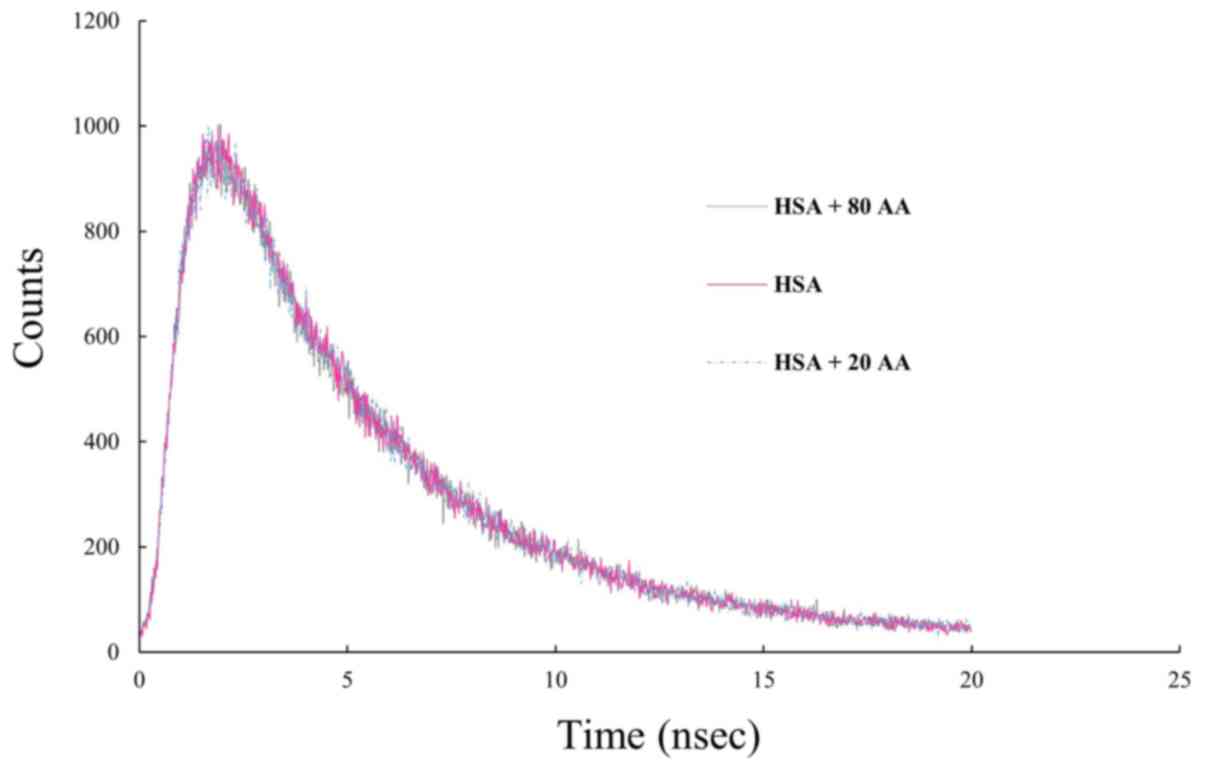
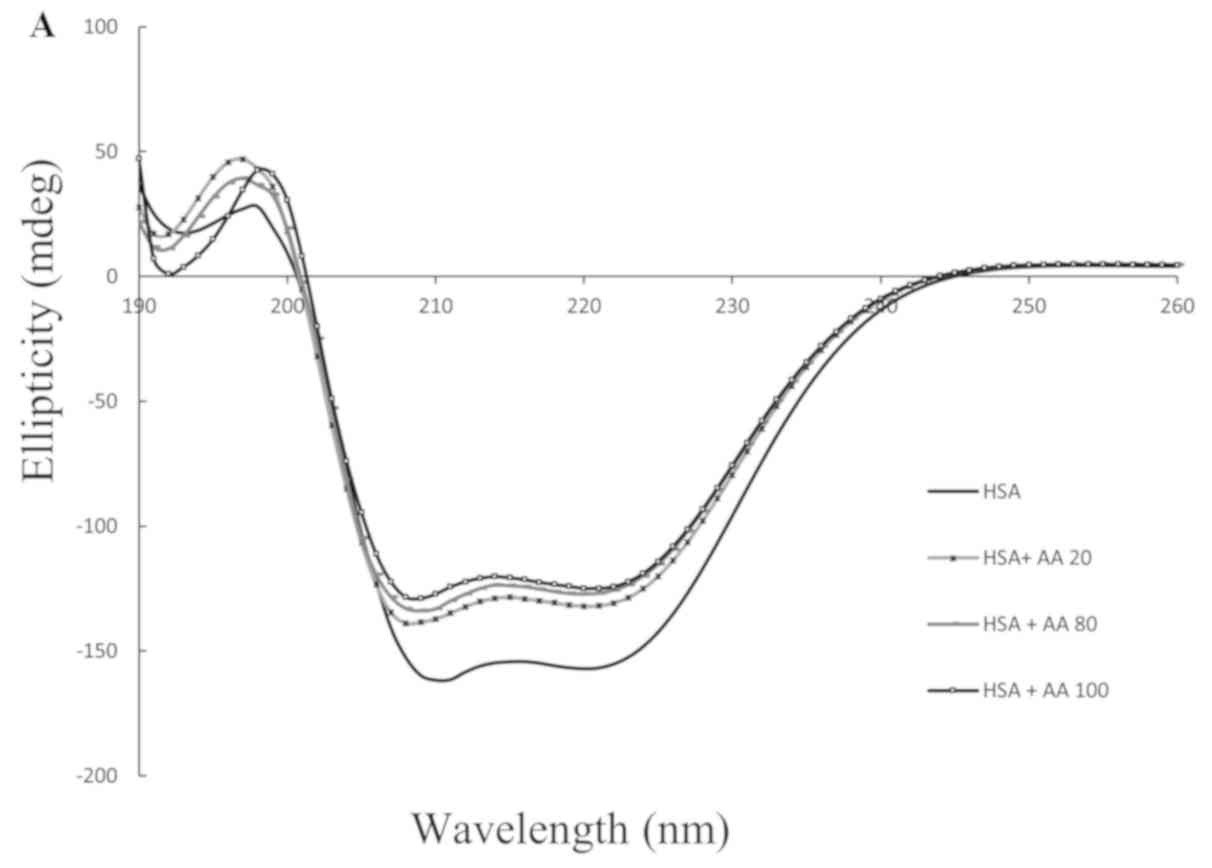
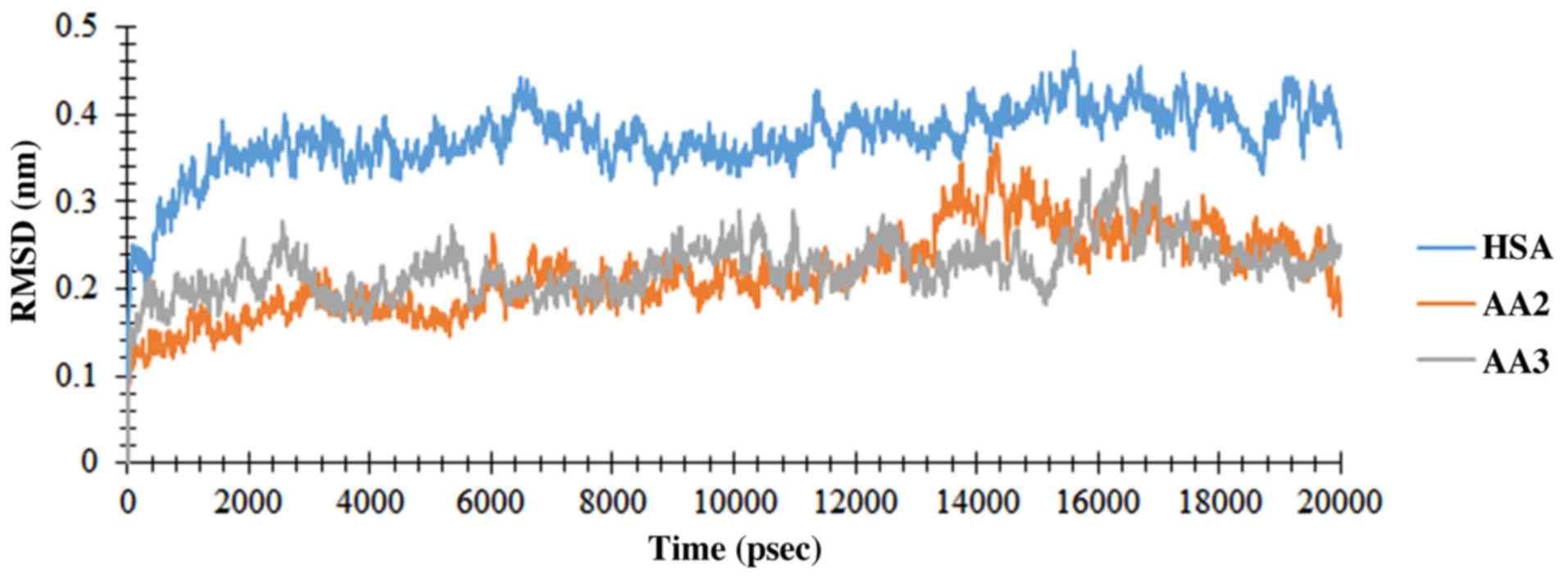
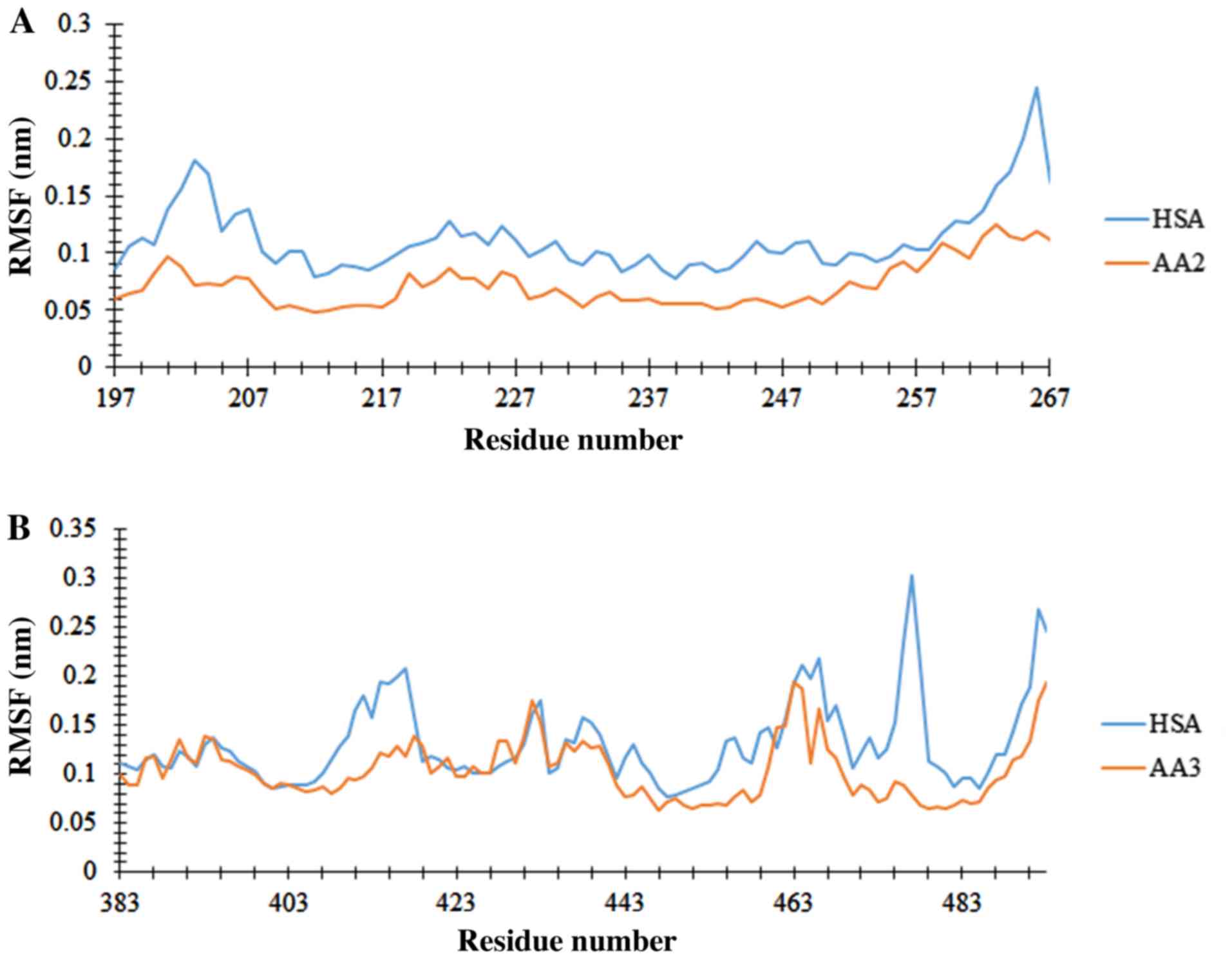
|
1
|
Kratz F: Albumin as a drug carrier: Design
of prodrugs, drug conjugates and nanoparticles. J Control Release.
132:171–183. 2008.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sarkar D, Mahata A, Das P, Girigoswami A,
Ghosh D and Chattopadhyay N: Deciphering the perturbation of serum
albumins by a ketocyanine dye: A spectroscopic approach. J
Photochem Photobiol B. 96:136–143. 2009.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Chatterjee T, Pal A, Dey S, Chatterjee BK
