Introduction
Liposomes are commonly used as pharmaceutical
delivery vehicles to improve therapeutic efficacy and reduce drug
toxicity (1). Encapsulation of
drugs into liposomes can prolong the time course of the drug effect
and improve the stability of drugs in vitro and in
vivo (2). However, rapid uptake
of liposomes in vivo by cells of the mononuclear phagocytic
system (MPS) has restricted their therapeutic utility (3). Surface modification of liposomes by
carbohydrates, glycolipids or polymers to form long-circulating
liposomes can alter their pharmacokinetic pattern and reduce MPS
uptake (4). Furthermore, liposomes
that have been modified with glycolipid-targeted ligands are
promising as a long-circulating and tumor-targeting carrier system
(5). Asialoglycoprotein receptor
(ASGPR) is a high-capacity galactose-binding receptor expressed on
the surface of hepatocytes (6) that
possesses characteristics that can specifically recognize
glycoproteins that possess exposed terminal galactose or lactose
residues (7). Liposomes modified
with galactose derivatives can be recognized and bound by ASGPR in
liver parenchyma cells, and significantly improve the
liver-targeted effect of liposome drugs (8). This is crucial in the development of
novel drugs in the treatment of hepatic carcinoma (9). Therefore, liposomes modified with
galactose esters not only avoid the uptake of MPS, but also improve
the targeting effect of liposomal carriers.
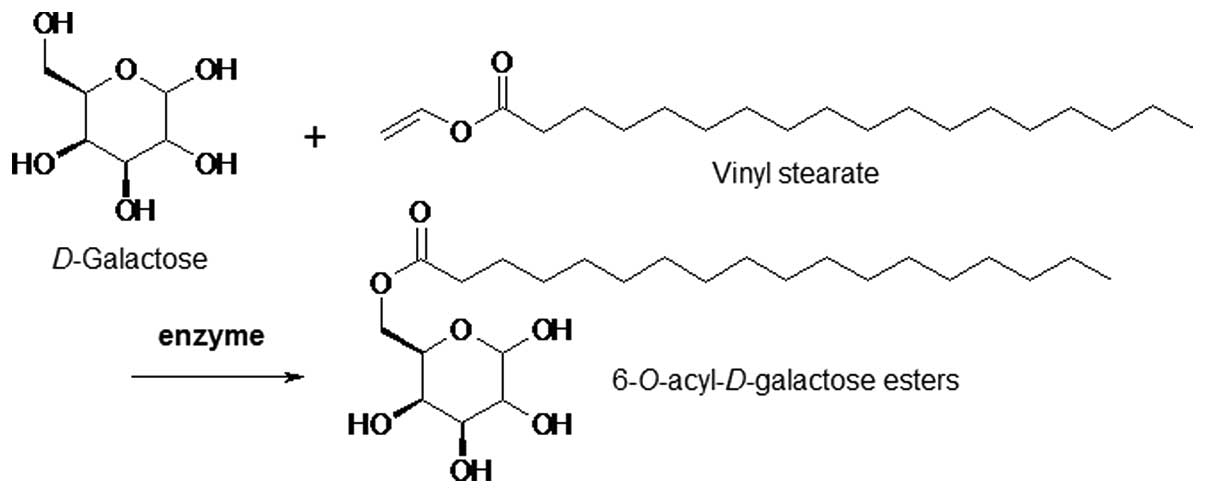
In our previous study (10), 6-O-acyl-D-galactose esters
were synthesized by the enzyme-catalyzed esterification of
D-galactose and vinyl stearate (Fig.
1).
The anti-cancer drug docetaxel was used as a model
drug, and docetaxel liposomes modified with
6-O-acyl-D-galactose esters (Gal-DOC-L) were successfully
prepared as a novel formulation for liver cancer treatment
(11). In the present study, the
pharmacokinetic characteristics of docetaxel following intravenous
administration of Gal-DOC-L in rabbits was investigated, and the
results were compared with docetaxel injection (DOC-I).
Materials and methods
Materials
Docetaxel (purity, 99%) was purchased from the
Beijing Norzer Pharmaceutical Co., Ltd. (Beijing, China).
Norethisterone was purchased from the National Institute for the
Control of Pharmaceutical and Biological Products (Beijing, China).
DOC-I was purchased from the Jiangsu Hengrui Medicine Co., Ltd.
