Introduction
Lung cancer is one of the leading causes of
cancer-related death in the world (1), and non-small cell lung cancer (NSCLC)
accounts for ~80% of lung cancer caused deaths (2). The 5-year survival rate of NSCLC
remains very low even though the mechanism studies of NSCLC
progresses rapidly (3). In patients
with NSCLC, regional lymph node, liver, contralateral lung, brain
and bone marrow are the most commonly metastatic sites and resulted
in the primary causes of death of NSCLC patients (4). Therefore, the major challenge in
treating NSCLC is to find the underlying mechanisms regulating
NSCLC metastasis.
Follistatin like-1 (FSTL1), also named TSC-36,
located on chromosome 3 (5), is a
BM-40/SPARC/osteonectin family protein which encodes a secreted
glycoprotein (6). After the
identification in mouse osteoblastic MC3T3E1 cells with TGF-β1
stimulation (7,8), FSTL1 was found to be downregulated by
v-myc and v-ras overexpression cell lines, implying a possible role
of FSTL1 in carcinogenesis (9).
FSTL1 is expressed in almost all cell types and enriched in normal
placenta, smooth muscle with various functions in different
biological processes, especially in cell proliferation and
migration (10–13). It has been reported that FSTL1
participated in the regulation of rheumatoid arthritis and other
autoimmune diseases (14). FSTL1
can regulate cell cycle entry to improve cardiac function and
cardio-renal communication (15).
In the kidney, FSTL1 is expressed in the loop of Henle and protects
the kidney from being attacked by acute nephrotoxic injury
(16). Moreover, myocytes produce
less FSTL1 to increase glomerular and tubulointerstitial damage in
the kidney (17). In the process of
lung morphogenesis, FSTL1 is essential for tracheal cartilage
formation and alveolar maturation through BMP signaling (18).
The function of FSTL1 in cancer was investigated in
many kinds of tumors such as lung cancer, colon cancer, stomach
cancer, breast cancer (7), renal
cell carcinoma (19), ovarian and
endometrial carcinoma (20). Also,
expression level of FSTL1 depends on the degree of malignancies in
cancer patients. However the function of FSTL1 in cancer is
controversial. Recent reports showed that high level of FSTL1 was
associated with poor prognosis of glioblastoma (21) and promoted cancer cell bone
metastasis by inhibiting antitumor immune responses (22). In contrast, in colon, stomach,
breast, kidney, and ovarian cancers, the expression of FSTL1 is
decreased suggesting a role of tumor suppressor as well (7,20,23,24).
In lung cancer cells, FSTL1 inhibition could promote mitotic cell
death by inactivated Erk1/2 (25).
In this study, we further explored the function of FSTL1 in NSCLC
tumor cell proliferation, migration, invasion as well as apoptosis.
Gain- and loss-of-function experiments demonstrated that FSTL1
suppressed tumor cell proliferation with altered the cell cycle.
FSTL1 inhibited cell survival, migration and invasion of NSCLC
cells. The key factors associated with cell apoptosis and invasion
including FAS/FASL, caspases and MMPs were changed with FSTL1. Our
results indicated the crucial functions of FSTL1 in NSCLC and
suggested that FSTL1 might by a new important factor in NSCLC
progression.
Materials and methods
Cell lines
The human NSCLC cell lines were purchased from ATCC.
Beas-2b, H446, H460, A549 and H1299 cells were maintained in
Dulbecco's modified Eagle's medium (DMEM). All the medium were
supplemented with 10% heat-inactivated fetal bovine serum (FBS;
Invitrogen, Carlsbad, CA, USA), penicillin (100 U/ml), and
streptomycin (100 µg/ml) in a humidified atmosphere of 5%
CO2 at 37°C. All the cells were confirmed to be free
from mycoplasma contamination.
Establishment of stable cell lines in
which FSTL1 was overexpressed or knocked down
The targeted knockdown sequence was the following:
SH1: 5′-tctgagaagttgaggcaaa-3′; SH4: 5′-gcagcaactacagtgaaatcc-3′.
The negative sequence was 5′-gtagcgcggtgtattatac-3′. Lentiviral
vectors encoding shRNA were constructed by Hanyin Co. (Shanghai,
China). The recombinant lentiviruses (SH) and the negative control
(NC) lentivirus (Hanyin Co.) were prepared and titered to
109 transfection units (TU)/ml. To obtain stable cell
lines, cells were seeded in 6-well plates and infected with virus
and polybrene the following day. Positive clones were selected with
puromycin for 14 days to establish the stable cell lines.
Additionally, the lentiviruses expressing the FSTL1 sequence (OE)
and the negative control lentivirus (NC) were constructed by Hanyin
Co. FSTL1-OE and control stable cell lines were then established.
The efficiency of FSTL1 knockdown and overexpression was confirmed
by qRT-PCR and western blot analysis.
Western blot analysis
Protein lysates (50 µg per lane) were separated by
sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis and
transferred onto nitrocellulose membranes. After blocking with 5%
fat-free milk, the membranes were incubated with primary antibodies
(1:500 dilution) at 4°C overnight, followed by horseradish
peroxidase-conjugated secondary antibodies (1:3,000 dilution).
Anti-human RBM8A (Santa Cruz, sc-32312), anti-human actin
(Proteintech, HRP-60008), anti-human snail (Cell Signaling
Technology, 3879), anti-human p-stat3 (Cell Signaling Technology,
9131), anti-human stat3 (Cell Signaling Technology, 12640),
anti-human fibronectin (Sigma-Aldrich, F3648), anti-human vimentin
(Cell Signaling Technology, 5741), anti-human Notch (Abways
Technology, CY52444) and donkey anti-rabbit IgG (Cell Signaling
Technology, 7074) were used in this study. Immune-reactive proteins
were visualized by enhanced chemiluminescence (ECL).
