Introduction
Ultraviolet (UV) radiation (200–400 nm) is
associated with a number of acute and chronic effects on the skin,
which may result in inflammation, immunosuppression, premature
aging and the development of malignancies (1). Ultraviolet A (UVA) radiation (320–400
nm), which is not absorbed by the ozone layer, comprises >95% of
the UV light that reaches the surface of the earth. UVA penetrates
the epidermis and affects the epidermal and dermal layers of the
skin. At the cellular level, UVA exposure causes significant
oxidative stress via generation of reactive oxygen species (ROS),
such as singlet oxygen, hydroxyl radical, superoxide anion and
hydrogen peroxide (2). ROS are
rapidly removed by non-enzymatic antioxidants, particularly
glutathione (γ-glutamylcysteinylglycine; GSH), as well as enzymatic
antioxidants, such as catalase, superoxide dismutase, GSH
peroxidase and GSH reductase, which maintain the pro-/antioxidant
balance resulting in cell and tissue stabilization. However, a
surplus of ROS may overwhelm the skin antioxidant defense
mechanisms causing a pro-/antioxidant disequilibrium. The
overproduction of ROS induces oxidation of nucleic acids, proteins
and membrane lipids, which may result in intracellular GSH and
NADH/NADPH depletion and, therefore, energy loss from the cell.
UV-generated ROS also affects the regulation of the gene expression
of signaling molecules/cascades, such as mitogen-activated protein
kinases, interrelated inflammatory cytokines, nuclear factor-κB and
activator protein-1 (3).
α-Tocopherol is the most active form of vitamin E, a
fat-soluble vitamin that exists in 8 different forms in humans, and
is a powerful biological antioxidant. α-Tocopherol is considered to
be the major membrane-bound antioxidant employed by cells (4). α-Tocopherol scavenges acylperoxyl
radicals and, thus, exerts a protective effect against cell
membrane lipid peroxidation induced by UVA (5–8).
Moreover, α-tocopherol and ascorbic acid work together in a cyclic
process (9). During the antioxidant
reaction, α-tocopherol is converted to an α-tocopherol radical by
the donation of a labile hydrogen to a lipid or lipid peroxyl
radical. The α-tocopherol radical is thus reduced to the original
α-tocopherol form by ascorbic acid (9).
Among the cutaneous antioxidants, the tripeptide
GSH, plays a pivotal role in protecting skin cells against
oxidative damage by directly scavenging ROS or by acting as a
coenzyme in GSH peroxidase- or GSH S-transferase-catalyzed
reactions (10,11). It was demonstrated that GSH is also
involved in DNA repair and cell apoptosis (12,13).
Moreover, GSH participates in a number of biological processes,
such as mitochondrial respiration, inflammatory response, signal
transduction, regulation of gene expression and cell proliferation
(14). GSH is able to regenerate
the most essential antioxidants, vitamins C and E, back to their
active forms and reduce the tocopherol radical of vitamin E
directly or indirectly, via the reduction of semidehydroascorbate
to ascorbate.
This study aimed to investigate the protective
activity of α-tocopherol against UVA in an experimental model of
HaCaT human keratinocytes. The effects of α-tocopherol on
UVA-induced cellular oxidative stress, particularly on the
intracellular levels of GSH, lipid peroxidation and ROS were
investigated.
Materials and methods
Materials
The HaCaT human keratinocyte cell line was obtained
from the Food Industry Research and Development Institute (Hsinchu,
Taiwan). Dulbecco’s modified Eagle’s medium (DMEM),
heat-inactivated fetal calf serum (FCS), penicillin-streptomycin
and trypsin-EDTA solutions were purchased from Gibco-BRL (Carlsbad,
CA, USA). Malondialdehyde (MDA) was purchased from Merck KGaA
(Darmstadt, Germany). Sterile dimethylsulfoxide (DMSO),
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT),
5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB), thiobarbituric acid
(TBA), dichlorodihydrofluorescein diacetate (H2DCFDA)
and α-tocopherol were purchased from Sigma-Aldrich (St. Louis, MO,
USA).
