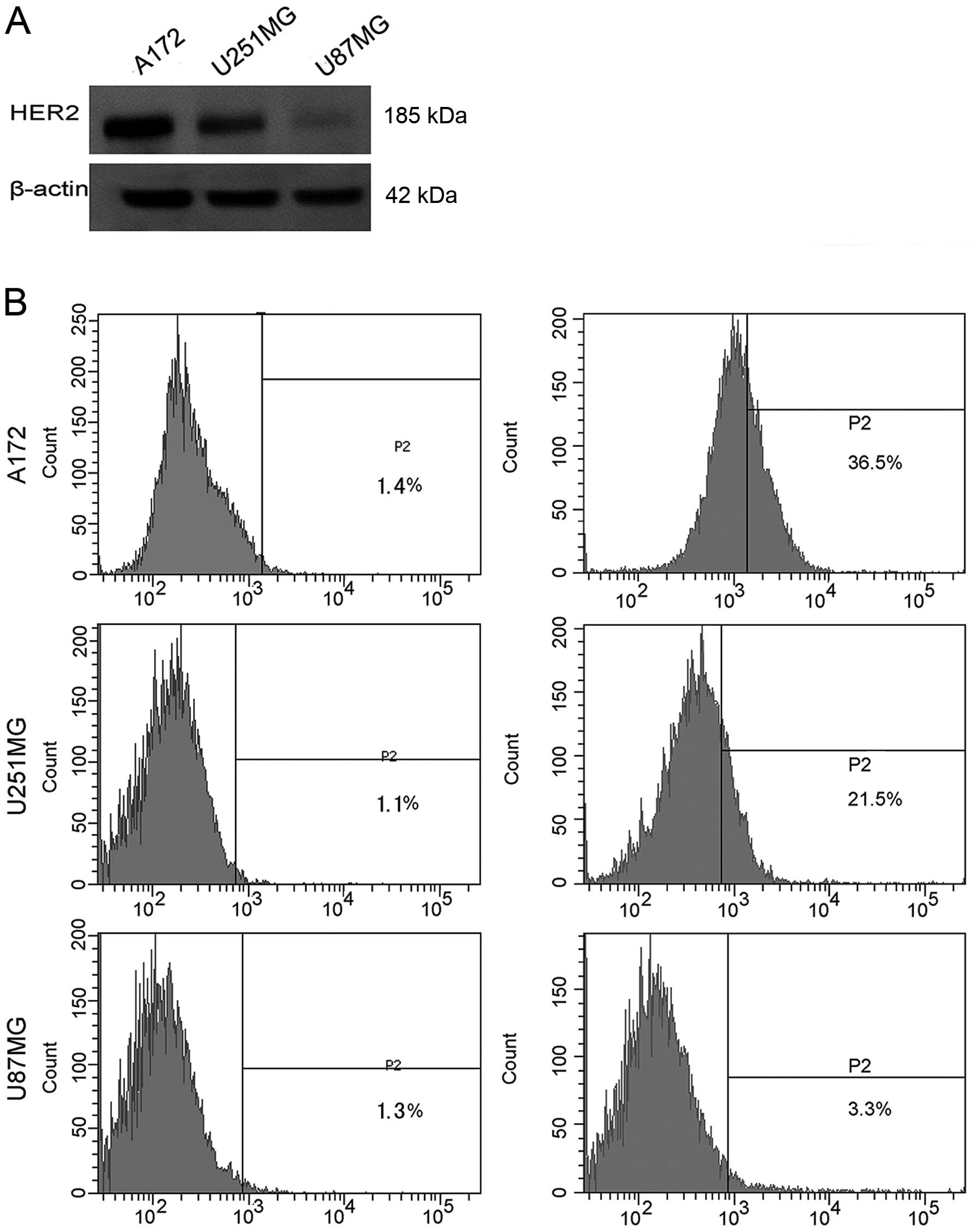
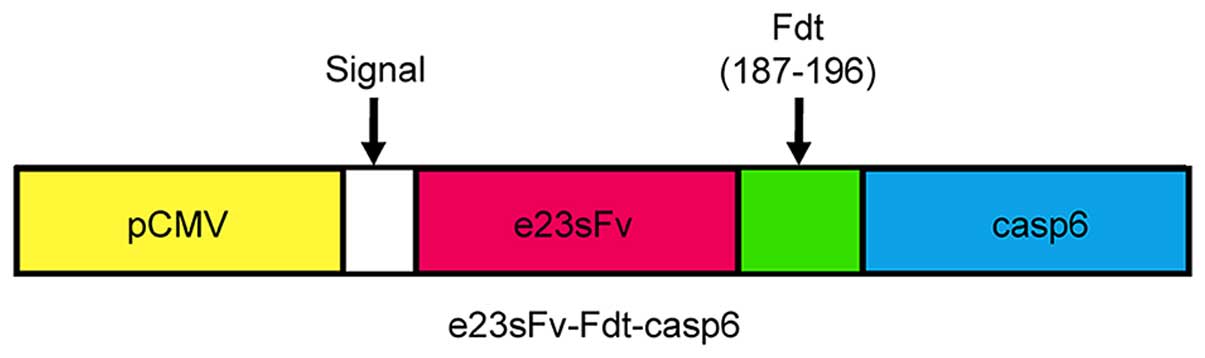
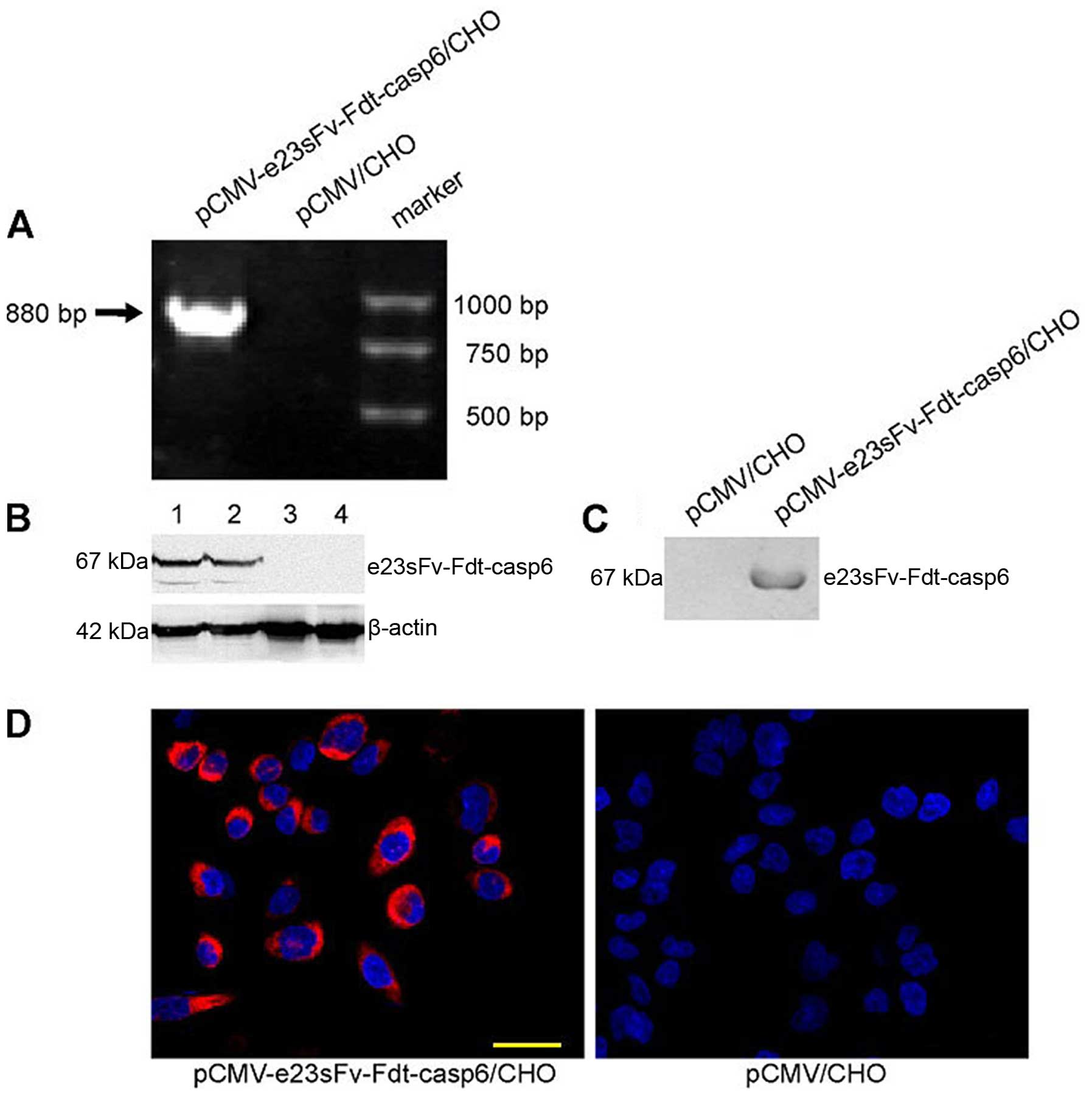
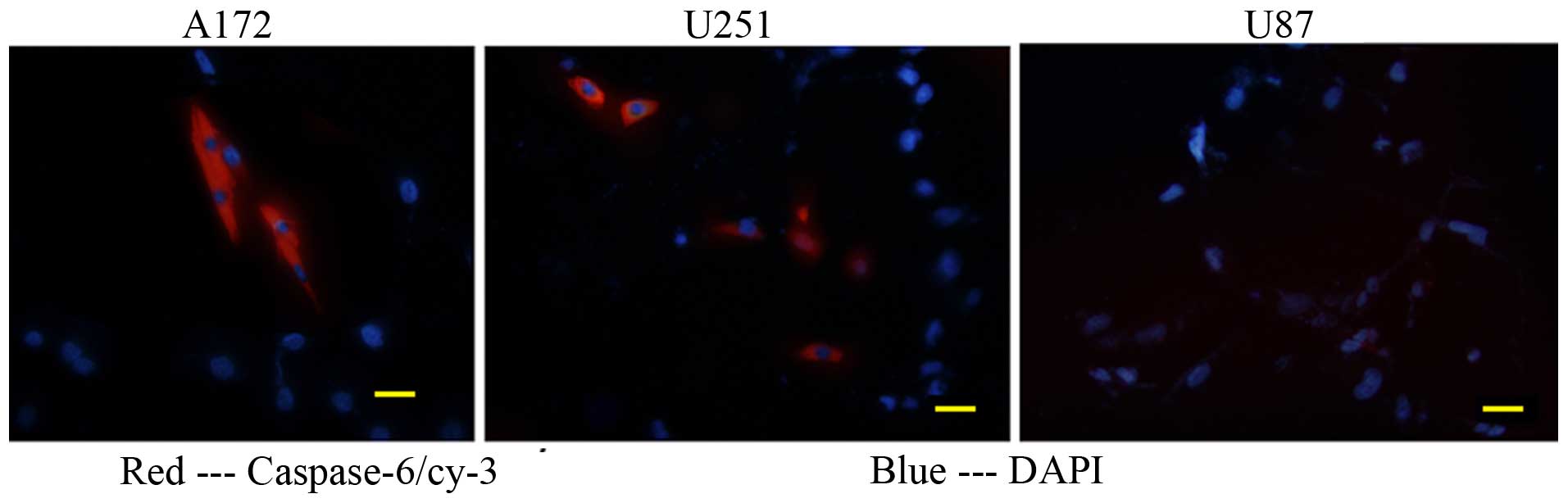
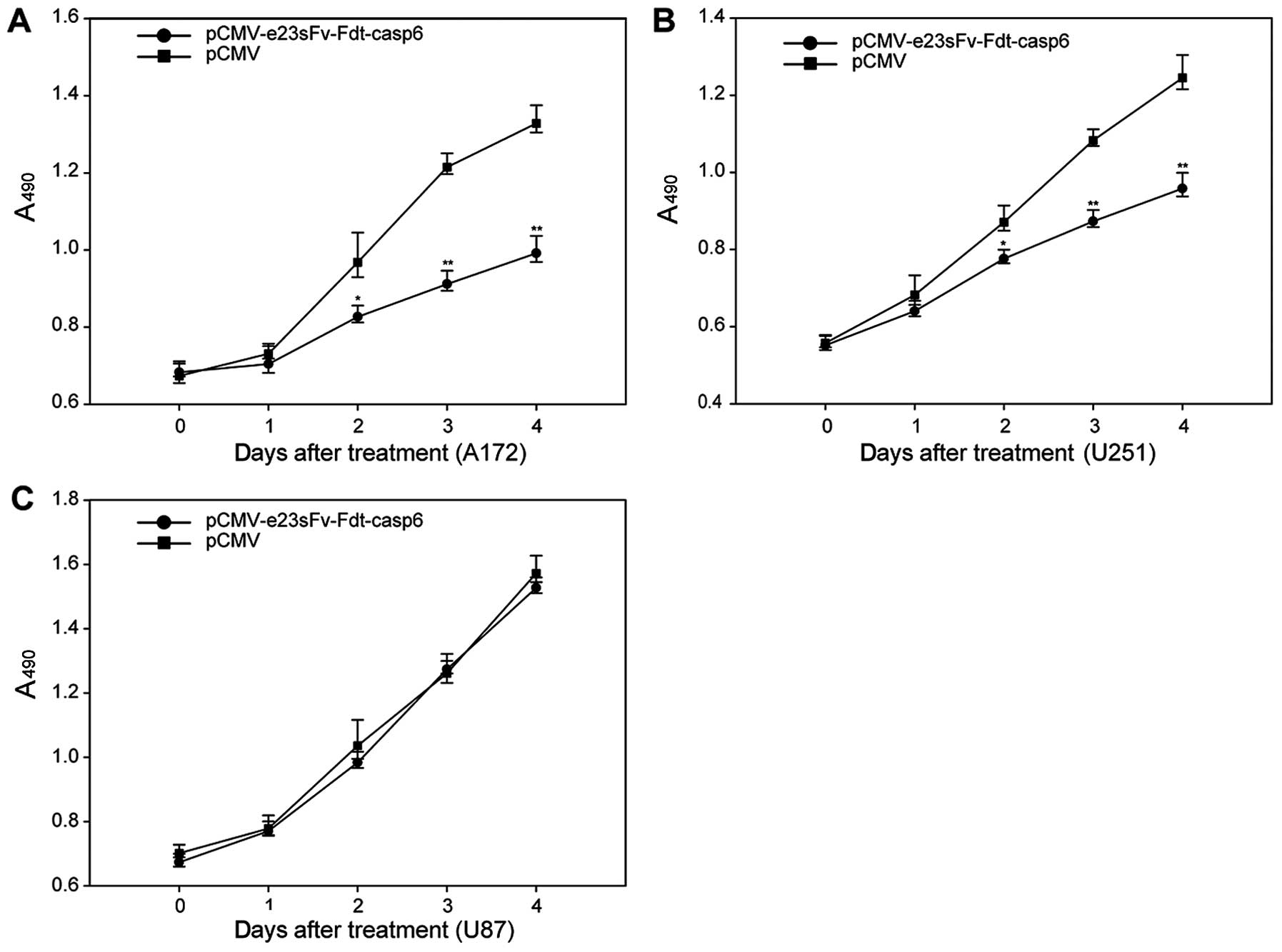
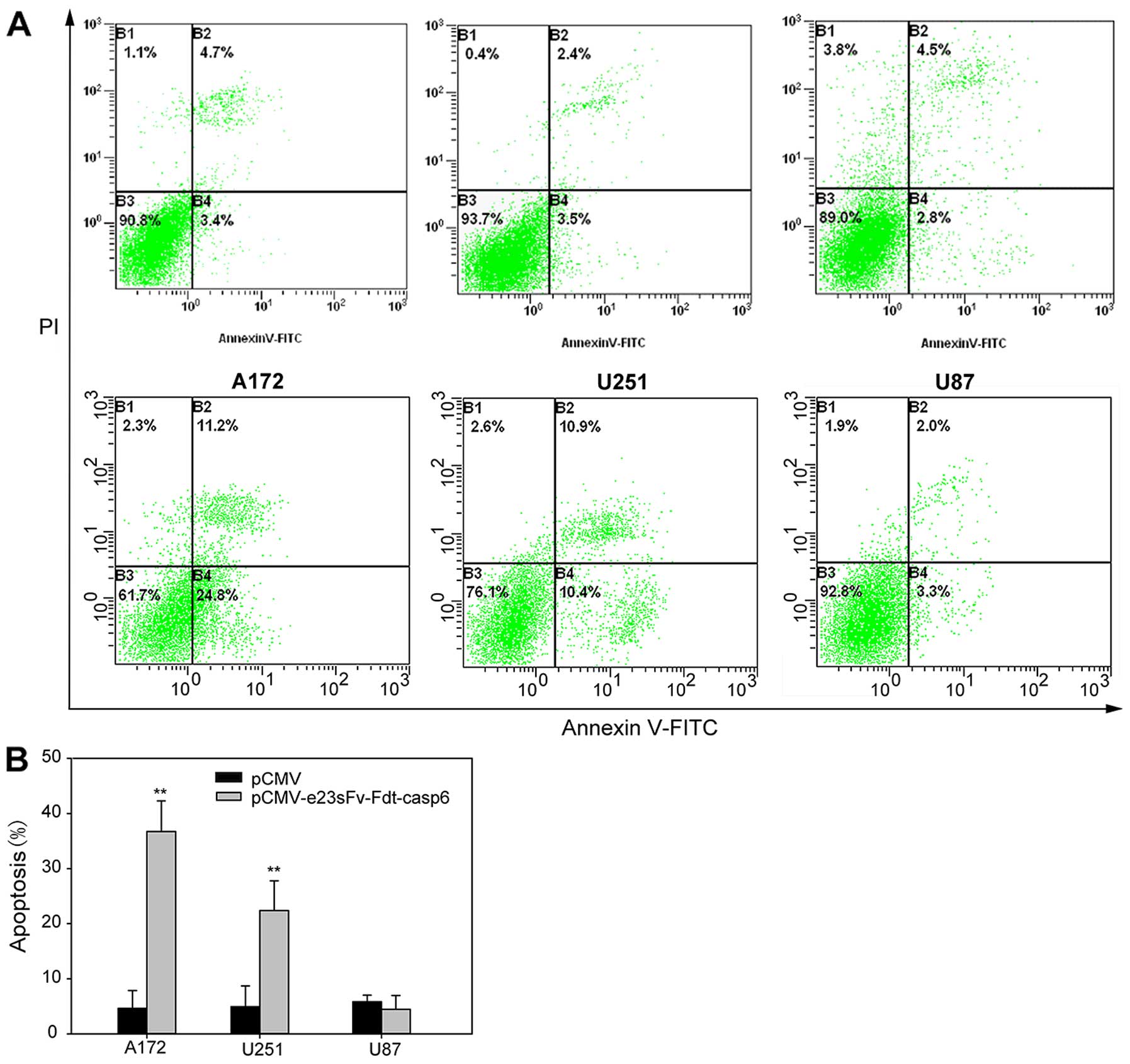
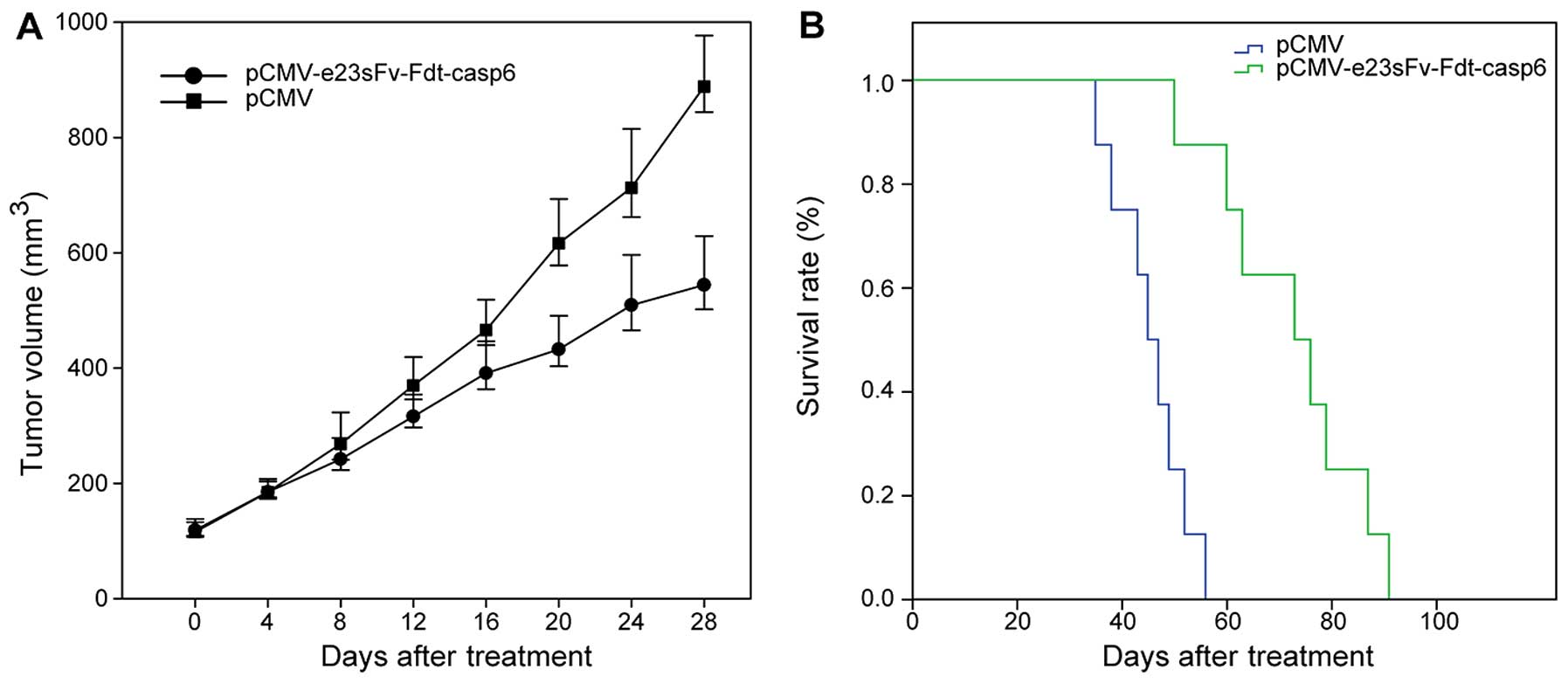
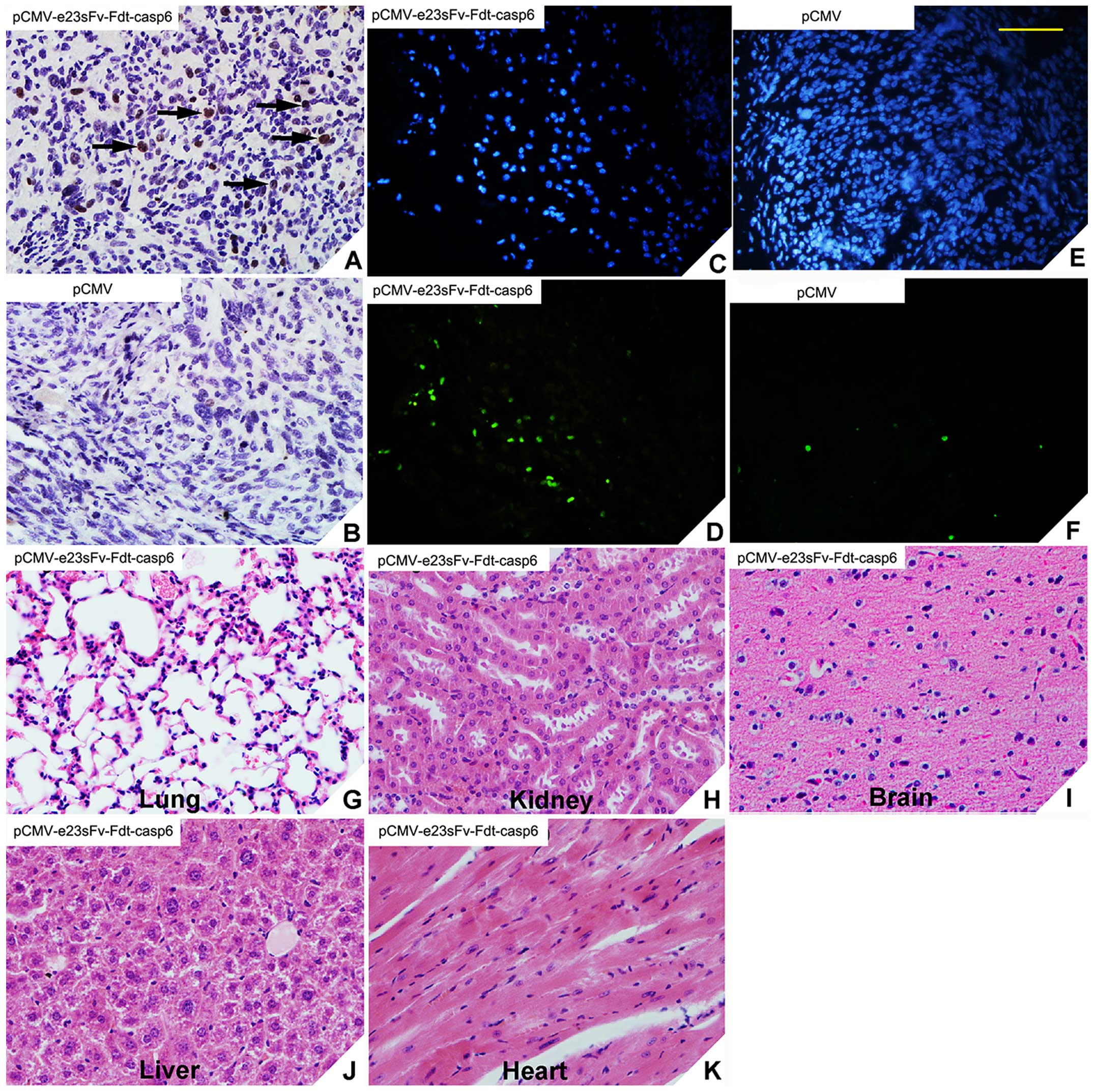
|
1
|
Wen PY and Kesari S: Malignant gliomas in
