Introduction
Osteosarcoma (OS) is the most common malignant bone
tumor in children and adolescents (1). Although the 5-year survival rate of
patients with OS is relatively favorable, at ~70%, OS is prone to
metastasis, and 15–20% of patients already have metastases at the
time of diagnosis (2,3). However, the 5-year survival rate of
patients with metastatic and recurrent OS is <30%, and treatment
strategies remain unsatisfactory (3). As innovative molecular targeted
therapies have not yet been developed, there are no effective
treatments for patients with OS, although extensive surgical
resection and chemotherapy are often used. To overcome this serious
issue, the identification of new therapeutic targets in OS and the
development of more effective treatment strategies are urgently
required.
p300 plays a vital role in the regulation of gene
expression through the chromatin remodeling mediated by histone
acetylation, and its expression level has been shown to be
associated with a poor prognosis of the patients with multiple
solid tumors (4–6). Curcumin, an active natural component
of turmeric extracted from the rhizome of Curcuma longa, has
long been used in Asian cuisine and traditional herbal medicine,
particularly in India (7). It has
been well documented that curcumin has anti-inflammatory and
antioxidant effects, as well as antitumor activity, targeting
distinct gene products dependent on tumor type (7,8).
Several lines of evidence suggest that curcumin inhibits p300
(9–11).
To further improve its antitumor potential, the
curcumin derivatives pentagamavunon-1 (PGV-1) (12) and chemoprevention curcumin
analog-1.1 (13,14) were created. Recently, Lestari et
al (15) reported that PGV-1
inhibits the proliferation of tumor cells derived from leukemia,
breast adenocarcinoma, cervical, uterine and pancreatic cancers at
a lower concentration than its parent curcumin through the
induction of M-phase cell cycle arrest, cellular senescence and
apoptosis, which are associated with the production of reactive
oxygen species (ROS). Similar to PGV-1, CCA-1.1 treatment resulted
in the accumulation of G2/M-phase cells, induction of cellular
senescence, and apoptosis in colon cancer cells, in association
with the generation of ROS (16).
Although a large body of evidence indicates that curcumin is also
effective in patients with OS (17–19),
the potential effects of PGV-1 and CCA-1.1 on OS have not yet been
validated.
In the present study, focus was addressed on OS, and
it was investigated whether PGV-1 and CCA-1.1 could suppress OS
through the inhibition of histone acetyltransferase (HAT) activity
of p300. The present results in the chicken egg chorioallantoic
membrane (CAM) model demonstrated that PGV-1 and CCA-1.1 inhibited
p300-HAT activity and thereby exerted a stronger suppressive effect
on OS than parental curcumin.
Materials and methods
Cell culture, p300 knockdown, green
fluorescent protein (GFP) expression, drug treatment
Human OS-derived U2OS, MG-63 and Saos-2 cells were
obtained from RIKEN BioResource Research Center and maintained in
DMEM (Nacalai Tesque, Inc.) supplemented with 10% heat-inactivated
fetal bovine serum (FBS; Serana) and 100 U/ml
penicillin-streptomycin (Nacalai Tesque, Inc.). The cells were
cultured in 5% CO2 at 37°C in a humidified atmosphere
(20). To transiently knock down
endogenous p300, OS cells were seeded in 6-well plates (50%
confluent at the time of transfection) and transfected with 10
nmol/l p300-specific small interfering RNAs (siRNAs; cat. no.
sc-29431; Santa Cruz Biotechnology; the sequences have not been
made public by the supplier) or control siRNA (cat. no. sc-37007;
Santa Cruz Biotechnology, Inc.; the sequences have not been made
public by the supplier.) using Opti-MEM (Thermo Fisher Scientific,
Inc.) and Lipofectamine RNAiMAX transfection reagent (Thermo Fisher
Scientific, Inc.) at room temperature for 20 min, according to the
manufacturer's protocol. The transfected cells were cultured for
additional 72 h in a humidified atmosphere with 5% CO2.
To measure the proliferation rate of OS cells after siRNA
transfection, the number of viable cells was determined using the
trypan blue exclusion test (21).
To establish MG-63 cells overexpressing GFP
(GFP-MG-63), MG-63 cells were infected with copGFP lentiviral
particles (cat. no. sc-108084; Santa Cruz Biotechnology, Inc.). A
total of 24 h after infection, the infected cells were cultured in
a medium containing 1 µg/ml puromycin (cat. no. sc-108071; Santa
Cruz Biotechnology, Inc.).
Curcumin and its analogs PGV-1 (MedChemExpress)
(12) and CCA-1.1 (13,14),
and another p300 HAT inhibitor, L002 (MilliporeSigma) (22) were dissolved in dimethyl sulfoxide
(DMSO; Nacalai Tesque, Inc.). OS cells were then cultured in medium
containing these inhibitors. A total of 96 h after treatment, cell
viability was measured using the Cell Count Reagent SF (Nacalai
Tesque, Inc.). The absorbance at 450 nm was measured using a
Multiskan Sky microplate spectrophotometer (Thermo Fisher
Scientific, Inc.).
WST-8 assay
To check the cellular viability, U2OS, MG63 and
SAOS-2 cells were seeded at a density of 0.5×104
cells/well on 96-well plate. After 24 h of incubation, L002,
Curcumin, PGV-1 or CCA-1.1 was added to the cell culture. After
treatment for 96 h, cells were incubated with 10 µl of Cell Count
Reagent SF (Nacalai Tesque, Inc.) for at at 37°C for 2 h and then
absorbance at 450 nm wavelength was detected using Multiskan
SkyHigh plate reader (Thermo Fisher Scientific, Inc.).
Dose-response curves were drawn and half maximal inhibitory drug
concentration (IC50) values were calculated based on
log-logistic model by R studio software (R-tools technology).
Kaplan-Meier curve
Kaplan-Meier curve analysis was performed using the
following database and dataset: R2 database; Genomics Analysis and
Visualization Platform; http://r2.amc.nl.
Data set: Mixed Osteosarcoma (Mesenchymal)-
Kuijjer-127-vst-ilmnhwg6v2 (Genome-wide gene expression analysis of
high-grade osteosarcoma); Gene Expression Omnibus ID: GSE42352.
Immunoblotting
OS cells were lysed in cell lysis buffer (cat. no.
9803; Cell Signaling Technology, Inc.) and whole cell lysates were
prepared and their protein concentration was measured by using
bicinchoninic acid (BCA) protein assay kit (Thermo Fisher
Scientific, Inc.). A total of 10 µg of protein were loaded per lane
and separated by SDS-PAGE (5–20% polyacrylamide gel; FUJIFILM Wako
Pure Chemical Corporation), then transferred onto PVDF membranes
(Immobilon-P; MilliporeSigma). The membranes were then blocked with
Blocking One (Nacalai Tesque, Inc.) overnight at 4°C as previously
described (23). The primary and
secondary antibodies used in the present study were as follows:
anti-p300 (cat. nos. sc-585 and sc-48343), anti-beta-actin (cat.
no. sc-47778; both from Santa Cruz Biotechnology, Inc.),
anti-phospho-histone H3 at Ser-10 (FUJIFILM Wako Pure Chemical
Corporation), anti-acetyl-histone H3 at Lys-27 (cat. no. 8173),
anti-histone H3 (cat. no. 4499), anti-poly (ADP-ribose) polymerase
1 (PARP1; cat. no. 9542), anti-cleaved-caspase-3 (cat. no. 9961),
anti-phospho-STAT3 at Tyr-705 (cat. no. 4113), anti-STAT3 (cat. no.
4904), anti-PCAF (cat. no. 3378) and anti-CBP (cat. no. 7389; all
from Cell Signaling Technology, Inc.). Horseradish peroxidase
(HRP)-conjugated anti-rabbit (cat. no. 7074) and anti-mouse (cat.
no. 7076) secondary antibodies were purchased from Cell Signaling
Technology, Inc. The membranes were incubated with primary
antibodies (1:1,000) at room temperature for 2 h and then with
secondary antibodies (1:1,500) at room temperature for 1 h. The
membranes were treated with Chemi-Lumi One (Nacalai Tesque, Inc.)
and signals were detected using a LAS-4000 Image Analyzer (GE
Healthcare).
Apoptosis detection
OS cells were harvested using trypsin (Thermo Fisher
Scientific, Inc.) and washed with phosphate-buffered saline (PBS)
(Takara Bio, Inc.). Apoptotic cells were labeled with ApoAlert™
Annexin V-FITC Apoptosis Kit (Takara Bio, Inc.) and then detected
by fluorescent microscopy. Stained images were analyzed using a
Leica TCS SP8 confocal microscope (Leica Microsystems GmbH).
Alternatively, the cells were labeled with an APC Annexin V
Apoptosis Detection Kit (BioLegend, Inc.) containing 7-AAD
(BioLegend, Inc.) according to the manufacturer's instructions. The
labeled cells were analyzed by fluorescence activated cell sorting
(FACS; BD FACSVerse; Becton, Dickinson and Company). The data
obtained were analyzed with FlowJo 10.9.0 (Becton, Dickinson and
Company). Annexin V-positive and propidium iodide (PI)-positive
cells were considered apoptotic.