Total RNA isolation and quantitative
real-time PCR (qRT-PCR) analysis
Total RNA was isolated from NSCLC cancer cells using
TRIzol reagent according to the manufacturer's instructions
(Invitrogen). cDNA was reverse transcripted from 1 mg total RNA.
qRT-PCR was performed with the SYBR Premix Ex Taq (Takara, Dalian,
China). PCR primers were as follows: F:
5′-TCGCATCATCCAGTGGCTGGAA-3′, R: 5′-TCACTGGAGTCCAGGCGAGAAT-3′.
The cycling conditions were as initial denaturation
at 95°C for 5 min, and then 36 cycles of denaturation at 95°C for
10 sec and annealing at 60°C for 30 sec. The relative mRNA
expression was calculated using the comparative Ct (∆∆Ct)
method.
Cell Counting Kit-8 (CCK8) assay
Cells were seeded into 96-well culture plates
(5×103 cells/well). At 24, 48, 72 and 96 h, 10 µl CCK-8
reagent (Beyotime Biotechnology) was added to each well and
incubated for 4 h. Then the absorbance values were read at a
wavelength of 450 nm using a microplate reader (SpectraMax 250; GE
Healthcare Life Sciences, Pittsburgh, PA, USA).
Scratch assay
The in vitro migration ability of NSCLC cells
was assessed by scratch assay. Cells were seeded in 6-well plates
and the monolayer was scratched with 10-µl pipette tips. The wound
areas were photographed 0 and 20 h after scratching and measured
using a caliper. The wound-closure percentages were calculated
using the following formula: [1-(current wound size/initial wound
size)] × 100.
Cell invasion assay
Cells were detached and re-suspended in a serum-free
medium and seeded on the upper chamber of Matrigel-coated Transwell
inserts with a pore size of 8 µm. The culture medium containing 10%
FBS as a chemo-attractant was added to the lower chamber. After
24-h incubation, the cells on the upper surface of the insert were
gently removed with a cotton swab. Invading cells (lower surface of
the insert) were fixed with 4% paraformaldehyde (Sigma-Aldrich),
stained with crystal violet, and counted under a microscope. Five
random microscopic fields were examined for each insert.
Flow cytometry analysis
Cells were seeded into 6-well plates at a density of
1×106 cells/well for 24 h. Subsequently, the cells were
collected and stained with the ANXA5 (Annexin V)-PE apoptosis
detection kit (4A Biotech Co. Ltd., FXP018-100) according to the
manufacturer's instructions and analyzed by flow cytometry
(FACSCalibur, BD Bioscience, San Jose, CA, USA).
Statistical analyses
Unless stated otherwise, data are presented as mean
± SD in the figures. A Student's t-test was performed to compare
the two groups of in vitro data. For more than two groups,
we analyzed with one-way ANOVA followed by Tukey's multiple
comparison test. All statistical tests were two-sided.
Results
FSTL1 is downregulated in NSCLC
cells
In order to explore the function of FSTL1 in NSCLC,
we collected an array of lung cancer cells and lung normal
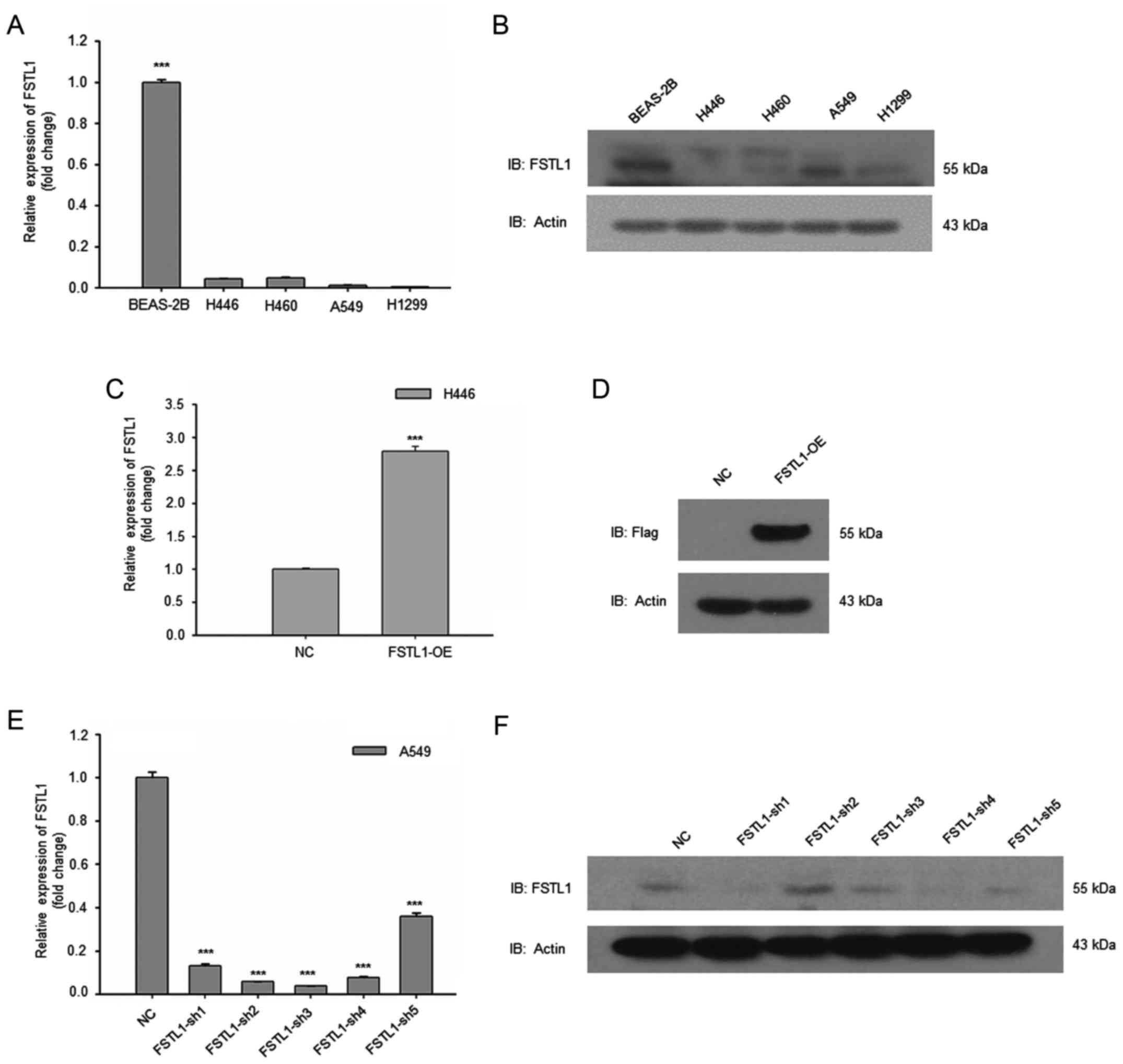
epithelial cell line, BEAS-2B. Expression of FSTL1 was examined by
qRT-PCR and western blot analysis. As shown in Fig. 1A, the mRNA levels of FSTL1 in NSCLC
cells were much lower than normal BEAS-2B cells. Consistently, the
protein level of FSTL1 in BEAS-2B was higher than NSCLC cells
(Fig. 1B). These results suggest
that FSTL1 is downregulated in NSCLC cells.