and Chakrabarti P: Interaction of virstatin with human serum
albumin: Spectroscopic analysis and molecular modeling. PLoS One.
7(e37468)2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Elsadek B and Kratz F: Impact of albumin
on drug delivery--new applications on the horizon. J Control
Release. 157:4–28. 2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lhiaubet-Vallet V, Sarabia Z, Boscá F and
Miranda MA: Human serum albumin-mediated stereodifferentiation in
the triplet state behavior of (S)- and (R)-carprofen. J Am Chem
Soc. 126:9538–9539. 2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Jiménez MC, Miranda MA and Vayá I: Triplet
excited States as chiral reporters for the binding of drugs to
transport proteins. J Am Chem Soc. 127:10134–10135. 2005.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Fasano M, Curry S, Terreno E, Galliano M,
Fanali G, Narciso P, Notari S and Ascenzi P: The extraordinary
ligand binding properties of human serum albumin. IUBMB Life.
57:787–796. 2005.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Ahmed-Ouameur A, Diamantoglou S,
Sedaghat-Herati MR, Nafisi Sh, Carpentier R and Tajmir-Riahi HA:
The effects of drug complexation on the stability and conformation
of human serum albumin: Protein unfolding. Cell Biochem Biophys.
45:203–213. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Varshney A, Sen P, Ahmad E, Rehan M,
Subbarao N and Khan RH: Ligand binding strategies of human serum
albumin: How can the cargo be utilized? Chirality. 22:77–87.
2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Anand U and Mukherjee S: Binding,
unfolding and refolding dynamics of serum albumins. Biochim Biophys
Acta. 1830:5394–5404. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Petitpas I, Petersen CE, Ha C-E,
Bhattacharya AA, Zunszain PA, Ghuman J, Bhagavan NV and Curry S:
Structural basis of albumin-thyroxine interactions and familial
dysalbuminemic hyperthyroxinemia. Proc Natl Acad Sci USA.
