Introduction
Prostate cancer was the second most frequently
diagnosed cancer in 2012 (1) and
the sixth leading cause of death due to cancer in males worldwide
(2). Research into the pathways of
prostate cancer growth suggests a novel therapeutic strategy to
enhance the survival of this group (3).
Since vessels do not form in tumors, adequate gas
exchange does not occur between the tumor cells and surrounding
environment. This leads to intratumoral hypoxia, known to promote
cancer cell proliferation, invasion, metastasis, and resistance to
radiation and chemotherapy (4,5). We
previously reported that chronic hypoxia induces
androgen-independent growth in the human prostate cancer cell line
LNCaP (6). Hypoxic conditions
upregulate the expression of some proteins including
angiopoietin-like protein 4 (ANGPTL4).
ANGPTL4 is a member of the angiopoietin family and
is highly expressed in adipose and gastrointestinal tissues
(7,8). Furthermore, ANGPTL4 is also known to
influence inflammation, angiogenesis, and tumorigenesis (9–12).
Several studies have reported a role of ANGPTL4 in cancer
development, and its suppression can impair tumor growth by
enhancing apoptosis (13).
Considering the fact that hypoxia is a feature of the tumor
microenvironment (5), it has been
suggested that ANGPTL4 has effects on prostate cancer growth and/or
malignant behavior. However, knowledge on the function of ANGPTL4
in human prostate cancer remains limited (14).
Thus in the present study, we examined ANGPTL4
expression in prostate cancer cell lines under hypoxia and
investigated the link between ANGPTL4 expression and associated
clinicopathological factors.
Materials and methods
Cell cultures
We purchased a human-androgen-dependent prostate
cancer cell line (LNCaP) from the American Type Culture Collection
(Rockville, MD, USA). These cells were cultured in RPMI-1640 medium
(Sigma-Aldrich, St. Louis, MO, USA) supplemented with 15% fetal
bovine serum (FBS, Sigma-Aldrich), 50 IU/ml penicillin, and 50
mg/ml streptomycin sulfate (Gibco, Grand Island, NY, USA) at 37°C
with 5% CO2 and 95% room air (normoxia), or 5%
CO2, 94% N2, and 1% O2 (hypoxia).
LNCaP/CH cells were cultured under hypoxia for at least 6
months.
Proliferation assay
The cells were seeded into 12-well plates at a
density of 1×105 cells per well. They were then
trypsinized, collected, and counted using a hemocytometer 24, 48,
and 72 h after seeding.
Water-soluble tetrazolium salt (WST-1)
assay
The cells were seeded into 96-well plates at density
of 5,000 cells per well. After culturing overnight, a fresh
standard medium containing various concentrations of ANGPTL4
recombinant proteins (rANGPTL4) (0–1000 pg/ml) was added to each
well. After incubation for 48 h, 5 mM WST-1 (Dojindo, Kumamoto,
Japan) and 3 nM 1-methoxy-5-methylphenazinium methyl sulfate
(Dojindo) were added to each well. After incubation for 1–4 h,
absorbance was measured at 450 nm with a microplate reader (Tecan
Japan Co., Ltd., Kanagawa, Japan).
Cell migration assay
Cell migration was assessed using a 24-well plate
with Corning® FluoroBlok Inserts (Corning Inc., Corning,
NY, USA). The cells were seeded at 5×104 cells per well
in 0.5-ml serum-free medium. The outer chambers were filled with
0.75 ml of media containing 15% FBS. After 24 or 48 h, migrating
cells were labeled using Cellstain®-Calcein-AM solution
(Dojindo). After incubation for 1 h under normoxia or hypoxia, the
cell fluorescence was detected using a microplate reader.
RNA isolation and quantitative
real-time PCR
This method was performed as previously described
(6). The primers comprised ANGPTL4
(Hs_ANGPTL4_1_SG QuantiTect Primer Assay, QT00003631) and β-actin
(HS_ACTB_1_SG QuantiTect Primer Assay, QT00095431). The
quantification of mRNA expression was normalized using β-actin.
Quantification of ANGPTL4 protein by
ELISA
The cells were cultured in six-well plates at a
density of 5×105 cells per well for 24, 48, and 72 h.
The medium was then collected before adding fresh medium to the
cultures. The collected medium was used to quantify ANGPTL4 protein
concentration using a commercial ELISA according to the
manufacturer's instructions (Human ANGPTL4 ELISA; Raybiotech, Inc.,
Norcross, GA, USA).
Establishment of
ANGPTL4-overexpressing cells
To create stably transfected lines, LNCaP cells were
transduced with either FLAG-tagged ANGPTL4-expressing (Addgene) or
control vectors (Cell Biolabs, Inc., San Diego, CA, USA) using
Lipofectamine 3000 (Invitrogen), according to the manufacturer's
protocol. The transfected cell lines were selected in 0.2-µg/ml
puromycin dihydrochloride (Santa Cruz Biotechnology, Dallas, TX,
USA).
Protein extraction and immunoblot
analysis
This method was performed as previously described
(6). The primary antibodies
included anti-Bcl-2 (cat. no. SASC509, Santa Cruz Biotechnology),
anti-Bad (cat. no. 9292T, Cell Signaling), anti-phospho-Akt
(Set473; cat. no. 9271S, Cell Signaling), anti-Akt (cat. no. 9272S,
Cell Signaling), anti-β-tubulin (cat. no. #MAB3408, EMD Millipore),
and anti-ANGPTL4 (cat. no. GTX114198, GeneTex).
ANGPTL4 knockdown by RNA
interference
LNCaP cells were transiently transfected with an
ANGPTL4 siRNA duplex [siANGPTL4, final concentration: 60 nmol/l
(Qiagen Inc.)] or control siRNA [random scrambled sequence: siScr,
final concentration: 60 nmol/l (Qiagen Inc.)] using Lipofectamine
RNAiMAX (Invitrogen Life Technologies) according to the
manufacturer's instructions. The siRNA sequences against ANGPTL4
generated by Qiagen were 5′-GGGACAAGAACUGCGCCAATT-3′ and
5′-UUGGCGCAGUUCUUGUCCCTG −3′.
Flow cytometry analysis (FACS)
This method was performed as previously described
(6). The cell cycles were analyzed
using a BD FACSVerse™ flow cytometer and BD FACSuite™ software
(Becton Dickinson, San Jose, CA, USA).