We then constructed FSTL1 overexpression in H446
cells. Both RT-PCR and western blot analysis revealed the
successful establishment of FSTL1 overexpression (Fig. 1C and D). Then FSTL1 expression was
knocked down in A549 cells. The results of qRT-PCR and western blot
analysis shown, FSTL1 was effectively suppressed by SH1 and SH4
(Fig. 1E and F).
FSTL1 reduced NSCLC cell proliferation
with altered cell cycle
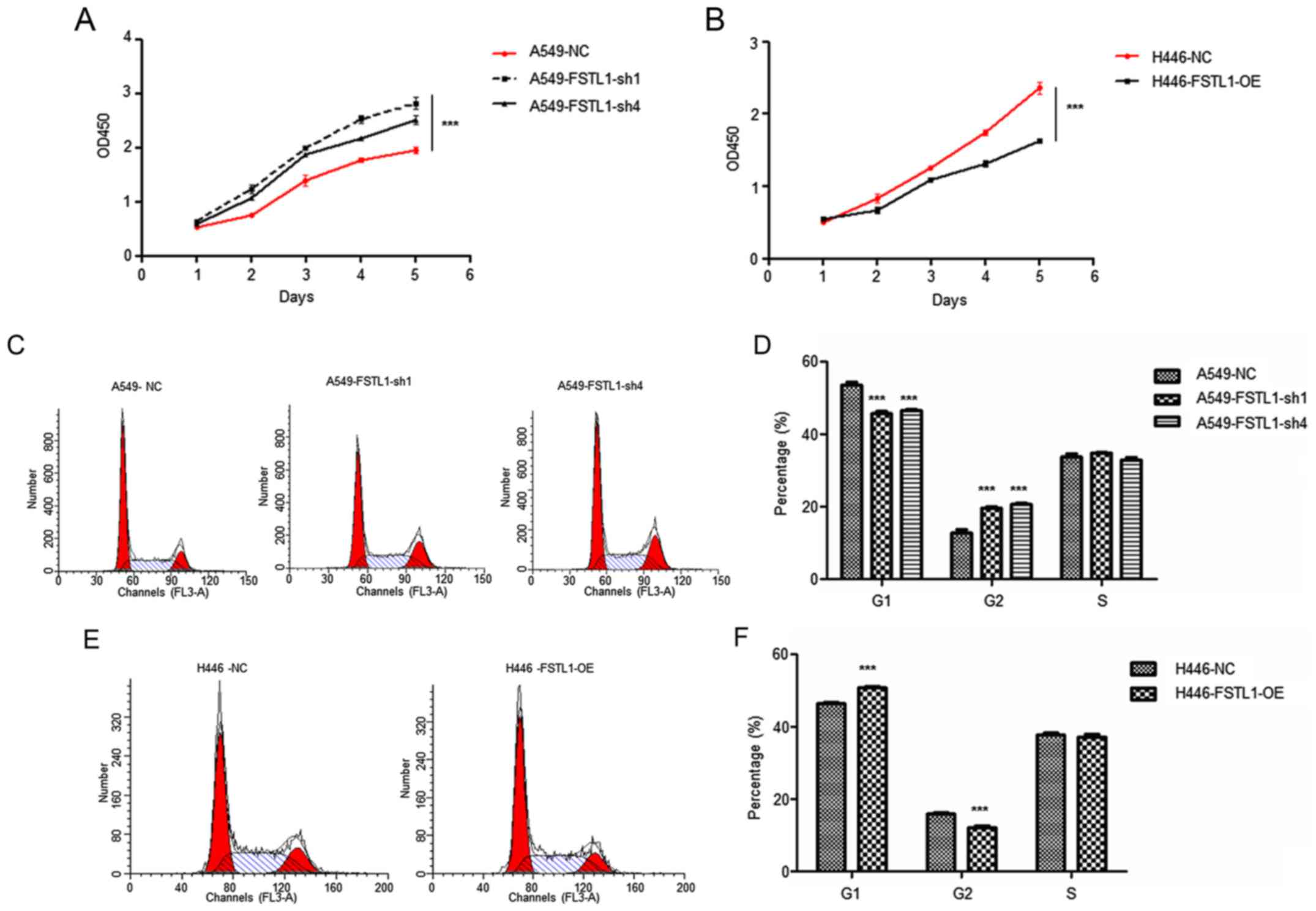
To analyze the function of FSTL1 in NSCLC cells, we
examined the cell proliferation ability using CCK8. The results
showed that A549 cells with FSTL1 knockdown proliferated faster
than control cells (Fig. 2A). On
the contrary, H446 cells with FSTL1 overexpression proliferated
slower than control cells (Fig.