100:6440–6445. 2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zajdel A, Wilczok A, Chodurek E, Gruchlik
A and Dzierzewicz Z: Polyunsaturated fatty acids inhibit melanoma
cell growth in vitro. Acta Pol Pharm. 70:365–369. 2013.PubMed/NCBI
|
|
13
|
Nelson GJ, Schmidt PC, Bartolini G, Kelley
DS and Kyle D: The effect of dietary arachidonic acid on platelet
function, platelet fatty acid composition, and blood coagulation in
humans. Lipids. 32:421–425. 1997.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Zhang C, Li A, Gao S, Zhang X and Xiao H:
The TIP30 protein complex, arachidonic acid and coenzyme A are
required for vesicle membrane fusion. PLoS One.
6(e21233)2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Høstmark AT and Haug A: Percentage oleic
acid is inversely related to percentage arachidonic acid in total
lipids of rat serum. Lipids Health Dis. 12(40)2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Guo X-J, Sun X-D and Xu S-K: Spectroscopic
investigation of the interaction between riboflavin and bovine
serum albumin. J Mol Struct. 931:55–59. 2009. View Article : Google Scholar
|
|
17
|
Gao H, Lei L, Liu J, Kong Q, Chen X and Hu
Z: The study on the interaction between human serum albumin and a
new reagent with antitumour activity by spectrophotometric methods.
J Photochem Photobiol Chem. 167:213–221. 2004. View Article : Google Scholar
|
|
18
|
Ahmad E, Sen P and Khan RH: Structural
stability as a probe for molecular evolution of homologous albumins
studied by spectroscopy and bioinformatics. Cell Biochem Biophys.
61:313–325. 2011.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ghosh KS, Sahoo BK and Dasgupta S:
Spectrophotometric studies on the interaction between
(-)-epigallocatechin gallate and lysozyme. Chem Phys Lett.
452:193–197. 2008. View Article : Google Scholar
|
|
20
|
Ross PD and Subramanian S: Thermodynamics
of protein association reactions: Forces contributing to stability.
Biochemistry. 20:3096–3102. 1981.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Nigen M, Le Tilly V, Croguennec T,
Drouin-Kucma D and Bouhallab S: Molecular interaction between apo
or holo α-lactalbumin and lysozyme: Formation of heterodimers as
assessed by fluorescence measurements. Biochim Biophys Acta.
1794:709–715. 2009.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Rusinova E, Tretyachenko-Ladokhina V, Vele
OE, Senear DF and Alexander Ross JB: Alexa and Oregon Green dyes as
fluorescence anisotropy probes for measuring protein-protein and
protein-nucleic acid interactions. Anal Biochem. 308:18–25.
2002.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Laskowski RA and Swindells MB: LigPlot+:
multiple ligand-protein interaction diagrams for drug discovery. J
Chem Inf Model. 51:2778–2786. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Trott O and Olson AJ: AutoDock Vina:
Improving the speed and accuracy of docking with a new scoring
function, efficient optimization, and multithreading. J Comput
Chem. 31:455–461. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Morris GM, Goodsell DS, Halliday RS, Huey
R, Hart WE, Belew RK and Olson AJ: Automated docking using a
Lamarckian genetic algorithm and an empirical binding free energy
function. J Comput Chem. 19:1639–1662. 1998.
|
|
26
|
Lindahl E, Hess B and Van Der Spoel D:
GROMACS 3.0: A package for molecular simulation and trajectory
analysis. Mol Model Annu. 7:306–317. 2001. View Article : Google Scholar
|
|
27
|
Farasat A, Rahbarizadeh F, Hosseinzadeh G,
Sajjadi S, Kamali M and Keihan AH: Affinity enhancement of nanobody
binding to EGFR: In silico site-directed mutagenesis and molecular
dynamics simulation approaches. J Biomol Struct Dyn. 35:1710–1728.
2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Hansson T, Oostenbrink C and van Gunsteren
W: Molecular dynamics simulations. Curr Opin Struct Biol.
12:190–196. 2002.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Schmittschmitt JP and Scholtz JM: The role
of protein stability, solubility, and net charge in amyloid fibril
formation. Protein Sci. 12:2374–2378. 2003.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ashoka S, Seetharamappa J, Kandagal P and
Shaikh S: Investigation of the interaction between trazodone
hydrochloride and bovine serum albumin. J Lumin. 121:179–186. 2006.
