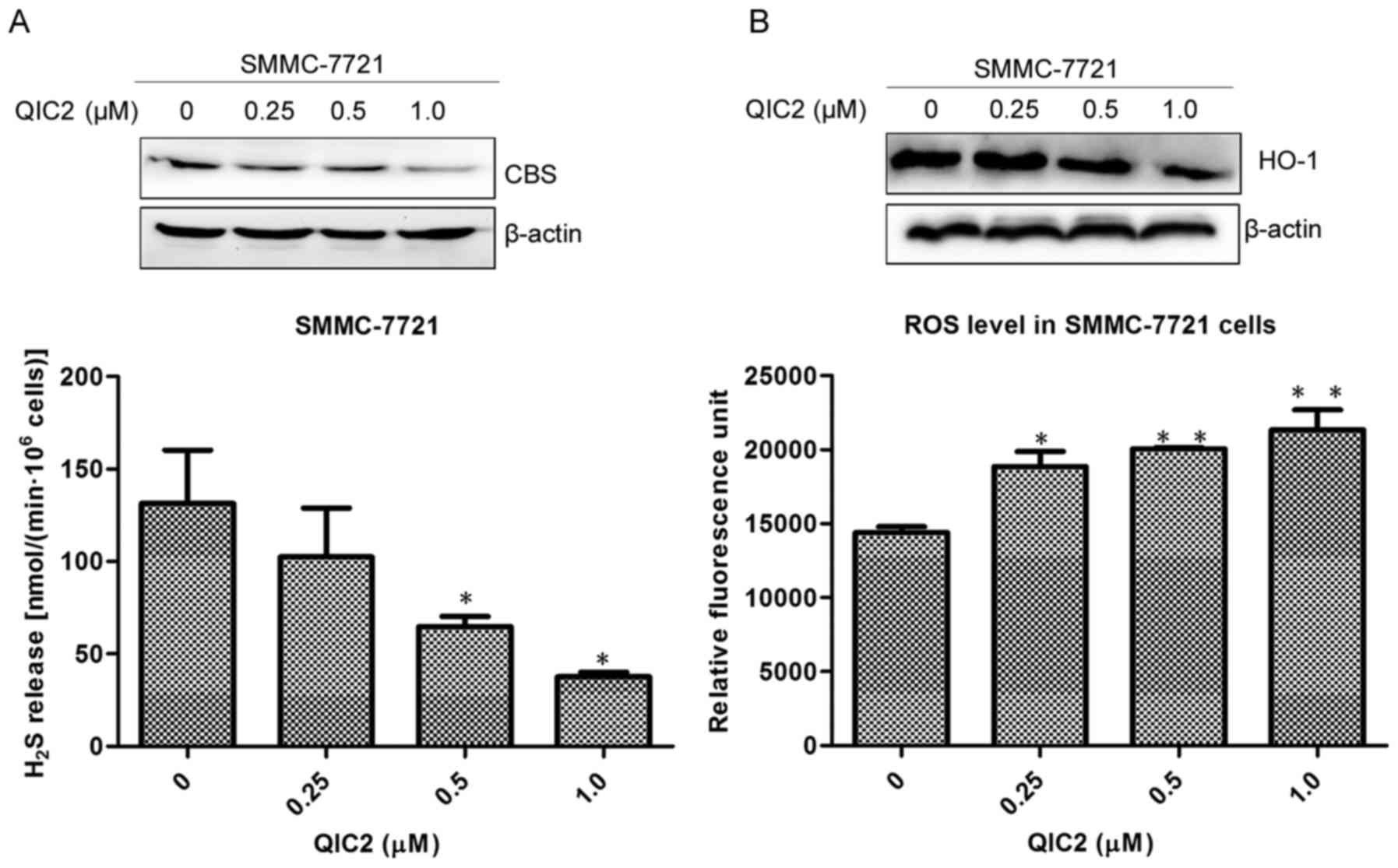
|
1
|
Coletta C, Papapetropoulos A, Erdelyi K,
Olah G, Módis K, Panopoulos P, Asimakopoulou A, Gerö D, Sharina I,
Martin E, et al: Hydrogen sulfide and nitric oxide are mutually
dependent in the regulation of angiogenesis and
endothelium-dependent vasorelaxation. Proc Natl Acad Sci USA.
109:9161–9166. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Szabo C, Coletta C, Chao C, Módis K,
Szczesny B, Papapetropoulos A and Hellmich MR: Tumor-derived
hydrogen sulfide, produced by cystathionine-β-synthase, stimulates
bioenergetics, cell proliferation, and angiogenesis in colon
cancer. Proc Natl Acad Sci USA. 110:12474–12479. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kimura Y, Goto Y and Kimura H: Hydrogen
sulfide increases glutathione production and suppresses oxidative
stress in mitochondria. Antioxid Redox Signal. 12:1–13. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sheng J, Shim W, Wei H, Lim SY, Liew R,
Lim TS, Ong BH, Chua YL and Wong P: Hydrogen sulphide suppresses
human atrial fibroblast proliferation and transformation to
myofibroblasts. J Cell Mol Med. 17:1345–1354. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Popov D: An outlook on vascular hydrogen
sulphide effects, signalling, and therapeutic potential. Arch
Physiol Biochem. 119:189–194. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yin P, Zhao C, Li Z, Mei C, Yao W, Liu Y,
Li N, Qi J, Wang L, Shi Y, et al: Sp1 is involved in regulation of
cystathionine γ-lyase gene expression and biological function by
PI3K/Akt pathway in human hepatocellular carcinoma cell lines. Cell
Signal. 24:1229–1240. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baskar R, Li L and Moore PK: Hydrogen
sulfide-induces DNA damage and changes in apoptotic gene expression
in human lung fibroblast cells. FASEB J. 21:247–255. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sivarajah A, Collino M, Yasin M, Benetti
E, Gallicchio M, Mazzon E, Cuzzocrea S, Fantozzi R and Thiemermann
C: Anti-apoptotic and anti-inflammatory effects of hydrogen sulfide
in a rat model of regional myocardial I/R. Shock. 31:267–274. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cao Y, Adhikari S, Ang AD, Moore PK and
Bhatia M: Mechanism of induction of pancreatic acinar cell
apoptosis by hydrogen sulfide. Am J Physiol Cell Physiol.