2B). In order to further clarify the function of FSTL1 in NSCLC
cells, we examined the cell cycle of A549 cells with FSTL1
knockdown. As shown in Fig. 2C and
D, the percentage of G1 phase in FSTL1 suppressed A549 cells
was reduced while the percentage of G2 phase was elevated. However,
the percentage of G1 phase in FSTL1 overexpressed H446 cells was
increased while the percentage of G2 phase was reduced (Fig. 2E and F).
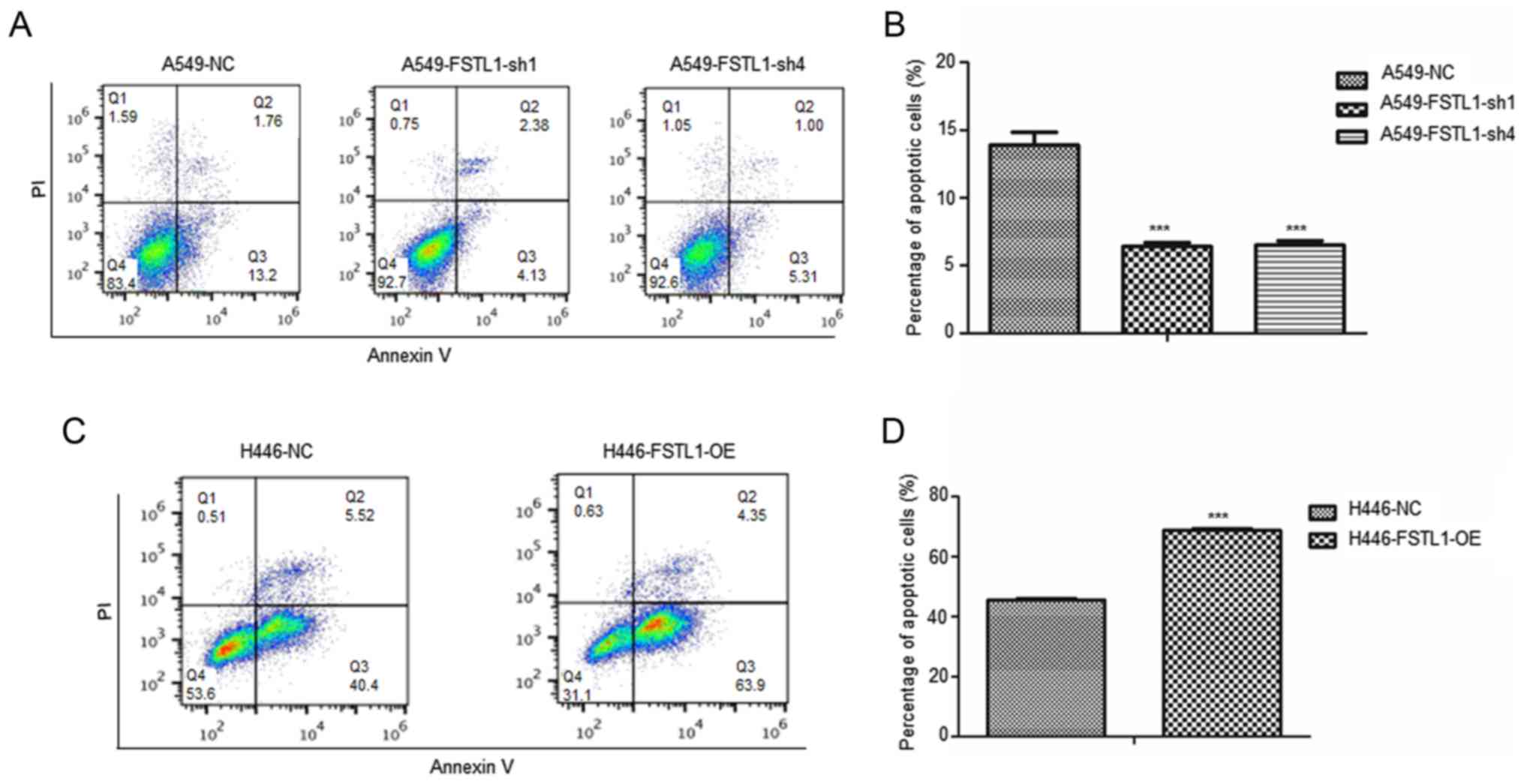
We also examined cell apoptosis after changing FSTL1
expression. As shown in Fig. 3A and
B, the percentage of apoptotic cells in FSTL1-suppressed A549
cells was reduced significantly. However, the percentage of
apoptotic cells in FSTL1 overexpressed H446 cells was increased
(Fig. 3C and D).
Taken together, these results indicate that FSTL1
reduced NSCLC cell proliferation with altered cell cycle and
elevated apoptosis.
