Introduction
Ovarian cancer is the leading cause of death from
gynecological cancers in highly developed countries, and
responsible for approximately 14,000 deaths each year in the United
States (1). The 5-year survival of
patients with ovarian cancer ranges from 30 to 50%. Despite
debulking surgery and standard adjuvant chemotherapy, most patients
with advanced disease relapse within 2 years after diagnosis
(2). In accordance with these data,
there is a pressing need for new approaches to the medical
management of this disease. Understanding of the specific
neoplastic process and the knowledge of cancer cell biology
(3) will allow the identification
and development of novel biological agents in clinical oncology
which could be used in addition to classical chemotherapy. This
also applies to ovarian cancer in which genomic and epigenetic
alterations have been frequently shown (4–6).
An interesting class of drugs targeting mechanisms
strictly related to cancer initiation and progression are histone
deacetylase (HDAC) inhibitors (7).
These agents are involved in the regulation of apoptosis and
differentiation of tumor cells, suppression of angiogenesis, cell
migration and motility, and immunomodulation (8,9).
Eighteen mammalian HDAC enzymes have been identified to date, which
can be subdivided into different families according to their
homology with yeast HDACs (10). To
date, 4 HDAC inhibitors have been approved in oncology (vorinostat,
panobinostat, belinostat and romidepsin) and many other drugs from
this group are currently being tested in clinical trials. Some HDAC
inhibitors have been found to be effective in preclinical studies
in ovarian carcinoma, either as monotherapy or in combination with
other agents (11–14). There has been a strong rationale to
test HDAC inhibitors as anticancer agents against ovarian cancer in
the clinic (15–17). An interesting pan-HDAC inhibitor
that has been poorly studied is scriptaid,
6-[1,3-dioxo-1H,3H-benzo(de)isoquinolin-2-yl]-hexanoic
acid hydroxyamide. This agent was shown to inhibit the growth and
induce differentiation and/or apoptosis in breast, glioma, colon
and also gynecological cancer cells (18–21).
Some data suggest that the antitumor effectiveness of scriptaid can
be strongly boosted, even synergistically, by its combination with
other agents/modalities (22).
Another group of agents that is being explored in
experimental and clinical oncology are proteasome inhibitors
(23). Blocking proteasome activity
leads to accumulation of damaged proteins, resulting in caspase
activation and cell death (23).
Bortezomib is the first proteasome inhibitor approved by the US
Food and Drug Administration for the treatment of
relapsed/refractory multiple myeloma. Recent research demonstrated
that combinations of HDAC3 inhibitor BG45 and bortezomib exerted a
synergistic therapeutic effect in a murine xenograft model of human
multiple melanoma (24). A
combination of HDAC inhibitor vorinostat with bortezomib was found
to improve survival in comparison with bortezomib alone in patients
with multiple myeloma (25).
In the present study, we investigated the antitumor
effects of scriptaid, used either alone or in combination with
bortezomib, as well as with standard chemotherapeutics: paclitaxel,
doxorubicin, carboplatin or etoposide, on representative ovarian
cancer cells in vitro. The rationale for combining scriptaid
together with bortezomib or with conventional chemotherapeutics was
that the former can sensitize ovarian cancer cells and can reverse
resistance of cancer cells to drugs used in currently established
therapeutic protocols (26–29).
Materials and methods
Tumor cells and reagents
Three ovarian cancer cell lines were studied: SKOV-3
(cat. no. HTB-77; ATCC, Manassas, VA, USA), MDAH 2774 (cat. no.
CRL-10303; ATCC), and OVP-10 (obtained from Dr Barbara Szaniawska,
Department of Cell Biology, The Maria Skłodowska Curie Memorial
Cancer Centre and Institute of Oncology, Warsaw, Poland). MDAH 2774
cells were cultured either in Dulbecco's MEM with 4.5 g/l glucose,
sodium pyruvate and L-glutamine (high glucose DMEM) (Corning,
Corning, NY, USA) and OVP-10 and SKOV-3 cells in RPMI-1640
(Sigma-Aldrich, Irvine, UK) [supplemented with
antibiotic-antimycotic (Corning), and 10% heat-inactivated fetal
bovine serum (FBS) (Gibco BRL, Paisley, UK)]. The cells were
maintained in 25-cm2 tissue flasks (Nunc, Roskdile,
Denmark) at 37°C in a humidified atmosphere of 5% CO2
and were passaged two to three times weekly. The following
antitumor agents were tested against ovarian cancer cells:
doxorubicin (Pfizer), etoposide (Ebeve), paclitaxel (Actavis),
carboplatin (Medac), scriptaid (Selleckchem), and bortezomib
(Millenium Pharm. Inc.). The drugs were first diluted in DMSO or
water for injection and then in growth medium.
MTT assay
The cytotoxic effect of scriptaid, bortezomib, and
other antitumor chemotherapeutics on SKOV-3, MDAH 2774 and OVP-10
ovarian cancer cells was tested in a standard
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
assay. The cells were placed in 96-well plates (flat bottom) at a
concentration of 12×103 (SKOV-3), or 4×103
(OVP-10, MDAH 2774) cells/well and drugs or solvent control were
added for a 72-h exposure in a final volume of 0.2 ml/well. At the
end of the incubation, 25 µl of MTT (2.5 mg/ml stock solution) was
added to each well. After a 4-h incubation with MTT, the cells were
centrifuged (350 × g, 10 min), supernatants were removed and
formazan crystals were dissolved in 100 µl of acid DMSO solution.
The absorbance in each well was measured using an ELISA reader
(Asys UVM 340 Microplate Reader, Biochrom Ltd., UK) using a 540-nm
filter. The means ± standard deviations (SDs) were determined for
triplicate samples. The cytotoxic/cytostatic effect was expressed
as the relative viability and was calculated according to the
formula: Relative viability = [(experimental absorbance -
background absorbance)/(absorbance of vehicle-treated cells -
background absorbance) × 100%.