Molecular docking simulation
Molecular docking was conducted using Molecular
Operating Environment software (Chemical Computing Group) to
estimate the binding affinities of the curcumin analogs toward
p300, PCAF and CBP. The well-known p300 inhibitor, L002, was used
as a positive control. Default settings for the molecular docking
protocols were applied unless specific factors required
customization. Representative crystal structures of p300 [Protein
Data Bank (PDB) ID: 4PZS], PCAF (PDB ID: 4NSQ), and CBP (PDB ID:
5U7G) were obtained from PDB (https://www.rcsb.org/). An acetyl-CoA-binding pocket
was employed as the target domain for molecular docking simulation.
The molecular docking screening system requires a flexible ligand
and a rigid receptor. Potential binding to each compound was
estimated by collecting the lowest docking score.
HAT assay
The inhibitory effect of curcumin and its analogs on
p300-HAT activity was examined using a HAT activity assay kit (Enzo
Life Sciences, Inc.) according to the manufacturer's instructions.
Briefly, 3 µg of recombinant human p300 catalytic domain (Enzo Life
Sciences, Inc.) was added to each reaction mixture. A reaction
mixture without the p300 catalytic domain was used as a reference.
L002 was used as a positive control. The reaction mixture was
incubated at 37°C for 3 h, and then absorbance at 440 nm was
detected by a Multiskan Sky microplate spectrophotometer.
Chicken egg CAM assay
Fertilized white chicken eggs purchased from Japan
Layer were hatched in a bird incubator (Autoelex) at 37.5°C in 65%
of humidity with egg rotation each hour. After 10 days of
incubation (embryonic day 10), GFP-MG-63 cells were transplanted.
To this end, a square window was opened on the eggshell using a
diamond cutter without damaging the shell membrane. Then,
2×106 of cells suspended in 20 µl of culture medium were
placed at the Y-shaped blood vessel on the CAM surface using a
sterile Teflon ring (Tokyo Garasu Kikai, Co., Ltd.). Subsequently,
the window was covered with Tegaderm film and the cells were
returned to the incubator. On embryonic day 13, the CAM tumors were
treated with curcumin, PGV-1 or CCA-1.1 (0.43 mg/kg) dissolved in
10% DMSO in PBS. CAM tumors treated with an equivalent volume of
10% DMSO in PBS were used as negative controls. A total of 2 days
after drug treatment (embryonic day 15), the treated CAM tumors
were observed under a fluorescent stereomicroscope (Leica
Microsystems GmbH). For further evaluation, after removal of the
CAM tumors from eggs, their pictures were captured using The Color
CMOS Camera K5C under the stereomicroscope (Leica Microsystems
GmbH) and their largest and shortest diameters were measured using
ImageJ (version 1.54g; National Institutes of Health). Their volume
was calculated as L (largest diameter) × S (shortest diameter) × S
(shortest diameter) × 1/2. Their weight was measured using XSR 105
(Mettler Toledo). For immunohistochemical analysis, the tumors were
fixed in 4% paraformaldehyde (Nacalai Tesque, Inc.) at 4°C
overnight, and subjected to paraffin block preparation with a
CT-Pro20 Cell & Tissue Processor (Genostaff Co., Ltd.).
Hematoxylin and eosin staining
Sections, ~4 µm in thickness, were prepared from the
paraffin-embedded blocks. After deparaffinization, hematoxylin and
eosin (H&E) staining was performed using the Hematoxylin and
Eosin Stain Kit (cat. no. H-3502; Vector Laboratories, Inc.)
according to the manufacturer's instructions. The H&E-stained
images were obtained using a BZ-X710 microscope (Keyence
Corporation).
Immunohistochemistry
Sections with around 4 µm in thickness, were
prepared from the paraffin-embedded blocks. After deparaffinization
by Tissue Tech Tissue Clear Xylene substitute, the primary
antigen-binding sites were exposed using Liberate Antibody Binding
Solution (cat. no. 24310; Polysciences, Inc.) for 15 min at room
temperature. Immunohistochemistry was performed using ImmPRESS HRP
Horse Anti-mouse IgG PLUS Polymer Kit (cat. no. MP-7802; Vector
Laboratories, Inc.) according to the manufacturer's protocol.
Briefly, glass slides were incubated with blocking solution plus
normal horse serum (cat. no. MP-7802; Vector Laboratories, Inc.) at
room temperature for 10 min and with anti-phospho-STAT3 antibody at
Tyr-705 (cat. no. 4113; Cell Signaling Technology, Inc.) diluted at
1/100 in Can Get Signal Solution 1 (cat. no. NKB-101; Toyobo Life
Science). After incubation, HRP-conjugated anti-mouse IgG secondary
antibody (concentration adjusted by supplier) was added, and the
color was developed with DAB substrate. Finally, the slides were
counterstained with Hematoxylin QS (cat. no. H-3404; Vector
Laboratories, Inc.). Images were obtained using a BZ-X710
microscope (Keyence Corporation).
In situ hybridization
In situ hybridization was performed using an
ISH Reagent Kit (Genostaff Co., Ltd.) according to the
manufacturer's instructions. The indicated sections (6 µm in
thickness) were deparaffinized with G-Nox, rehydrated through the
ethanol series, and placed in PBS. The sections were then fixed
with 4% paraformaldehyde in PBS (Nacalai Tesque, Inc.) for 30 min
at 37°C. After fixation, the sections were extensively washed in
distilled water, placed in 0.2% HCl for 10 min at 37°C, and washed
in PBS followed by the treatment with 4 µg/ml of proteinase K
(FUJIFILM Wako Pure Chemical Corporation) in PBS for 10 min at
37°C. After washing in PBS, the sections were heat-treated in PBS
for 10 min at 95°C, immediately cooled down in PBS at room
temperature, and placed within a Coplin jar containing 1X G-Wash
(Genostaff Co., Ltd.), which was equal to 1X SSC. The concentration
of Human Cytoplasm Stain Probe (Genostaff Co., Ltd.) was adjusted
to 25 nM by using G-Hybo-L (Genostaff Co., Ltd.). The probe was
heat-treated for 15 min at 80°C, and then immediately cooled down
on ice. The tissue sections were hybridized with the probe for 16 h
at 40°C. After the hybridization, the sections were washed three
times in 50% of formamide in 2X G-Wash for 30 min at 40°C, followed
by five washes in TBST (0.1% Tween 20 in TBS) at room temperature.
After exposure to 1X G-Block (Genostaff Co., Ltd.) for 15 min at
room temperature, the sections were incubated with anti-DIG AP
conjugate (Roche Diagnostics) diluted 2,000-fold with G-Block
(diluted 1/50) in TBST for 1 h at room temperature. The sections
were washed twice with TBST and incubated with a solution composed
of 100 mM NaCl, 50 mM MgCl2, 0.1% Tween 20 and 100 mM
Tris-HCl (pH 9.5). Coloring reactions were performed using NBT/BCIP
solution (Merck KGaA). Sections were counterstained with
Kernechtrot Stain Solution (Muto) and mounted with G-Mount
(Genostaff Co., Ltd.) and Malinol (Muto Pure Chemicals Co.,Ltd.).
Images were captured using a NanoZoomer S210 digital slide scanner
(C13239-01; Hamamatsu Photonics) and NDP.view2 Plus viewing
software (U12388-02; Hamamatsu Photonics).
Statistical analysis
Each experiment was performed at least thrice. The
error bars represent standard deviations (SD) or standard errors
(SE). The statical analysis on chicken egg CAM assay (Fig. 7C) was performed using one-way ANOVA
and Tukey-Kramer methods. Two-sample unpaired Student's t-test was
applied to the comparison between two groups. P-values were
calculated using Excel software (v16.77.1; Microsoft
Corporation).
Ethics and study approval
All procedures and studies were conducted in
accordance with the Regulations on Animal Experimentation at Chiba
Cancer Center Research Institute, based on the International
Guiding Principles for Biomedical Research Involving Animals. All
CAM procedures in the present study were approved (approval no.
24-1) by the Chiba Cancer Center Research Institute Animal
Experimentation Committee. (Chiba, Japan).