FSTL1 suppressed NSCLC cell migration
and invasion
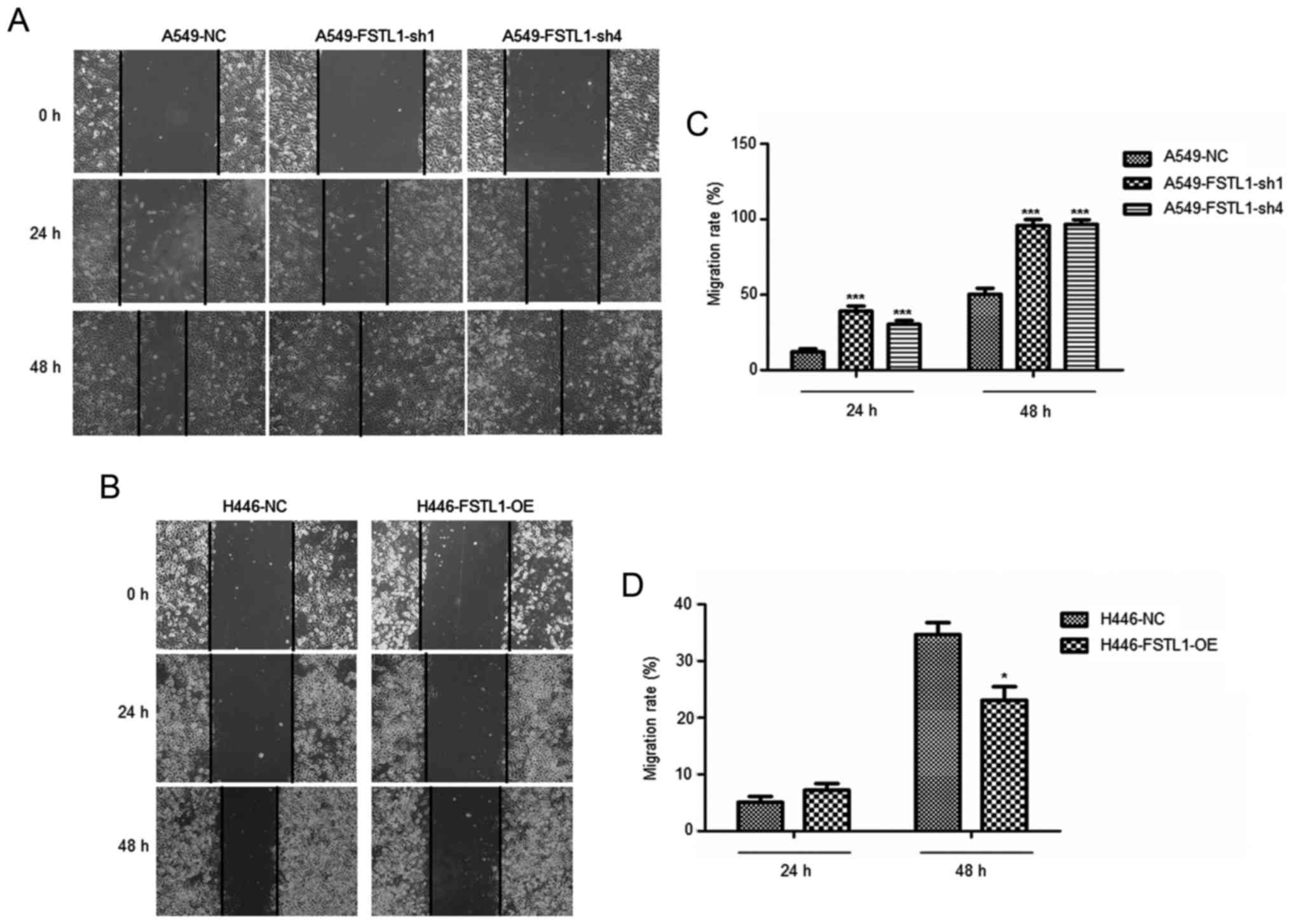
To investigate the effect of FSTL1 on NSCLC cell
migration and invasion, control cells, FSTL1 knockdown cells were
submitted to scratch assay. Our results showed that the migration
ability of A549 cells was significantly increased upon FSTL1
knockdown (Fig. 4A and B). However,
the FSTL1 overexpression strongly reduced the migration ability of
H446 cells.
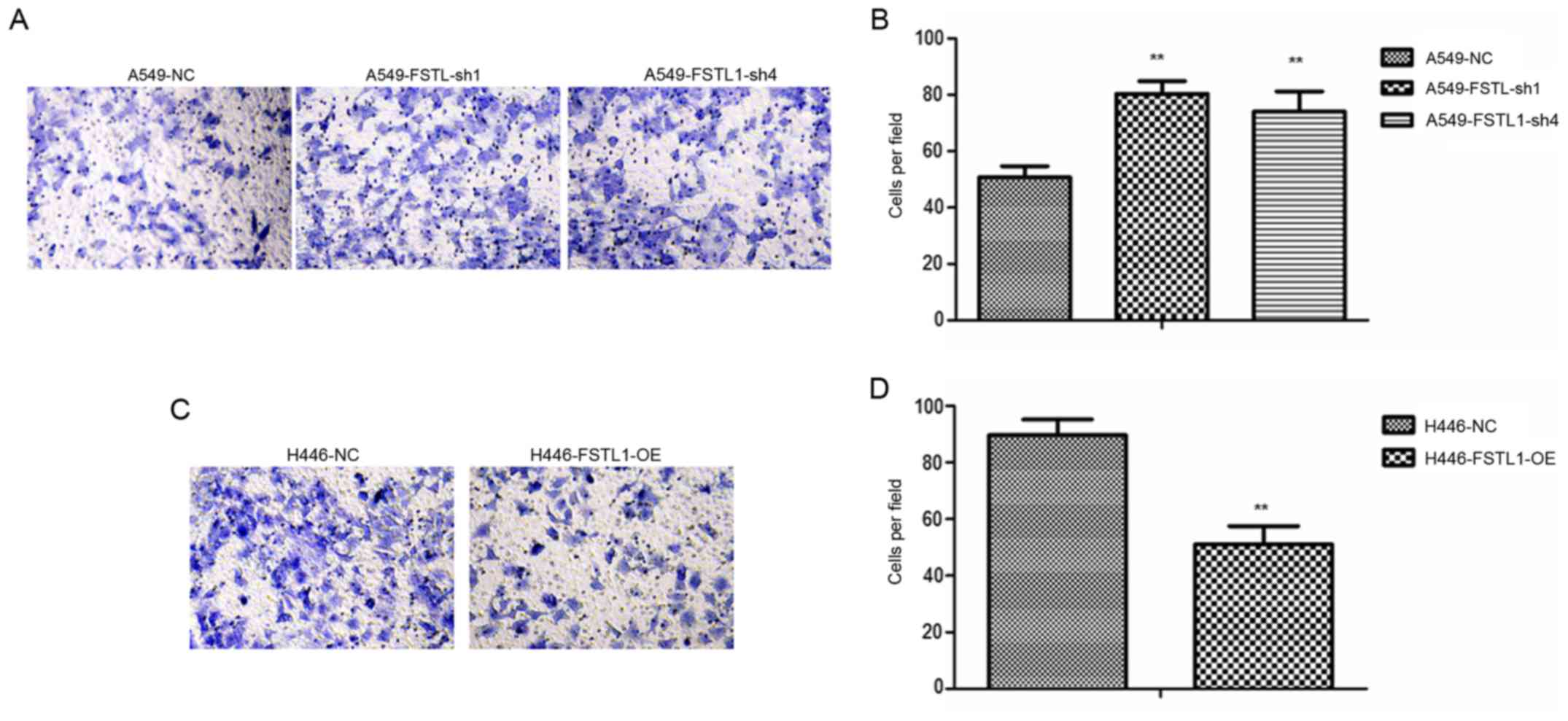
Further, the Transwell assay showed that the
invasion ability of A549 cells was significantly increased after
FSTL1 knockdown (Fig. 5A and B). On
the contrary, ectopic expression of FSTL1 reduced the invasion of
H446 cells (Fig. 5C and D). Thus,
our results demonstrated that FSTL1 suppressed NSCLC cell migration
and invasion.
