Introduction
Specificity protein 1 (Sp1) is a transcription
factor that binds to GC-rich motifs including GC-boxes, CACCC-boxes
(also called GT-boxes), and basic transcription elements; Sp1 binds
with high affinity and regulates the expression of numerous
mammalian genes (1). Sp1 is an
ideal target for cancer treatment given its important role in
prostate cancer (PCa) progression, including cell proliferation,
angiogenesis, differentiation, apoptosis, migration and invasion
(2,3). Recent evidence indicates that Sp
transcription factors act in concert with Akt to regulate the
abnormal metabolism of cancer cells (4). In addition, Sp1 inhibition
significantly suppresses fatty acid synthase expression in PCa
cells (5). However, the regulatory
role of Sp1 in PCa cell metabolism remains unknown.
Autophagy promotes disease progression and
therapeutic resistance in advanced PCa (6). Sp1 interacts with hypoxia-inducible
factor (HIF-1), transactivates vascular endothelial growth factor
(VEGF), regulates the VEGF pathway and influences autophagy
(7). PKM2, a modulator of
glycolysis, also regulates the autophagic process by upregulating
LC3B or Beclin-1 in glioma cells or in cancer-associated
fibroblasts (8,9). We speculate that autophagy in PCa may
be modulated by the Sp1/PKM2 axis given that Sp1 regulates the
transcriptional activity of PKM2 (10).
In this study, we illustrated that Sp1 plays a vital
role in PCa metabolism and autophagy by positively regulating PKM2.
In addition, miR-361-5p directly targets Sp1/PKM2 signaling,
thereby modulating metabolism and autophagy in PCa.
Materials and methods
Data mining and bioinformatics
analysis
The GSE35988 dataset was downloaded from GEO
database (http://www.ncbi.nlm.nih.gov/geo) and re-analyzed with
R (11). Pearson's correlation in
MATLAB was used to analyze the relationship between SP1 and all
identified genes (P<0.05).
Cell culture
Two human PCa cell lines, namely, DU145 and PC3,
were purchased from Nanjing KeyGen Biotech, Co., Ltd. (Nanjing,
China). The cells were cultured at 37°C in a 20% O2
humidified atmosphere with 5% CO2 in RPMI-1640 medium
that was supplemented with 10% fetal bovine serum (FBS; 100–700;
Gemini Bio Products, Woodland, CA, USA), 50 U/ml penicillin and 50
mg/ml streptomycin (15140-122; Gibco Laboratories, Gaithersburg,
MD, USA). Hypoxia experiments were performed in a hypoxic chamber
(Coy Laboratory Products, Inc., Grass Lake, MI, USA) at 1%
O2 at 37°C in a 5% CO2 humidified
environment.
Oligonucleotides, plasmid and cell
transfection
Oligonucleotides were chemically synthesized by
Shanghai GenePharma, Co., Ltd., (Shanghai, China), based on the
following sequences: Sp1 siRNA, 5-GGUCAGUUGGCAGA CUCUATT-3 (sense);
siRNA negative control(NC), 5-UUC UCCGAACGUGUCACGUTT-3 (sense);
hsa-miR-361-5p mimics: 5-ACCCCUGGAGAUUCUGAUAAUU-3; negative control
(NC): 5-UUCUCCGAACGUGUCACGUTT-3. Cells that were 60–70% confluent
were transfected with Lipofectamine 2000 (11668-019; Invitrogen,
Albuquerque, NM, USA). Oligonucleotides or plasmids formed
transfection complexes with Lipofectamine 2000. The transfection
complexes were then added to DU145 and PC3 cells at different
concentrations and incubated for 6–8 h before changing the
medium.
RNA isolation and RT-qPCR
Total RNA was extracted from cells using TRIzol
(15596018; Invitrogen) in accordance with the manufacturers
instructions. RNA concentration was measured using NanoDrop 2000c
(Thermo Scientific, Wilmington, DE, USA). For miR-361-5p, the
RT-qPCR reaction was performed with a HiScript RT kit (R132-01;
Vazyme Biotech, Co., Ltd., Nanjing, China) and AceQ SYBR-Green PCR
Master Mix (Q141-02/03; Vazyme Biotech) following the manufacturers
protocols. Primers were chemically synthesized by Nanjing Springen
Biotech, Co., Ltd. (Nanjing, China). The miR-361-5p primers for
amplification were as follows: stem-loop reverse transcription
primer: 5-CTCAACTGGT GTCGTGGAGTCGGCAATTCAGTTGAGGTACCCCT-3; specific
forward primer, 5-ACACTCCAGCTGGGTTATC AGAATCTCC-3 and specific
reverse primer: 5-TGGTGTCG TGGAGTCG-3. The U6 snRNA primers were as
follows: forward: 5-CTCGCTTCGGCAGCACA-3 and reverse, 5-AA
CGCTTCACGAATTTGCGT-3. PCR was conducted with a 20 µl reaction
volume that contained 2 µl of RT products, 1 µl of 10 µM miR-361-5p
or U6 primer set, 10 µl of 2X SYBR-Green PCR Master Mix, and 7 µl
of ddH2O. Cycling conditions were as follows: 95°C for 3
min, 95°C for 15 sec, and 60°C for 60 sec. The last two steps were
conducted with 40 cycles. RT-qPCR was performed using 7500
Real-Time PCR System (Applied Biosystems, Inc., Carlsbad, CA, USA).
The gene expression threshold cycle (CT) values of miRNAs were
calculated by normalizing to the internal control U6. Relative
quantification values were calculated via the 2−ΔΔCt
method (12).
Cell proliferation assay
DU145 and PC3 cells were seeded in a 6-well plate
and cultured overnight. The cells were transfected with
oligonucleotides and cultured overnight. Subsequently, the cells
were trypsinized and seeded at 2,000 cells/well (200 µl/well) in a
96-well plate. At 24-h post-transfection, cell proliferation was
measured with a CCK-8 assay kit (KGA317; Nanjing KeyGen Biotech) in
accordance with the manufacturers instructions. Absorbance was
detected at a wavelength of 450 nm. Five wells were measured for
cell viability in each treatment group. All independent treatments
were performed in triplicate.
Cell cycle assay
Cell cycle distributions were analyzed using FACS
(Becton-Dickinson, Franklin Lakes, NJ, USA). Cell cycle assay kits
were procured from Nanjing KeyGen Biotech (KGA514). At 48 h
post-transfection, cells were harvested and fixed with 75% ethanol
at −20°C overnight. Subsequently, the cells were incubated in 50
µg/ml propidium iodide and 1 mg/ml RNase for 30 min at room
temperature and analyzed using flow cytometry. The experiments were
performed in triplicate.