Docetaxel or LY294002 treatment
The cells were seeded into 12-well plates at a
density of 1×105 cells per well. After culturing
overnight, a fresh standard medium containing 2 nM docetaxel
(Sigma-Aldrich) was added to the plates. The phosphoinositide
3-kinase (PI3K) inhibitor, LY294002, was obtained from Calbiochem
(Darmstadt, Germany). Docetaxel is the standard chemotherapy for
men with advanced prostate cancer.
Patients
As this study was a retrospective chart review, no
patient consent was required. From June 2009 to December 2012, 92
patients underwent curative surgery involving a radical
prostatectomy for localized prostate cancer at Oita University
Hospital (Oita, Japan). Patients who received therapy before and/or
immediately after surgery were excluded, and data from the
remaining 70 patients were analyzed (Table I). After surgery, the serum prostate
specific antigen (PSA) levels were monitored, and PSA recurrence
was defined as an elevation in serum PSA levels (≥0.2 ng/ml) in two
consecutive measurements.
 | Table I.Characteristics of the patients and
tumors. |
Table I.
Characteristics of the patients and
tumors.
| Characteristics | Value |
|---|
| Median age, years
(range) | 67.5 (49–76) |
| Median follow-up,
months (range) | 53.5 (13–82) |
| Median PSA at
diagnosis, ng/ml (range) | 9.0 (3.7–23.9) |
| PSA |
|
|
<10 | 43 |
| ≥10 | 27 |
| pT |
|
| ≤2 | 61 |
| ≥3 | 9 |
| Gleason score |
| ≤6 | 20 |
| ≥7 | 47 |
| Unremarkable | 3 |
| Extraprostatic
extension |
| 0 | 50 |
| 1 | 20 |
| Resection
margin |
| 0 | 54 |
| 1 | 14 |
| Unremarkable | 2 |
| Biochemical
recurrence |
| – | 51 |
| + | 19 |
Immunohistochemistry
This method was performed as previously described
(15). The primary polyclonal goat
anti-human ANGPTL4 (cat. no. 18374-1-AP, Proteintech™) antibody
diluted to 100X with PBS containing 1% bovine serum albumin was
applied.
Evaluation of immunostaining
To evaluate ANGPTL4 staining, we counted the
proportion of positively stained cells out of 1,000 cells from five
randomly selected areas. ANGPTL4 expression was divided according
to the percentage of positive tumor cells (negative, <20%; weak:
20–50%; and strong, >50%). Two authors independently evaluated
in a blinded manner to the clinical findings, the level of
immunoreactivity using an ECLIPSE E600 research microscope (Nikon
Corp., Tokyo, Japan).
Statistical analysis
Statistical analyses were performed using a
Student's t-test, Fisher's exact test, Mann-Whitney U-test, or
Spearman's rank correlation test. A two-tailed test was used for
all analyses. Biochemical recurrence-free survival was estimated
using the Kaplan-Meier method and compared the groups using the
log-rank test. A multivariate analysis was performed using a Cox's
proportional hazards model. The P-values of <0.05 were
considered to be statistically significant. All statistical
analyses were performed using EZR (Saitama Medical Center, Jichi
Medical University, Saitama, Japan), a graphical user interface for
R (The R Foundation for Statistical Computing, Vienna, Austria)
(16).
Results
ANGPTL4 expression levels during
chronic hypoxia
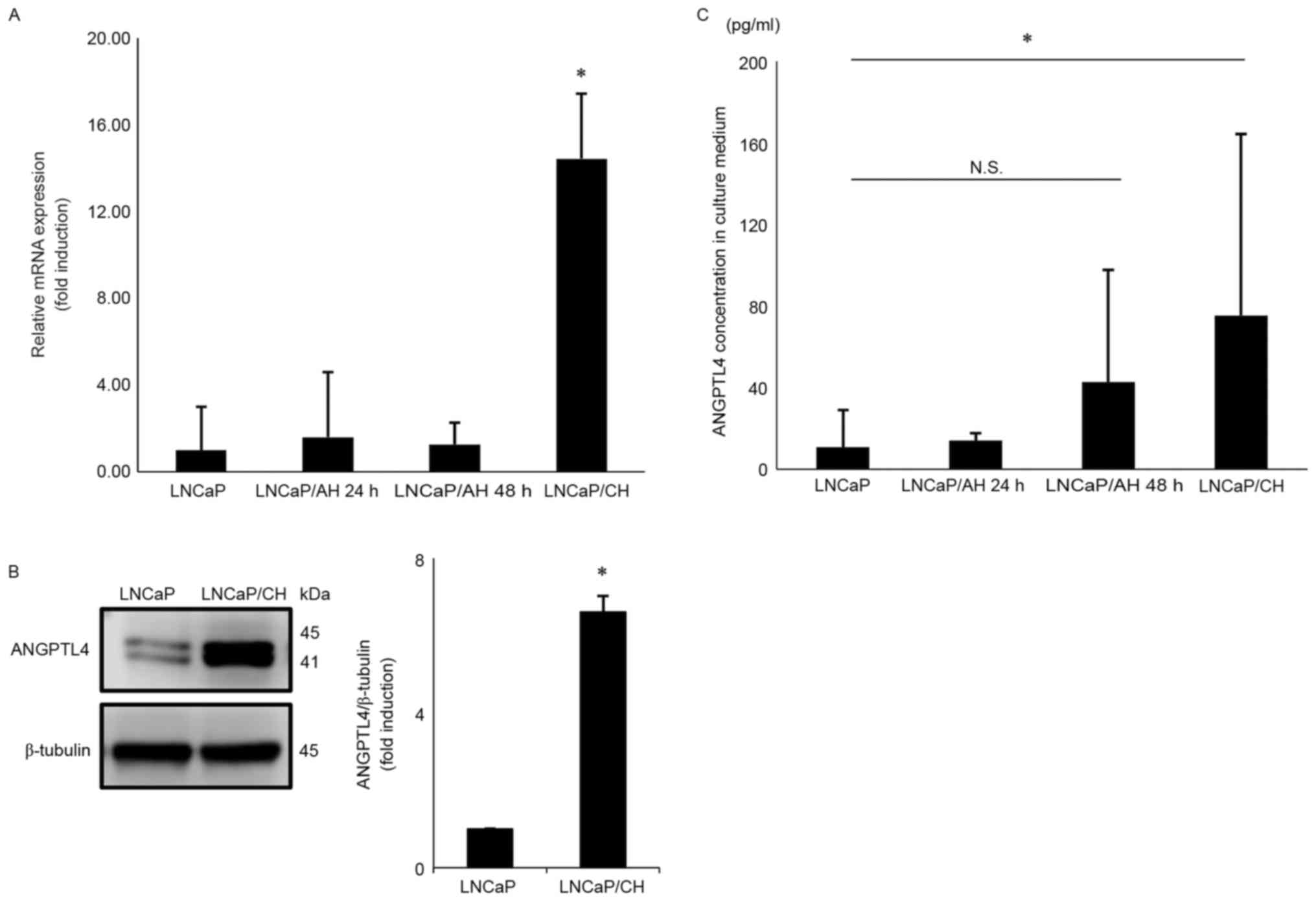
LNCaP/CH cells were grown under hypoxic conditions
for over 6 months, and relative ANGPTL4 mRNA expression was 14.4-,
9.0-, and 11.4-fold higher in LNCaP/CH cells than in LNCaP,
LNCaP/AH 24 h and LNCaP/AH 48 h cells, respectively (Fig. 1A). Similarly, ANGPTL4 protein
expression was 6.6-fold higher in LNCaP/CH cells than in LNCaP
cells (Fig. 1B). Fig. 1B showed a doublet band and the
molecular weight of the detected bands was 41 and 45 kDa. ANGPTL4
could be cleaved to an active form and the lower band indicates the
active form of ANGPTL4.