Apoptosis assay by flow cytometry
Flow cytometric analysis was performed using
propidium iodide (PI) and fluorescein isothiocyanate (FITC)-labeled
Annexin V (eBioscience, Vienna, Austria). SKOV-3, MDAH 2774 or
OVP-10 cells, after the incubation with agents for 72 h, were
washed with cold binding buffer at 4°C and centrifuged at 500 × g
for 5 min. Then, cells were resuspended in binding buffer at a
concentration of 4×106 cells/ml. Cell suspensions
(2×105 cells in 50 µl) were mixed with 1 µl FITC Annexin
V and 1 µl propidium iodide (PI) and incubated for 15 min at room
temperature in the dark. Finally, the cells were analyzed by flow
cytometry (BD Accuri™ C6 Plus flow cytometer).
Western blotting
At the end of the incubation with the agents (72 h),
SKOV-3 cells were washed three times with PBS, pelleted and lysed
in RIPA buffer containing protease inhibitor cocktail and
phosphatase inhibitor cocktail. The lysates were centrifuged at
15,000 rpm for 30 min at 4°C and the supernatant was collected. The
protein concentration in the lysates was determined by
bicinchoninic acid assay (BCA assay). Next, the cell extracts were
separated on 10% SDS-polyacrylamide gel (cleaved-caspase 9) or 12%
SDS-polyacrylamide gel (p21 and procaspase 3). The primary
antibodies (all from Cell Signaling, at 1:1,000 dilutions) were
used for the overnight incubation at 4°C in the presence of 5%
non-fat dry milk. After washing with TBST, membranes were incubated
with anti-rabbit HRP-coupled secondary antibody (Jackson
ImmunoResearch) in 5% dry milk in TBST for 1 h at room temperature.
The expression of targeted proteins was detected with the enhanced
chemiluminescent detection system and visualized with Stella 8300
bioimager (Raytest, Straubenhardt, Germany). The blots were