Metabolism assay
DU145 and PC3 cells were seeded in a 24-well plate
that contained 200 µl of media/well. To determine glucose and
lactate levels in cells, the supernatants of the cell culture media
were collected and assayed for glucose and lactate levels by using
a glucose assay kit (K686; BioVision, Inc., San Francisco, CA, USA)
and lactate assay kit (K627; BioVision), respectively, in
accordance with the manufacturers instructions. OD values of the
supernatants were analyzed at 72 h after different treatments. The
remaining glucose and lactate production were calculated based on
the standard curve and normalized to the cell number.
Transmission electron microscopy
(TEM)
Cells were harvested 48 h post-transfection with Sp1
siRNA and NC siRNA. The samples were then pelleted and fixed in
2.5% glutaraldehyde (G5882; Sigma-Aldrich, St. Louis, MO, USA) in
phosphate buffer (pH 7.4). Next, the samples were rinsed with
phosphate buffer (pH 7.4) and postfixed with 2% OsO4
(75633; Sigma-Aldrich) for 1 h. The samples were then rinsed with
water, dehydrated in a graded ethanol series [70% ethanol (20 min),
96% ethanol (20 min), and then 100% ethanol (2×20 min)] followed by
propylene oxide (2×10 min) (SAJ-24-6210; Sigma-Aldrich), and
maintained overnight in Epon 812 (45347-1L-F; Sigma-Aldrich).
Subsequently, the samples were embedded in Epon 812 and then
fumigated in a microwave oven at 60°C. Ultrathin sections were
obtained using Reichert Ultracut E microtome and then stained with
uranyl acetate and lead citrate. Samples were imaged using an
H-7650 transmission electron microscope (Hitachi, Ltd., Tokyo,
Japan).
Western blot analysis
Radioimmunoprecipitation assay buffer (KGP701;
Keygentec, Inc., Nanjing, China) was used to lyse cells for total
proteins. Protein concentration was determined via the BCA method
(KGP902; Keygentec). Protein samples (30 µg) were electrophoresed
on 10% sodium dodecyl sulphate-polyacrylamide electrophoresis gels
(KGP113K; Keygentec) and transferred onto polyvinylidene fluoride
membranes (IPVH00010; Millipore, Billerica, MA, USA). The membranes
were blocked with 5% skim milk at room temperature for 1 h and
incubated overnight at 4°C with diluted primary antibodies.
Subsequently, the membranes were washed thrice with TBST buffer (pH
7.6, 20 mM Tris-HCl, 137 mM NaCl, 0.01% Tween-20) and then
incubated with HRP-conjugated secondary antibodies at 37°C for 1 h.
After being washed with TBST buffer, the membranes were visualized
using enhanced chemiluminescence ECL (WBKLS0050; Millipore).
Protein levels were normalized to GAPDH levels. The related
antibodies were HRP-conjugated monoclonal mouse anti-GAPDH (KC-5G5,
1:1,000; Shanghai Kangchen), rabbit polyclonal anti-LC3B
(18725-1-AP, 1:1,000; Proteintech Group, Inc., Chicago, IL, USA),
rabbit monoclonal anti-Beclin-1 (3495, 1:1,000; Cell Signaling
Technology, Danvers, MA, USA), rabbit monoclonal anti-PKM2 (4053,
1:1,000; Cell Signaling Technology), rabbit monoclonal anti-SP1
(9389S, 1:1,000; Cell Signaling Technology), HRP-labeled goat
anti-rabbit secondary antibody (ZB-2301, 1:3000; Beijing Zhongshan
Golden Bridge Biotechnology, Co., Ltd., Beijing, China).
Plasmid construction and luciferase
reporter assay
The 3-UTR segment of Sp1 mRNA with miR-361-5p
binding site was amplified via PCR with human genomic DNA. To
generate the Sp1 3-UTR reporter, PCR products were cloned into the
XhoI and NotI restriction sites that were downstream
of the open reading frame of luciferase in the psiCHECK-2™ Vector
(c8021; Promega, Madison, WI, USA). Deleting the binding site for
miR-361-5p generated the mutant reporter. For reporter assays, the
cells were transfected with Sp1 3′UTR, mutant reporter plasmid and
miR-361-5p mimics, or NC. Luciferase activity was measured by
Dual-luciferase assays (E1910; Promega) 48 h after
co-transfection.
Statistical analysis
Each value in this study was obtained from at least
three independent experiments and presented as mean ± SD. The
significance of differences was calculated using t-tests for
two-group comparisons with the SPSS 22.0 (IBM Corp., Armonk, NY,
USA). A probability of P<0.05 was considered statistically
significant.