ANGPTL4 secretion levels during
chronic hypoxia
After 72 h, ANGPTL4 secretion was 7.0-fold higher in
LNCaP/CH cells than in LNCaP cells (Fig. 1C); however, no statistically
significant differences were found after 48 h (data not shown).
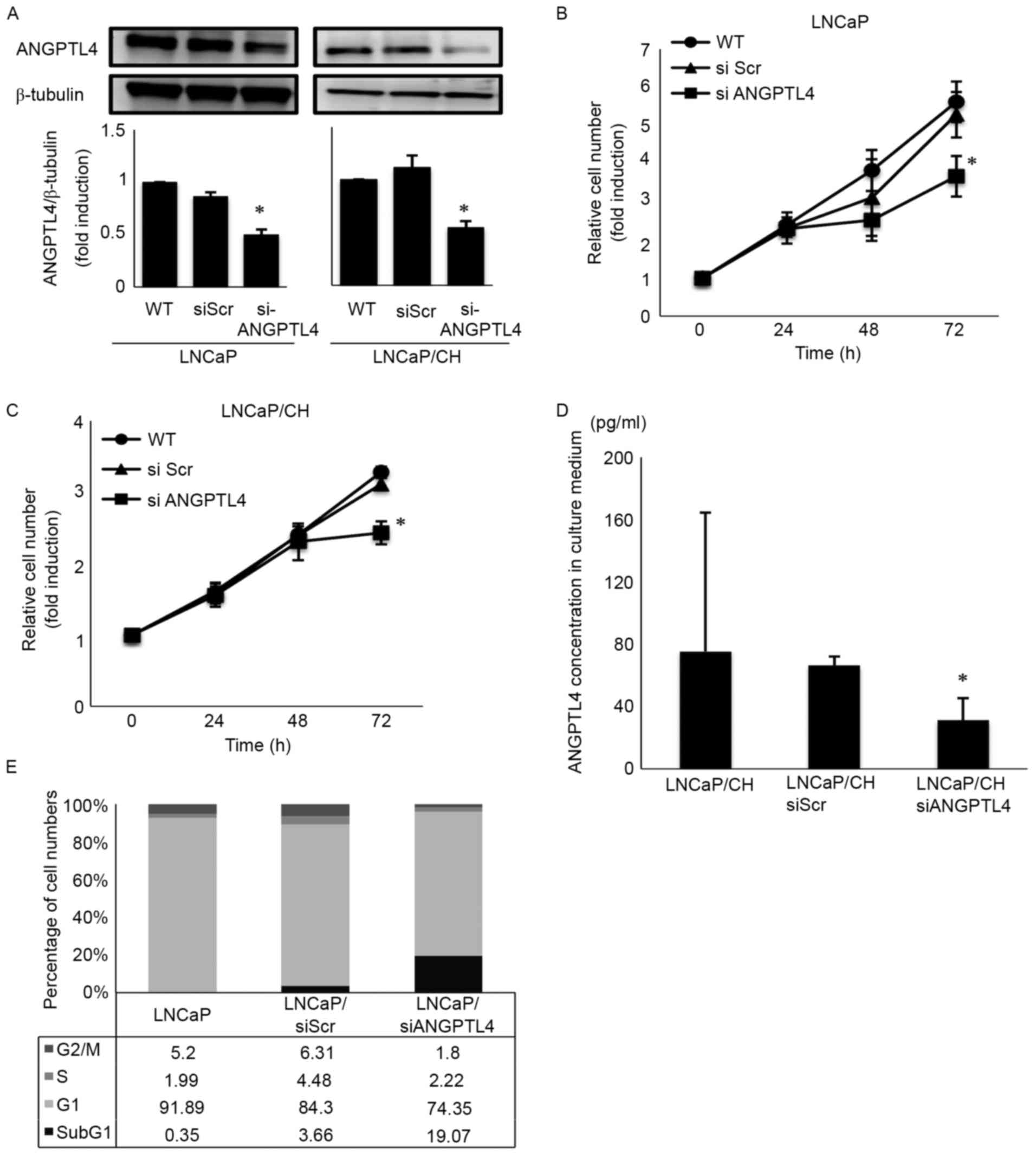
Effect of ANGPTL4 siRNA on LNCaP and
LNCaP/CH cell behavior
ANGPTL4 siRNA (siANGPTL4) effectively downregulated
ANGPTL4 expression in both LNCaP and LNCaP/CH cells (Fig. 2A). After incubation for 72 h with
siANGPTL4, cellular proliferation decreased to 82.0 and 74.2% in
LNCaP and LNCaP/CH cells, respectively (Fig. 2B and C). Similarly, siANGPTL4
decreased ANGPTL4 secretion to 41.2% after 72 h in LNCaP/CH cells
(Fig. 2D). Flow cytometry revealed
an increase in hypodiploid DNA (sub-G1 population) cells among the
siANGPTL4 transfected cells, concomitant to a significant growth
inhibition (Fig. 2E).