Results
p300 is significantly associated with
a poor survival of patients with OS
Although curcumin, with its intrinsic inhibitory
activity against p300-HAT, has been shown to suppress OS (9–11), it
remains unclear whether p300 is the most effective HAT for
shortening the survival of patients with OS. To adequately address
this issue, three representative HATs, p300, CBP and PCAF, were
selected; and their possible involvement in the survival of
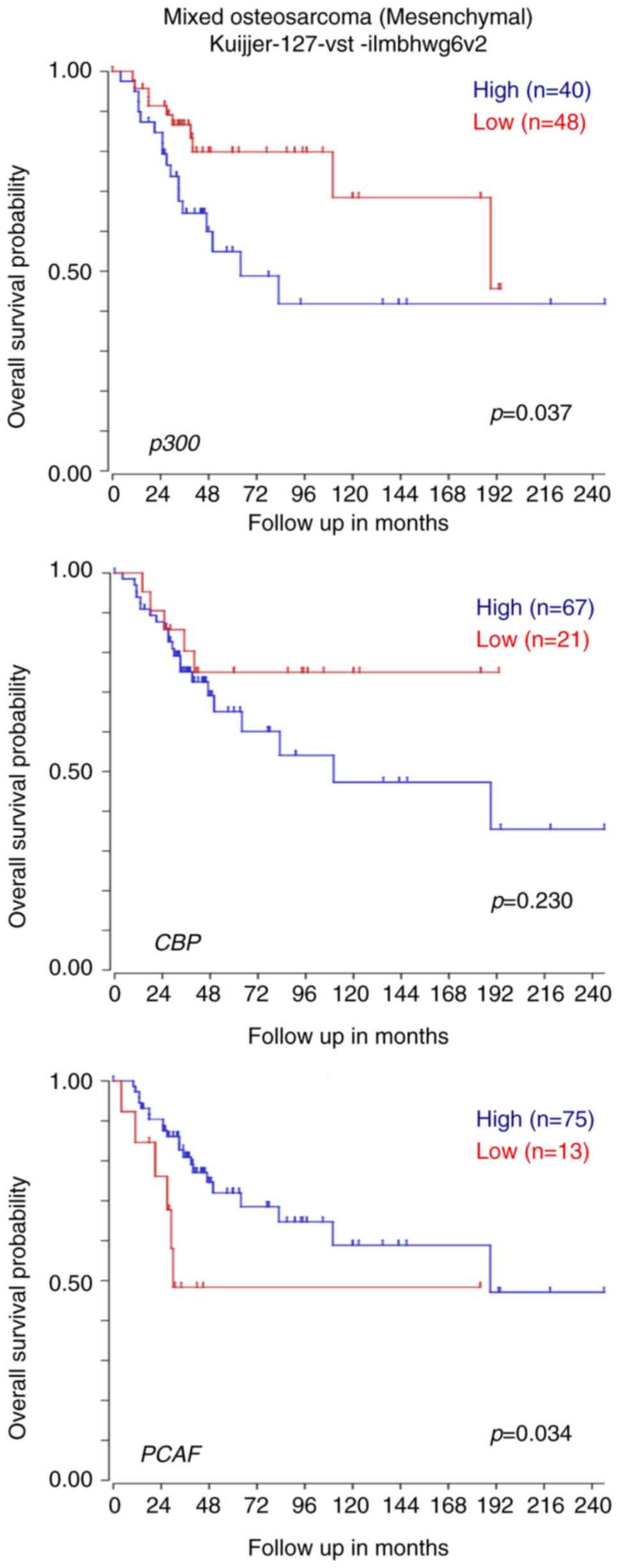
patients with OS was compared. As revealed in Fig. 1, the Kaplan-Meier curve based on a
public database (R2 database; Genomics Analysis and Visualization
Platform; http://r2.amc.nl) demonstrated that a
higher expression level of p300 but not of CBP or PCAF, was
significantly correlated with poor survival in patients with OS.
Since p300 was strongly implicated in the poor survival of patients
with OS, p300 was used for further experiments.
To examine the possible effects of p300 on OS, a
siRNA-mediated transient knockdown of p300 was performed in OS
cells, including p53-wild-type U2OS, p53-deficient MG-63 and
p53-deficient Saos-2 cells. As demonstrated in Fig. S1, siRNA against p300 reduced the
amount of endogenous p300 but not CBP and PCAF, indicating that
p300-specific knockdown was successfully performed. As expected,
p300 depletion caused a significant reduction in the proliferation
rate (Fig. S2). Under our
experimental conditions, U2OS and MG-63 cells underwent apoptosis,
as indicated by the induction of cleavage of PARP and caspase-3,
and an increase in the number of annexin-V/PI-positive cells
(Fig. S3). Similar results were
obtained in Saos-2 cells (data not shown). Knockdown of p300
induced apoptosis in p53-proficient U2OS and p53-deficient MG-63
cells, indicating that p300 depletion-mediated apoptosis is
regulated in a p53-independent manner. Thus, the results suggest
that p300 is required, at least in part, for OS cell survival and
that stronger inhibition of p300-HAT activity could suppress
OS.
Curcumin analogs bind to the HAT
domain of p300 and reduce its HAT activity
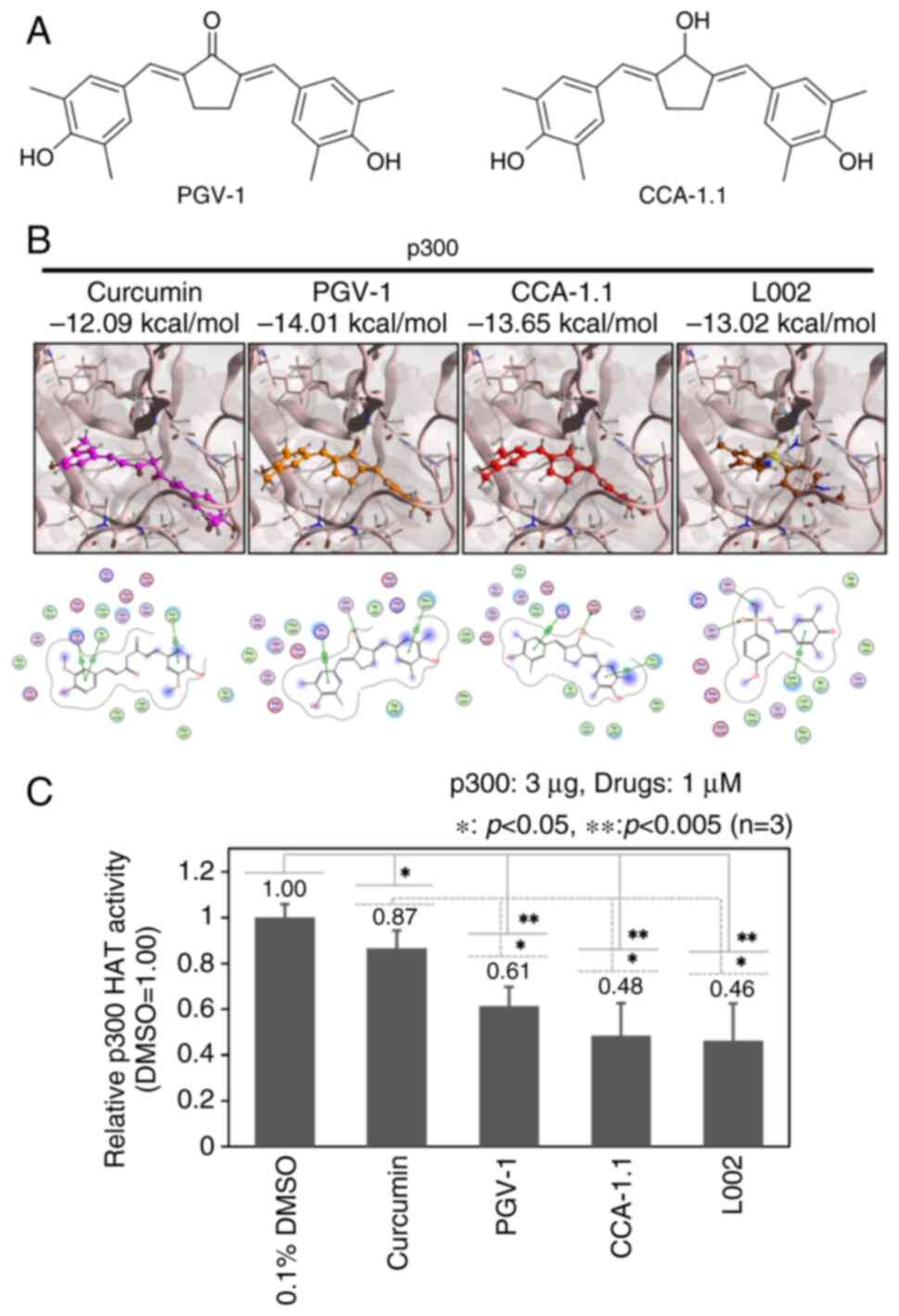
The curcumin analogs PGV-1 and CCA-1.1 were created
to enhance the native tumor-suppressive activity of curcumin
(Fig. 2A) (12,13).
Based on the results obtained from the docking simulation modeling
analysis, it was found that both PGV-1 (−14.01 kcal/mol) and
CCA-1.1 (−13.65 kcal/mol) bound more strongly to the HAT domain of
p300 than curcumin (−12.09 kcal/mol) and the general p300-HAT
inhibitor L002 (−13.02 kcal/mol) (Fig.
2B). In addition, PGV-1 (−15.64 kcal/mol) and CCA-1.1 (−15.93
kcal/mol), but not L002 (−12.28 kcal/mol), were predicted to
associate with the HAT domain of CBP more strongly than curcumin
(−13.95 kcal/mol) (Fig. S4A).
PGV-1 (−8.03 kcal/mol), CCA-1.1 (−8.06 kcal/mol) and L002 (−8.99
kcal/mol) were estimated to bind to the HAT domain of PCAF weaker
than curcumin (−9.21 kcal/mol) (Fig.