Results
Sp1 affects castration-resistant
prostate cancer (CRPC) in vitro
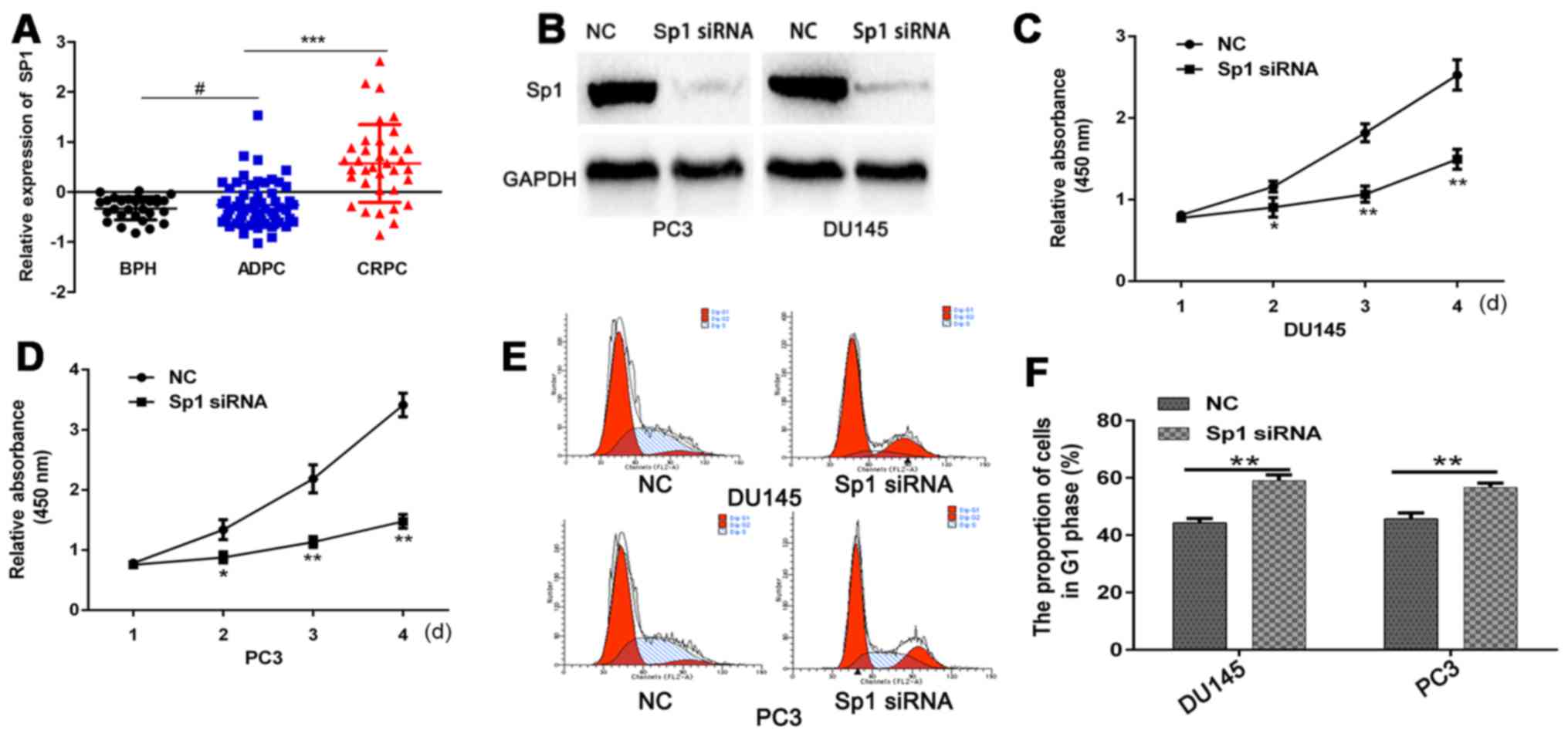
First, we used the GEO database to analyze the SP1
expression in benign prostatic hyperplasia tissues and PCa tissues
at different stages. The results indicated that SP1 expression is
significantly higher in CRPC than in androgen-dependent prostate
cancer (ADPC) (Fig. 1A). To verify
the function of Sp1 in CRPC, we transfected CRPC cells with Sp1
siRNA or NC. SP1 protein was detected by western blot analysis, and
results showed that Sp1 siRNA efficiently silenced Sp1 gene
expression (Fig. 1B). CCK-8 assays
demonstrated that Sp1 depletion significantly decreased
proliferative index of PC3 and DU145 cells compared with NC
(Fig. 1C and D). Flow cytometry
assay revealed that CRPC cells that were transfected with Sp1 siRNA
demonstrated a higher rate of G1 cell cycle arrest (Fig. 1E and F). Moreover, Sp1 siRNA
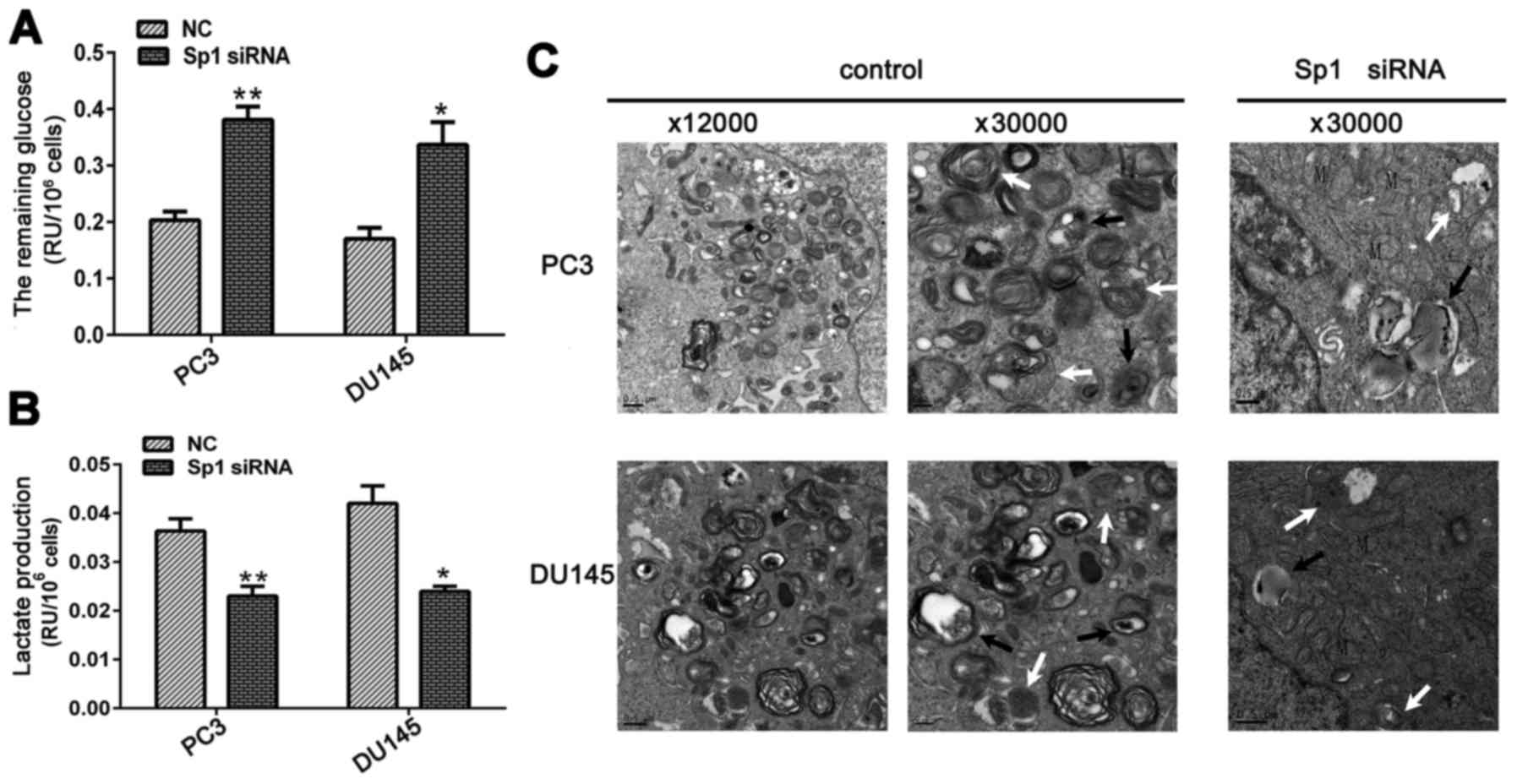
significantly decreased glucose consumption and lactate production
in CRPC cells compared with those in the NC group (Fig. 2A and B).