re-probed with anti-β-actin-peroxidase purified immunoglobulin
(clone AC-15; Sigma-Aldrich) at 1:50,000 dilution for 45 min.
Statistical analysis
The data in the figures are presented as the mean ±
SD. The combination index (CI) for drug interaction (synergism) was
calculated using CompuSyn software (ComboSyn, Inc.) (30). According to Chou-Talalay method, if
CI is <1, the two drugs show synergism; if CI is >1, the two
drugs show antagonism (31).
Results
Synergistic inhibitory effects of
scriptaid and bortezomib on the viability of SKOV-3, OVP-10, and
MDAH 2774 cells
In our preliminary experiments, cytotoxic effects of
single agents: scriptaid, bortezomib, doxorubicin, paclitaxel,
carboplatin or etoposide were determined in a 72-h MTT assay
against SKOV-3, OVP-10, and MDAH 2774 cells. A dose-dependent
decrease in cell viability was observed in all cultures (data not
shown). In the next step, combinations of scriptaid with other
agents were tested. Incubation of SKOV-3 cells with scriptaid
(0.5–16 µM) or bortezomib (15–1,500 nM) resulted in a
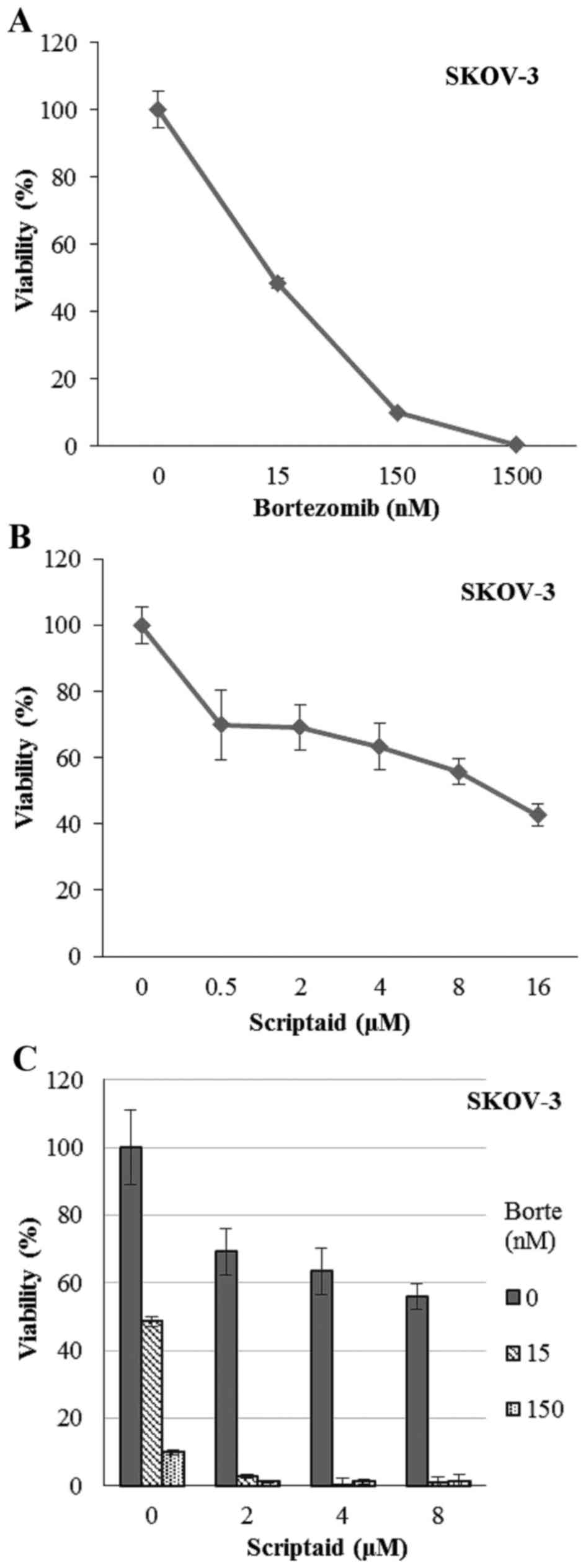
dose-dependent decreasing viability (Fig. 1A and B). In fact, the highest
concentrations of bortezomib completely killed SKOV-3 cells. The
combination of scriptaid and bortezomib caused a synergistic effect
(CI <1, between 0.009 and 0.69) and a concentration as low as 15
nM of bortezomib and 2 µM dose of scriptaid led to killing of
almost all cells (Fig. 1C). Similar
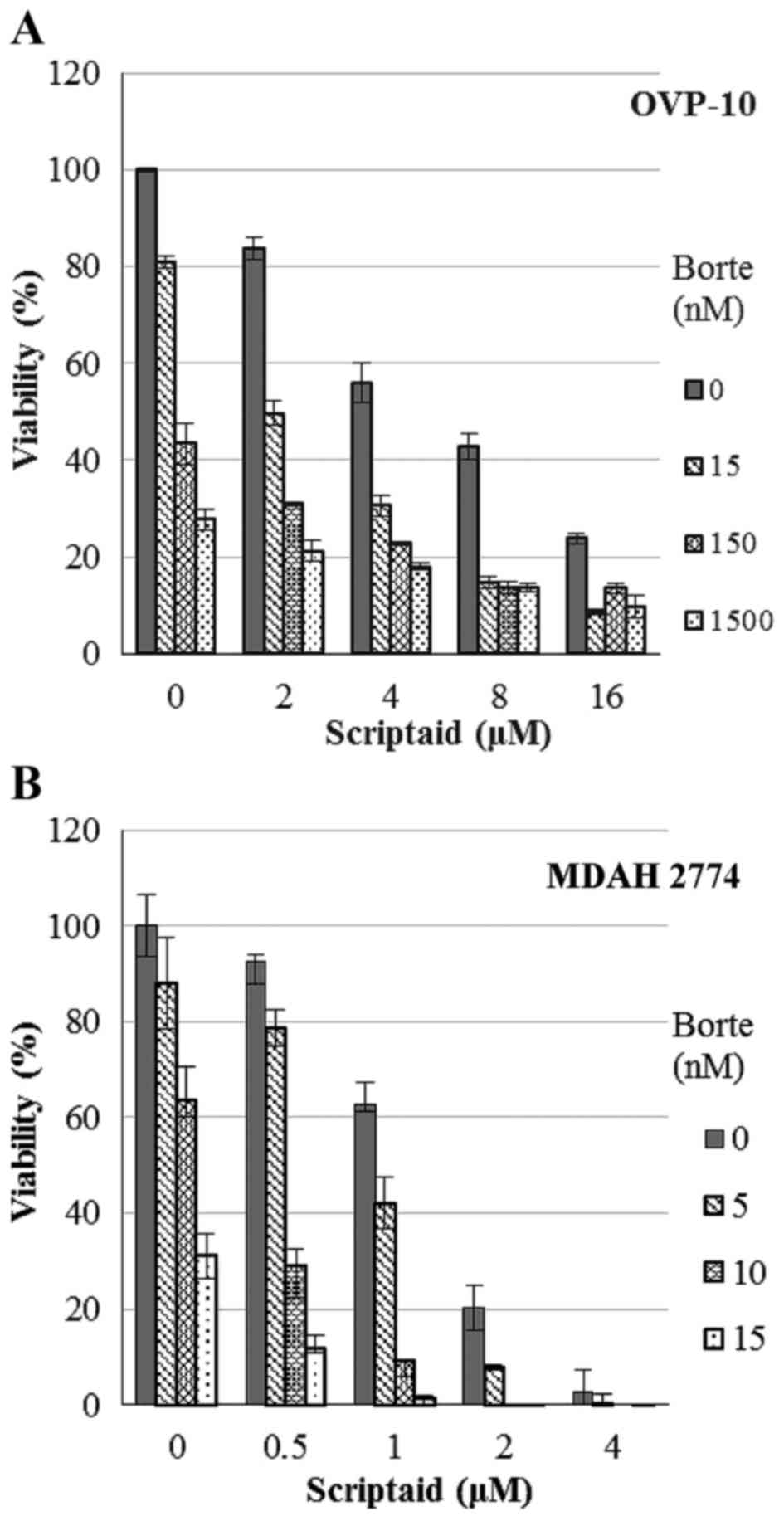
effects were observed in cultures of OVP-10 and MDAH 2774 ovarian
cancer cells. Combination treatment induced stronger antitumor
effect than the treatment with either scriptaid or bortezomib and
resulted in a synergistic effect with CI between 0.335 and 0.819
(OVP-10) and between 0.183 and 0.917 (MDAH 2774) (Fig. 2).