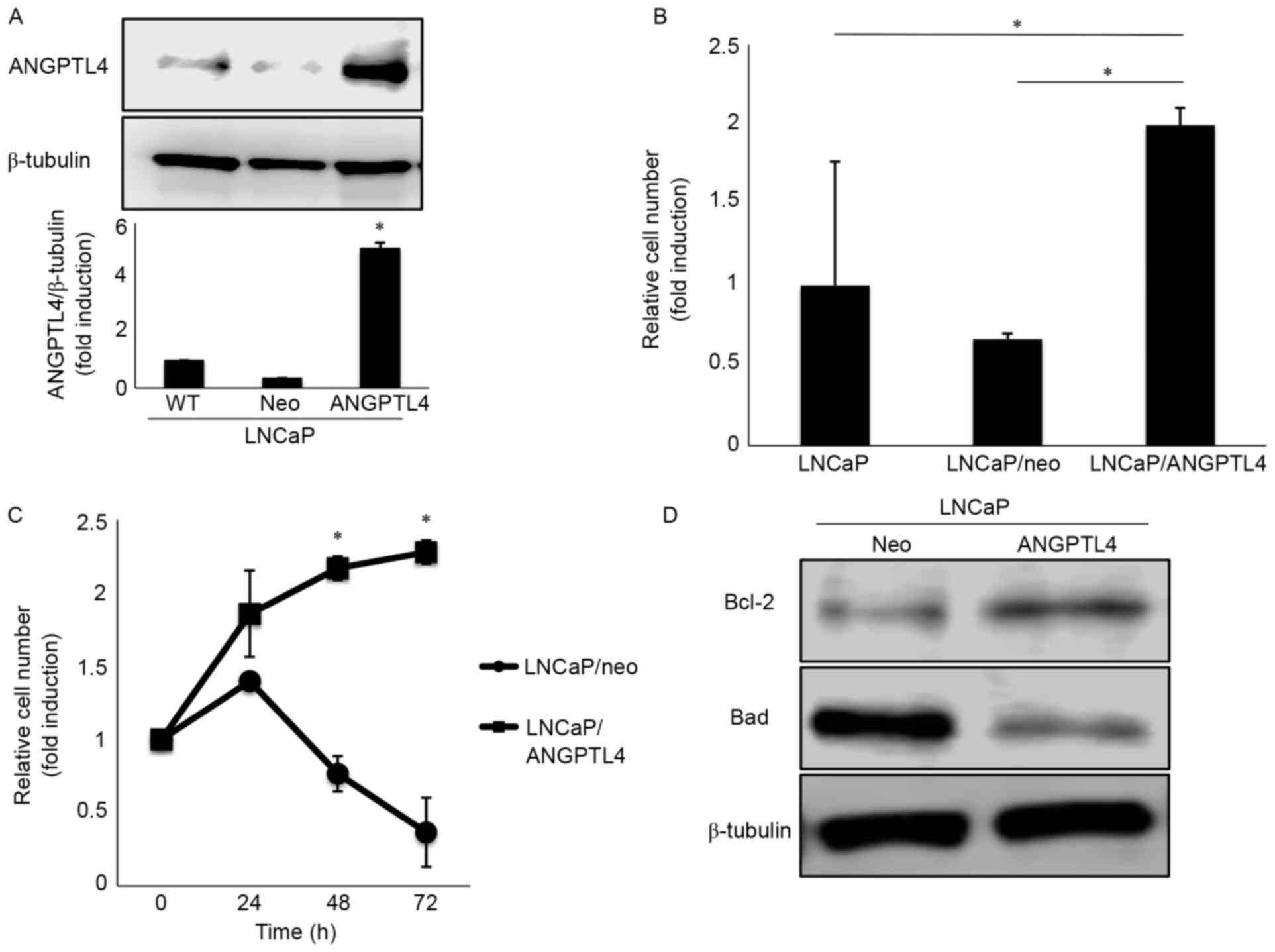
ANGPTL4 overexpression accelerates the
cellular migration of LNCaP cells
To investigate the precise behavior of ANGPTL4 in
prostate cancer, we established a stable cell line overexpressing
ANGPTL4 (LNCaP/ANGPTL4). LNCaP/ANGPTL4 increased ANGPTL4 expression
by 5.1-fold compared to the control (Fig. 3A). Furthermore, cellular migration
was 2.0-fold and 3.0-fold higher in LNCaP/ANGPTL4 cells than in
LNCaP and LNCaP/neo-transfected cells with a control vector,
respectively (Fig. 3B). In
contrast, cellular proliferation did not differ significantly among
the cell lines (data not shown).