Cell culture
The HaCaT cells were grown in DMEM supplemented with
heat-inactivated FCS (10%, v/v), streptomycin (100 U/ml) and
penicillin (0.1 mg/ml) in a humidified atmosphere of 5%
CO2 at 37°C. The culture medium was changed 3 times per
week. The cells were subcultured following trypsinization and
seeded in 6-well plates at a density of 1×105
cells/cm2.
UVA irradiation and pretreatment with
α-tocopherol
The keratinocytes were pretreated with α-tocopherol
(2.9–14.7 IU/ml) at 37°C for 24 h, irradiated and incubated in
serum-free medium at 37°C for an additional 24 h. Stock solutions
of α-tocopherol were dissolved in DMSO, with a final concentration
of DMSO in the medium of 1% (v/v). Irradiated and non-irradiated
control cells were treated with serum-free medium. Prior to UVA
irradiation, the cells were washed with phosphate-buffered saline
(PBS) and covered with a layer of PBS. Dishes with keratinocytes
were irradiated (8 J/cm2 UVA) on ice-cold plates to
eliminate UVA thermal stimulation. Non-irradiated cells were
treated similarly and were incubated in the dark. For the
irradiation, a solar simulator system (Bio-Sun; Vilber Lourmat,
Marne-la-Vallée, France) with a fixed wavelength of 365 nm was
used.
MTT assay
Cell viability was monitored following UVA
irradiation and pretreatment with α-tocopherol. MTT was used to
quantify the metabolically active living cells. Mitochondrial
dehydrogenases metabolize MTT to a purple formazan dye, which is
measured photometrically at 570 nm using a spectrophotometer
(15).
Intracellular GSH levels
Intracellular GSH was measured using a reaction with
DTNB (16). The keratinocytes were
rinsed with PBS, scraped into cooled perchloric acid (1%, v/v) and
sonicated. The aliquots were frozen for protein determination using
the Bradford assay. The suspension was centrifuged (13,000 rpm for
10 min at 4°C) and the supernatant was used to measure GSH in the
reaction with the reaction mixture [800 mmol/l Tris-HCl, 20 mmol/l
EDTA (pH 8.2) and 20 mg/ml DTNB)]. A dilution series of GSH (0.15–1
mM) were prepared as standard. The absorbance was measured on a
microplate reader at 412 nm.
Lipid peroxidation
The levels of lipid peroxidation were determined by
measuring MDA in the cell extract. MDA is the end product of lipid
peroxidation, which is induced by free radicals or activated oxygen
species when cells are irradiated by UVA. The amount of MDA was
measured using the TBA assay (17).
TBA reacts with MDA, yielding red complexes, which absorb at 535
nm. A dilution series of MDA (0.1–10 μM) were prepared as standard.
The cells were washed with cooled PBS, scraped into trichloroacetic
acid (2.8%, w/v), sonicated and aliquots were used for protein
determination with the Bradford assay. The suspension was mixed
with TBA (1%, w/v) in a ratio of 2:1, heated for 30 min at 95°C and
centrifuged (13,000 rpm for 10 min at 4°C). The amount of MDA was
determined spectrophotometrically.
ROS generation
The radical-scavenging efficacy of α-tocopherol in
irradiated cells was monitored by the dichlorodihydrofluorescein
assay. The polar, pre-fluorescent dichlorodihydrofluorescein
diacetate (H2DCFDA) undergoes deacetylation by cytosolic
esterases to form dichlorodihydrofluorescein, which reacts with ROS
to produce fluorescein. The fluorescence is monitored at specific
excitation/emission wavelengths of 488/525 nm. Following
irradiation and pretreatment with α-tocopherol, the cells were
incubated with H2DCFDA (5 nmol/l) for 15 min at 37°C.
The cells were then washed with PBS, scraped into 2 ml of PBS and
sonicated (18). The fluorescence
was measured using a spectrophotometer (LS 50B; PerkinElmer,
Waltham, MA, USA) and the protein concentration was determined with
the Bradford assay.