291:C503–C510. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bhattacharyya S, Saha S, Giri K, Lanza IR,
Nair KS, Jennings NB, Rodriguez-Aguayo C, Lopez-Berestein G, Basal
E, Weaver AL, et al: Cystathionine beta-synthase (CBS) contributes
to advanced ovarian cancer progression and drug resistance. PLoS
One. 8:e791672013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Szabo C, Coletta C, Chao C, Módis K,
Szczesny B, Papapetropoulos A and Hellmich MR: Tumor-derived
hydrogen sulfide, produced by cystathionine-β-synthase, stimulates
bioenergetics, cell proliferation, and angiogenesis in colon
cancer. Proc Natl Acad Sci USA. 110:12474–12479. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang R: Hydrogen sulfide: The third
gasotransmitter in biology and medicine. Antioxid Redox Signal.
12:1061–1064. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu YH, Wei XL, Hu GQ and Wang TX:
Quinolone-indolone conjugate induces apoptosis by inhibiting the
EGFR-STAT3-HK2 pathway in human cancer cells. Mol Med Rep.
12:2749–2756. 2015.PubMed/NCBI
|
|
14
|
Maclean KN, Janosík M, Kraus E, Kozich V,
Allen RH, Raab BK and Kraus JP: Cystathionine beta-synthase is
coordinately regulated with proliferation through a redox-sensitive
mechanism in cultured human cells and Saccharomyces
cerevisiae. J Cell Physiol. 192:81–92. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sanokawa-Akakura R, Ostrakhovitch EA,
Akakura S, Goodwin S and Tabibzadeh S: A H2S-Nampt
dependent energetic circuit is critical to survival and
cytoprotection from damage in cancer cells. PLoS One.
9:e1085372014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hua W, Chen Q, Gong F, Xie C, Zhou S and
Gao L: Cardioprotection of H2S by downregulating iNOS
and upregulating HO-1 expression in mice with CVB3-induced
myocarditis. Life Sci. 93:949–954. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Danial NN and Korsmeyer SJ: Cell death:
Critical control points. Cell. 116:205–219. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fulda S and Debatin KM: Sensitization for
anticancer drug-induced apoptosis by the chemopreventive agent
resveratrol. Oncogene. 23:6702–6711. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu Z, Li D, Zheng X, Wang E and Wang J:
Selective induction of apoptosis: Promising therapy in pancreatic
cancer. Curr Pharm Des. 19:2259–2268. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kaufmann SH and Earnshaw WC: Induction of
apoptosis by cancer chemotherapy. Exp Cell Res. 256:42–49. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shan T, Ma Q, Zhang D, Guo K, Liu H, Wang
F and Wu E: β2-adrenoceptor blocker synergizes with gemcitabine to
inhibit the proliferation of pancreatic cancer cells via apoptosis
induction. Eur J Pharmacol. 665:1–7. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Circu ML and Aw TY: Reactive oxygen
species, cellular redox systems, and apoptosis. Free Radic Biol
Med. 48:749–762. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kuranaga Y, Yamada N, Kashiwaya M,
Nakamura M, Cui L, Kumazaki M, Shinohara H, Sugito N, Taniguchi K,
Ito Y, et al: Anti-oncogenic gem-dihydroperoxides induce
apoptosis in cancer cells by trapping reactive oxygen species. Int
J Mol Sci. 17:E712016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
He G, He G, Zhou R, Pi Z, Zhu T, Jiang L
and Xie Y: Enhancement of cisplatin-induced colon cancer cells
apoptosis by shikonin, a natural inducer of ROS in vitro and in
vivo. Biochem Biophys Res Commun. 469:1075–1082. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Manna A, Saha P, Sarkar A, Mukhopadhyay D,
Bauri AK, Kumar D, Das P, Chattopadhyay S and Chatterjee M:
Malabaricone-A induces a redox imbalance that mediates apoptosis in
U937 cell line. PLoS One. 7:e369382012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ahamed M, Akhtar MJ, Raja M, Ahmad I,
Siddiqui MK, AlSalhi MS and Alrokayan SA: ZnO nanorod-induced
apoptosis in human alveolar adenocarcinoma cells via p53, survivin
and bax/bcl-2 pathways: Role of oxidative stress. Nanomedicine.
7:904–913. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mileo AM and Miccadei S: Polyphenols as
modulator of oxidative stress in cancer disease: New therapeutic
strategies. Oxid Med Cell Longev. 2016:64756242016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee G, Oh TI, Um KB, Yoon H, Son J, Kim
BM, Kim HI, Kim H, Kim YJ, Lee CS, et al: Small-molecule inhibitors
of USP7 induce apoptosis through oxidative and endoplasmic
reticulum stress in cancer cells. Biochem Biophys Res Commun.
470:181–186. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pan Y, Ye S, Yuan D, Zhang J, Bai Y and
Shao C: Hydrogen sulfide (H2S)/cystathionine γ-lyase
(CSE) pathway contributes to the proliferation of hepatoma cells.
Mutat Res. 763–764:10–18. 2014. View Article : Google Scholar
|