FSTL1 regulates crucial factors in
apoptosis and invasion
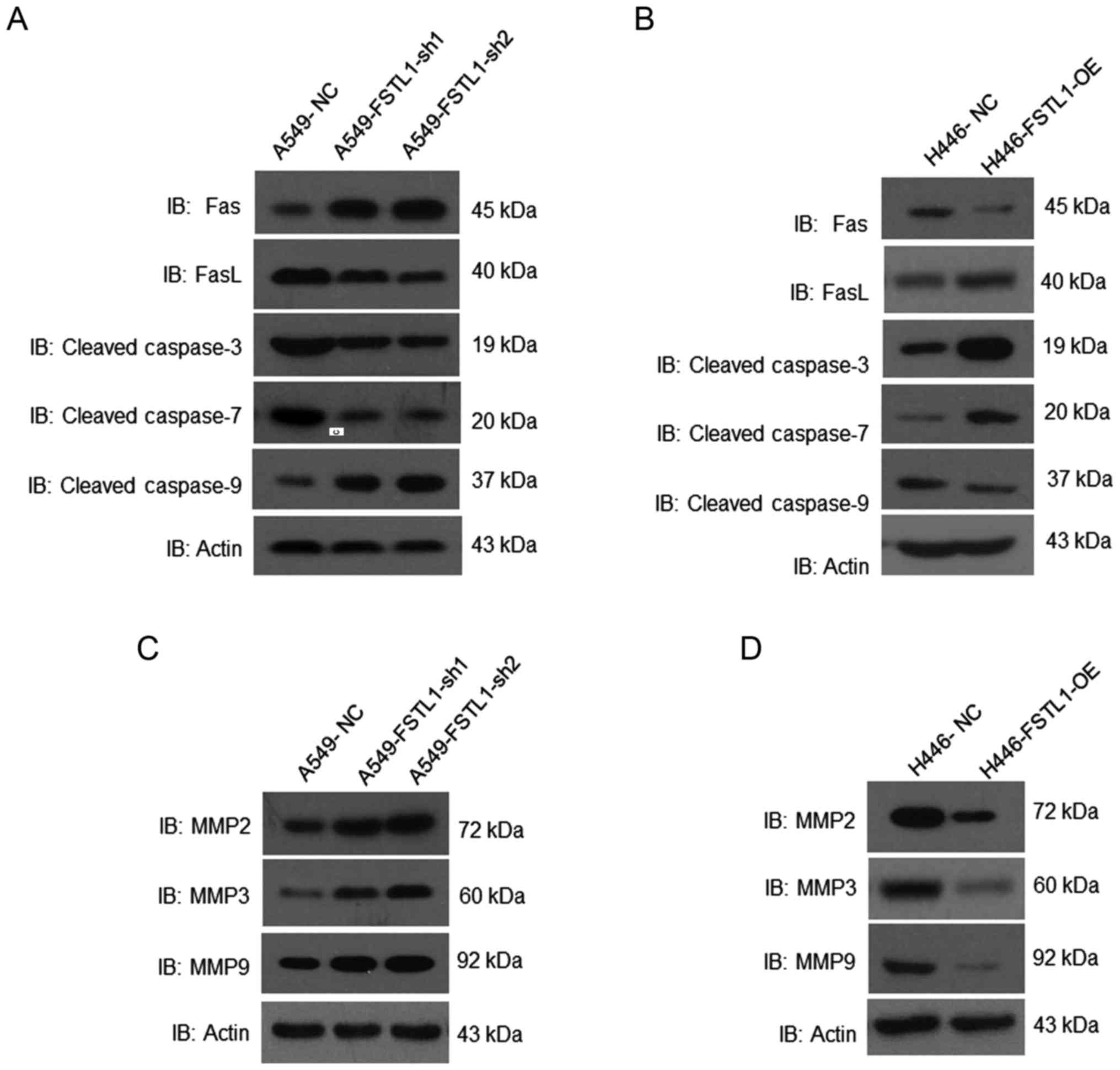
In order to elucidate the underlying mechanisms of
FSTL1 mediated NSCLC cell apoptosis, we investigated the critical
factors in cell apoptosis. We found a significant reduction of
FASL, cleaved caspase-3 and −7, and upregulation of FAS and cleaved
caspase-9 in FSTL1 knockdown A549 cells (Fig. 6A). On the contrary, in FSTL1 over
expressed H446 cells, we observed an increase of FASL, cleaved
caspase-3 and −7 (Fig. 6B) compared
with control cells.