(Jiangsu, China). Gal-DOC-L were prepared in our laboratory
(Department of Pharmaceutics, School of Chinese Materia Medica,
Guangzhou University of Traditional Chinese Medicine, Guangzhou,
Guangdong, China). Methanol and acetonitrile from the Shandong
Yuwang Industrial Co., Ltd. (Shandong, China) were of
chromatographic grade. All other reagents were of analytical grade
and used without further purification.
Instruments
The Dionex high-performance liquid chromatography
(HPLC) system consisted of a PDA-100 detector, a P680 pump and an
ASI-100 autosampler (Dionex, Sunnyvale, CA, USA). The chromeleon
chromatography workstation software was used for data acquisition
and processing.
Chromatographic conditions
Chromatographic separation was carried out on a
Diamonsil C18 column (250×4.6 mm, 5 μm; Dikma Technologies, Inc.,
Radnor, PA, USA) with a EasyGuard Cl8 Security guard column (8×4.0
mm internal diameter; Dikma Technologies, Inc.) maintained at 30°C.
The mobile phase consisted of acetonitrile:water (55:45, v/v), at a
flow rate of 1.0 ml/min, and the wavelength was set at 230 nm.
Experimental animals
New Zealand rabbits were purchased from the
Laboratory Animal Center of Guangzhou University of Chinese
Medicine (Guangzhou, Guangdong, China). The rabbits were
acclimatized to the laboratory conditions one week prior to the
experiment. The rabbits were deprived of food but provided with
water ad libitum for 12 h before the experiment. All
experiments that included blood collection from the rabbit ear
marginal veins were approved by Guilin Medical University Animal
Ethics Committee (Guilin, China).
Sample preparation
In the present study, methyl tertiary-butyl ether
was used as a solvent for the extraction of drugs from plasma
(12,13). The blood samples (0.9 ml) were
collected from the rabbit ear marginal veins at a dose of 10.0 mg
docetaxel eq/kg. Plasma was obtained by centrifugation of the blood
at 1,000 × g for 10 min. Norethisterone (2.0 mg/ml, 100 μl) as an
internal standard was added into 900 μl of plasma, and vortexed
with 3.0 ml tert-butyl methyl ether for 10 min. Following
centrifugation of the mixture at 1,000 × g for 5 min, the upper
organic layer was collected and evaporated to dryness with
N2 at 30°C. The residue was reconstituted in 10 ml of
HPLC mobile phase, vortexed for 1 min and centrifuged at 11,200 × g
for 5 min. The plasma concentration of docetaxel was determined by
HPLC.
HPLC method validation
The method of HPLC was validated according to the
currently accepted USA Food and Drug Administration (FDA)
bioanalytical method validation guidance (14). The following validation
characteristics were determined: Specificity, linearity, precision
and accuracy.
The specificity was evaluated by comparing retention
times obtained in the standard solutions of docetaxel using various
samples (blank plasma sample, blank plasma sample spiked with
docetaxel and plasma sample following administration of Gal-DOC-L).
The calibration curves were constructed with seven concentrations
(simultaneously prepared) ranging from 0.1 to 10 mg/ml for standard
solutions of docetaxel. Each concentration level was prepared in
triplicate and analyzed three times. Calibration curves were
constructed by plotting the concentration of compounds vs. the peak
area response. The linearity was evaluated by the least square
regression method. The precision was determined by repeatability
(intra-day) and intermediate precision (inter-day) and was
expressed as the relative standard deviation (%). The accuracy was
tested by replicate analysis of different samples at known
concentrations and then compared with the true concentration of a
standard, and accuracy was assessed by the recovery percentage.
Pharmacokinetics investigation
Gal-DOC-L or DOC-I were intravenously administered
into the ear marginal veins of rabbits at a dose of 10.0 mg
docetaxel eq/kg. The blood samples (0.9 ml) were collected from the
ear marginal veins with a heparinized syringe at 0.083, 0.25, 0.5,
l, 2, 4, 6, 8, 12, 16 and 24 h after administration. Plasma was
obtained by centrifugation of the blood at 1,000 × g for 10 min.
Norethisterone (2.0 mg/ml, 100 μl) was added into 900 μl of plasma,
and vortexed with 3.0 ml tert-butyl methyl ether for 10 min.
Following centrifugation of the mixture at 1,000 × g for 5 min, the
upper organic layer was collected and evaporated to dryness with
N2 at 30°C. The residue was reconstituted in 10 ml of
HPLC mobile phase, vortexed for 1 min and centrifuged at 11,200 × g
for 5 min. The plasma concentration of docetaxel was determined by
HPLC and pharmacokinetic parameters were calculated with DAS 2.0
software (Mathematical Pharmacology Professional Committee of
China, Shanghai, China).