Statistical analysis
The means ± standard error of the mean were
calculated from at least 3 repeated groups in all the experiments.
Statistical significance between groups was determined with the
Student’s t-test. P<0.05 was considered to indicate a
statistically significant difference between two groups.
Results
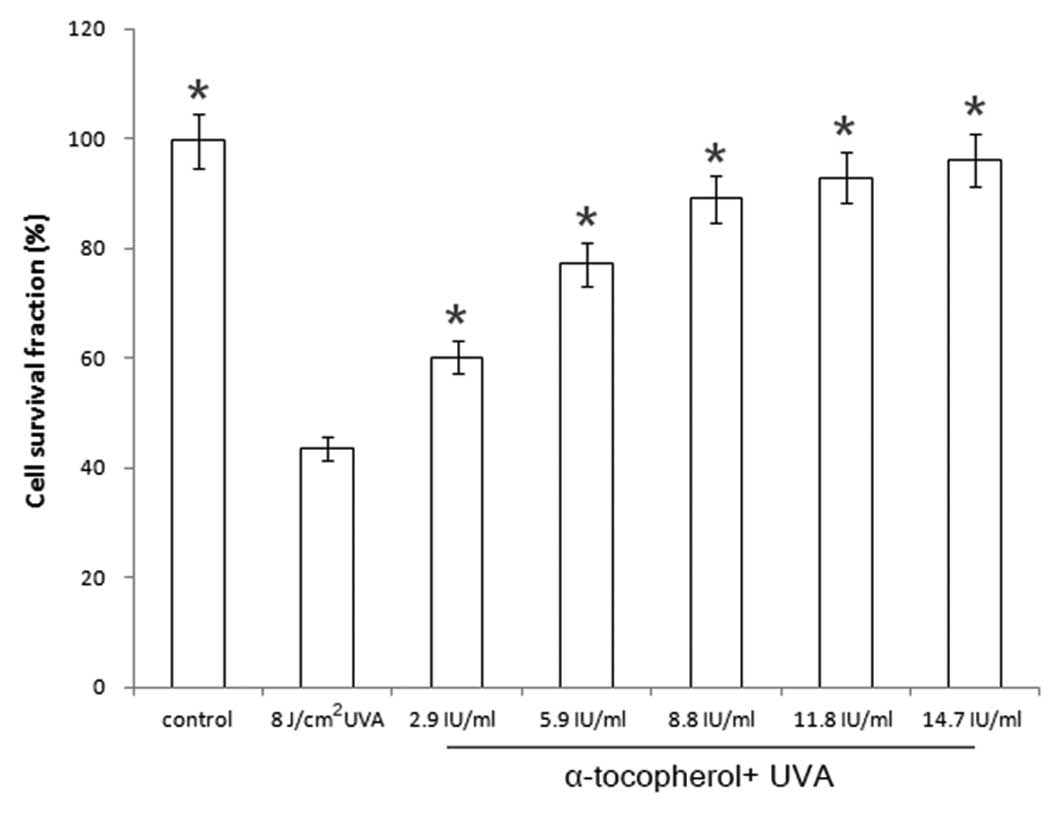
Modulation of cell viability in
UVA-irradiated cells by α-tocopherol
The cell survival fraction was 43.6% when
keratinocytes were irradiated with UVA at a dose of 8
J/cm2. The keratinocytes were pretreated with
α-tocopherol (2.9–14.7 IU/ml) prior to UVA irradiation. The cell
survival fractions were 60.2, 77.1, 89.0, 92.9 and 96.2% following
addition of α-tocopherol at concentrations of 2.9, 5.9, 8.8, 11.8
and 14.7 IU/ml, respectively (Fig.
1). α-Tocopherol pretreatment suppressed the UVA-induced
decrease in cell viability in a concentration-dependent manner.
These findings suggest that α-tocopherol is capable of protecting
keratinocytes against UVA irradiation.
