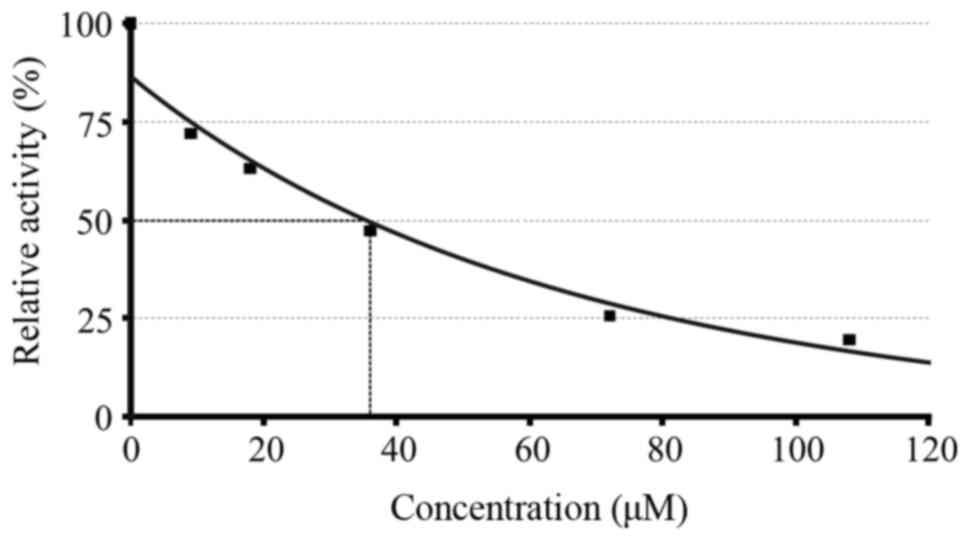
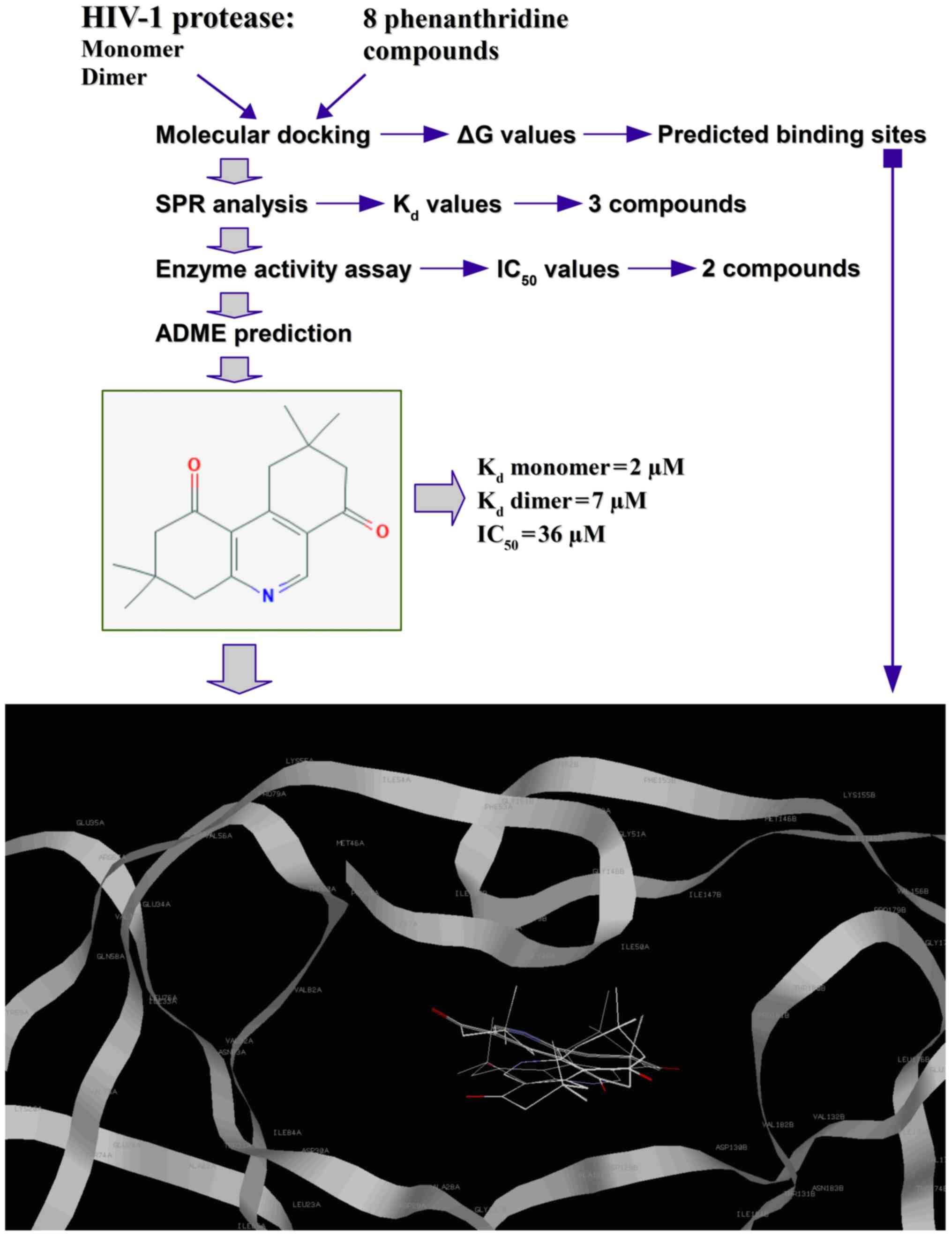
|
1
|
Azad I, Ahmad R, Khan T, Saquib M, Hassan
F, Akhter Y, Khan AR and Nasibullah M: Phenanthridine derivatives
as promising new anticancer agents: Synthesis, biological
evaluation and binding studies. Future Med Chem. 12:709–739.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Tumir LM, Radić Stojković M and Piantanida
I: Come-back of phenanthridine and phenanthridinium derivatives in
the 21st century. Beilstein J Org Chem. 10:2930–2954.
2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fuchino H, Kawano M, Mori-Yasumoto K,
Sekita S, Satake M, Ishikawa T, Kiuchi F and Kawahara N: In vitro
leishmanicidal activity of benzophenanthridine alkaloids from
Bocconia pearcei and related compounds. Chem Pharm Bull (Tokyo).
58:1047–1050. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wan M, Zhang L, Chen Y, Li Q, Fan W, Xue
Q, Yan F and Song W: Synthesis and anticancer activity evaluation
of novel phenanthridine derivatives. Front Oncol.
9(274)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Bernardo PH, Wan KF, Sivaraman T, Xu J,