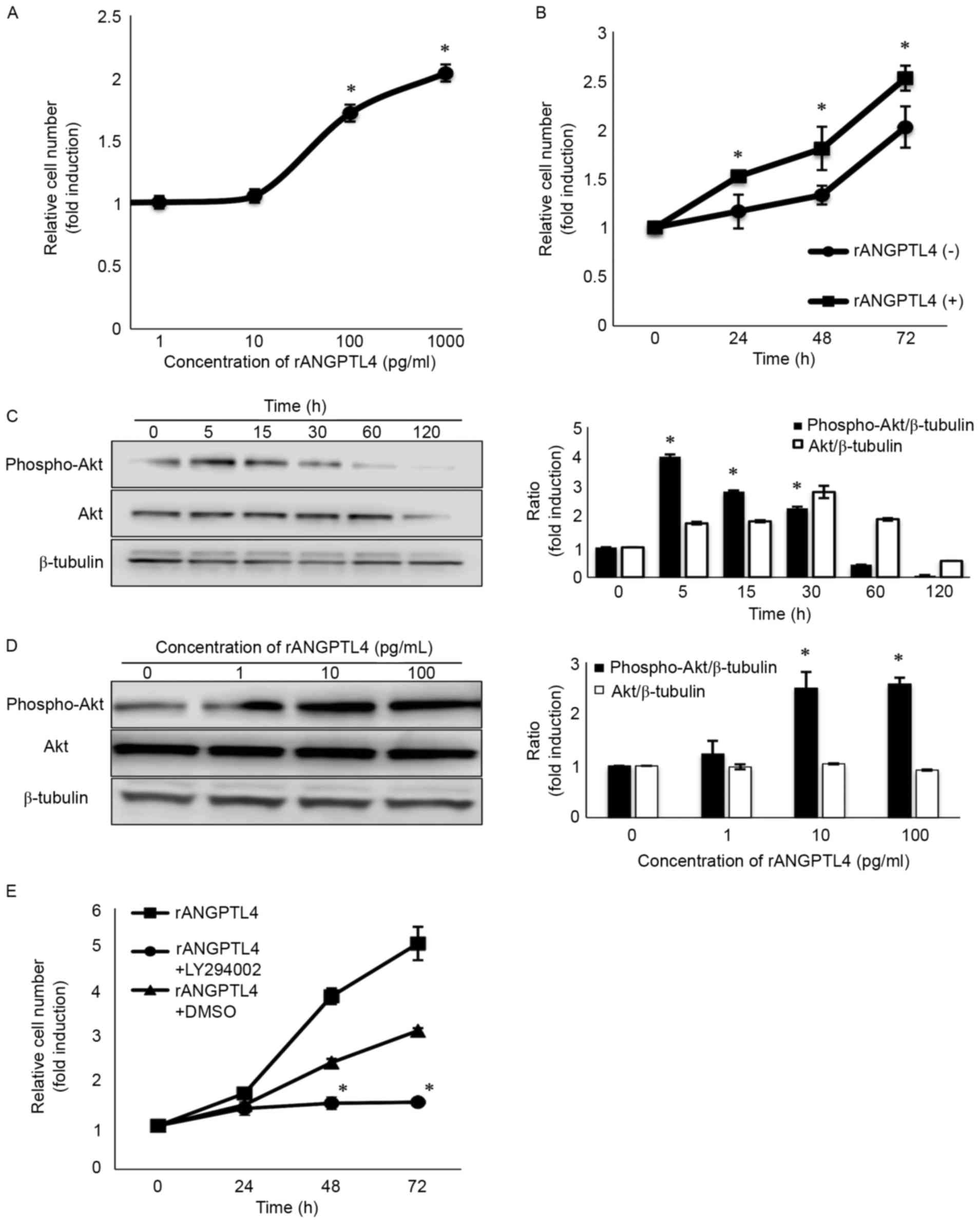
ANGPTL4 activates the PI3K/Akt
pathway, and LY294002 inhibits the cellular proliferation of LNCaP
cells
To assess the potential role of ANGPTL4, we added
different concentrations of rANGPTL4 to LNCaP cells (Fig. 4A). Treatment with 100 pg/ml of
rANGPTL4 for 72 h increased cellular proliferation 1.72-fold
(Fig. 4B). The immunoblot assay
also revealed that Akt phosphorylation at Ser 473, a site required
for Akt activation, was upregulated both time- and dose-dependently
(Fig. 4C and D). However, cellular
proliferation was suppressed in the cells subsequently treated with
LY294002 (a PI3K inhibitor) 6 h later (Fig. 4E). Akt phosphorylation peaked at
5–15 min (Fig. 4C), but the effect
on cellular proliferation was observed 48–72 h later (Fig. 4B). It is considered that with the
activation of Akt, various signaling molecules are activated
resulting in cell survival.
ANGPTL4 overexpression and drug
resistance
Cellular proliferation was successfully repressed in
LNCaP/neo cells following 2 nM docetaxel treatment; however,
LNCaP/ANGPTL4 cells maintained their proliferative capabilities
even after treatment (Fig. 3C). The
immunoblot assay revealed that anti-apoptotic B-cell lymphoma 2
(Bcl-2) expression was upregulated and that of Bcl-2-associated
death promoter (Bad) was downregulated in LNCaP/ANGPTL4 cells
compared with that in the controls (Fig. 3D). Thus, these results indicate that
ANGPTL4 expression is linked to docetaxel resistance by inhibiting
apoptosis.
Patient characteristics
Table I presents the
clinicopathological characteristics of the patients included in
this study. The median age of the patients was 67.5 (range: 49–76)
years. The median PSA at diagnosis was 9.0 (range: 3.7–23.9) ng/ml.
The pathological T (pT) stage was ≤pT2 in 61 cases (87.1%) and ≥pT3
in nine cases (12.9%). The Gleason score (GS) was ≤6 in 20 cases
(28.6%) and ≥7 in 47 cases (67.1%). The remaining three cases were
unremarkable. During a median follow-up of 53.5 (range: 13–82)
months, 19 patients (27.1%) had PSA recurrence.