S4B). To evaluate the inhibitory activities of PGV-1, CCA-1.1,
curcumin and L002 on p300-HAT, in vitro HAT assays were
performed. As shown in Fig. 2C,
both PGV-1 and CCA-1.1, exhibited stronger inhibitory activities
against p300-HAT than curcumin. The inhibitory activities of PGV-1
and CCA-1.1 against p300-HAT were lower than those of L002 and
almost similar to those of L002. These results suggest that PGV-1
and CCA-1.1 might effectively suppress OS compared with curcumin
because of their higher inhibitory activity against p300.
PGV-1- and CCA-1.1-mediated inhibition
of OS cell viability
Since it has been identified that PGV-1 and CCA-1.1
have antitumor effects in breast cancer, pancreatic cancer and
glioblastoma multiforme (24–29),
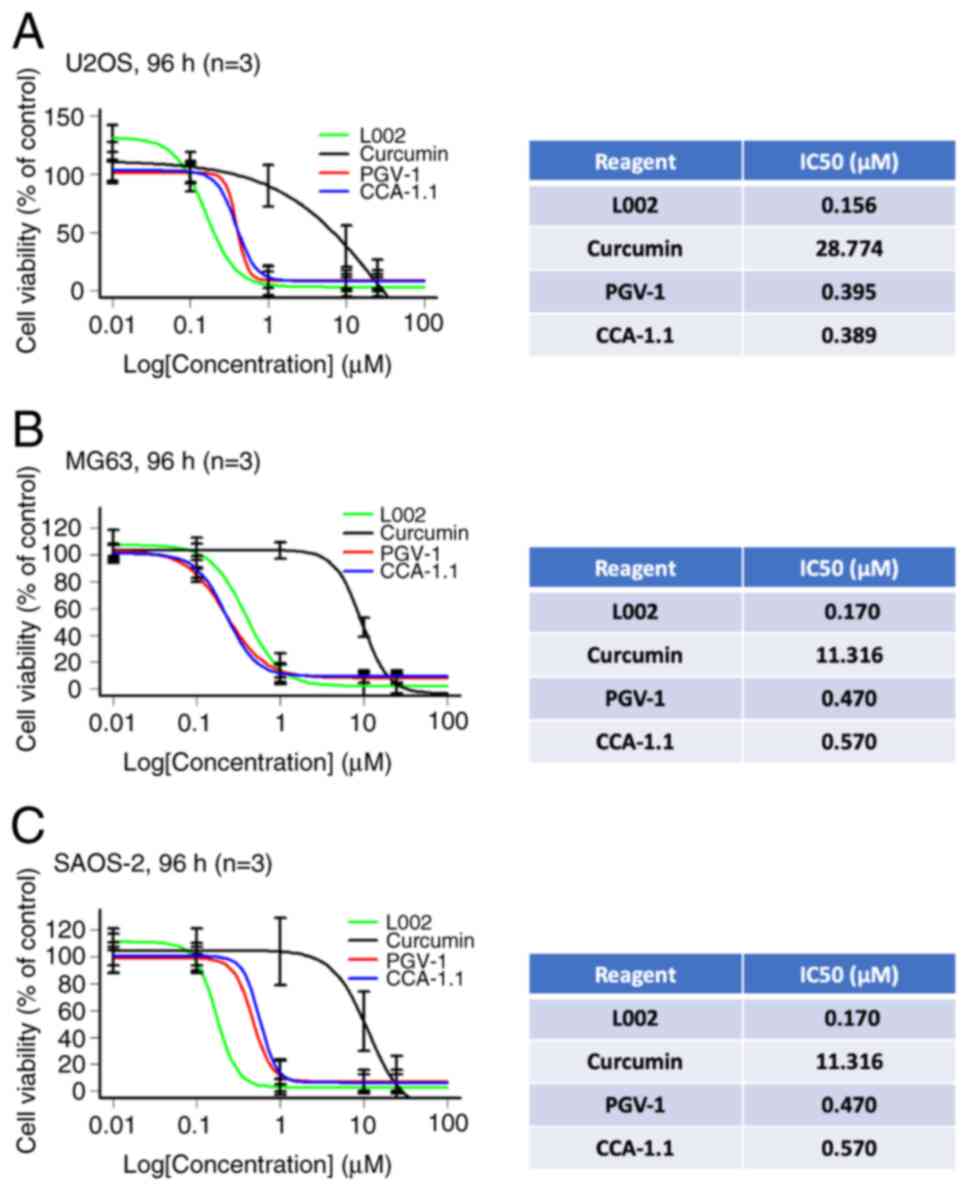
their antitumor effect was evaluated on OS. The cytotoxic effects
of curcumin, PGV-1, CCA-1.1 and L002 were compared on U2OS, MG-63
and Saos-2 cells. As shown in Fig.
3A-C, a remarkable decrease in cell viability was observed in
all cells treated with curcumin, PGV-1, CCA-1.1 or L002. The
IC50 values of these HAT inhibitors were determined
based on graphs showing the viability curves of the
inhibitor-treated cells relative to the cells exposed to DMSO.
Notably, PGV-1 and CCA-1.1 exhibited ~20-100-fold stronger
inhibitory activity against OS cell proliferation than curcumin.
Suppressive activities of PGV-1 and CCA-1.1 were almost similar to
those of L002. Collectively, these results indicate that the
reduction in the proliferation rate of OS cells due to PGV-1 and
CCA-1.1 is related to their inhibitory activity against
p300-HAT.
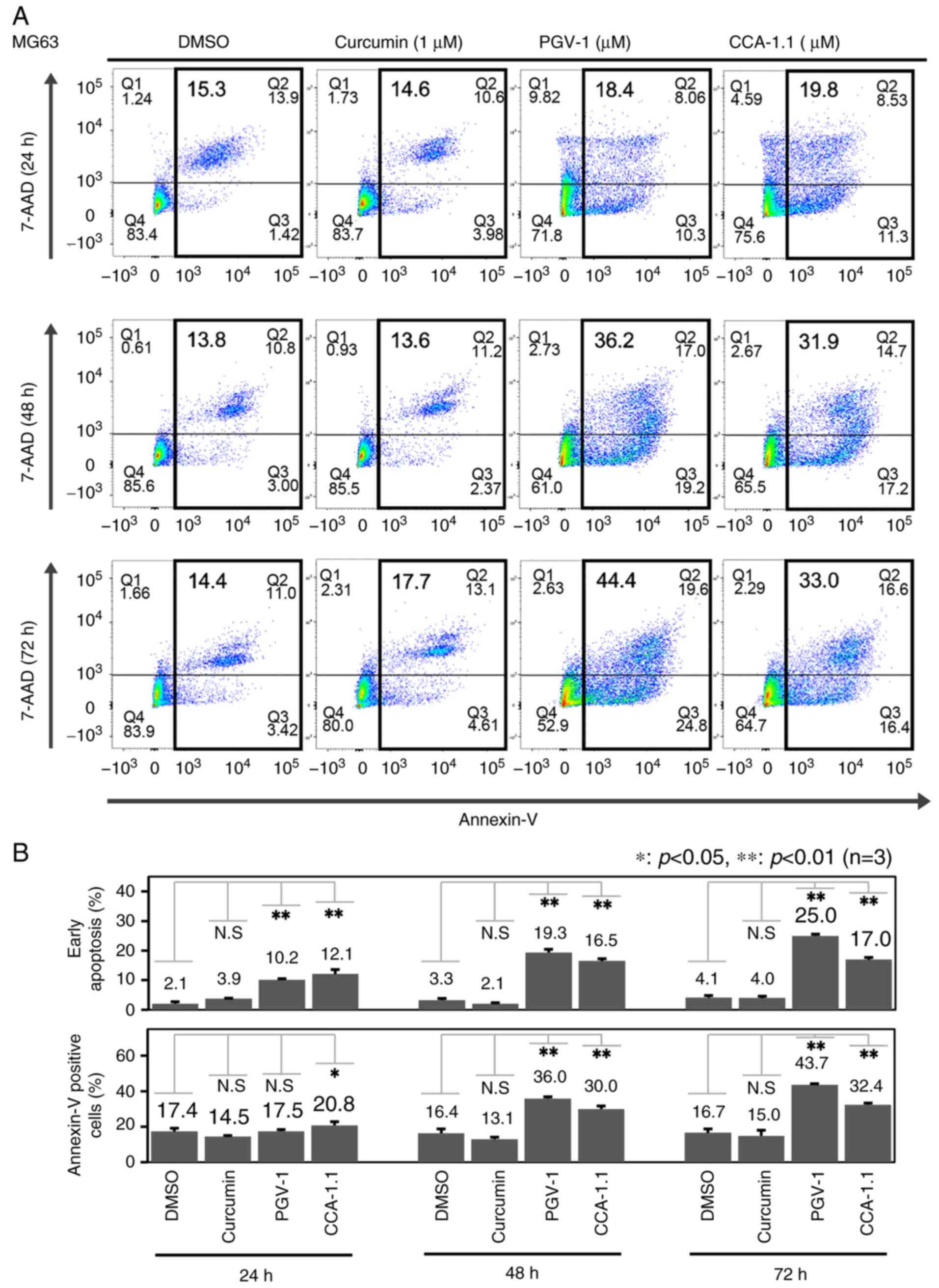
PGV-1 and CCA-1.1 stimulate OS cell
apoptosis
Our results prompted us to elucidate the molecular
mechanisms by which PGV-1 and CCA-1.1 slowed OS cell proliferation.