View Article : Google Scholar
|
|
31
|
Vekshin IL: Separation of the tyrosine and
tryptophan components of fluorescence using synchronous scanning
method. Biofizika. 41:1176–1179. 1996.(In Russian). PubMed/NCBI
|
|
32
|
Gokara M, Sudhamalla B, Amooru DG and
Subramanyam R: Molecular interaction studies of trimethoxy flavone
with human serum albumin. PLoS One. 5(e8834)2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhong D, Douhal A and Zewail AH:
Femtosecond studies of protein-ligand hydrophobic binding and
dynamics: Human serum albumin. Proc Natl Acad Sci USA.
97:14056–14061. 2000.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Gokara M, Narayana VV, Sadarangani V,
Chowdhury SR, Varkala S, Ramachary DB and Subramanyam R:
Unravelling the binding mechanism and protein stability of human
serum albumin while interacting with nefopam analogues: A
biophysical and insilico approach. J Biomol Struct Dyn.
35:2280–2292. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Abreu RM, Froufe HJ, Queiroz MJR and
Ferreira IC: Selective flexibility of side-chain residues improves
VEGFR-2 docking score using AutoDock Vina. Chem Biol Drug Des.
79:530–534. 2012.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Mohan V, Gibbs AC, Cummings MD, Jaeger EP
and DesJarlais RL: Docking: Successes and challenges. Curr Pharm
Des. 11:323–333. 2005.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Gou Y, Zhang Z, Li D, Zhao L, Cai M, Sun
Z, Li Y, Zhang Y, Khan H, Sun H, et al: HSA-based multi-target
combination therapy: Regulating drugs' release from HSA and
overcoming single drug resistance in a breast cancer model. Drug
Deliv. 25:321–329. 2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Shahabadi N, Fili SM and Kashanian S:
Human serum albumin interaction studies of a new copper (II)
complex containing ceftobiprole drug using molecular modeling and
multispectroscopic methods. J Coord Chem. 71:329–341. 2018.
View Article : Google Scholar
|
|
39
|
Shahabadi N, Bazvandi B and Taherpour A:
Synthesis, structural determination and HSA interaction studies of
a new water-soluble Cu (II) complex derived from 1,
10-phenanthroline and ranitidine drug. J Coord Chem. 70:3186–3198.
2017. View Article : Google Scholar
|
|
40
|
Wang N, Ye L, Zhao BQ and Yu JX:
Spectroscopic studies on the interaction of efonidipine with bovine
serum albumin. Braz J Med Biol Res. 41:589–595. 2008.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Lori C, Lantella A, Pasquo A, Alexander
LT, Knapp S, Chiaraluce R and Consalvi V: Effect of single amino
acid substitution observed in cancer on Pim-1 kinase thermodynamic
stability and structure. PLoS One. 8(e64824)2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yuan T, Weljie AM and Vogel HJ: Tryptophan
fluorescence quenching by methionine and selenomethionine residues
of calmodulin: Orientation of peptide and protein binding.
Biochemistry. 37:3187–3195. 1998.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Li S, Huang K, Zhong M, Guo J, Wang WZ and
Zhu R: Comparative studies on the interaction of caffeic acid,
chlorogenic acid and ferulic acid with bovine serum albumin.
Spectrochim Acta A Mol Biomol Spectrosc. 77:680–686.
2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
OAbou-Zied OK and Al-Shihi OI:
Characterization of Subdomain IIA Binding Site of Human Serum
Albumin in its Native, Unfolded, and Refolded States Using Small
Molecular Probes. J Am Chem Soc. 130:10793–10801. 2008.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Hou HN, Qi ZD, Ouyang YW, Liao FL, Zhang Y
and Liu Y: Studies on interaction between Vitamin B12 and human
serum albumin. J Pharm Biomed Anal. 47:134–139. 2008.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Zhang HM, Chen TT, Zhou QH and Wang YQ:
Binding of caffeine, theophylline, and theobromine with human serum
albumin: A spectroscopic study. J Mol Struct. 938:221–228. 2009.
View Article : Google Scholar
|