Synergistic inhibitory effects of
scriptaid and doxorubicin on the viability of SKOV-3, OVP-10, and
MDAH 2774 cells
Paclitaxel or carboplatin, when used in combination
with scriptaid, presented additive antitumor effects against
ovarian cancer cells while etoposide did not significantly affect
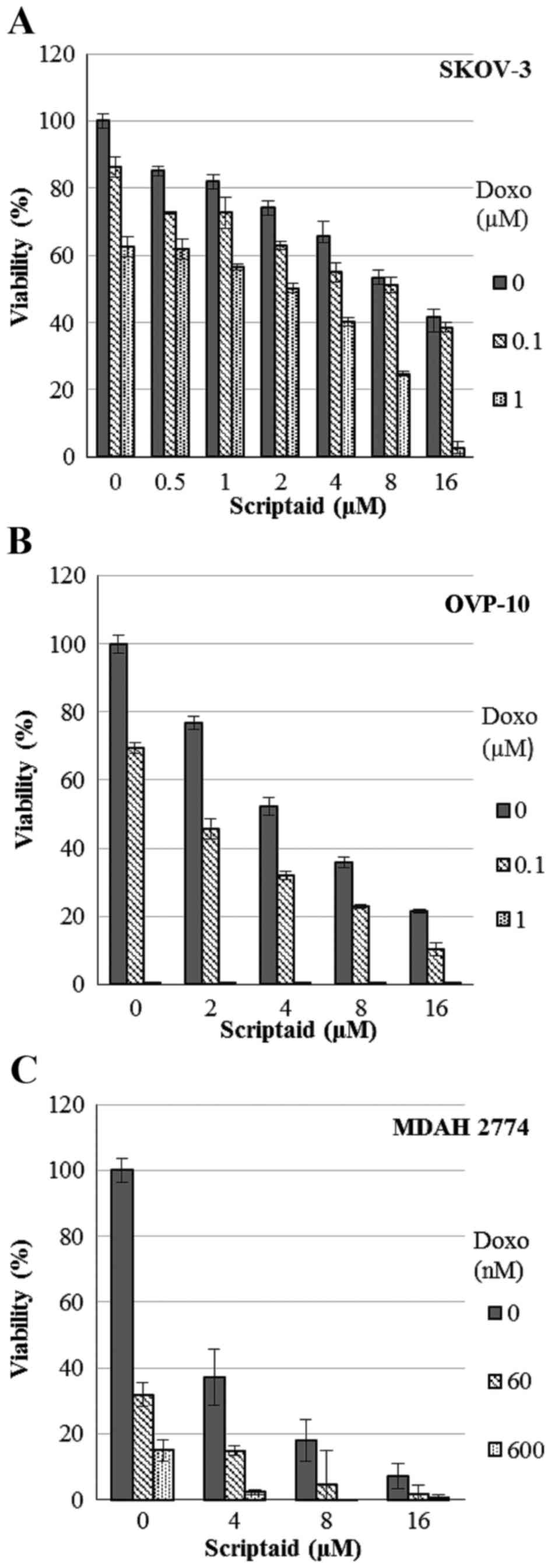
cell viability (data not shown). In contrast, joint administration
of scriptaid (concentration range 0.5–16 µM) with doxorubicin (60
nM - 1 µM) induced synergistic cytotoxic activity in cultures of
SKOV-3, OVP-10 and MDAH 2774 ovarian cancer cell lines. In these
cases, the combined treatment caused synergistic effects with a CI
between 0.027 and 0.727 (SKOV-3), 0.059 and 0.456 (OVP-10)
(Fig. 3A and B). In MDAH 2774
cells, a synergistic effect was observed in the combinations with
the highest concentrations of scriptaid (8–16 µM) with CI between
0.188 and 0.987 (Fig. 3C).
Effects of combined treatment on
cellular apoptosis
To determine the effects of scriptaid, bortezomib,
doxorubicin and combinations of these agents on cellular apoptosis
of SKOV-3, OVP-10 and MDAH 2774 cells, Annexin V-FITC/PI apoptosis
assay was used. Treatment with scriptaid (0.5–16 µM), bortezomib
(5–1,500 nM) or doxorubicin (0.06–10 µM) for 72 h induced a
concentration-dependent increase in cellular apoptosis of each cell
line (data not shown). Combined treatment with scriptaid and
bortezomib or scriptaid and doxorubicin resulted in a significant
increase in cellular apoptosis of cells when compared with the
single agents and the control. Representative results are shown in
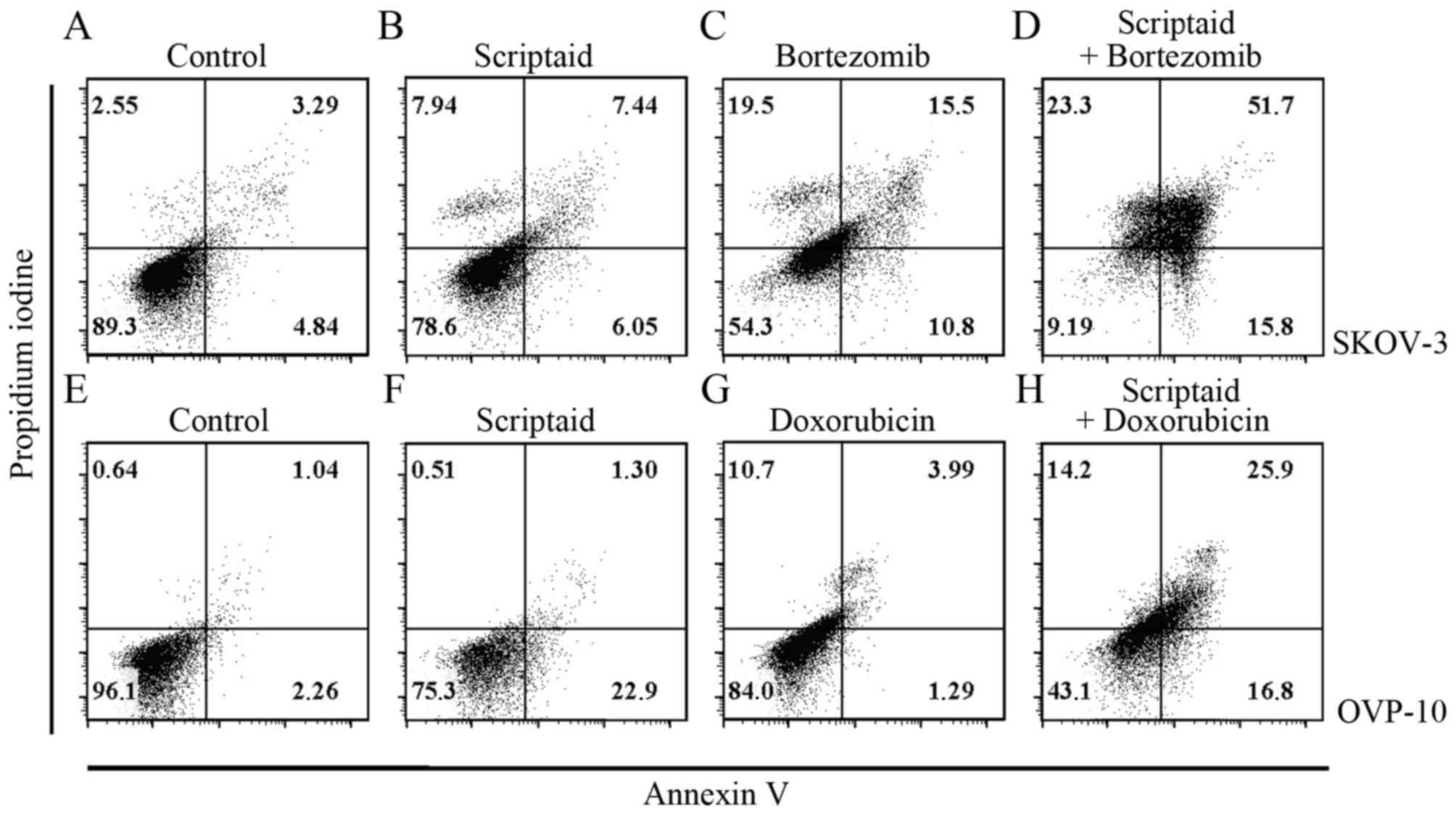