adults. N Engl J Med. 359:492–507. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Furnari FB, Fenton T, Bachoo RM, Mukasa A,
Stommel JM, Stegh A, Hahn WC, Ligon KL, Louis DN, Brennan C, et al:
Malignant astrocytic glioma: Genetics, biology, and paths to
treatment. Genes Dev. 21:2683–2710. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ohgaki H and Kleihues P: Genetic pathways
to primary and secondary glioblastoma. Am J Pathol. 170:1445–1453.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lupu R, Colomer R, Kannan B and Lippman
ME: Characterization of a growth factor that binds exclusively to
the erbB-2 receptor and induces cellular responses. Proc Natl Acad
Sci USA. 89:2287–2291. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Koka V, Potti A, Forseen SE, Pervez H,
Fraiman GN, Koch M and Levitt R: Role of Her-2/neu overexpression
and clinical determinants of early mortality in glioblastoma
multiforme. Am J Clin Oncol. 26:332–335. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ahmed N, Salsman VS, Kew Y, Shaffer D,
Powell S, Zhang YJ, Grossman RG, Heslop HE and Gottschalk S:
HER2-specific T cells target primary glioblastoma stem cells and
induce regression of autologous experimental tumors. Clin Cancer
Res. 16:474–485. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu G, Ying H, Zeng G, Wheeler CJ, Black
KL and Yu JS: HER-2, gp100, and MAGE-1 are expressed in human
glioblastoma and recognized by cytotoxic T cells. Cancer Res.
64:4980–4986. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bian XW, Shi JQ and Liu FX: Pathologic
significance of proliferative activity and oncoprotein expression
in astrocytic tumors. Anal Quant Cytol Histol. 22:429–437.
2000.
|
|
9
|
Forseen SE, Potti A, Koka V, Koch M,
Fraiman G and Levitt R: Identification and relationship of
HER-2/neu overexpression to short-term mortality in primary
malignant brain tumors. Anticancer Res. 22:1599–1602.
2002.PubMed/NCBI
|
|
10
|
Mineo JF, Bordron A, Quintin-Roué I,
Loisel S, Ster KL, Buhé V, Lagarde N and Berthou C: Recombinant
humanised anti-HER2/neu antibody (Herceptin) induces cellular death
of glioblastomas. Br J Cancer. 91:1195–1199. 2004.PubMed/NCBI
|
|
11
|
Press MF, Cordon-Cardo C and Slamon DJ:
Expression of the HER-2/neu proto-oncogene in normal human adult
and fetal tissues. Oncogene. 5:953–962. 1990.PubMed/NCBI
|
|
12
|
Bold RJ, Termuhlen PM and McConkey DJ:
Apoptosis, cancer and cancer therapy. Surg Oncol. 6:133–142. 1997.