Moore FK, Hung AW, Mok HY, Yu VC and Chai CL: Structure-activity
relationship studies of Phenanthridine-based Bcl-XL inhibitors. J
Med Chem. 51:6699–6710. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Thai KM, Bui QH, Tran TD and Huynh TN:
QSAR modeling on benzo[c]phenanthridine analogues as topoisomerase
I inhibitors and anti-cancer agents. Molecules. 17:5690–5712.
2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Inbar-Rozensal D, Castiel A, Visochek L,
Castel D, Dantzer F, Izraeli S and Cohen-Armon M: A selective
eradication of human nonhereditary breast cancer cells by
phenanthridine-derived polyADP-ribose polymerase inhibitors. Breast
Cancer Res. 11(R78)2009.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Ershov PV, Gnedenko OV, Molnar AA, Lisitsa
AV, Ivanov AS and Archakov AI: Biosensor analysis of the
interaction of potential dimerization inhibitors with HIV-1
protease. Biomed Khim. 55:462–478. 2009.PubMed/NCBI(In Russian).
|
|
9
|
Ershov PV, Gnedenko OV, Molnar AA, Lisitsa
AV, Ivanov AS and Archakov AI: Kinetic and thermodynamic analysis
of dimerization inhibitors binding to HIV protease monomers by
surface plasmon resonance. Biomeditsinskaya Khimiya. 58:43–49.