MG-63 cells were exposed to curcumin, PGV-1 or CCA-1.1, and
subjected to FACS analysis. As revealed in Fig. 4, the number of PI-positive and
annexin V-negative early apoptotic cells, as well as annexin
V-positive apoptotic cells, was obviously increased following PGV-1
or CCA-1.1 exposure in a time-dependent manner, whereas the
curcumin-mediated increase in the number of apoptotic cells was
small. Similar results were obtained in U2OS and Saos-2 cells (data
not shown). Thus, the aforementioned results suggested that PGV-1
and CCA-1.1 efficiently promote apoptosis in OS cells.
To further examine the mechanism of action,
whole-cell lysates were prepared from OS cells exposed to curcumin,
PGV-1 or CCA-1.1, and cell cycle- and apoptosis-related proteins
were analyzed. As demonstrated in Fig.
5, marked accumulation of phosphorylated histone H3 at Ser-10
was observed in OS cells treated with PGV-1 or CCA-1.1, indicating
that G2/M cell cycle arrest might occur following PGV-1 or CCA-1.1
exposure. By contrast, curcumin and L002 had no detectable effect
on the phosphorylation of histone H3 at Ser-10 in these OS cells.
Taken together, these results suggested that OS cells undergo
apoptosis, at least in part, through the induction of G2/M cell
cycle arrest in response to PGV-1 or CCA-1.1.
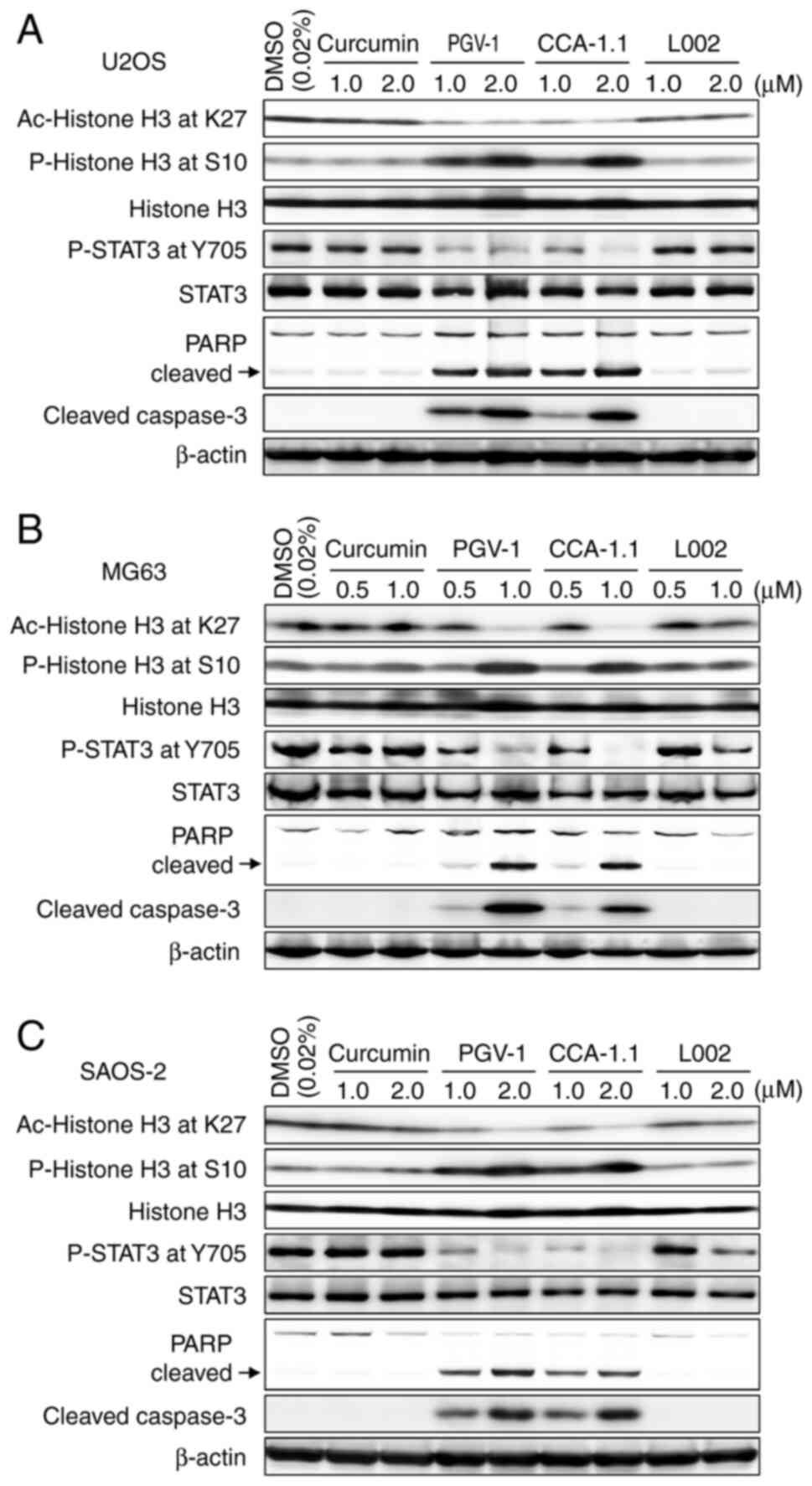 | Figure 5.PGV-1- and CCA-1.1-induced
osteosarcoma cell apoptosis accompanies a decrease in acetylated
histone H3 at Lys-27 and phosphorylated histone H3 at Tyr-705.
(A-C) Western blotting of (A) U2OS, (B) MG-63 and (C) Saos-2 cells
treated with the indicated concentrations of curcumin, PGV-1,
CCA-1.1 or with L002. A total of 24 h after treatment, whole cell
lysates were prepared and analyzed for acetylated histone H3 at
Lys-27, phospho-histone H3 at Ser-10, histone H3, phospho-STAT3 at
Tyr-705, STAT3, cleaved PARP and cleaved caspase-3 by western
blotting. DMSO was used as a negative control. PGV-1,
pentagamavunon-1; CCA-1.1, chemoprevention curcumin analog-1.1;
PARP, poly (ADP-ribose) polymerase. |
PGV-1- and CCA-1.1-mediated OS cell
apoptosis is accompanied by a reduction in the acetylated histone
H3 at Lys-27 and phosphorylated STAT3 at Tyr-705
Consistent with our in vitro HAT assay shown
in Fig. 2C, a significant reduction
in histone H3 acetylation at Lys-27 was observed in MG-63, U2OS and
Saos-2 cells following PGV-1 or CCA-1.1 treatment (Fig. 5). By contrast, there was negligible
downregulation of histone H3 acetylation at Lys-27 from L002.
Similarly, curcumin had a negligible effect on acetylated histone
H3 at Lys-27. It is worth noting that the phosphorylation level of
pro-oncogenic STAT3 at Tyr-705, whose increased amount is tightly
linked to the poor prognosis of patients with OS (30), was markedly downregulated in PGV-1-
or CCA-1.1-treated OS cells. By contrast, curcumin had no
detectable effect on the phosphorylation level of STAT3 at Tyr-705.
Thus, in addition to p300, STAT3 might be implicated in PGV-1- or
CCA-1.1-mediated induction of OS cell apoptosis.