Glycolysis positively regulates autophagy via
various mechanisms (13). The
transcription factor HIF-1α is an important regulator of
glycolysis, and its upregulation is the mechanism through which
autophagy is induced under hypoxia or nutrient starvation (14,15).
Given that previous studies have indicated that decreased
glycolysis decreases HIF-1 and inhibits autophagy (16), we investigated whether the
inhibition of metabolism by Sp1 depletion affects hypoxia-induced
autophagy in CRPC.
PC3 and DU145 cells were cultured at 37°C in 1%
O2 and 5% CO2 humidified environment and were
simultaneously transfected with Sp1 siRNA or NC. The cells were
harvested for TEM at 48 h post-transfection. Autophagosomes,
spherical structures with mono- or bilayer membranes, engulf
degraded or abandoned organelles and deliver cytoplasmic components
to lysosomes. As expected, hypoxic conditions significantly induced
autophagy in PCa cells, as evidenced by large areas of
autophagosomes or autolysosomes that were revealed by TEM. By
contrast, Sp1 downregulation markedly inhibited hypoxia-induced
autophagy, as indicated by the significantly decreased number of
autophagosomes or autolysosomes in the Sp1 siRNA group in both CRPC
cell lines (Fig. 2C).
Sp1 modulates metabolism and autophagy
by directly targeting PKM2 in CRPC
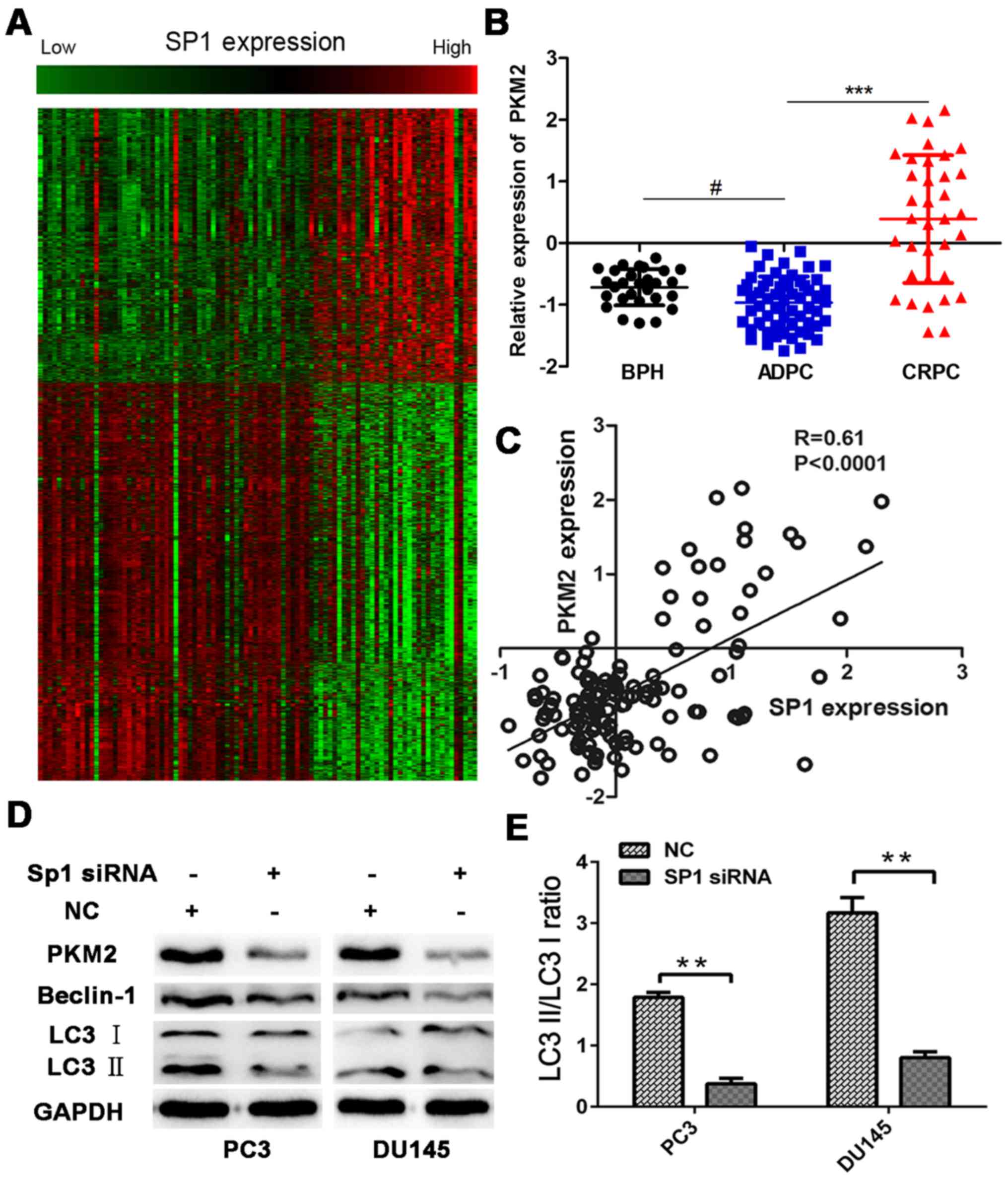
To further evaluate the potential role of aberrantly
expressed SP1, Pearson correlation was used to analyze the
relationship between Sp1 and all identified genes (Fig. 3A). The SP1-specific gene signature
included 2,357 genes that are positively correlated with SP1
expression (R>0.4, P<0.05) and 3,418 genes that are
negatively correlated with SP1 expression (R≤0.4, P<0.05) (data
available upon request). Among these genes, a set of
glycometabolism-related genes are positively correlated with SP1
expression. PKM2, a metabolic enzyme that executes the last step of
aerobic glycolysis, is included in this set of genes, and its
expression is significantly higher in CRPC than in
androgen-dependent prostate cancer (Fig. 3B and C). Previous studies that
identified Sp1 binding motifs in PKM2 promoter sequences have also
asserted that Sp1 activates PKM2 promoter activity (10,17);
thus, western blot analysis was conducted in this study to confirm
if Sp1 inhibition could decrease PKM2 expression. After 48 h of
hypoxia experiments with Sp1 siRNA transfection, the cells were
harvested for western blot analysis. Results indicated that
transfection with Sp1 siRNA downregulated PKM2.