Results and Discussion
Method validation
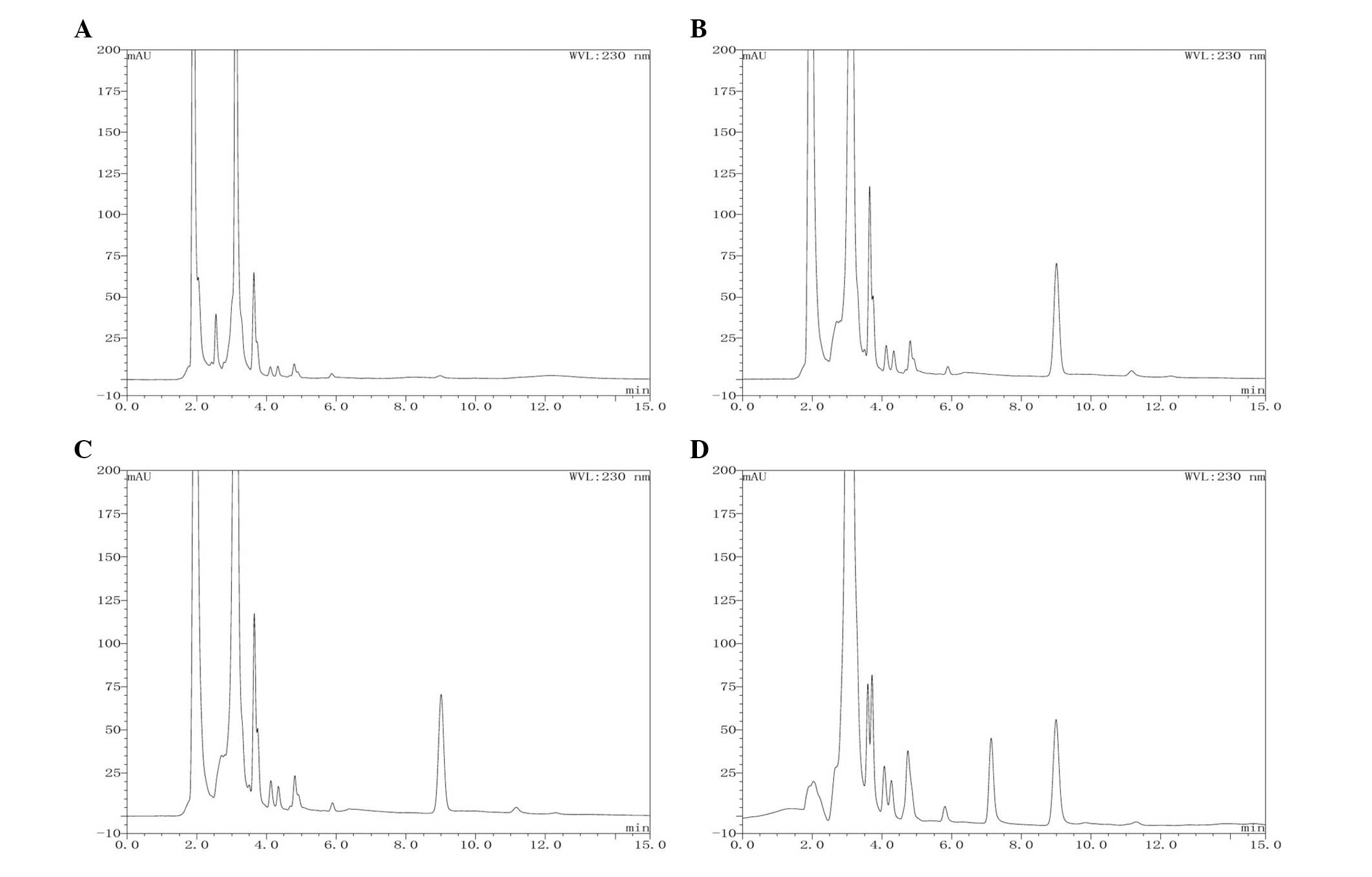
The specificity analysis revealed that the developed
HPLC method was not affected by interference at the retention time
of the analytes. The typical chromatograms of the docetaxel, blank
plasma, blank plasma spiked with docetaxel and plasma samples
following administration of Gal-DOC-L are shown in Fig. 2.
There was a good linearity in the tested range. The
area response was y=0.2728×−0.0066 with a correlation coefficient
of 0.9992 and confidence intervals at P=0.05. The precision
obtained for the repeatability studies and for intermediate
precision (expressed as RSD) was in the range between 2 and 8%,
which comply with the acceptance criteria proposed (not more 15%).