The important factors in cell migration and invasion
were analyzed. MMP2, MMP3 and MMP9 were increased in FSTL1
knockdown A549 cells (Fig. 6C).
However, MMP2, MMP3 and MMP9 were reduced in FSTL1 overexpressing
H446 cells (Fig. 6D).
Taken together, our results demonstrated that FSTL1
might functioned as a tumor suppressor in NSCLC, including
suppressing tumor cell proliferation, invasion and survival.
Discussion
FSTL1 has been found downregulated in many human
cancer cell lines such as lung cancer, colon cancer, stomach
cancer, breast cancer (7) and tumor
patient samples like renal cell carcinoma (19), ovarian and endometrial carcinoma
(20). Our studies revealed that
FSTL1 is significantly downregulated in NSCLC cells. FSTL1 is also
significantly downregulated in SCLC cell line H446. The relatively
different levels of FSTL1 among these lung cancer cells might be
associated with the heterogeneity of lung cancer. Being a
tumor-suppressor gene, FSTL1 was first cloned from a mouse
osteoblastic cell line and can be induced by TGF-β1 (26). In lung cancer cells, FSTL1 secretion
can be influenced by connexin 43, which is also a tumor suppressor
to suppress lung cancer cell invasion and metastasis through
regulating histone H3 and H4 acetylation and MMP-2 expression
(27). Downregulation of MMP-2 was
also detected upon FSTL1 expression (20). In nasopharyngeal cancer cell lines
and tumor biopsies, FSTL1 gene was found downregulated by promoter
hypermethylation (28).
Overexpression of FSTL1 in nasopharyngeal cancer cell lines
significantly suppressed tumor cell proliferation, migration and
invasion ability and induced cell apoptosis.
The downregulated FSTL1 protein level in tumors was
associated with downregulated interleukin 1β and tumor necrosis
factor α, therefore, FSTL1 could activate macrophages and attenuate
the immune evasion (28).
Furthermore, FSTL1 deficiency induced the cellular arrest of G2/M
phase in human lung cancer cells through the accumulation of
cyclin-dependent kinase 1-cyclin B1 complex (25). Besides, based on the study of a
multiple-organ metastasis model of human small cell lung cancer in
natural killer cell-depleted SCID mice, FSTL1 was proved to play a
critical role in the production of multiple-organ metastasis via
inhibiting angiogenesis (29).
FSTL1 has been found regulating many signaling pathways including
AKT, NF-κB, SMAD and others (18,30–32).
The receptor for FSTL1 has not been reported. However, FSTL1 can
interact directly with BMP4 and influence the downstream signaling
of BMP4/SMAD (18), suggesting that
as a secreted glycoprotein, FSTL1 might trigger downstream
signaling through binding to other ligands.
In this study, we found that FSTL1 was downregulated
in NSCLC cells compared with normal control. FSTL1 overexpression
suppressed tumor cell proliferation with altered cell cycle and
induced cell apoptosis. In addition, FSTL1 inhibited cell survival,
migration and invasion of NSCLC cells. The proteins associated with
cell apoptosis and invasion including FAS/FASL, caspases and MMPs
were regulated by FSTL1. Altogether, our results revealed the
critical tumor-suppression function of FSTL1 in NSCLC progression,
suggesting that FSTL1 might be an important factor in NSCLC
progression.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wood SL, Pernemalm M, Crosbie PA and
Whetton AD: The role of the tumor-microenvironment in lung
cancer-metastasis and its relationship to potential therapeutic
targets. Cancer Treat Rev. 40:558–566. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fidler IJ: Critical determinants of cancer
metastasis: Rationale for therapy. Cancer Chemother Pharmacol. 43
Suppl:S3–S10. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shimasaki S, Koga M, Esch F, Cooksey K,
Mercado M, Koba A, Ueno N, Ying SY, Ling N and Guillemin R: Primary
structure of the human follistatin precursor and its genomic
organization. Proc Natl Acad Sci USA. 85:pp. 4218–4222. 1988;
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Engel J, Taylor W, Paulsson M, Sage H and
Hogan B: Calcium binding domains and calcium-induced conformational
transition of SPARC/BM-40/osteonectin, an extracellular
glycoprotein expressed in mineralized and nonmineralized tissues.
Biochemistry. 26:6958–6965. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mashimo J, Maniwa R, Sugino H and Nose K:
Decrease in the expression of a novel TGF beta1-inducible and
ras-recision gene, TSC-36, in human cancer cells. Cancer Lett.
113:213–219. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ohba M, Shibanuma M, Kuroki T and Nose K:
Production of hydrogen peroxide by transforming growth factor-beta
1 and its involvement in induction of egr-1 in mouse osteoblastic
cells. J Cell Biol. 126:1079–1088. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Johnston IM, Spence HJ, Winnie JN, McGarry
L, Vass JK, Meagher L, Stapleton G and Ozanne BW: Regulation of a
multigenic invasion programme by the transcription factor, AP-1:
Re-expression of a down-regulated gene, TSC-36, inhibits invasion.
Oncogene. 19:5348–5358. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu S, Wang L, Wang W, Lin J, Han J, Sun
H, Guo H, Sun R and Wu Q: TSC-36/FRP inhibits vascular smooth
muscle cell proliferation and migration. Exp Mol Pathol.
80:132–140. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Esterberg R, Delalande JM and Fritz A:
Tailbud-derived Bmp4 drives proliferation and inhibits maturation
of zebrafish chordamesoderm. Development. 135:3891–3901. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Oshima Y, Ouchi N, Sato K, Izumiya Y,
Pimentel DR and Walsh K: Follistatin-like 1 is an Akt-regulated
cardioprotective factor that is secreted by the heart. Circulation.