Microtubule-associated protein 1 LC3, a specific marker of
autophagy, is converted from LC3-I (inactivated form) to LC3-II
(cleaved form) during autophagy. Western blot analysis showed that
the conversion from LC3-I to LC3-II was inhibited in the Sp1 siRNA
group, and that Beclin-1 expression also decreased (Fig. 3D). The signal ratio between LC3-II
and LC3-I indirectly showed the extent of autophagy in PCa. The
result indicated that the LC3-II/LC3-I ratio decreased in the Sp1
siRNA group and that autophagy decreased with Sp1 depletion
(Fig. 3E).
Sp1 is a direct target of
miR-361-5p
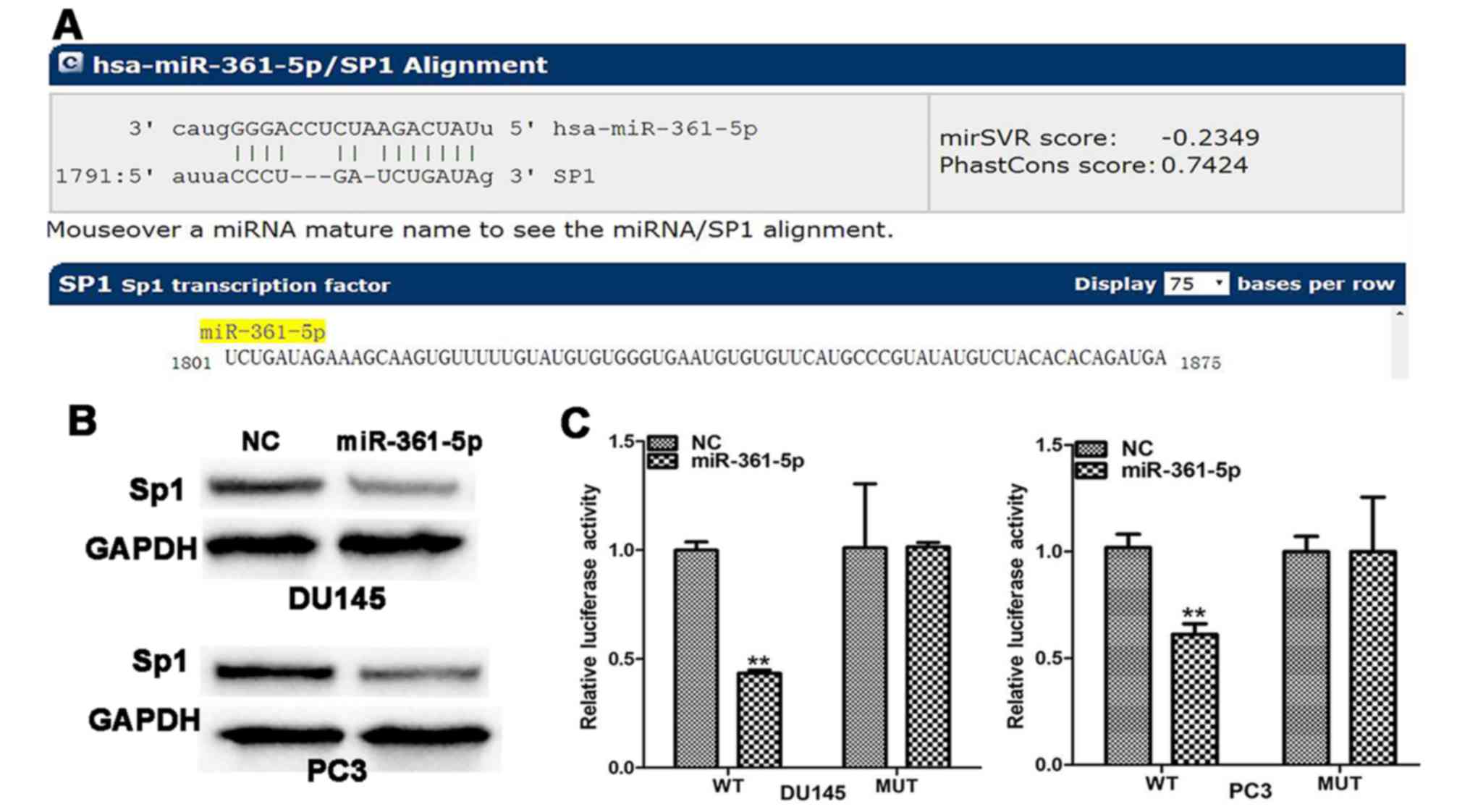
To identify the potential targets of miR-361-5p in
PCa, bioinformatics analysis was performed with the website
microRNA.org. Bioinformatics analysis revealed that
miR-361-5p could bind to target sequences in the 3′UTR of Sp1 mRNA
(Fig. 4A). Moreover, western blot
analysis revealed that Sp1 protein expression was inhibited by
treatment with miR-361-5p mimics (Fig.
4B). To confirm the direct miRNA-target interaction, Sp1 3-UTR
was cloned into a Dual-luciferase reporter cloning site in the
psiCHECK-2 Dual-luciferase vector. Luciferase activities remarkably
decreased with increasing miR-361-5p levels (Fig. 4C).