The accuracy was expressed as percent recoveries obtained for
different docetaxel concentrations. The results indicated that the
analytical method has acceptable precision and accuracy (Table I).
 | Table IIntra- and inter-day precision and
accuracy for determination of docetaxel (n=5). |
Table I
Intra- and inter-day precision and
accuracy for determination of docetaxel (n=5).
| Concentration,
mg/ml | Added, μg/ml | RSD, % | Recovery (%) |
|---|
|
|---|
| Intra-day | Inter-day |
|---|
| 0.1 | 0.1 | 4.56 | 7.13 | 82.45±3.01 |
| 1.0 | 1.0 | 2.17 | 6.39 | 93.22±0.95 |
| 10.0 | 10.0 | 3.88 | 4.24 | 86.97±1.64 |
Pharmacokinetics investigation
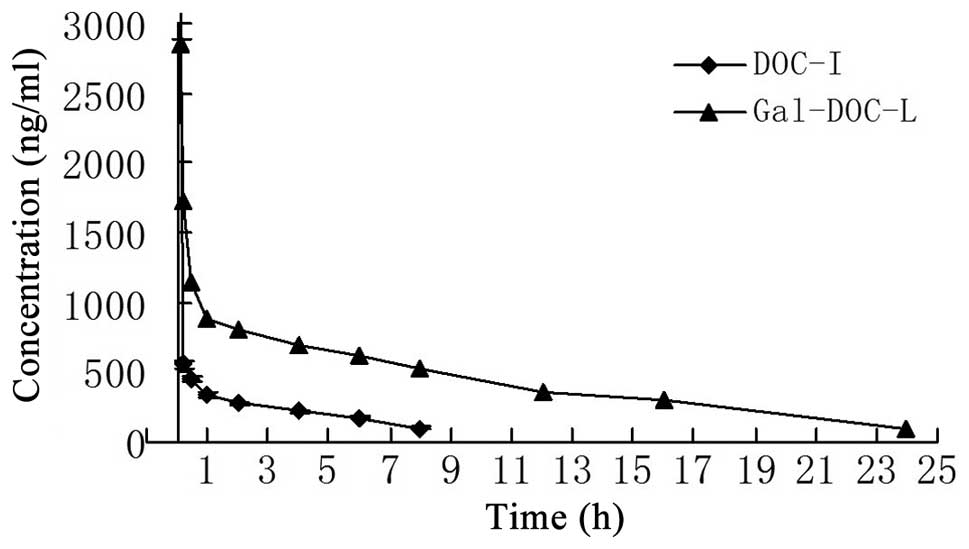
The change in concentration over the time period for
docetaxel in plasma following intravenous injection of Gal-DOC-L
and DOC-I in rabbits is shown in Fig.
3.
A peak plasma concentration was achieved rapidly
following intravenous injection of the docetaxel formulation, with
Gal-DOC-L having a drug concentration-time curve similar to DOC-I.
The results indicated that docetaxel was rapidly removed from the
circulation. By contrast, Gal-DOC-L showed a markedly delayed
clearance from the plasma. DOC-I was rapidly removed from the
circulating system and was not detected in the plasma 8 h after
intravenous injection. The concentration-time curves for Gal-DOC-L
and DOC-I in rabbits were fitted by the two-compartment model, and
their pharmacokinetic parameters are shown in Table II. The results showed that
Gal-DOC-L prolonged t1/2β of docetaxel by 2.31-fold in
plasma, and improved the area under the curve(0-∞)
values of docetaxel by 4.03-fold compared to DOC-I. The docetaxel
in Gal-DOC-L was eliminated gradually.
 | Table IIPharmacokinetic parameters of
docetaxel following a single intravenous injection of Gal-DOC-L and
DOC-I in rabbits with a dose of 10.0 mg/kg (n=6). |
Table II
Pharmacokinetic parameters of
docetaxel following a single intravenous injection of Gal-DOC-L and
DOC-I in rabbits with a dose of 10.0 mg/kg (n=6).