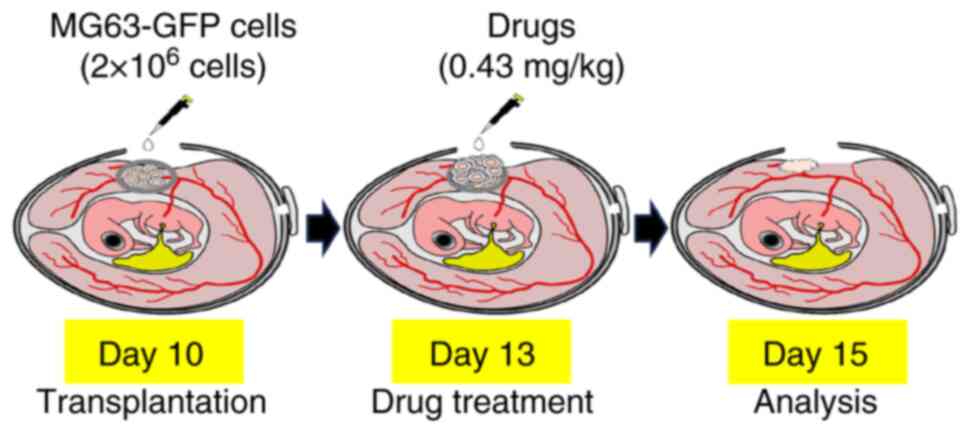
PGV-1 and CCA-1.1 attenuate the growth
of MG-63 cell-derived tumor developed on chicken CAM
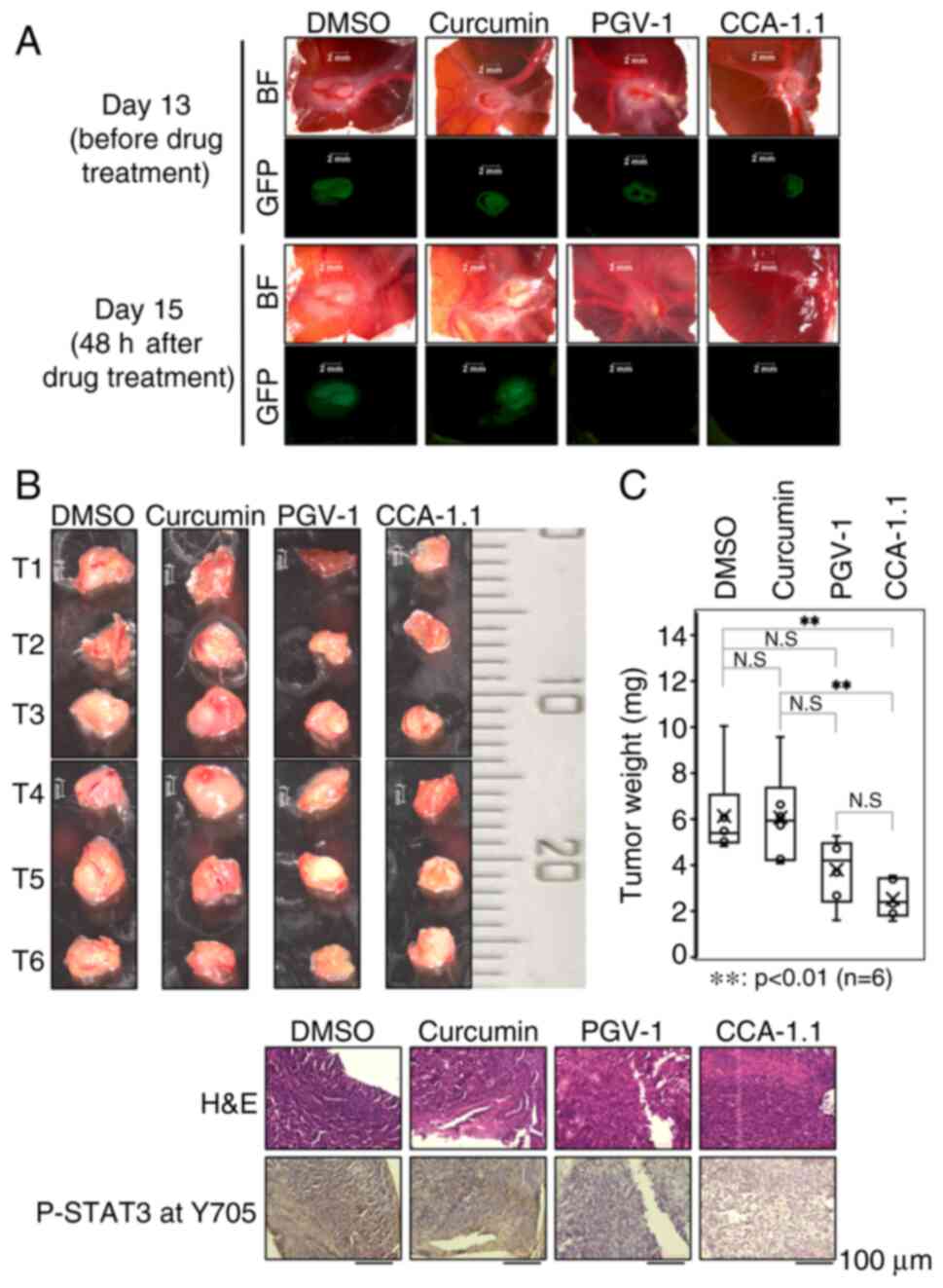
Finally, the chicken CAM system was used to evaluate
the possible antitumor effects of PGV-1 and CCA-1.1 on OS in
vivo (Fig. 6). For this
purpose, the GFP expression plasmid was transfected into MG-63
cells (GFP-MG-63), and the GFP-MG-63 cells were transplanted from
an eggshell window onto the CAM on embryonic day 10. On embryonic
day 13, GFP-MG-63 cell-derived tumors formed on the CAM (CAM
tumors) (Fig. 7A, upper panels)
were treated with DMSO, curcumin, PGV-1 or with CCA-1.1. A total of
2 days after treatment (embryonic day 15), the CAM tumors were
examined under a fluorescence stereomicroscope. Based on the signal
emitted from GFP, CAM tumors were easily distinguished from
original chicken tissues. As expected, in situ hybridization
of CAM tumors with a human-specific probe demonstrated that the
tumors that developed on the CAM largely consisted of human cells
(Fig. S5). Notably, H&E
counterstaining showed that the CAM tumors grew around the blood
vessels on the CAM. As demonstrated in the lower panels of Fig. 7A, the GFP signal disappeared in the
presence of PGV-1 or CCA-1.1, whereas GFP signal remained unchanged
in the presence of DMSO or curcumin. Based on these results, PGV-1-
or CCA-1.1-treated CAM tumors did not emit GFP signal, whereas GFP
signal remained unchanged in the CAM tumors exposed to DMSO or
curcumin. The largest diameters of the CAM tumors treated with
DMSO, curcumin, PGV-1 or CCA-1.1 were 4.49, 3.82, 3.97 and 3.2 mm,
respectively. The volumes of the CAM tumors exposed to DMSO,
curcumin, PGV-1 or CCA-1.1 were 26.4 mm3, 20.8
mm3, 9.51 mm3 and 8.84 mm3,
respectively. The extensive statistical analysis on these results
suggest that the growth of the CAM tumors treated with CCA-1.1 but
not with PGV-1 is efficiently attenuated as compared with curcumin
treated CAM tumors. Indeed, a significant reduction in the size and
weight of the CAM tumors was observed in response to PGV-1 or only
CCA-1.1 relative to curcumin-exposed CAM tumors (Fig. 7B and C; and Figs. S6 and S7). Moreover, CCA-1.1 but not curcumin,
decreased the amount of phosphorylated STAT3 at Tyr-705 in CAM
tumors (Fig. 7D). Collectively,
these results strongly suggest that CCA-1.1 has a stronger anti-OS
activity than curcumin, and thus might be a promising antitumor
drug against OS.
Discussion
In the present study, using a CAM model, it was
found for the first time, to the best of our knowledge, that the
curcumin derivatives PGV-1 and only CCA-1.1 attenuates OS cell
proliferation and growth more efficiently than curcumin. Although
an innovative molecular targeted therapy for patients with OS has
not yet been established, our current findings might provide
valuable clues for the development of a novel and effective
therapeutic strategy for patients with OS.
According to the present results, both PGV-1 and
CCA-1.1 slowed OS cell proliferation more strongly than curcumin
in vitro. The PGV-1- and CCA-1.1-mediated decrease in the
proliferation rate was accompanied by an increase in cleaved PARP,
cleaved caspase-3 and phosphorylated histone H3 at Ser-10. Thus, it
is suggestive that the PGV-1- and CCA-1.1-dependent reduction in
the OS cell proliferation rate might be due to the induction of
apoptosis and/or G2/M cell cycle arrest. In support of our results,
Kelly et al (31) reported
that the curcumin analog RL71 potentiates G2/M cell cycle arrest
and apoptosis in OS cells. In contrast to PGV-1 and CCA-1.1,
curcumin had a negligible effect on OS cell apoptosis under our
experimental conditions. Although it has been demonstrated that
curcumin efficiently inhibits OS cell proliferation in association
with the accumulation of cleaved PARP and cleaved caspase-3, the
concentration used was markedly higher than that used in the
present study (32). Thus, it is
likely that PGV-1 and CCA-1.1 attenuate OS cell proliferation more
effectively than curcumin in vitro, which may be at least in
part due to the induction of apoptosis.
Accumulating evidence suggests that curcumin has
antitumor activity against colorectal carcinoma, pancreatic cancer
and OS (33,34) Notably, Balasubramanyam et al
(9) found that curcumin
specifically inhibits p300-HAT activity. Consistent with these
observations, Marcu et al (35) demonstrated that curcumin prevents
the p300-dependent acetylation of histone H3, indicating that
curcumin-dependent inhibition of p300-HAT activity could
participate in its tumor-suppressive mechanism. The current results
showed that PGV-1 and CCA-1.1 bound to the p300-HAT domain more
strongly than curcumin, as estimated by docking simulation modeling
analysis. Indeed, their inhibitory effects on p300-HAT activity
were greater than that of curcumin alone. Additionally, PGV-1 and
CCA-1.1 displayed weaker affinities toward PCAF-HAT than curcumin.