Immunohistochemistry for ANGPTL4
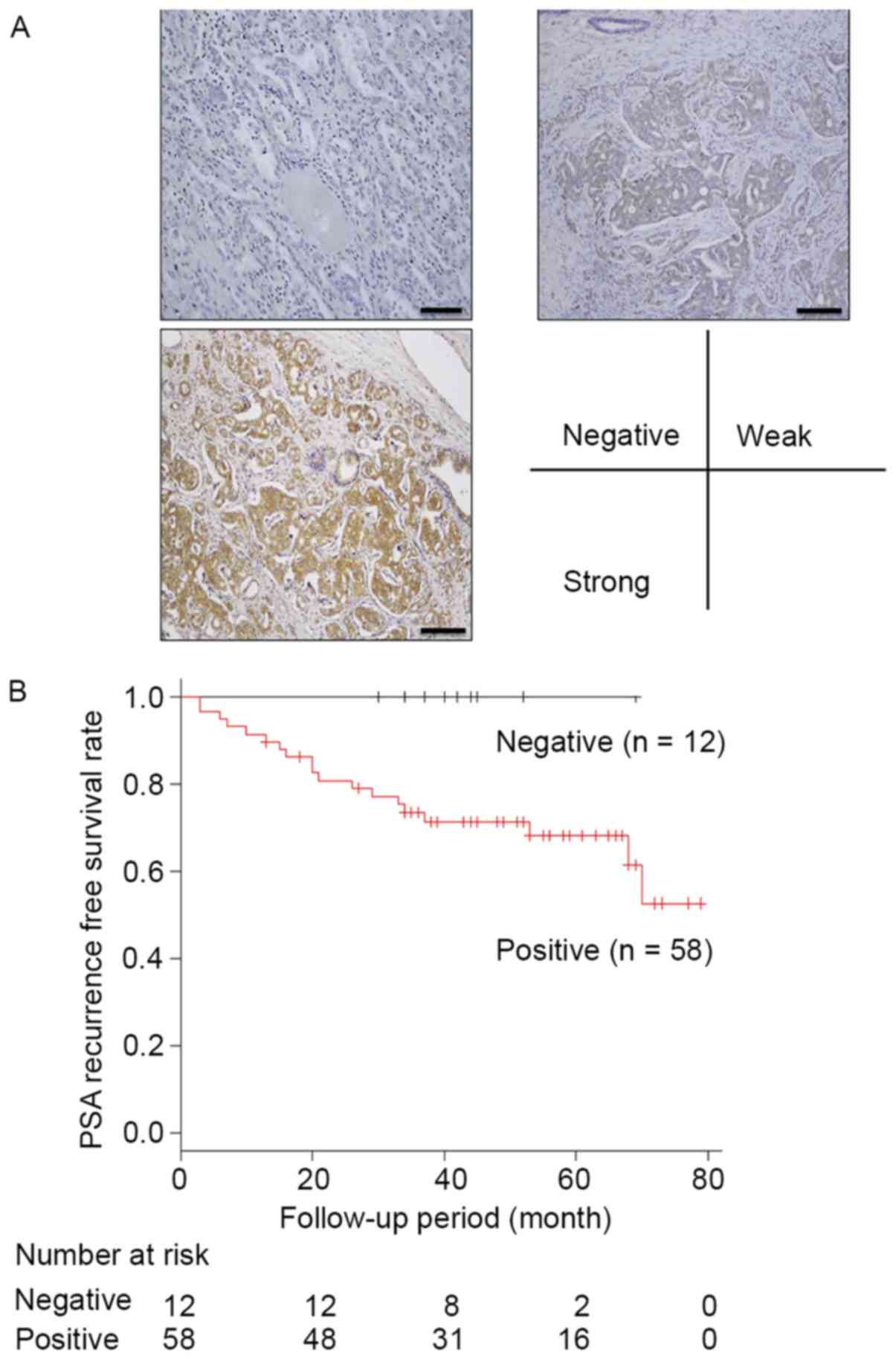
We evaluated ANGPTL4 expression by
immunohistochemistry in 70 prostate tissue samples. Normal prostate
gland tissue did not express ANGPTL4. In contrast, we observed
negative staining in 12 of the 70 prostate cancer samples (17.1%)
and positive staining in 58 samples (82.9%). ANGPTL4 expression was
particularly strong in 11 samples. Fig.
5A shows the representative images.
Association between ANGPTL4 expression
and clinicopathological parameters
Table II lists the
correlation between ANGPTL4 expression levels and different
clinicopathological factors that were analyzed. ANGPTL4 expression
levels were significantly correlated with PSA recurrence (P=0.02).
However, PSA at diagnosis, GS, pT stage, and the resection margin
were not correlated with ANGPTL4 expression. An extraprostatic
extension tended to be higher in cells with positive ANGPTL4
expression than in cells with negative ANGPTL4 expression; however,
this difference was not significant (P=0.091).
 | Table II.Correlation between ANGPTL4
expression and clinicopathological factors. |
Table II.
Correlation between ANGPTL4
expression and clinicopathological factors.
|
| ANGPTL4 |
|
|
|---|
|
|
|
|
|
|---|
| Factors | Negative
(n=12) | Positive
(n=58) | R | P-value |
|---|
| Age, years |
|
| −0.049 | 0.687 |
|
<65 | 5 | 21 |
|
|
|
≥65 | 7 | 37 |
|
|
| PSA at diagnosis,
ng/ml |
|
| 0.049 | 0.687 |
|
<10 | 8 | 35 |
|
|
|
≥10 | 4 | 23 |
|
|
| Gleason score |
|
| −0.182 | 0.141 |
| ≤6 | 1 | 17 |
|
|
| ≥7 | 9 | 40 |
|
|
| Unremarkable | 2 | 1 |
|
|
| pT stage |
|
| 0.166 | 0.172 |
| ≤2 | 12 | 49 |
|
|
| ≥3 | 0 | 9 |
|
|
| Extraprostatic
extension |
|
| 0.204 | 0.091 |
| 0 | 11 | 39 |
|
|
| 1 | 1 | 19 |
|
|
| Resection
margin |
|
| −0.051 | 0.683 |
| 0 | 9 | 47 |
|
|
| 1 | 3 | 11 |
|
|
| Biochemical
recurrence |
|
| 0.28 | 0.02 |
| – | 12 | 39 |
|
|
| + | 0 | 19 |
|
|
ANGPTL4 expression levels and patient
outcomes
We performed univariate and multivariate analyses to
determine the factors that could be used as indicators of PSA
recurrence after surgery (Table
III). A univariate analysis revealed that both pT stage
(P=0.007) and positive ANGPTL4 expression of the tumor (P=0.04)
were indicators of PSA recurrence. The multivariate analysis using
the Cox's proportional hazards model revealed that positive ANGPTL4
expression was an independent prognostic indicator of PSA
recurrence (P=0.03, hazard ratio = 2.02). The 3-year PSA
recurrence-free survival rate for patients with positive ANGPTL4
expression was significantly lower than that for other patients
(P=0.01) (Fig. 5B).
 | Table III.Univariate and multivariate analysis
of PSA recurrence-free rates. |
Table III.
Univariate and multivariate analysis
of PSA recurrence-free rates.
|
| Recurrence-free
rates |
|---|
|
|
|
|---|
|
| Univariate | Multivariate |
|---|
|
|
|
|
|---|
| Factors | P-value | HR (95% CI) | P-value |
|---|
| Age | 0.244 |
|
|
|
<65 |
|
|
|
|
≥65 |
|
|
|
| PSA at
diagnosis | 0.497 |
|
|
|
<10 |
|
|
|
|
≥10 |
|
|
|
| Gleason score | 0.119 |
|
|
| ≤6 |
|
|
|
| ≥7 |
|
|
|
| pT | 0.007 |
| 0.1 |
| ≤2 |
|
|
|
| ≥3 |
| 3.95
(0.76–20.54) |
|
| Extra prostatic
extension | 0.181 |
|
|
| 0 |
|
|
|
| 1 |
|
|
|
| Resection
margin | 0.393 |
|
|
| 0 |
|
|
|
| 1 |
|
|
|
| ANGPTL4 | 0.04 |
| 0.032 |
| – |
|
|
|
| + |
| 2.02
(1.06–3.86) |
|
Discussion
Herein, we examined ANGPTL4 expression in prostate
cancer cell lines under hypoxic conditions. In addition, we
assessed the behavior of ANGPTL4 in prostate cancer cell lines. We
also assessed whether ANGPTL4 rendered prostate cancer cells
resistant to chemotherapy. Finally, we investigated how ANGPTL4
expression correlated with clinicopathological factors.