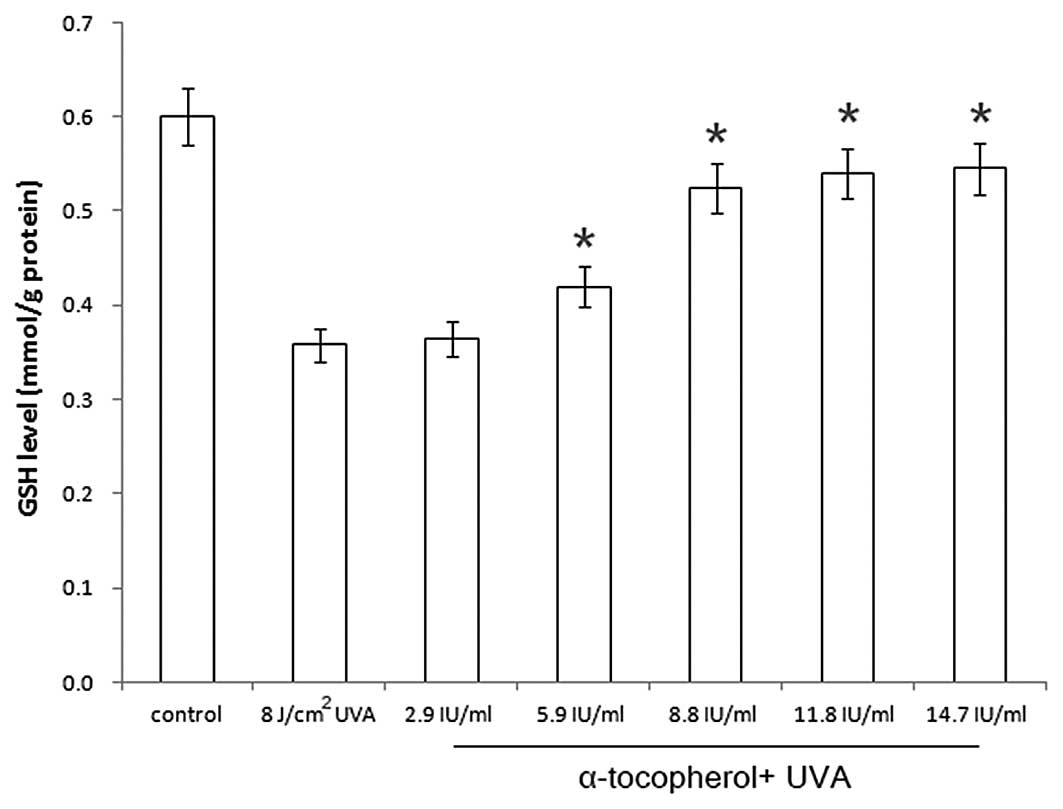
Prevention of UVA-induced GSH depletion
by α-tocopherol
As illustrated in Fig.
2, the GSH level in UVA-irradiated HaCaT cells (8
J/cm2) was decreased to ~50% of that in control cells
(0.600→0.354 mmol/g protein). When α-tocopherol was added prior to
UVA irradiation, the GSH levels in the cells were 0.364, 0.420,
0.525, 0.540 and 0.545 mmol/g protein at α-tocopherol
concentrations of 2.9, 5.9, 8.8, 11.8 and 14.7 IU/ml, respectively.
Therefore, the application of α-tocopherol to UVA-irradiated
keratinocytes led to a dose-dependent prevention of GSH
depletion.