2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Darke PL: Stability of dimeric retroviral
proteases. Meth Enzymol. 241:104–127. 1994.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Weber IT, Kneller DW and Wong-Sam A:
Highly resistant HIV-1 proteases and strategies for their
inhibition. Future Med Chem. 7:1023–1038. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Weber IT and Agniswamy J: HIV-1 Protease:
Structural perspectives on drug resistance. Viruses. 1:1110–1136.
2009.PubMed/NCBI View
Article : Google Scholar
|
|
13
|
Hao P, Ren Y and Xie Y: Label-free
relative quantification method for Low-abundance glycoproteins in
human serum by micrOTOF-Q. J Chromatogr B: Analyt Technol Biomed
Life Sci. 877:1657–1666. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Purushothaman AK and Pemiah B: Ultra high
performance liquid chromatography-ultraviolet-electrospray
ionization-micrOTOF-Q II analysis of flavonoid fractions from
Jatropha tanjorensis. Pharmacogn Mag. 10 (Suppl 3):S472–S479.
2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Richards AD, Roberts R, Dunn BM, Graves MC
and Kay J: Effective blocking of HIV-1 proteinase activity by
characteristic inhibitors of aspartic proteinases. FEBS Lett.
247:113–117. 1989.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Daina A, Michielin O and Zoete V:
SwissADME: A free web tool to evaluate pharmacokinetics,
drug-likeness and medicinal chemistry friendliness of small
molecules. Sci Rep. 7(42717)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Qureshi A, Rajput A, Kaur G and Kumar M:
HIVprotI: An integrated web based platform for prediction and
design of HIV proteins inhibitors. J Cheminform.
10(12)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Pettersen EF, Goddard TD, Huang CC, Couch
GS, Greenblatt DM, Meng EC and Ferrin TE: UCSF Chimera-a
visualization system for exploratory research and analysis. J
Comput Chem. 25:1605–1612. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang J, Wang W, Kollman PA and Case DA:
Automatic atom type and bond type perception in molecular
mechanical calculations. J Mol Graph Model. 25:247–260.
2006.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shapovalov MV and Dunbrack RL: A smoothed
Backbone-dependent rotamer library for proteins derived from
adaptive kernel density estimates and regressions. Structure.
19:844–858. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Martin JL, Begun J, Schindeler A,
Wickramasinghe WA, Alewood D, Alewood PF, Bergman DA, Brinkworth
RI, Abbenante G, March DR, et al: Molecular recognition of
macrocyclic peptidomimetic inhibitors by HIV-1 protease.
Biochemistry. 38:7978–7988. 1999.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ortiz D, delToro D, Ordyan M, Pajak J,
Sippy J, Catala A, Oh CS, Vu A, Arya G, Feiss M, et al: Evidence
that a catalytic glutamate and an ‘Arginine Toggle’ act in concert
to mediate ATP hydrolysis and mechanochemical coupling in a viral
DNA packaging motor. Nucl Acid Res. 47:1404–1415. 2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Hsu KC, Chen YF, Lin SR and Yang JM:
iGEMDOCK: A graphical environment of enhancing GEMDOCK using
pharmacological interactions and Post-screening analysis. BMC
Bioinformatics. 12 (Suppl 1)(S33)2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Scott WR and Schiffer CA: Curling of flap
tips in HIV-1 protease as a mechanism for substrate entry and
tolerance of drug resistance. Structure. 8:1259–1265.
2000.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yu Y, Wang J, Chen Z, Wang G, Shao Q, Shi
J and Zhu W: Structural insights into HIV-1 protease flap opening
processes and key intermediates. RSC Adv. 7:45121–45128. 2017.
|
|
26
|
Babé LM, Pichuantes S and Craik CS:
Inhibition of HIV protease activity by heterodimer formation.
Biochemistry. 30:106–111. 1991.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Miller M, Schneider J, Sathyanarayana BK,
Toth MV, Marshall GR, Clawson L, Selk L, Kent SB and Wlodawer A:
Structure of complex of synthetic HIV-1 protease with a
substrate-based inhibitor at 2.3 A resolution. Science.
246:1149–1152. 1989.PubMed/NCBI View Article : Google Scholar
|