117:3099–3108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ouchi N, Oshima Y, Ohashi K, Higuchi A,
Ikegami C, Izumiya Y and Walsh K: Follistatin-like 1, a secreted
muscle protein, promotes endothelial cell function and
revascularization in ischemic tissue through a nitric-oxide
synthase-dependent mechanism. J Biol Chem. 283:32802–32811. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wilson DC, Marinov AD, Blair HC, Bushnell
DS, Thompson SD, Chaly Y and Hirsch R: Follistatin-like protein 1
is a mesenchyme-derived inflammatory protein and may represent a
biomarker for systemic-onset juvenile rheumatoid arthritis.
Arthritis Rheum. 62:2510–2516. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wei K, Serpooshan V, Hurtado C,
Diez-Cuñado M, Zhao M, Maruyama S, Zhu W, Fajardo G, Noseda M,
Nakamura K, et al: Epicardial FSTL1 reconstitution regenerates the
adult mammalian heart. Nature. 525:479–485. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Adams DC, Karolak MJ, Larman BW, Liaw L,
Nolin JD and Oxburgh L: Follistatin-like 1 regulates renal IL-1β
expression in cisplatin nephrotoxicity. Am J Physiol Renal Physiol.
299:F1320–F1327. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hayakawa S, Ohashi K, Shibata R, Kataoka
Y, Miyabe M, Enomoto T, Joki Y, Shimizu Y, Kambara T, Uemura Y, et
al: Cardiac myocyte-derived follistatin-like 1 prevents renal
injury in a subtotal nephrectomy model. J Am Soc Nephrol.
26:636–646. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Geng Y, Dong Y, Yu M, Zhang L, Yan X, Sun
J, Qiao L, Geng H, Nakajima M, Furuichi T, et al: Follistatin-like
1 (Fstl1) is a bone morphogenetic protein (BMP) 4 signaling
antagonist in controlling mouse lung development. Proc Natl Acad
Sci USA. 108:pp. 7058–7063. 2011; View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu Y, Han X, Yu Y, Ding Y, Ni C, Liu W,
Hou X, Li Z, Hou J, Shen D, et al: A genetic polymorphism affects
the risk and prognosis of renal cell carcinoma: Association with
follistatin-like protein 1 expression. Sci Rep. 6:266892016.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chan QK, Ngan HY, Ip PP, Liu VW, Xue WC
and Cheung AN: Tumor suppressor effect of follistatin-like 1 in
ovarian and endometrial carcinogenesis: A differential expression
and functional analysis. Carcinogenesis. 30:114–121. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Reddy SP, Britto R, Vinnakota K, Aparna H,
Sreepathi HK, Thota B, Kumari A, Shilpa BM, Vrinda M, Umesh S, et
al: Novel glioblastoma markers with diagnostic and prognostic value
identified through transcriptome analysis. Clin Cancer Res.
14:2978–2987. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kudo-Saito C, Fuwa T, Murakami K and
Kawakami Y: Targeting FSTL1 prevents tumor bone metastasis and
consequent immune dysfunction. Cancer Res. 73:6185–6193. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tan X, Zhai Y, Chang W, Hou J, He S, Lin
L, Yu Y, Xu D, Xiao J, Ma L, et al: Global analysis of
metastasis-associated gene expression in primary cultures from
clinical specimens of clear-cell renal-cell carcinoma. Int J
Cancer. 123:1080–1088. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sumitomo K, Kurisaki A, Yamakawa N,
Tsuchida K, Shimizu E, Sone S and Sugino H: Expression of a
TGF-beta1 inducible gene, TSC-36, causes growth inhibition in human
lung cancer cell lines. Cancer Lett. 155:37–46. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bae K, Park KE, Han J, Kim J, Kim K and
Yoon KA: Mitotic cell death caused by follistatin-like 1 inhibition
is associated with up-regulated Bim by inactivated Erk1/2 in human
lung cancer cells. Oncotarget. 7:18076–18084. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shibanuma M, Mashimo J, Mita A, Kuroki T
and Nose K: Cloning from a mouse osteoblastic cell line of a set of
transforming-growth-factor-beta 1-regulated genes, one of which
seems to encode a follistatin-related polypeptide. Eur J Biochem.
217:13–19. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao W, Han HB and Zhang ZQ: Suppression
of lung cancer cell invasion and metastasis by connexin43 involves
the secretion of follistatin-like 1 mediated via histone
acetylation. Int J Biochem Cell Biol. 43:1459–1468. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhou X, Xiao X, Huang T, Du C, Wang S, Mo
Y, Ma N, Murata M, Li B, Wen W, et al: Epigenetic inactivation of
follistatin-like 1 mediates tumor immune evasion in nasopharyngeal
carcinoma. Oncotarget. 7:16433–16444. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ogino H, Yano S, Kakiuchi S, Muguruma H,
Ikuta K, Hanibuchi M, Uehara H, Tsuchida K, Sugino H and Sone S:
Follistatin suppresses the production of experimental
multiple-organ metastasis by small cell lung cancer cells in
natural killer cell-depleted SCID mice. Clin Cancer Res.
14:660–667. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liang X, Hu Q, Li B, McBride D, Bian H,
Spagnoli P, Chen D, Tang J and Zhang JH: Follistatin-like 1
attenuates apoptosis via disco-interacting protein 2 homolog A/Akt
pathway after middle cerebral artery occlusion in rats. Stroke.
45:3048–3054. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen W, Xia J, Hu P, Zhou F, Chen Y, Wu J,
Lei W and Shen Z: Follistatin-like 1 protects cardiomyoblasts from
injury induced by sodium nitroprusside through modulating Akt and
Smad1/5/9 signaling. Biochem Biophys Res Commun. 469:418–423. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang H, Wu S, Huang S, Yin S, Zou G, Huang
K, Zhang Z, Tang A and Wen W: Follistatin-like protein 1
contributes to dendritic cell and T-lymphocyte activation in
nasopharyngeal carcinoma patients by altering nuclear factor κb and
Jun N-terminal kinase expression. Cell Biochem Funct. 34:554–562.
2016. View
Article : Google Scholar : PubMed/NCBI
|