Fig. 4. For example, in the SKOV-3
cell culture incubated with 2 µM scriptaid + 15 nM bortezomib, late
apoptotic and dead cells constituted 51.7% of the cell population
in comparison with 7.44, 15.5 and 3.29% in scriptaid, bortezomib,
and control cultures, respectively (Fig. 4A-D). Only 9.19% cells remained fully
alive in a double-treated culture (Fig.
4D). Similar effect - the lowest percentage of living cells was
observed in culture of MDAH 2774 cells treated with both scriptaid
and bortezomib (data not shown). Results of scriptaid and
doxorubicin combination were very much alike. In the case of the
OVP-10 ovarian cancer cells incubated with 2 µM scriptaid + 50 nM
doxorubicin, the percentage of late apoptotic/dead cells was
highest and the number of living cells was smallest, in comparison
with the single agent-treated cultures (Fig. 4E-H).
Effects of the combined treatment on
the expression of proapoptotic and cell cycle arrest proteins
HDAC inhibitors and bortezomib, as well as
conventional antitumor chemotherapeutics, have been reported to
regulate the level of different proteins that control the cell
cycle and apoptosis (26,32,33).
In the present study, we analyzed the influence of scriptaid in
combination with either bortezomib or doxorubicin on the expression
levels of apoptotic proteins caspase 3 and 9, and the key marker of
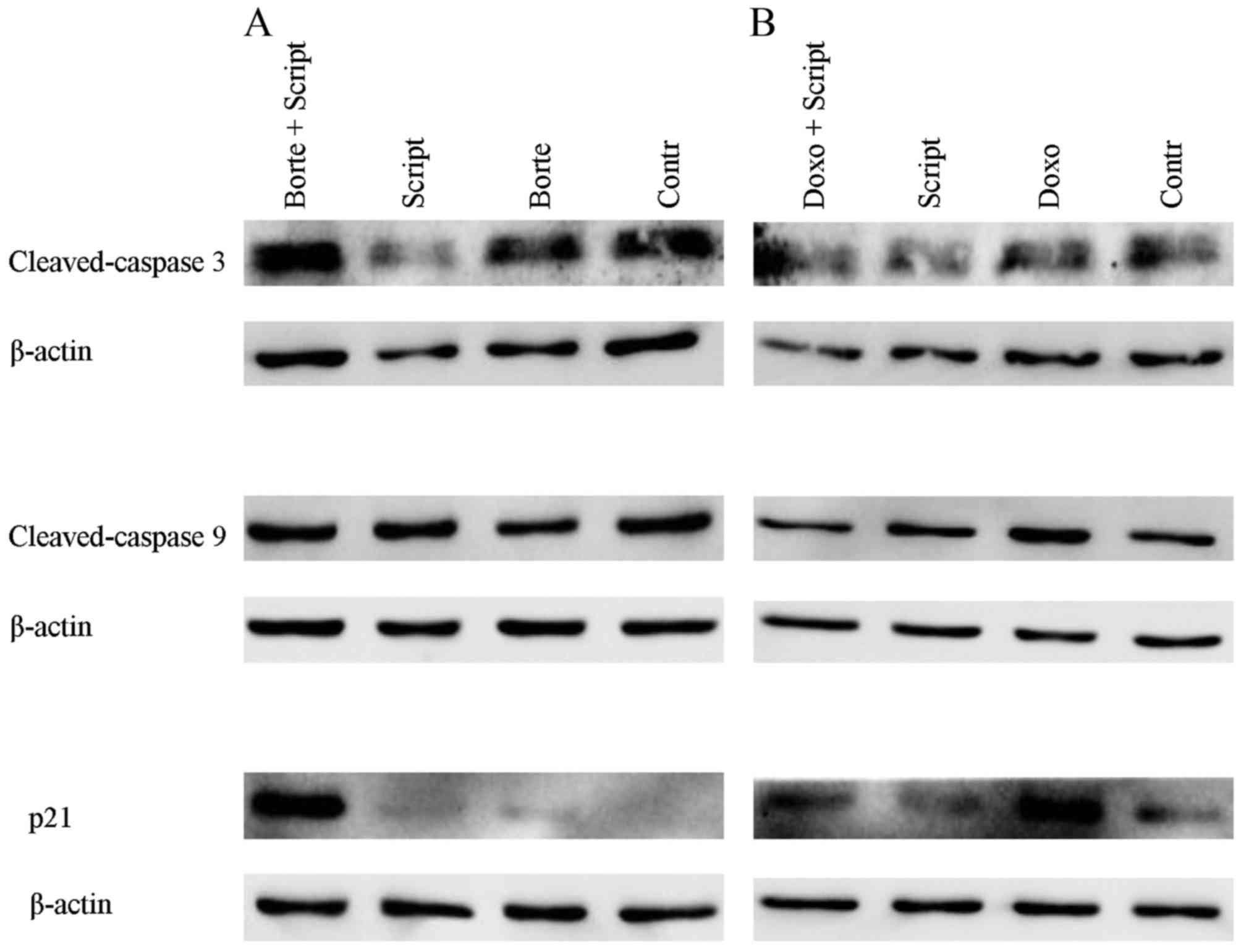
cell cycle arrest protein p21. As shown in Fig. 5, scriptaid, bortezomib, and
doxorubicin, either alone or in combination, did not change
expression of caspase 3 and 9 in the 72-h cultures of SKOV-3 cells.