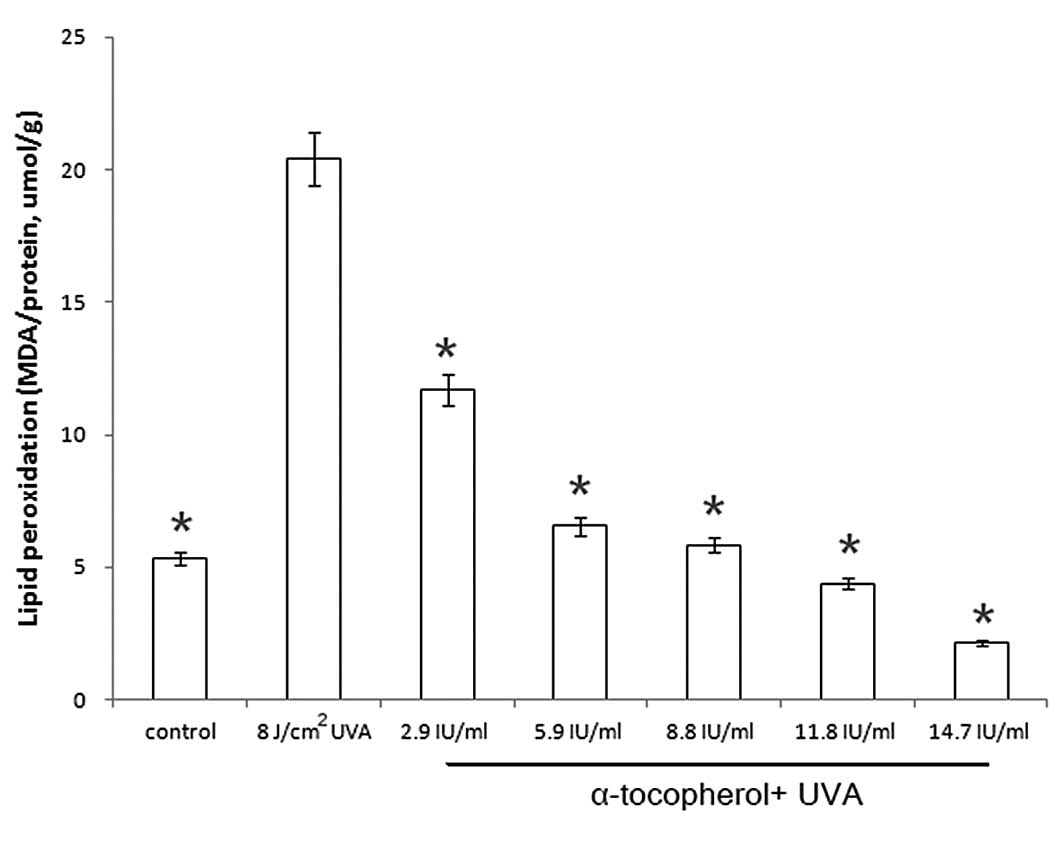
Modulation of UVA-induced lipid
peroxidation by α-tocopherol
The development of lipid peroxidation in irradiated
HaCaT cells was measured using the TBA assay. Under our
experimental conditions, there was a distinct increase in lipid
peroxidation in the UVA-irradiated cells compared to the control
cells (5.238→20.401 MDA/protein, μmol/g). As illustrated in
Fig. 3, the amount of MDA was
significantly reduced by α-tocopherol pretreatment.