miR-361-5p inhibits cell growth,
glucose metabolism and autophagy in CRPC cells in vitro
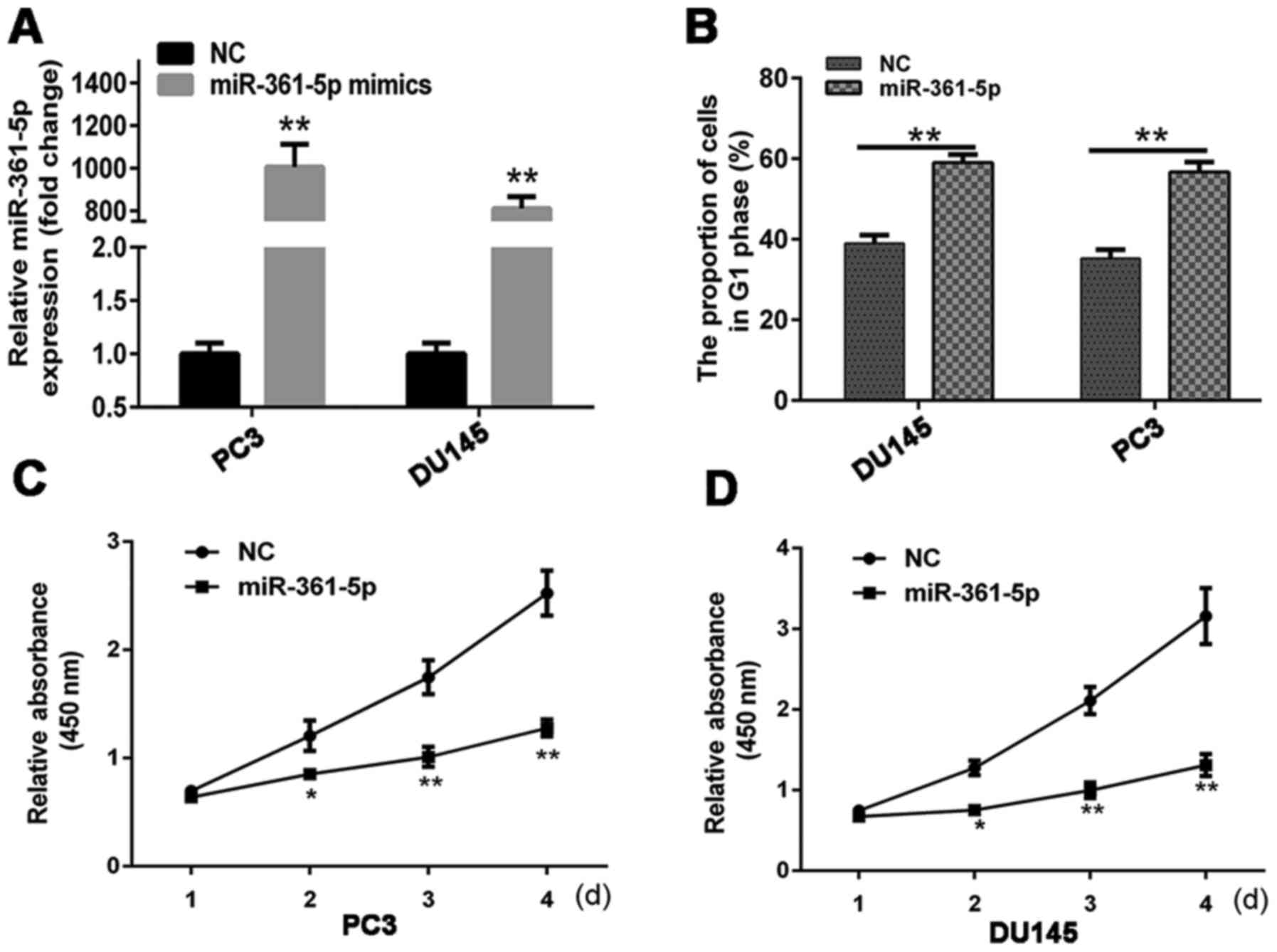
RT-qPCR was utilized to examine miR-361-5p
expression in cells at 48 h post-transfection with miR-361-5p
mimics or NC into DU145 (PC3). miR-361-5p was expressed at higher
levels in the miR-361-5p mimic group than in the NC group (Fig. 5A). Cell cycle analysis was conducted
to determine the effect of miR-361-5p on cell proliferation. At 48
h post-transfection, the percentage of miR-361-5p-transfected cells
in the G1 phase was higher than that of the NC-transfected cells
(Fig. 5B). This result indicated
that miR-361-5p induced G1 cell cycle arrest in CRPC cell lines.
CCK-8 assays revealed that miR-361-5p overexpression significantly
inhibited DU145 (PC3) proliferation (Fig. 5C and D). Moreover, miR-361-5p
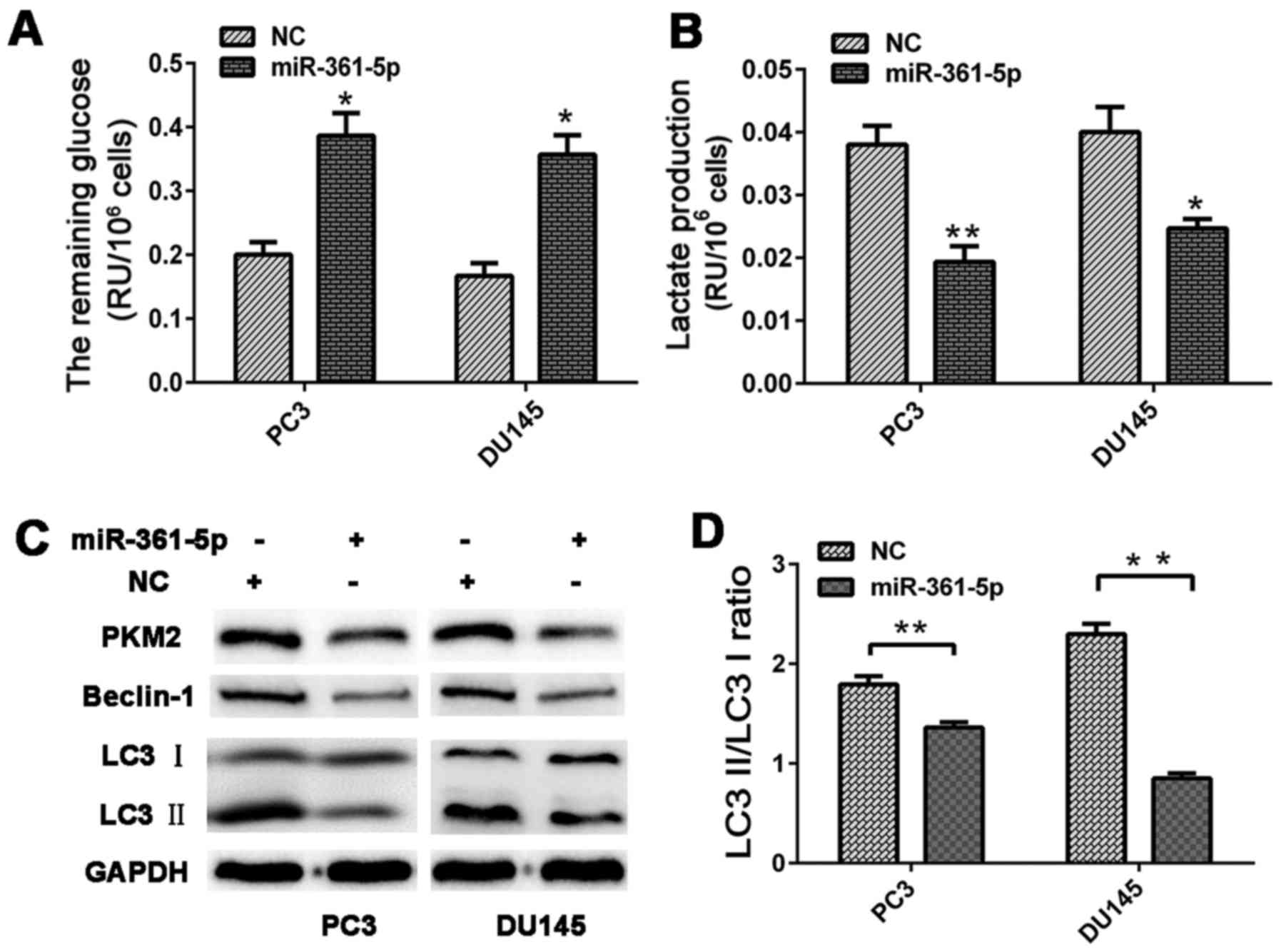
significantly reduced glucose consumption and lactate production in
the CRPC cells relative to that in the NC group (Fig. 6A and B).