View Article : Google Scholar
|
|
13
|
Riedl SJ and Shi Y: Molecular mechanisms
of caspase regulation during apoptosis. Nat Rev Mol Cell Biol.
5:897–907. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Srinivasula SM, Ahmad M, MacFarlane M, Luo
Z, Huang Z, Fernandes-Alnemri T and Alnemri ES: Generation of
constitutively active recombinant caspases-3 and -6 by
rearrangement of their subunits. J Biol Chem. 273:10107–10111.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Komata T, Kondo Y, Kanzawa T, Hirohata S,
Koga S, Sumiyoshi H, Srinivasula SM, Barna BP, Germano IM, Takakura
M, et al: Treatment of malignant glioma cells with the transfer of
constitutively active caspase-6 using the human telomerase
catalytic subunit (human telomerase reverse transcriptase) gene
promoter. Cancer Res. 61:5796–5802. 2001.PubMed/NCBI
|
|
16
|
Ren JL, Meng YL, Hu B, Jia LT, Zhang R, Xu
YM, Xie QS, Zhang YQ, Jin BQ, Chen SY, et al: The effect of direct
translocation across endosomes on the cytotoxicity of the
recombinant protein e23sFv-Fdt-casp6 to HER2 positive gastric
cancer cells. Biomaterials. 32:7641–7650. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang T, Zhao J, Ren JL, Zhang L, Wen WH,
Zhang R, Qin WW, Jia LT, Yao LB, Zhang YQ, et al: Recombinant
immunoproapoptotic proteins with furin site can translocate and
kill HER2-positive cancer cells. Cancer Res. 67:11830–11839. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Omasa T, Onitsuka M and Kim WD: Cell
engineering and cultivation of chinese hamster ovary (CHO) cells.
Curr Pharm Biotechnol. 11:233–240. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mineo JF, Bordron A, Quintin-Roué I,
Maurage CA, Buhé V, Loisel S, Dubois F, Blond S and Berthou C:
Increasing of HER2 membranar density in human glioblastoma U251MG
cell line established in a new nude mice model. J Neurooncol.
76:249–255. 2006. View Article : Google Scholar
|
|
20
|
Vajkoczy P, Schilling L, Ullrich A,
Schmiedek P and Menger MD: Characterization of angiogenesis and
microcirculation of high-grade glioma: An intravital
multifluorescence microscopic approach in the athymic nude mouse. J
Cereb Blood Flow Metab. 18:510–520. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hegde M, Corder A, Chow KK, Mukherjee M,
Ashoori A, Kew Y, Zhang YJ, Baskin DS, Merchant FA, Brawley VS, et
al: Combinational targeting offsets antigen escape and enhances
effector functions of adoptively transferred T cells in
glioblastoma. Mol Ther. 21:2087–2101. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Akiyama Y, Oshita C, Kume A, Iizuka A,
Miyata H, Komiyama M, Ashizawa T, Yagoto M, Abe Y, Mitsuya K, et
al: α-type-1 polarized dendritic cell-based vaccination in
recurrent high-grade glioma: A phase I clinical trial. BMC Cancer.
12:6232012. View Article : Google Scholar
|
|
23
|
Phuphanich S, Wheeler CJ, Rudnick JD,
Mazer M, Wang H, Nuño MA, Richardson JE, Fan X, Ji J, Chu RM, et
al: Phase I trial of a multi-epitope-pulsed dendritic cell vaccine
for patients with newly diagnosed glioblastoma. Cancer Immunol
Immunother. 62:125–135. 2013. View Article : Google Scholar :
|
|
24
|
Sasaki S, Takeshita F, Xin KQ, Ishii N and
Okuda K: Adjuvant formulations and delivery systems for DNA
vaccines. Methods. 31:243–254. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Robinson HL and Pertmer TM: DNA vaccines
for viral infections: Basic studies and applications. Adv Virus
Res. 55:1–74. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sun Y, Hu YH, Liu CS and Sun L:
Construction and analysis of an experimental Streptococcus iniae
DNA vaccine. Vaccine. 28:3905–3912. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kim D, Hung CF, Wu TC and Park YM: DNA
vaccine with α-galactosylceramide at prime phase enhances
anti-tumor immunity after boosting with antigen-expressing
dendritic cells. Vaccine. 28:7297–7305. 2010. View Article : Google Scholar : PubMed/NCBI
|