In contrast, treatment with scriptaid and bortezomib resulted in a
marked increase in p21, suggesting that cell cycle arrest
mechanisms significantly contributed to the cytotoxic/cytostatic
effects of this combination.
Discussion
Over the last two decades, through improved surgical
cytoreduction and more chemotherapy options, the survival of
patients with advanced ovarian cancer and the chance to cure this
disease have increased significantly (34). However, current conventional
chemotherapy is non-selective and often results in a marked
toxicity. Due to increased knowledge of the molecular, genetic and
epigenetic background of ovarian cancer, novel treatment approaches
are being investigated (5). Recent
implementation of olaparib to the drug armamentarium used in
ovarian cancer (to treat selected patients) is the example of such
a successful search (35). Since
epigenetic changes may play a role in the pathogenesis of many
ovarian cancers, there have been studies focusing on the
interference with histone modification and DNA methylation
processes.
In the present study, we demonstrated unique
combinations of the experimental agent, HDAC inhibitor scriptaid,
with bortezomib (the proteasome inhibitor used in patients with
hematological malignancies) or with conventional chemotherapeutics
used to treat gynecological cancers. The most significant
combination, scriptaid and bortezomib, was found to act
synergistically against cancer cells of three representative
ovarian cancer cell lines. Notably, a growth inhibitory effect of
this combination was manifested at suboptimal concentrations of the
single agents. This effect can be attributed both to induction of
apoptosis (Fig. 4) and cell cycle
inhibition (Fig. 5). Our results
are in agreement with studies of other authors who tested various
combinations of HDAC and proteasome inhibitors in colon (36), hepatocellular (32), and lung (26) cancer models in vitro.
HDAC inhibitors (including scriptaid) are recognized
as promising drugs in oncology, since they frequently reverse
epigenetic changes in different types of tumors (7). It was recently shown that there are
other proteins, in addition to histones, whose activity is affected
by acetylation (9). Four HDAC
inhibitors have been approved in clinical oncology to date:
vorinostat, panobinostat, belinostat (hydroxamate-based pan-HDAC
inhibitor), and romidepsin (cyclic tetrapeptide HDAC inhibitor).
All these drugs are used in hematological malignancies. However,
despite their evident benefit in lymphoma and myeloma, these drugs
have not been found effective in studies with different solid
tumors, including ovarian cancer (16,37).
The general view is that HDAC inhibitors are promising drugs in
solid malignancies but only when combined with other anticancer
drugs/agent or radiotherapy (7,38).
Scriptaid is an HDAC inhibitor that was identified
by screening a library of 16,320 compounds (DIVERSet; Chembridge,
San Diego, CA, USA) in 2000 (39).
The advantage of this agent over other known HDAC inhibitors is its
relative non-toxic effect on normal cells (19,21),
and possible preferential activity against gynecological cancers
(18,21,40).
This was one of the rationales for selecting scriptaid in our study
using combination treatment with proteasome inhibitor bortezomib
and other chemotherapeutics on ovarian cancer cell lines. We were
additionally encouraged by the observation of the synergistic
antitumor and chemosensitization effect of scriptaid and various
proteasome inhibitors on human colorectal cancer cells (41) and antitumor effects (in renal cancer
model in mice) of the combination of pan-HDAC inhibitor vorinostat
(SAHA) with bortezomib (42).
Furthermore, vorinostat and bortezomib inhibited tumor growth in a
prostate tumor model in mice (43).
Of note, there have been numerous clinical trials to improve the
treatment of myeloma (and also lymphoma), in which bortezomib is
approved as a drug, by combining HDAC inhibitors vorinostat or
panobinostat with different proteasome inhibitors (25,44,45).
Recently, the synergistic activity of HDAC inhibitor trichostatin
(TSA) and bortezomib against taxan-resistant ovarian cancer cell
lines has been reported (46).
Since scriptaid has been found less toxic than TSA, the former
seems to be more suitable for further in vivo investigation
of combination protocols.
In conclusion, our data suggest that the use of
scriptaid may enhance the effectiveness of conventional
chemotherapy of ovarian cancer and that the new combination:
scriptaid + bortezomib is worth considering as a treatment option
for heavily pretreated patients. This combination may be favorable,
by analogy to olaparib (35), in a
selected group of patients, for example in protocols of
intraperitoneal administration in women with recurrent disease.
Encouraging results of a recent phase I trial of intraperitoneal
treatment of bortezomib in combination with carboplatin in patients
with persistent/recurrent ovarian cancer argue for this assumption
(47).
Acknowledgements
The authors are grateful to Drs Małgorzata Firczuk
and Małgorzata Wańczyk for providing various reagents.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Giornelli GH: Management of relapsed
ovarian cancer: A review. Springerplus. 5:11972016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bast RC Jr, Hennessy B and Mills GB: The
biology of ovarian cancer: New opportunities for translation. Nat
Rev Cancer. 9:415–428. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dong A, Lu Y and Lu B: Genomic/epigenomic
alterations in ovarian carcinoma: Translational insight into
clinical practice. J Cancer. 7:1441–1451. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Grunewald T and Ledermann JA: Targeted
therapies for ovarian cancer. Best Pract Res Clin Obstet Gynaecol.