Therefore, PGV-1 and CCA-1.1. may be more specific to p300-HAT than
curcumin. In accordance with these results, the amount of
acetylated histone H3 at Lys-27 was significantly decreased in OS
cells exposed to PGV-1 or CCA-1.1 as compared with curcumin. It has
been well established that p300-mediated acetylation of histone H3
at Lys-27 at its target gene promoter, enhancer, and super-enhancer
causes the activation of its target gene transcription (36–39).
To further improve the effectiveness of PGV-1 and CCA-1.1 on OS
cells, it is important to identify the p300-target gene(s)
implicated in PGV-1- and CCA-1.1-mediated attenuation of OS cell
proliferation.
Lestari et al (15) found that PGV-1 binds to several ROS
metabolic enzymes and increases intracellular ROS levels, thereby
reducing the proliferation rate of various tumor cells. Similarly,
Wulandari et al (16)
revealed that CCA-1.1 promotes G2/M cell cycle arrest and apoptosis
through ROS production in colon carcinoma cells, raising a
possibility that PGV-1- or CCA-1.1-induced G2/M cell cycle arrest
and apoptosis in OS cells may also depend on ROS generation. In
addition, Chen et al (40)
reported that the curcumin derivative, WZ26, potentiates G2/M cell
cycle arrest and apoptosis in cholangiocarcinoma cells through the
production of ROS. These findings strongly suggest that curcumin
derivative-induced ROS production triggers G2/M cell cycle arrest
and apoptosis in tumor cells. Several lines of evidence have
provided clues to clarify the molecular mechanisms underlying these
observations. For example, Li et al (41) demonstrated that ROS promotes tumor
cell apoptosis by downregulating phosphorylated STAT3. Alsamri
et al (42) reported that
ROS-dependent degradation of p300 triggers apoptosis in highly
invasive breast cancer cells. Consistent with these results, it was
found that PGV-1 and CCA-1.1 efficiently prohibit p300-HAT activity
and reduce STAT3 phosphorylation at Tyr-705 in OS cells. Thus, the
precise elucidation of the functional interplay between p300-HAT,
ROS, and STAT3 in response to PGV-1 or CCA-1.1 might contribute to
the development of more effective curcumin derivatives. However,
further studies should be required to validate these
hypotheses.
To evaluate whether PGV-1 and CCA-1.1 could suppress
OS growth in vivo, it was examined whether OS cells could
produce tumors after inoculation into immunodeficient mice.
Unfortunately, U2OS and MG-63 cells did not produce tumors after
transplantation (data not shown). Therefore, an alternative CAM
model was employed in the present study. The treatment with CCA-1.1
remarkably reduced CAM tumor volume, whereas curcumin and PVG-1
suppressed CAM tumor growth but to a lesser degree, suggesting that
CCA-1.1 has a more potent tumor-suppressive activity against OS
than curcumin and PGV-1. As shown in Fig. 7D, CCA-1.1, but not curcumin strongly
reduced decreased the amount of phosphorylated STAT3 at Tyr-705 in
CAM tumors in vivo, while the effect of PGV-1 was markedly
weaker than that of CCA1.1. It remains unknown why the suppressive
potential of PGV-1 against CAM tumors arising from OS cells could
be less than CCA-1.1. To address this issue, further study should
be required, which might provide a specific clue to produce more
effective curcumin derivatives against OS.
Because of the signal emitted from GFP-MG-63 cells,
it was easy to distinguish between CAM tumors and native chicken
tissues. Unlike the standard immunodeficient mouse model
accompanied by cost and efficiency constraints, the CAM model has
unique advantages, such as low cost, visibility, high
reproducibility and shorter experimental duration, which enable
tumor models that are difficult to produce to be established, and
to allow the screening of a large number of candidate antitumor
agents in a short time.
In conclusion, the present results strongly suggest
that curcumin derivatives, CCA-1.1 is promising candidates as
antitumor drug against OS patients and that the chicken CAM model
might be an alternative approach complementary to the
immunodeficient mouse model.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Mr Sohei Urakami
(S-POOL Co., Ltd.) for his understanding of the project and great
support in overcoming intractable cancers and sarcomas.
Funding
The present study was supported in part by a Grant-in-Aid from
the Japan Agency for Medical Research and Development (Basic
Science and Platform Technology Program for Innovative Biological
Medicine, grant no. 19am0101101j0003) and a Grant-in-Aid for
Scientific Research (JSPS KAKENHI, grant nos. 22K07168, 17H03597,
20K08199 and 20K07710).
Availability of data and materials
The data generated in the present study are included
in the figures and/or tables of this article.
Authors' contributions
YT and TM performed the experiments, analyzed the
data and wrote the manuscript. TW, RYU and UMZ contributed to data
collection. YT, TM, TW and YK confirm the authenticity of all the
raw data. EM synthesized curcumin derivatives. TO participated in
the discussion and interpretation of the data and helped write the
manuscript. YS participated in the discussion and interpretation of
data. YK initiated and designed the study, led the entire project
and approved the final submission. All authors read and approved
the final version of the manuscript.
Ethics approval and consent to
participate
All procedures and studies were conducted in
accordance with the Regulations on Animal Experimentation at Chiba
Cancer Center Research Institute, based on the International
Guiding Principles for Biomedical Research Involving Animals. All
CAM procedures in the present study were approved (approval no.
24-1) by the Chiba Cancer Center Research Institute Animal
Experimentation Committee (Chiba, Japan).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CAM
|
chorioallantoic membrane
|
|
CCA-1.1
|
chemoprevention curcumin
analog-1.1
|
|
FACS
|
fluorescence activated cell
sorting
|
|
FBS
|
fetal bovine serum
|
|
GFP
|
green fluorescent protein
|
|
HAT
|
histone acetyltransferase
|
|
H&E
|
hematoxylin and eosin
|
|
HRP
|
horseradish peroxidase
|
|
OS
|
osteosarcoma
|
|
PARP1
|
poly (ADP-ribose) polymerase 1
|
|
PBS
|
phosphate-buffered saline
|
|
PGV-1
|
pentagamavunon-1
|
|
PI
|
propidium iodide
|
|
ROS
|
reactive oxygen species
|
References
|
1
|
Longhi A, Errani C, De Paolis M, Mercuri M
and Bacci G: Primary bone osteosarcoma in the pediatric age: State
of the art. Cancer Treat Rev. 32:4234362006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
He H, Ni J and Huang J: Molecular
mechanisms of chemoresistance in osteosarcoma (Review). Oncol Lett.
7:135213622014. View Article : Google Scholar
|
|
3
|
Isakoff MS, Bielack SS, Meltzer P and
Gorlick R: Osteosarcoma: Current treatment and a collaborative
pathway to success. J Clin Oncol. 33:302930352015. View Article : Google Scholar
|
|
4
|
Santer FR, Höschele PP, Oh SJ, Erb HHH,
Bouchal J, Cavarretta IT, Parson W, Meyers DJ, Cole PA and Culig Z:
Inhibition of the acetyltransferases p300 and CBP reveals a
targetable function for p300 in the survival and invasion pathways
of prostate cancer cell lines. Mol Cancer Ther. 10:1644–1655. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hou X, Li Y, Luo RZ, Fu JH, He JH, Zhang
LJ and Yang HX: High expression of the transcriptional co-activator
p300 predicts poor survival in resectable non-small cell lung
cancers. Eur J Surg Oncol. 38:523–530. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang J, Wu M, Zheng D, Zhang H, Lv Y,
Zhang L, Tan HS, Zhou H, Lao YZ and Xu HX: Garcinol inhibits
esophageal cancer metastasis by suppressing the p300 and TGF-β1
signaling pathways. Acta Pharmacol Sin. 41:82–92. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang W, Li M, Wang L, Chen L and Goh BC:
Curcumin in cancer therapy: Exploring molecular mechanisms and
overcoming clinical challenges. Cancer Lett. 570:2163322023.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mundekkad D and Cho WC: Applications of
curcumin and its nanoforms in the treatment of cancer.
Pharmaceutics. 15:22232023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Balasubramanyam K, Varier RA, Altaf M,
Swaminathan V, Siddappa NB, Ranga U and Kundu TK: Curcumin, a novel
p300/CREB-binding protein-specific inhibitor of acetyltransferase,
represses the acetylation of histone/nonhistone proteins and
histone acetyltransferase-dependent chromatin transcription. J Biol
Chem. 279:51163–51171. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhu X, Li Q, Chang R, Yang D, Song Z, Guo
Q and Huang C: Curcumin alleviates neuropathic pain by inhibiting
p300/CBP histone acetyltransferase activity-regulated expression of
BDNF and cox-2 in a rat model. PLoS One. 9:e913032014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sunagawa Y, Funamoto M, Shimizu K, Shimizu
S, Sari N, Katanasaka Y, Miyazaki Y, Kakeya H, Hasegawa K and
Morimoto T: Curcumin, an inhibitor of p300-HAT activity, suppresses
the development of hypertension-induced left ventricular
hypertrophy with preserved ejection fraction in dahl rats.