Our results showed that chronic hypoxia induces
ANGPTL4 expression and promotes cancer progression via the
activated PI3K/Akt pathway. Previously, we reported that LNCaP
cells conditioned under chronic hypoxia grew in an
androgen-independent manner, presented accelerated G1 to S phases
of the cell cycle, and accelerated cell migration and invasion. In
addition, PI3K/Akt, JAK/STAT, and hypoxia-inducible factor 1
(HIF-1) pathways were activated in LNCaP cells conditioned under
chronic hypoxia (6). In similar
studies, Kim et al reported a synergistic effect of
prostaglandin E2 (PGE2) and hypoxia on
enhancing ANGPTL4 expression, and that increased ANGPTL4 expression
promoted colorectal cancer growth. Moreover, these authors showed
that hypoxia induced a PGE2 receptor, the E prostanoid
receptor (EP) 1. Activation of EP1 enhanced ANGPTL4 expression,
whereas blockage of EP1 by an antagonist inhibited PGE2
induction of ANGPTL4 under hypoxic conditions (17). Thus, our combined results suggest
that ANGPTL4 plays an important role in cancer progression under
hypoxia.
In addition, our results showed that ANGPTL4
overexpression plays a role in drug resistance via the
anti-apoptotic effect. A correlation between hypoxia and
chemoresistance has been previously reported in a number of tumor
cell types (18). To this end, many
reports have suggested that HIF-1-induced genes mediated
chemoresistance either directly or indirectly (19). Furthermore, some studies have
reported that hypoxia induced the expression of ANGPTL4, and that
the upregulation of ANGPTL4 is induced by the transcription factor
HIF-1 (20,21). Our results complement previous
research, and they show that ANGPTL4 directly mediates
chemoresistance by inhibiting apoptosis in prostate cancer.
Negative ANGPTL4 expression was significantly
related to longer PSA recurrence-free survival rates. To the best
of our knowledge, these results are the first that suggest ANGPTL4
as a novel marker for PSA recurrence after radical prostatectomy.
Generally, curative surgery or radiation therapy is used to treat
localized prostate cancer. However, 25–35% of the patients develop
evidence of biochemical recurrence after radical prostatectomy for
clinically localized prostate cancer (22,23).
Given the increased incidence of prostate cancer and the continued
need for improved diagnostic markers to detect prostate cancer
recurrence, research has focused on looking for specific blood and
tumor markers. Our data indicate that ANGPTL4 can be used as a new
prognostic marker. In the same analysis, other standard factors
such as pT stage, PSA at diagnosis, and GS were not significantly
associated with PSA recurrence. However, this lack of association
might be related to the small sample size, and the fact that the
patients who immediately received neo-adjuvant or adjuvant therapy
after surgery, based on pathological findings, were excluded from
the study potentially limited the scope of our results.
The role of ANGPTL4 in cancer remains controversial.
For example, Ng et al reported that ANGPTL4 could represent
a potential therapeutic agent to suppress hepatocellular carcinoma
growth, angiogenesis, and metastasis (24). On the other hand, a series of
studies have reported the role of ANGPTL4 in promoting cancer
progression (12,25,26).
Previously, ANGPTL4 has been shown to promote tumor cells for lung
metastasis in breast cancer patients, and to trigger the disruption
of vascular endothelial cell-cell junctions (27). The origin of the discrepancy among
these results is also unclear. The inferred cause is that ANGPTL4
is degraded in the human body. ANGPTL4 contains an N-terminal
coiled domain (nANGPTL4) and a C-terminal fibrinogen-like domain
(cANGPTL4); however, little is known about the relative expression
of different ANGPTL4 fragments in each tissue. To this end, a
recent study shows that cANGPTL4, but not nANGPTL4, is highly
expressed in major epithelial tumors such as adenocarcinoma. The
study examined ANGPTL4 expression using immunofluorescence with a
monoclonal antibody against cANGPTL4 (mAb11F6C4) (13). Thus, post-translational
modifications of ANGPTL4 such as N-glycosylation might be behind
different roles played by this protein in various cancers (28,29).
The number of uncertainties regarding the role and biological
mechanism of ANGPTL4 in human prostate cancer highlights the need
of further studies in this front.
In conclusion, we show that hypoxia-induced ANGPTL4
promotes prostate cancer progression via the activated PI3K/Akt
pathway. In addition, ANGPTL4 expression in surgically resected
prostate cancer specimens can represent a novel prognostic marker.
Finally, ANGPTL4 could also be suggested as a potential novel
therapeutic target in prostate cancer.
Acknowledgements
We thank Ms. N. Hamamatsu and Ms. S. Kato for
technical assistance in this work. The authors would like to thank
Enago (www.enago.jp) for the English language
review.
References
|
1
|
Stewart BW and Wild CP: World Cancer
Report 2014. International Agency for Research on Cancer World
Health Organization; 2014
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Scardino PT, Linehan W Marston, Zelefsky
MJ and Vogelzang NJ: Comprehensive Textbook of Genitourinary
Oncology (4th). Lippincott Williams and Wilkins. Philadelphia, PA:
2011.
|
|
4
|
Höckel M and Vaupel P: Tumor hypoxia:
Definitions and current clinical, biologic, and molecular aspects.
J Natl Cancer Inst. 93:266–276. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Harris AL: Hypoxia - a key regulatory
factor in tumour growth. Nat Rev Cancer. 2:38–47. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yamasaki M, Nomura T, Sato F and Mimata H:
Chronic hypoxia induces androgen-independent and invasive behavior
in LNCaP human prostate cancer cells. Urol Oncol. 31:1124–1131.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tan MJ, Teo Z, Sng MK, Zhu P and Tan NS:
Emerging roles of angiopoietin-like 4 in human cancer. Mol Cancer
Res. 10:677–688. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhu P, Goh YY, Chin HF, Kersten S and Tan
NS: Angio-poietin-like 4: A decade of research. Biosci Rep.