miR-361-5p mimics or NC were transfected into CRPC
cells, which were cultured under hypoxic conditions. The cells were
harvested for western blot analysis at 48 h post-transfection.
miR-361-5p overexpression significantly decreased PKM2, Beclin-1
and LC3B expression levels. Moreover, the LC3-II/LC3-I ratio of the
miR-361-5p group was also lower than that in the NC group,
indicating that miR-361-5p negatively regulates autophagy in PCa
cells (Fig. 6C and D).
Discussion
Altered energy metabolism is proving to be as
widespread in cancer cells as many of the other cancer-associated
traits that have been accepted as cancer hallmarks. Previous
studies have reported that sarcosine induces invasion and
intravasation via elevated metabolism in an in vivo PCa
model (18), and that androgens
regulate PCa cell growth via an AMPK-PGC-1α-mediated metabolic
switch (19). Autophagy can meet
the increased nutritional demand of proliferative cancer cells and
is responsible for resistance to therapies, including radiotherapy,
chemotherapy and androgen deprivation, in PCa (20–22).
Recent findings also indicated that the increased expression of
autophagy protein is associated with a high Gleason score and
extraprostatic invasion (23).
Therefore, targeting metabolism and autophagy could be a novel
route for managing CRPC.
Sp1 is a transcription factor that is an essential
target for regulating the translation rates and additional
metabolic processes of cell growth (17,24,25).
Consistent with a previously reported finding that Sp1 is
upregulated in numerous cancers with poor prognosis (3), we demonstrated that Sp1 upregulation
is common in CRPC tissues and further explored the roles of Sp1 in
CRPC. After determining that Sp1 expression increased in CRPC, we
further investigated the functions and underlying mechanisms of Sp1
by inhibiting its expression.
The present study indicated that Sp1 depletion
inhibits cell growth, metabolism and autophagy in CRPC. Western
blot analysis revealed that suppressing Sp1 decreased the
expression levels of PKM2 and of the autophagy markers Beclin-1 and
LC3B-II in PCa cells that were cultured under hypoxia. A previous
study (26) reported that five
putative binding regions for Sp1 in the PKM promoter region and the
dephosphorylation of Sp1 can enhance DNA-binding activity and
increase the expression of PKM2 (16). We believe that Sp1 regulates PKM2 at
the transcriptional level, thus modulating metabolism and cellular
growth in PCa. In view of the interplay between metabolism and
autophagy, the downregulation of PKM2 induces autophagy impairment
in 2fTGH cells (27,28). HIF-1α-induced autophagy promotes
tumor progression (29).PKM2 acts
as a HIF-1α co-activator to promote Warburg effects in cancer cells
and the PKM2 downregulation impairs autophagy (30). Our results illustrated that Sp1
plays a vital role in PCa cell metabolism and autophagy by
positively regulating PKM2.
We have previously proved that miR-361-5p was
downregulated and played a tumor suppressor role in CRPC (31), and we further illustrated that
miR-361-5p was involved in the regulation of autophagy and
metabolism in CRPC. Moreover, we showed that Sp1 was highly
expressed and could modulate autophagy and metabolism in CRPC. Via
bioinformatics analysis, it revealed that miR-361-5p could bind to
target sequences in the 3′UTR of Sp1 mRNA. Then, we further
explored whether miR-361-5p could regulate autophagy and metabolism
via inverse regulation of Sp1. Western blot analysis and luciferase
reporter assay demonstrated that miR-361-5p can reversely regulate
Sp1 by directly targeting the mRNA of Sp1, as predicted by
bioinformatics analysis. miR-361-5p overexpression in PCa cell
lines presented biological functions that are similar to those of
Sp1 depletion; thus, miR-361-5p modulates cell proliferation,
metabolism and autophagy in PCa in an Sp1-dependent manner.
In summary, our findings revealed that miR-361-5p is
a tumor suppressor. By contrast, Sp1 is an oncogene as it
positively regulates PKM2 in PCa. Notably, miR-361-5p inhibits CRPC
cell proliferation, metabolism, and autophagy by directly targeting
Sp1/PKM2 signaling, which is a potential target in PCa therapy.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (81272557), the Natural Science
Foundation of Jiangsu Province (BK20151158), the Talent Peak
Project of Jiangsu Province (2014-WSW-067), the Innovation Team
Project of Xuzhou Central Hospital(XZB201610), the Fundamental
Research Funds for the Central Universities and the Scientific
Research Innovation Program for College and University Graduates of
Jiangsu Province(KYZZ16_0135), and the Natural Science Foundation
of Anhui Province (1708085QH202).
References
|
1
|
Imataka H, Sogawa K, Yasumoto K, Kikuchi
Y, Sasano K, Kobayashi A, Hayami M and Fujii-Kuriyama Y: Two
regulatory proteins that bind to the basic transcription element
(BTE), a GC box sequence in the promoter region of the rat P-4501A1
gene. EMBO J. 11:3663–3671. 1992.PubMed/NCBI
|
|
2
|
Black AR, Black JD and Azizkhan-Clifford
J: Sp1 and krüppel-like factor family of transcription factors in
cell growth regulation and cancer. J Cell Physiol. 188:143–160.
2001. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sankpal UT, Goodison S, Abdelrahim M and
Basha R: Targeting Sp1 transcription factors in prostate cancer
therapy. Med Chem. 7:518–525. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Archer MC: Role of sp transcription
factors in the regulation of cancer cell metabolism. Genes Cancer.