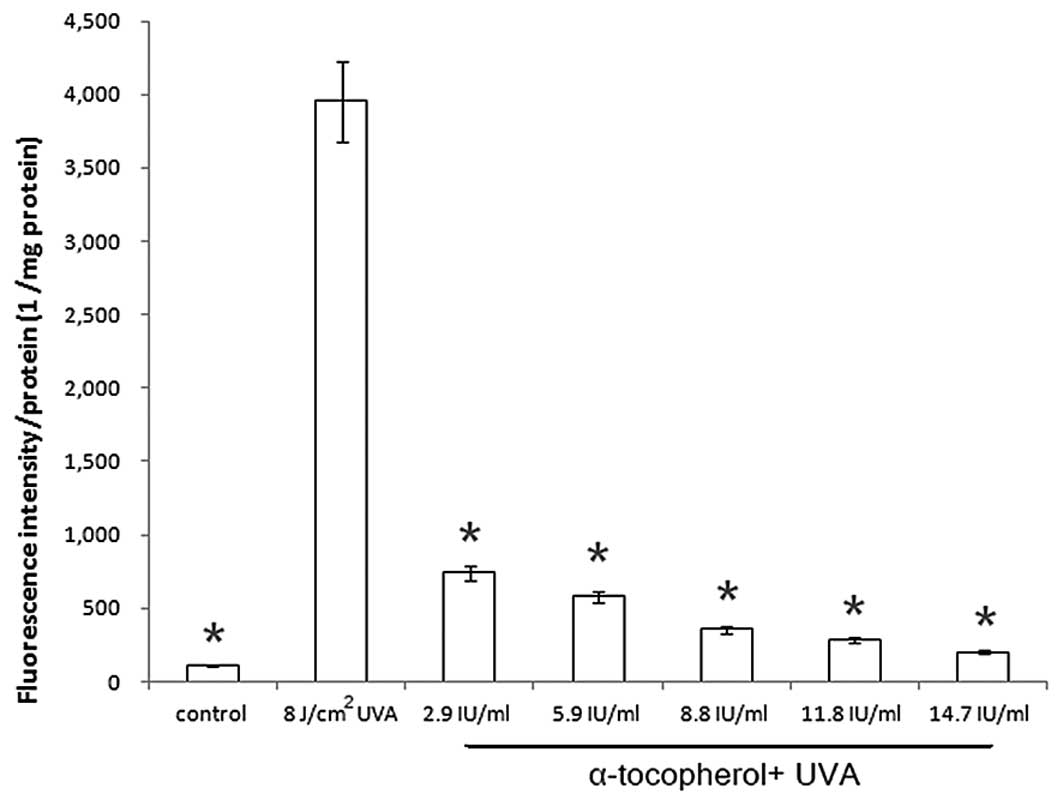
Modulation of UVA-induced ROS generation
by α-tocopherol
To investigate whether the addition of α-tocopherol
affects UVA-induced ROS generation, a fluorescence assay using
H2DCFDA was performed. α-Tocopherol significantly
reduced ROS generation in UVA-irradiated keratinocytes in a
dose-dependent manner (Fig. 4).
Discussion
UV radiation is the principal factor causing skin
cancer in humans. Several studies demonstrated that supplementation
with antioxidants decreases UV-induced skin damage in vitro
and in vivo (19). In this
study, we demonstrated the ability of α-tocopherol to prevent and
reduce UVA-related damage at the cellular level in human
keratinocytes. Treatment of HaCaT cells with α-tocopherol prior to
UVA exposure increased cell viability and suppressed intracellular
GSH depletion, lipid peroxidation and ROS generation. The cell
viability assay demonstrated that α-tocopherol protects HaCaT human
keratinocytes against UVA-induced apoptosis. It is well known that
during and following UVA irradiation, the generation of ROS is
significantly increased in exposed cells (20,21).
As UVA-related biological effects are primarily mediated by ROS,
their elimination is essential for protection against UVA damage.
The application of α-tocopherol led to a significant increase in
cell survival in irradiated HaCaT cells. α-Tocopherol pretreatment
exhibited maximal protection at the highest concentration
tested.
Pretreatment of cells with α-tocopherol resulted in
a concentration-dependent reduction in GSH depletion. The role of
GSH in protecting the skin from oxidative damage caused by various
chemicals and UV exposure has been well documented. Among
non-enzymatic antioxidants, GSH is considered to be the most
important, as it serves as a substrate for 2 major antioxidant
enzymes, GSH-peroxidase and GSH-transferase, and it also
participates in vitamin C and E regeneration (22). The GSH level is directly associated
with the degree of lipid peroxidation in the cell membrane
(23), as GSH participates in
eliminating lipid peroxidation products, including
4-hydroxynonenal, by forming a GSH conjugate (24).
The cutaneous antioxidant system is complex and
incompletely understood. We previously demonstrated that magnesium
ascorbyl phosphate and coenzyme Q10 increased
intracellular GSH levels (25).
Kagan et al (26) reported
that vitamin C is able to regenerate vitamin E from the
α-tocopheroxyl radical. Furthermore, α-tocopherol and ascorbic acid
work together in a cyclic process. During the antioxidant reaction,
α-tocopherol is converted to an α-tocopherol radical through the
donation of a labile hydrogen to a lipid or lipid peroxyl radical.