32:211–219. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kersten S, Lichtenstein L, Steenbergen E,
Mudde K, Hendriks HF, Hesselink MK, Schrauwen P and Müller M:
Caloric restriction and exercise increase plasma ANGPTL4 levels in
humans via elevated free fatty acids. Arterioscler Thromb Vasc
Biol. 29:969–974. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mandard S, Zandbergen F, van Straten E,
Wahli W, Kuipers F, Müller M and Kersten S: The fasting-induced
adipose factor/angiopoietin-like protein 4 is physically associated
with lipoproteins and governs plasma lipid levels and adiposity. J
Biol Chem. 281:934–944. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lu B, Moser A, Shigenaga JK, Grunfeld C
and Feingold KR: The acute phase response stimulates the expression
of angiopoietin like protein 4. Biochem Biophys Res Commun.
391:1737–1741. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kubo H, Kitajima Y, Kai K, Nakamura J,
Miyake S, Yanagihara K, Morito K, Tanaka T, Shida M and Noshiro H:
Regulation and clinical significance of the hypoxia-induced
expression of ANGPTL4 in gastric cancer. Oncol Lett. 11:1026–1034.
2016.PubMed/NCBI
|
|
13
|
Zhu P, Tan MJ, Huang RL, Tan CK, Chong HC,
Pal M, Lam CR, Boukamp P, Pan JY, Tan SH, et al: Angiopoietin-like
4 protein elevates the prosurvival intracellular O2(−):H2O2 ratio
and confers anoikis resistance to tumors. Cancer Cell. 19:401–415.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ifon ET, Pang AL, Johnson W, Cashman K,
Zimmerman S, Muralidhar S, Chan WY, Casey J and Rosenthal LJ: U94
alters FN1 and ANGPTL4 gene expression and inhibits tumorigenesis
of prostate cancer cell line PC3. Cancer Cell Int. 5:19–31. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sato R, Yamasaki M, Hirai K, Matsubara T,
Nomura T, Sato F and Mimata H: Angiopoietin-like protein 2 induces
androgen-independent and malignant behavior in human prostate
cancer cells. Oncol Rep. 33:58–66. 2015.PubMed/NCBI
|
|
16
|
Kanda Y: Investigation of the freely
available easy-to-use software ‘EZR’ for medical statistics. Bone
Marrow Transplant. 48:452–458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim SH, Park YY, Kim SW, Lee JS, Wang D
and DuBois RN: ANGPTL4 induction by prostaglandin E2 under hypoxic
conditions promotes colorectal cancer progression. Cancer Res.
71:7010–7020. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rohwer N and Cramer T: Hypoxia-mediated
drug resistance: Novel insights on the functional interaction of
HIFs and cell death pathways. Drug Resist Updat. 14:191–201. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Karakashev SV and Reginato MJ: Progress
toward overcoming hypoxia-induced resistance to solid tumor
therapy. Cancer Manag Res. 7:253–264. 2015.PubMed/NCBI
|
|
20
|
Zhang H, Wong CC, Wei H, Gilkes DM,
Korangath P, Chaturvedi P, Schito L, Chen J, Krishnamachary B,
Winnard PT Jr, et al: HIF-1-dependent expression of
angiopoietin-like 4 and L1CAM mediates vascular metastasis of
hypoxic breast cancer cells to the lungs. Oncogene. 31:1757–1770.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li H, Ge C, Zhao F, Yan M, Hu C, Jia D,
Tian H, Zhu M, Chen T, Jiang G, et al: Hypoxia-inducible factor 1
alpha-activated angiopoietin-like protein 4 contributes to tumor
metastasis via vascular cell adhesion molecule-1/integrin β1
signaling in human hepatocellular carcinoma. Hepatology.
54:910–919. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hull GW, Rabbani F, Abbas F, Wheeler TM,
Kattan MW and Scardino PT: Cancer control with radical
prostatectomy alone in 1,000 consecutive patients. J Urol.
167:528–534. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Roehl KA, Han M, Ramos CG, Antenor JA and
Catalona WJ: Cancer progression and survival rates following
anatomical radical retropubic prostatectomy in 3,478 consecutive
patients: Long-term results. J Urol. 172:910–914. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ng KT, Xu A, Cheng Q, Guo DY, Lim ZX, Sun
CK, Fung JH, Poon RT, Fan ST, Lo CM, et al: Clinical relevance and
therapeutic potential of angiopoietin-like protein 4 in
hepatocellular carcinoma. Mol Cancer. 13:196–212. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nakayama T, Hirakawa H, Shibata K, Nazneen
A, Abe K, Nagayasu T and Taguchi T: Expression of angiopoietin-like
4 (ANGPTL4) in human colorectal cancer: ANGPTL4 promotes venous
invasion and distant metastasis. Oncol Rep. 25:929–935. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shibata K, Nakayama T, Hirakawa H, Hidaka
S and Nagayasu T: Clinicopathological significance of
angiopoietin-like protein 4 expression in oesophageal squamous cell
carcinoma. J Clin Pathol. 63:1054–1058. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Padua D, Zhang XH, Wang Q, Nadal C, Gerald
WL, Gomis RR and Massagué J: TGFbeta primes breast tumors for lung
metastasis seeding through angiopoietin-like 4. Cell. 133:66–77.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim I, Kim HG, Kim H, Kim HH, Park SK, Uhm
CS, Lee ZH and Koh GY: Hepatic expression, synthesis and secretion
of a novel fibrinogen/angiopoietin-related protein that prevents
endothelial-cell apoptosis. Biochem J. 346:603–610. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang YH, Wang Y, Lam KS, Yau MH, Cheng KK,
Zhang J, Zhu W, Wu D and Xu A: Suppression of the Raf/MEK/ERK
signaling cascade and inhibition of angiogenesis by the carboxyl
terminus of angiopoietin-like protein 4. Arterioscler Thromb Vasc
Biol. 28:835–840. 2008. View Article : Google Scholar : PubMed/NCBI
|