| Parameters | Units | DOC-I | Gal-DOC-L |
|---|
| t1/2α | h | 0.039±0.005 | 0.134±0.017 |
| t1/2β | h | 3.848±0.751 | 8.891±1.280 |
| V1 | l/kg | 0.847±0.099 | 2.575±0.304 |
| CL | l/kg
·h−1 | 3.001±0.346 | 0.745±0.101 |
|
AUC(0-t) | mg·h/l | 2.747±0.357 | 11.126±1.643 |
|
AUC(0-∞) | mg·h/l | 3.332±0.402 | 13.417±1.468 |
| K10
h−1 | 3.544±0.485 | 0.289±0.036 | |
| K12
h−1 | 13.591±1.590 | 3.644±0.397 | |
| K21
h−1 | 0.839±0.123 | 1.329±0.178 | |
In the present study, the prolonged circulation time
of Gal-DOC-L in the plasma is possibly due to a reduced interaction
with plasma and cell-surface proteins. A possible explanation for
the reduced interaction is the steric hindrance effect, which is
generated by the surface-grafted 6-O-acyl-D-galactose esters
molecules (15). As galactose
esters exist on the surface of liposomes, a longer persistence of
the liposomes in the blood enabled an improved targeting effect,
which enhanced the active targeting function of the liposome. In
order to further evaluate the liver targeting effect of Gal-DOC-L,
analysis of the tissue distribution in mice is required.
Acknowledgements
The present study was supported by the Natural
Science Foundation of China (NSFC; grant no. 30772790). The authors
gratefully acknowledge the financial support by the NSFC.
References
|
1
|
Samad A, Sultana Y and Aqil M: Liposomal
drug delivery systems: an update review. Curr Drug Deliv.
4:297–305. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gabizon A, Shmeeda H and Barenholz Y:
Pharmacokinetics of pegylated liposomal Doxorubicin: review of
animal and human studies. Clin Pharmacokinet. 42:419–436. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Woodle MC and Lasic DD: Sterically
stabilized liposomes. Biochim Biophys Acta. 1113:171–199. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Allen TM, Hansen CB and de Menezes DEL:
Pharmacokinetics of long-circulating liposomes. Adv Drug Deliv Rev.
16:267–284. 1995. View Article : Google Scholar
|
|
5
|
Cai L, Wang X, Wang W, et al: Peptide
ligand and PEG-mediated long-circulating liposome targeted to FGFR
overexpressing tumor in vivo. Int J Nanomedicine. 7:4499–4510.
2012.PubMed/NCBI
|
|
6
|
Mamidyala SK, Dutta S, Chrunyk BA, et al:
Glycomimetic ligands for the human asialoglycoprotein receptor. J
Am Chem Soc. 134:1978–1981. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ye Z, Houssein HS and Mahato RI:
Bioconjugation of oligonucleotides for treating liver fibrosis.
Oligonucleotides. 17:349–404. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li Y, Huang G, Diakur J and Wiebe LI:
Targeted delivery of macromolecular drugs: asialoglycoprotein
receptor (ASGPR) expression by selected hepatoma cell lines used in
antiviral drug development. Curr Drug Deliv. 5:299–302. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhou X, Zhang M, Yung B, Li H, Zhou C, Lee
LJ and Lee RJ: Lactosylated liposomes for targeted delivery of
doxorubicin to hepatocellular carcinoma. Int J Nanomedicine.
7:5465–5474. 2012.PubMed/NCBI
|
|
10
|
Cheng Y, Wu W, Zhang D and Mai YZ:
Enzyme-catalyzed synthesis of ASGPR ligand-targeted modifier in
non-aqueous medium. Acta Pharmaceutica Sinica. 45:1134–1138.
2010.(In Chinese).
|
|
11
|
Guo BH, Cheng Y, Lin LP, Lin DH and Wu W:
Preparation and characterization of galactose-modified liposomes by
a nonaqueous enzymatic reaction. J Liposome Res. 21:255–260. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kuyukina MS, Ivshina IB, Philp JC,
Christofi N, Dunbar SA and Ritchkova MI: Recovery of Rhodococcus
biosurfactants using methyl tertiary-butyl ether extraction. J
Microbiol Methods. 46:149–156. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Matyash V, Liebisch G, Kurzchalia TV,
Shevchenko A and Schwudke D: Lipid extraction by methyl-tert-butyl
ether for high-throughput lipidomics. J Lipid Res. 49:1137–1146.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
FDA C: Guidance for industry:
bioanalytical method validation. US Department of Health and Human
Services, Food and Drug Administration, Center for Drug Evaluation
and Research (CDER), Center for Veterinary Medicine (CV); 2001
|
|
15
|
Immordino ML, Dosio F and Cattel L:
Stealth liposomes: review of the basic science, rationale, and
clinical applications, existing and potential. Int J Nanomedicine.
1:297–315. 2006.PubMed/NCBI
|