Nutrients. 13:26082021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Meiyanto E, Putri DD, Susidarti RA,
Murwanti R, Sardjiman Fitriasari A, Husnaa U, Purnomo H and
Kawaichi M: Curcumin and its analogues (PGV-0 and PGV-1) enhance
sensitivity of resistant MCF-7 cells to doxorubicin through
inhibition of HER2 and NF-kB activation. Asian Pac J Cancer Prev.
15:179–184. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Novitasari D, Jenie RI, Kato JY and
Meiyanto E: The integrative bioinformatic analysis deciphers the
predicted molecular target gene and pathway from curcumin
derivative CCA-1.1 against triple-negative breast cancer (TNBC). J
Egypt Natl Canc Inst. 33:192021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Utomo RY, Wulandari F, Novitasari D,
Lestari B, Susidarti RA, Jenie RI, Kato JY, Sardjiman S and
Meiyanto E: Preparation and cytotoxic evaluation of PGV-1
derivative, CCA-1.1, as a new curcumin analog with
improved-physicochemical and pharmacological properties. Adv Pharm
Bull. 12:6036122022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lestari B, Nakamae I, Yoneda-Kato N,
Morimoto T, Kanaya S, Yokoyama T, Shionyu M, Shirai T, Meiyanto E
and Kato JY: Pentagamavunon-1 (PGV-1) inhibits ROS metabolic
enzymes and suppresses tumor cell growth by inducing M phase
(prometaphase) arrest and cell senescence. Sci Rep. 9:148672019.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wulandari F, Ikawati M, Widyarini S,
Kirihata M, Novitasari D, Kato JY and Meiyanto E:
Tumour-suppressive effects of curcumin analogs CCA-1.1 and
Pentagamavunone-1 in colon cancer: In vivo and in vitro studies. J
Adv Pharm Technol Res. 14:317–324. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu C, Wang M, Guo W, Sun W and Liu Y:
Curcumin in osteosarcoma therapy: Combining with immunotherapy,
chemotherapeutics, bone tissue engineering materials and potential
synergism with photodynamic therapy. Front Oncol. 11:6724902021.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zahedipour F, Bolourinezhad M, Teng Y and
Sahebkar A: The multifaceted therapeutic mechanisms of curcumin in
osteosarcoma: State-of-the-art. J Oncol. 2021:30068532021.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lu KH, Lu PW, Lin CW and Yang SF: Curcumin
in human osteosarcoma: From analogs to carriers. Drug Discov Today.
28:1034372023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Iwata S, Tatsumi Y, Yonemoto T, Araki A,
Itami M, Kamoda H, Tsukanishi T, Hagiwara Y, Kinoshita H, Ishii T,
et al: CDK4 overexpression is a predictive biomarker for resistance
to conventional chemotherapy in patients with osteosarcoma. Oncol
Rep. 46:1352021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Strober W: Trypan blue exclusion test of
cell viability. Curr Protoc Immunol Appendix 3: Appendix 3B.
2001.PubMed/NCBI
|
|
22
|
Yang H, Pinello CE, Luo J, Li D, Wang Y,
Zhao LY, Jahn SC, Saldanha SA, Chase P, Planck J, et al:
Small-molecule inhibitors of acetyltransferase p300 identified by
high-throughput screening are potent anticancer agents. Mol Cancer
Ther. 12:610–620. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tatsumi Y, Takano R, Islam MS, Yokochi T,
Itami M, Nakamura Y and Nakagawara A: BMCC1, which is an
interacting partner of BCL2, attenuates AKT activity, accompanied
by apoptosis. Cell Death Dis. 6:e16072015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Meiyanto E, Putri H, Arum Larasati Y, Yudi
Utomo R, Istighfari Jenie R, Ikawati M, Lestari B, Yoneda-Kato N,
Nakamae I, Kawaichi M and Kato JY: Anti-proliferative and
anti-metastatic potential of curcumin analogue, pentagamavunon-1
(PGV-1), toward highly metastatic breast cancer cells in
correlation with ROS generation. Adv Pharm Bull. 9:445–452. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Meiyanto E, Husnaa U, Kastian RF, Putri H,
Larasati YA, Khumaira A, Pamungkas DDP, Jenie RI, Kawaichi M,
Lestari B, et al: The target differences of anti-tumorigenesis
potential of curcumin and its analogues against HER-2 positive and
triple-negative breast cancer cells. Adv Pharm Bull. 11:188–196.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Endah E, Wulandari F, Putri Y, Jenie RI
and Meiyanto E: Piperine increases Pentagamavunon-1 anti-cancer
activity on 4T1 breast cancer through mitotic catastrophe mechanism
and senescence with sharing targeting on mitotic regulatory
proteins. Iran J Pharm Res. 21:e1238202022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kamitani N, Nakamae I, Yoneda-Kato N, Kato
JY and Sho M: Preclinical evaluation of pentagamavunone-1 as
monotherapy and combination therapy for pancreatic cancer in
multiple xenograft models. Sci Rep. 12:224192022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hermawan A, Wulandari F, Hanif N, Utomo
RY, Jenie RI, Ikawati M and Tafrihani AS: Identification of
potential targets of the curcumin analog CCA-1.1 for glioblastoma
treatment: Integrated computational analysis and in vitro study.
Sci Rep. 12:139282022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Novitasari D, Jenie RI, Kato JY and
Meiyanto E: Chemoprevention curcumin analog 1.1 promotes metaphase
arrest and enhances intracellular reactive oxygen species levels on
TNBC MDA-MB-231 and HER2-positive HCC1954 cells. Res Pharm Sci.
18:358–370. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ryu K, Choy E, Yang C, Susa M, Hornicek
FJ, Mankin H and Duan Z: Activation of signal transducer and
activator of transcription 3 (Stat3) pathway in osteosarcoma cells
and overexpression of phosphorylated-Stat3 correlates with poor
prognosis. J Orthop Res. 28:971–978. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kelly B, Thamm D and Rosengren RJ: The
second-generation curcumin analogue RL71 elicits G2/M cell cycle
arrest and apoptosis in canine osteosarcoma cells. Vet Comp Oncol.
21:595–604. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lee DS, Lee MK and Kim JH: Curcumin
induces cell cycle arrest and apoptosis in human osteosarcoma (HOS)
cells. Anticancer Res. 29:5039–5044. 2009.PubMed/NCBI
|
|
33
|
Kabir MT, Rahman MH, Akter R, Behl T,
Kaushik D, Mittal V, Pandey P, Akhtar MF, Saleem A, Albadrani GM,
et al: Potential role of curcumin and its nanoformulations to treat
various types of cancers. Biomolecules. 11:3922021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lu KH, Lu PW, Lu EW, Lin CW and Yang SF:
Curcumin and its analogs and carriers: Potential therapeutic
strategies for human osteosarcoma. Int J Biol Sci. 19:1241–1265.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Marcu MG, Jung YJ, Lee S, Chung EJ, Lee
MJ, Trepel J and Neckers L: Curcumin is an inhibitor of p300
histone acetylatransferase. Med Chem. 2:169–174. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Szerlong HJ, Prenni JE, Nyborg JK and
Hansen JC: Activator-dependent p300 acetylation of chromatin in
vitro: Enhancement of transcription by disruption of repressive
nucleosome-nucleosome interactions. J Biol Chem.
285:31954319642010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Raisner R, Kharbanda S, Jin L, Jeng E,
Chan E, Merchant M, Haverty PM, Bainer R, Cheung T, Arnott D, et
al: Enhancer activity requires CBP/P300 bromodomain-dependent
histone H3K27 acetylation. Cell Rep. 24:1722–1729. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hogg SJ, Motorna O, Cluse LA, Johanson TM,
Coughlan HD, Raviram R, Myers RM, Costacurta M, Todorovski I,
Pijpers L, et al: Targeting histone acetylation dynamics and
oncogenic transcription by catalytic P300/CBP inhibition. Mol Cell.
81:2183–2200.e13. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Narita T, Ito S, Higashijima Y, Chu WK,
Neumann K, Walter J, Satpathy S, Liebner T, Hamilton WB, Maskey E,
et al: Enhancers are activated by p300/CBP activity-dependent PIC
assembly, RNAPII recruitment, and pause release. Mol Cell.
81:2166–2182.e6. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen M, Qian C, Jin B, Hu C, Zhang L, Wang
M, Zhou B, Zuo W, Huang L and Wang Y: Curcumin analog WZ26 induces
ROS and cell death via inhibition of STAT3 in cholangiocarcinoma.
Cancer Biol Ther. 24:21628072023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li L, Chen M, Li G and Cai R: Raddeanin A
induced apoptosis of non-small cell lung cancer cells by promoting
ROS-mediated STAT3 inactivation. Tissue Cell. 71:1015772021.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Alsamri H, Hasasna HE, Baby B, Alneyadi A,
Dhaheri YA, Ayoub MA, Eid AH, Vijayan R and Iratni R: Canosol is
novel inhibitor of p300 acetyltransferase in breast cancer. Front
Oncol. 11:6644032021. View Article : Google Scholar : PubMed/NCBI
|