2:712–719. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lu S and Archer MC: Sp1 coordinately
regulates de novo lipogenesis and proliferation in cancer cells.
Int J Cancer. 126:416–425. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Farrow JM, Yang JC and Evans CP: Autophagy
as a modulator and target in prostate cancer. Nat Rev Urol.
11:508–516. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Alam H, Maizels ET, Park Y, Ghaey S,
Feiger ZJ, Chandel NS and Hunzicker-Dunn M: Follicle-stimulating
hormone activation of hypoxia-inducible factor-1 by the
phosphatidylinositol 3-kinase/AKT/Ras homolog enriched in brain
(Rheb)/mammalian target of rapamycin (mTOR) pathway is necessary
for induction of select protein markers of follicular
differentiation. J Biol Chem. 279:19431–19440. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chiavarina B, Whitaker-Menezes D,
Martinez-Outschoorn UE, Witkiewicz AK, Birbe R, Howell A, Pestell
RG, Smith J, Daniel R, Sotgia F, et al: Pyruvate kinase expression
(PKM1 and PKM2) in cancer-associated fibroblasts drives stromal
nutrient production and tumor growth. Cancer Biol Ther.
12:1101–1113. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ahmad F, Dixit D, Joshi SD and Sen E: G9a
inhibition induced PKM2 regulates autophagic responses. Int J
Biochem Cell Biol. 78:87–95. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang W and Lu Z: Regulation and function
of pyruvate kinase M2 in cancer. Cancer Lett. 339:153–158. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Grasso CS, Wu YM, Robinson DR, Cao X,
Dhanasekaran SM, Khan AP, Quist MJ, Jing X, Lonigro RJ, Brenner JC,
et al: The mutational landscape of lethal castration-resistant
prostate cancer. Nature. 487:239–243. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Eng CH and Abraham RT: The autophagy
conundrum in cancer: Influence of tumorigenic metabolic
reprogramming. Oncogene. 30:4687–4696. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Peng X, Gong F, Chen Y, Jiang Y, Liu J, Yu
M, Zhang S, Wang M, Xiao G and Liao H: Autophagy promotes
paclitaxel resistance of cervical cancer cells: Involvement of
Warburg effect activated hypoxia-induced factor 1-α-mediated
signaling. Cell Death Dis. 5:e13672014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sun Y, Xing X, Liu Q, Wang Z, Xin Y, Zhang
P, Hu C and Liu Y: Hypoxia-induced autophagy reduces
radiosensitivity by the HIF-1α/miR-210/Bcl-2 pathway in colon
cancer cells. Int J Oncol. 46:750–756. 2015.PubMed/NCBI
|
|
16
|
Schäfer D, Hamm-Künzelmann B and Brand K:
Glucose regulates the promoter activity of aldolase A and pyruvate
kinase M2 via dephosphorylation of Sp1. FEBS Lett. 417:325–328.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Discher DJ, Bishopric NH, Wu X, Peterson
CA and Webster KA: Hypoxia regulates beta-enolase and pyruvate
kinase-M promoters by modulating Sp1/Sp3 binding to a conserved GC
element. J Biol Chem. 273:26087–26093. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Khan AP, Rajendiran TM, Ateeq B, Asangani
IA, Athanikar JN, Yocum AK, Mehra R, Siddiqui J, Palapattu G, Wei
JT, et al: The role of sarcosine metabolism in prostate cancer
progression. Neoplasia. 15:491–501. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tennakoon JB, Shi Y, Han JJ, Tsouko E,
White MA, Burns AR, Zhang A, Xia X, Ilkayeva OR, Xin L, et al:
Androgens regulate prostate cancer cell growth via an
AMPK-PGC-1α-mediated metabolic switch. Oncogene. 33:5251–5261.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cao C, Subhawong T, Albert JM, Kim KW,
Geng L, Sekhar KR, Gi YJ and Lu B: Inhibition of mammalian target
of rapamycin or apoptotic pathway induces autophagy and
radiosensitizes PTEN null prostate cancer cells. Cancer Res.
66:10040–10047. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bennett HL, Stockley J, Fleming JT, Mandal
R, OPrey J, Ryan KM, Robson CN and Leung HY: Does androgen-ablation
therapy (AAT) associated autophagy have a pro-survival effect in
LNCaP human prostate cancer cells? BJU Int. 111:672–682. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li M, Jiang X, Liu D, Na Y, Gao GF and Xi
Z: Autophagy protects LNCaP cells under androgen deprivation
conditions. Autophagy. 4:54–60. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Giatromanolaki A, Sivridis E, Mendrinos S,
Koutsopoulos AV and Koukourakis MI: Autophagy proteins in prostate
cancer: relation with anaerobic metabolism and Gleason score. Urol
Oncol. 32:39.e11–38. 2014. View Article : Google Scholar
|
|
24
|
Wong N, De Melo J and Tang D: PKM2, a
central point of regulation in cancer metabolism. Int J Cell Biol.
2013:2425132013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Netzker R, Weigert C and Brand K: Role of
the stimulatory proteins Sp1 and Sp3 in the regulation of
transcription of the rat pyruvate kinase M gene. Eur J Biochem.
245:174–181. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jiang M: Interplay between autophagy and
metabolism in Ras mutation-induced tumorigenesis. Asian J Androl.
13:610–611. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Altuntas S, Rossin F, Marsella C, DEletto
M, Diaz-Hidalgo L, Farrace MG, Campanella M, Antonioli M, Fimia GM
and Piacentini M: The transglutaminase type 2 and pyruvate kinase
isoenzyme M2 interplay in autophagy regulation. Oncotarget.
6:44941–44954. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bellot G, Garcia-Medina R, Gounon P,
Chiche J, Roux D, Pouysségur J and Mazure NM: Hypoxia-induced
autophagy is mediated through hypoxia-inducible factor induction of
BNIP3 and BNIP3L via their BH3 domains. Mol Cell Biol.
29:2570–2581. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Luo W, Hu H, Chang R, Zhong J, Knabel M,
OMeally R, Cole RN, Pandey A and Semenza GL: Pyruvate kinase M2 is
a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell.
145:732–744. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu D, Tao T, Xu B, Chen S, Liu C, Zhang
L, Lu K, Huang Y, Jiang L, Zhang X, et al: MiR-361-5p acts as a
tumor suppressor in prostate cancer by targeting signal transducer
and activator of transcription-6 (STAT6). Biochem Biophys Res
Commun. 445:151–156. 2014. View Article : Google Scholar : PubMed/NCBI
|