The α-tocopherol radical is thus be reduced to the original
α-tocopherol form by ascorbic acid (9). α-Lipoic acid was previously shown to
elevate intracellular GSH levels in vitro by increasing
de novo synthesis (27), an
effect dependent upon the metabolic reduction of lipoic to
dihydrolipoic acid. Dihydrolipoic acid is then released into the
culture medium, where it reduces cystine to cysteine. Cysteine is
readily taken up by the neutral amino acid transport system and
utilized for GSH synthesis. Through this mechanism, lipoic acid
enables cysteine to bypass the Xc− transport system,
which is weakly expressed in lymphocytes and inhibited by
glutamate. Thereby, lipoic acid enables the key enzyme of GSH
synthesis, γ-glutamylcysteine synthetase, which is regulated by an
uptake-limited cysteine supply, to work at optimum conditions.
However, the precise mechanism underlying the α-tocopherol-induced
increase in intracellular GSH levels requires elucidation by
further studies.
α-Tocopherol is the most active form of vitamin E in
humans and is a powerful biological antioxidant, considered to be
the major membrane-bound antioxidant employed by cells (4). The main antioxidant function of
α-tocopherol is protection against lipid peroxidation (28). The overall process of lipid
peroxidation includes three stages: initiation, propagation and
termination. Once formed, peroxyl radicals are rearranged via a
cyclization reaction to endoperoxides (precursors of MDA), with MDA
as the final product (29). Our
findings demonstrated that the amount of MDA was markedly reduced
by treatment with α-tocopherol.
In conclusion, α-tocopherol protects keratinocytes
against UVA irradiation, possibly through increasing the levels of
GSH or decreasing the levels of lipid peroxidation and ROS
generation.
Acknowledgements
This study was supported by grants from the National
Science Council (no. 98-2314-B-238-001) and the Vanung University
(no. VIT-98-CM-01), Taiwan, R.O.C..
References
|
1
|
Melnikova VO and Ananthaswamy HN: Cellular
and molecular events leading to the development of skin cancer.
Mutat Res. 571:91–106. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Svobodova A, Psotova J and Walterova D:
Natural phenolics in the prevention of UV-induced skin damage. A
review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub.
147:137–145. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Svobodova A, Walterova D and Vostalova J:
Ultraviolet light induced alteration to the skin. Biomed Pap Med
Fac Univ Palacky Olomouc Czech Repub. 150:25–38. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Burton GW and Ingold KU: Vitamin E as an
in vitro and in vivo antioxidant. Ann NY Acad Sci. 570:7–22. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Burton GW, Page YJ, Gabe EJ and Ingold KU:
Antioxidant activity of vitamin E and related phenols. Importance
of stereoelectronic factors. J Am Chem Soc. 102:7792–7794. 1980.
View Article : Google Scholar
|
|
6
|
Fryer MJ: Evidence for the photoprotective
effects of vitamin E. Photochem Photobiol. 58:304–312. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nachbar F and Korting HC: The role of
vitamin E in normal and damaged skin. J Mol Med (Berl). 73:7–17.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Norkus EP, Bryce GF and Bhagavan HN:
Uptake and bioconversion of alpha-tocopheryl acetate to
alpha-tocopherol in skin of hairless mice. Photochem Photobiol.
57:613–615. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kojo S: Vitamin C: basic metabolism and
its function as an index of oxidative stress. Curr Med Chem.
11:1041–1064. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Afaq F and Mukhtar H: Effects of solar
radiation on cutaneous detoxification pathways. J Photochem
Photobiol B. 63:61–69. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hayes JD and McLellan LI: Glutathione and
glutathione-dependent enzymes represent a co-ordinately regulated
defence against oxidative stress. Free Radic Res. 31:273–300. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fonnum F and Lock EA: The contributions of
excitotoxicity, glutathione depletion and DNA repair in chemically
induced injury to neurones: exemplified with toxic effects on
cerebellar granule cells. J Neurochem. 88:513–531. 2004. View Article : Google Scholar
|
|
13
|
Hall AG: Review: The role of glutathione
in the regulation of apoptosis. Eur J Clin Invest. 29:238–245.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sies H: Glutathione and its role in
cellular functions. Free Radic Biol Med. 27:916–921. 1999.