41:139–152. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Smith HJ, Straughn JM, Buchsbaum DJ and
Arend RC: Epigenetic therapy for the treatment of epithelial
ovarian cancer: A clinical review. Gynecol Oncol Rep. 20:81–86.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Eckschlager T, Plch J, Stiborova M and
Hrabeta J: Histone deacety-lase inhibitors as anticancer drugs. Int
J Mol Sci. 18:E14142017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Manal M, Chandrasekar MJ, Gomathi Priya J
and Nanjan MJ: Inhibitors of histone deacetylase as antitumor
agents: A critical review. Bioorg Chem. 67:18–42. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mottamal M, Zheng S, Huang TL and Wang G:
Histone deacetylase inhibitors in clinical studies as templates for
new anticancer agents. Molecules. 20:3898–3941. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ropero S and Esteller M: The role of
histone deacetylases (HDACs) in human cancer. Mol Oncol. 1:19–25.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Budman DR, Tai J, Calabro A and John V:
The histone deacetylase inhibitor panobinostat demonstrates marked
synergy with conventional chemotherapeutic agents in human ovarian
cancer cell lines. Invest New Drugs. 29:1224–1229. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chao H, Wang L, Hao J, Ni J, Chang L,
Graham PH, Kearsley JH and Li Y: Low dose histone deacetylase
inhibitor, LBH589, potentiates anticancer effect of docetaxel in
epithelial ovarian cancer via PI3K/Akt pathway in vitro. Cancer
Lett. 329:17–26. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cooper AL, Greenberg VL, Lancaster PS, van
Nagell JR Jr, Zimmer SG and Modesitt SC: In vitro and in vivo
histone deacetylase inhibitor therapy with suberoylanilide
hydroxamic acid (SAHA) and paclitaxel in ovarian cancer. Gynecol
Oncol. 104:596–601. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Konstantinopoulos PA, Wilson AJ, Saskowski
J, Wass E and Khabele D: Suberoylanilide hydroxamic acid (SAHA)
enhances olaparib activity by targeting homologous recombination
DNA repair in ovarian cancer. Gynecol Oncol. 133:599–606. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cassier PA, Floquet A, Penel N, Derbel O,
Bui N'guyen B, Guastalla JP, Pissaloux D, Treilleux I, Saba CE,
Blay JY, et al: The histone deacetylase inhibitor panobinostat is
active in patients with advanced pretreated ovarian sex-cord
tumors. Ann Oncol. 25:1074–1075. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Matulonis U, Berlin S, Lee H, Whalen C,
Obermayer E, Penson R, Liu J, Campos S, Krasner C and Horowitz N:
Phase I study of combination of vorinostat, carboplatin, and
gemcitabine in women with recurrent, platinum-sensitive epithelial
ovarian, fallopian tube, or peritoneal cancer. Cancer Chemother
Pharmacol. 76:417–423. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Modesitt SC, Sill M, Hoffman JS and Bender
DP; Gynecologic Oncology Group, : A phase II study of vorinostat in
the treatment of persistent or recurrent epithelial ovarian or
primary peritoneal carcinoma: A Gynecologic Oncology Group study.
Gynecol Oncol. 109:182–186. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Giacinti L, Giacinti C, Gabellini C,
Rizzuto E, Lopez M and Giordano A: Scriptaid effects on breast
cancer cell lines. J Cell Physiol. 227:3426–3433. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Janaki Ramaiah M, Naushad SM, Lavanya A,
Srinivas C, Anjana Devi T, Sampathkumar S, Dharan DB and Bhadra MP:
Scriptaid cause histone deacetylase inhibition and cell cycle
arrest in HeLa cancer cells: A study on structural and functional
aspects. Gene. 627:379–386. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sharma V, Koul N, Joseph C, Dixit D, Ghosh
S and Sen E: HDAC inhibitor, scriptaid, induces glioma cell
apoptosis through JNK activation and inhibits telomerase activity.
J Cell Mol Med. 14:2151–2161. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Takai N, Ueda T, Nishida M, Nasu K and
Narahara H: A novel histone deacetylase inhibitor, Scriptaid,
induces growth inhibition, cell cycle arrest and apoptosis in human
endometrial cancer and ovarian cancer cells. Int J Mol Med.
17:323–329. 2006.PubMed/NCBI
|
|
22
|
Berghauser Pont LM, Kleijn A, Kloezeman
JJ, van den Bossche W, Kaufmann JK, de Vrij J, Leenstra S, Dirven
CM and Lamfers ML: The HDAC inhibitors scriptaid and LBH589
combined with the oncolytic virus Delta24-RGD exert enhanced
anti-tumor efficacy in patient-derived glioblastoma cells. PLoS
One. 10:e01270582015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Manasanch EE and Orlowski RZ: Proteasome
inhibitors in cancer therapy. Nat Rev Clin Oncol. 14:417–433. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Minami J, Suzuki R, Mazitschek R, Gorgun
G, Ghosh B, Cirstea D, Hu Y, Mimura N, Ohguchi H, Cottini F, et al:
Histone deacetylase 3 as a novel therapeutic target in multiple
myeloma. Leukemia. 28:680–689. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dimopoulos M, Siegel DS, Lonial S, Qi J,
Hajek R, Facon T, Rosinol L, Williams C, Blacklock H, Goldschmidt
H, et al: Vorinostat or placebo in combination with bortezomib in
patients with multiple myeloma (VANTAGE 088): A multicentre,
randomised, double-blind study. Lancet Oncol. 14:1129–1140. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Karthik S, Sankar R, Varunkumar K and
Ravikumar V: Romidepsin induces cell cycle arrest, apoptosis,
histone hyperacetylation and reduces matrix metalloproteinases 2
and 9 expression in bortezomib sensitized non-small cell lung
cancer cells. Biomed Pharmacother. 68:327–334. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma YY, Lin H, Moh JS, Chen KD, Wang IW, Ou
YC, You YS and Lung CC: Low-dose LBH589 increases the sensitivity
of cisplatin to cisplatin-resistant ovarian cancer cells. Taiwan J
Obstet Gynecol. 50:165–171. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ong PS, Wang XQ, Lin HS, Chan SY and Ho
PC: Synergistic effects of suberoylanilide hydroxamic acid combined
with cisplatin causing cell cycle arrest independent apoptosis in
platinum-resistant ovarian cancer cells. Int J Oncol. 40:1705–1713.