View Article : Google Scholar
|
|
15
|
Green LM, Reade JL and Ware CF: Rapid
colorimetric assay for cell viability: application to the
quantitation of cytotoxic and growth inhibitory lymphokines. J
Immunol Methods. 70:257–268. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sedlak J and Lindsay RH: Estimation of
total, protein-bound, and nonprotein sulfhydryl groups in tissue
with Ellman’s reagent. Anal Biochem. 25:192–205. 1968.PubMed/NCBI
|
|
17
|
Buege JA and Aust SD: Microsomal lipid
peroxidation. Methods Enzymol. 52:302–310. 1978. View Article : Google Scholar
|
|
18
|
D’Angelo S, Ingrosso D, Migliardi V, et
al: Hydroxytyrosol, a natural antioxidant from olive oil, prevents
protein damage induced by long-wave ultraviolet radiation in
melanoma cells. Free Radic Biol Med. 38:908–919. 2005.PubMed/NCBI
|
|
19
|
Swindells K and Rhodes LE: Influence of
oral antioxidants on ultraviolet radiation-induced skin damage in
humans. Photodermatol Photoimmunol Photomed. 20:297–304. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tyrrell RM: The molecular and cellular
pathology of solar ultraviolet radiation. Mol Aspects Med. 15:1–77.
1994.PubMed/NCBI
|
|
21
|
Morita A and Krutmann J: Ultraviolet A
radiation-induced apoptosis. Methods Enzymol. 319:302–309. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Svobodova A, Rambouskova J, Walterova D
and Vostalova J: Protective effects of phenolic fraction of blue
honeysuckle fruits against UVA-induced damage to human
keratinocytes. Arch Dermatol Res. 300:225–233. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schneider LA, Dissemond J, Brenneisen P,
Hainzl A, Briviba K, Wlaschek M and Scharffetter-Kochanek K:
Adaptive cellular protection against UVA-1-induced lipid
peroxidation in human dermal fibroblasts shows donor-to-donor
variability and is glutathione dependent. Arch Dermatol Res.
297:324–328. 2006. View Article : Google Scholar
|
|
24
|
Tarozzi A, Marchesi A, Hrelia S, Angeloni
C, Andrisano V, Fiori J, Cantelli-Forti G and Hrelia P: Protective
effects of cyanidin-3-O-beta-glucopyranoside against
UVA-induced oxidative stress in human keratinocytes. Photochem
Photobiol. 81:623–629. 2005.
|
|
25
|
Hwang TL, Tsai CJ, Chen JL, Changchien TT,
Wang CC and Wu CM: Magnesium ascorbyl phosphate and coenzyme
Q10protect the keratinocytes against UVA irradiation by
suppressing glutathione depletion. Mol Med Rep. 6:375–378.
2012.PubMed/NCBI
|
|
26
|
Kagan V, Witt E, Goldman R, Scita G and
Packer L: Ultraviolet light-induced generation of vitamin E
radicals and their recycling. A possible photosensitizing effect of
vitamin E in skin. Free Radic Res Commun. 16:51–64. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Han D, Handelman G, Marcocci L, Sen CK,
Roy S, Kobuchi H, Tritschler HJ, Flohe L and Packer L: Lipoic acid
increases de novo synthesis of cellular glutathione by improving
cystine utilization. Biofactors. 6:321–338. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pryor WA: Vitamin E and heart disease:
basic science to clinical intervention trials. Free Radic Biol Med.
28:141–164. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Marnett LJ: Lipid peroxidation-DNA damage
by malondialdehyde. Mutat Res. 424:83–95. 1999. View Article : Google Scholar : PubMed/NCBI
|