2012.PubMed/NCBI
|
|
29
|
Pradhan S, Mahajan D, Kaur P, Pandey N,
Sharma C and Srivastava T: Scriptaid overcomes hypoxia-induced
cisplatin resistance in both wild-type and mutant p53 lung cancer
cells. Oncotarget. 7:71841–71855. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chou TC and Talalay P: Quantitative
analysis of dose-effect relationships: The combined effects of
multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 22:27–55.
1984. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chou TC: Theoretical basis, experimental
design, and computerized simulation of synergism and antagonism in
drug combination studies. Pharmacol Rev. 58:621–681. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Spratlin JL, Pitts TM, Kulikowski GN,
Morelli MP, Tentler JJ, Serkova NJ and Eckhardt SG: Synergistic
activity of histone deacetylase and proteasome inhibition against
pancreatic and hepatocellular cancer cell lines. Anticancer Res.
31:1093–1103. 2011.PubMed/NCBI
|
|
33
|
Zhang H, Dong L, Chen Q, Kong L, Meng B,
Wang H, Fu K, Wang X, Pan-Hammarström Q, Wang P, et al: Synergistic
antitumor effect of histone deacetylase inhibitor and Doxorubicin
in peripheral T-cell lymphoma. Leuk Res. 56:29–35. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jessmon P, Boulanger T, Zhou W and
Patwardhan P: Epidemiology and treatment patterns of epithelial
ovarian cancer. Expert Rev Anticancer Ther. 17:427–437. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Meehan RS and Chen AP: New treatment
option for ovarian cancer: PARP inhibitors. Gynecol Oncol Res
Pract. 3:32016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Pitts TM, Morrow M, Kaufman SA, Tentler JJ
and Eckhardt SG: Vorinostat and bortezomib exert synergistic
antiproliferative and proapoptotic effects in colon cancer cell
models. Mol Cancer Ther. 8:342–349. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Modesitt SC and Parsons SJ: In vitro and
in vivo histone deacetylase inhibitor therapy with vorinostat and
paclitaxel in ovarian cancer models: Does timing matter? Gynecol
Oncol. 119:351–357. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Grassadonia A, Cioffi P, Simiele F, Iezzi
L, Zilli M and Natoli C: Role of hydroxamate-based histone
deacetylase inhibitors (Hb-HDACIs) in the treatment of solid
malignancies. Cancers (Basel). 5:919–942. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Su GH, Sohn TA, Ryu B and Kern SE: A novel
histone deacetylase inhibitor identified by high-throughput
transcriptional screening of a compound library. Cancer Res.
60:3137–3142. 2000.PubMed/NCBI
|
|
40
|
Keen JC, Yan L, Mack KM, Pettit C, Smith
D, Sharma D and Davidson NE: A novel histone deacetylase inhibitor,
scriptaid, enhances expression of functional estrogen receptor
alpha (ER) in ER negative human breast cancer cells in combination
with 5-aza 2′-deoxycytidine. Breast Cancer Res Treat. 81:177–186.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Abaza MS, Bahman AM, Al-Attiyah RJ and
Kollamparambil AM: Synergistic induction of apoptosis and
chemosensitization of human colorectal cancer cells by histone
deacetylase inhibitor, scriptaid, and proteasome inhibitors:
Potential mechanisms of action. Tumour Biol. 33:1951–1972. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sato A and Asano T, Ito K, Sumitomo M and
Asano T: Suberoylanilide hydroxamic acid (SAHA) combined with
bortezomib inhibits renal cancer growth by enhancing histone
acetylation and protein ubiquitination synergistically. BJU Int.
109:1258–1268. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sato A and Asano T, Ito K and Asano T:
Vorinostat and bortezomib synergistically cause ubiquitinated
protein accumulation in prostate cancer cells. J Urol.
188:2410–2418. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chhabra S: Novel proteasome inhibitors and
histone deacetylase inhibitors: Progress in myeloma therapeutics.
Pharmaceuticals (Basel). 10:402017. View Article : Google Scholar :
|
|
45
|
Tan D, Diong CP, Loh Y and Goh YT: Histone
deacetylase (HDAC) inhibitors when combined with a proteasome
inhibitor are safe and effective in patients with extranodal
natural killer/T-cell lymphoma (ENKTL). Ann Oncol. 27:1811–1812.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Jin X, Fang Y, Hu Y, Chen J, Liu W, Chen
G, Gong M, Wu P, Zhu T, Wang S, et al: Synergistic activity of the
histone deacetylase inhibitor trichostatin A and the proteasome
inhibitor PS-341 against taxane-resistant ovarian cancer cell
lines. Oncol Lett. 13:4619–4626. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jandial DA, Brady WE, Howell SB, Lankes
HA, Schilder RJ, Beumer JH, Christner SM, Strychor S, Powell MA,
Hagemann AR, et al: A phase I pharmacokinetic study of
intraperitoneal bortezomib and carboplatin in patients with
persistent or recurrent ovarian cancer: An NRG Oncology/Gynecologic
Oncology Group study. Gynecol Oncol. 145:236–242. 2017. View Article : Google Scholar : PubMed/NCBI
|