1. Introduction
Portal vein (PV) thrombosis (PVT) is a disease that
is relatively common in patients with liver cirrhosis, especially
in those with advanced-stage disease (1). Thrombosis in these patients is more
frequently chronic and asymptomatic and, in most cases, diagnosed
incidentally during routine examinations or pre-transplant
evaluation. Until recently, PVT was considered an absolute
contraindication for orthotopic liver transplantation (OLT) since
it was associated with decreased graft survival and increased
recipient mortality (2).
Nevertheless, advances in surgical and medical strategies have
contributed towards overcoming this problem, and therefore patients
with PVT are no longer excluded from OLT waiting lists (3). The prevalence of PVT in patients on a
waiting list for OLT may vary from 5-26%, with reported 1-year
de novo PVT rates of 7.4-8.4% (4,5). The
objective of the present review was to briefly discuss and
critically appraise the available treatment options for cirrhotic
patients enlisted for liver transplantation in the setting of
PVT.
2. Pathophysiology
Τhe pathophysiology of PVT is multifactorial. Liver
fibrosis causes increased intra-hepatic resistance, which results
in decreased portal blood flow, and this in-turn predisposes
patients to PV stasis and clot formation. Furthermore, portal
hypertension can lead to endothelial damage, which constitutes an
additional critical risk factor for thrombosis. These factors,
combined with a dysregulated hemostasis mechanism, are commonly
observed in patients with cirrhosis and may result in PVT (6-8).
In patients with cirrhosis, the presence of hepatocellular
carcinoma is also considered an important factor associated with
PVT formation, not only due to the direct invasion and compression
caused by the tumor mass, which is common, but also due to the
cancer-induced thrombotic state (6). Moreover, endotoxemia, previous
splenectomy and placement of transjugular intrahepatic
portosystemic shunt (TIPS), are considered triggering risk factors
for thrombus development due to the associated endothelial cell
injury and alterations caused to the portal circulation (6). Other thrombotic risk factors that have
been shown to result in PVT include sex, diabetes mellitus,
obesity, non-alcoholic steatohepatitis and decreased portal flow
velocity (<15 cm/sec) (9).
3. Diagnosis and classification
PVT may be acute or chronic. Patients with cirrhosis
and PVT are typically asymptomatic due to the gradual presentation
and slower evolution of the disease that results in the formation
of collateral circulation (10).
The majority of the cases are diagnosed incidentally during routine
examinations or pre-OLT evaluation. Ultrasound and Doppler
ultrasound are the imaging methods of choice that are used to
diagnose PVT (11). Ultrasound
indicates hyperechoic material within the vein and absent flow on
pulsed Doppler, which is suggestive of chronic PVT (12). The application of these methods
provides information on the direction of the flow and may be used
to measure the exact velocity of the PV trunk and intrahepatic
branches (8). Alternatively,
co-axial tomography and magnetic resonance imaging angiography can
also be used, providing additional information regarding the PV and
the neighboring vessels (Table I).
Given the high incidence and clinical impact of PVT on OLT, in
conjunction with the often unavoidable prolonged period on a
waiting list, repetitive screening of enlisted patients (every 6
months)is strongly recommended (13). Doppler ultrasound appears to be the
most feasible modality for such narrow screening (12). Over the past 20 years, several
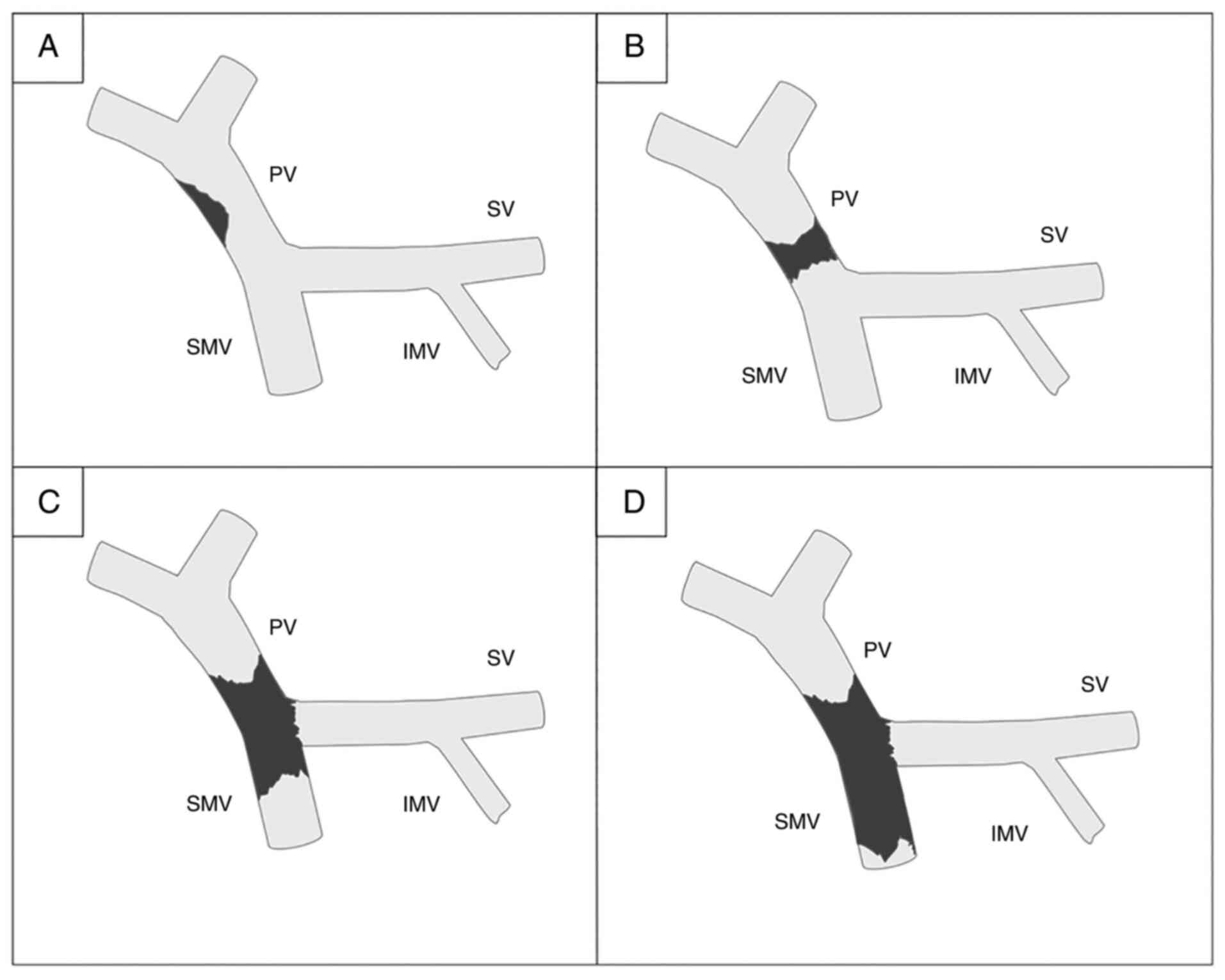
classification systems have been proposed for PVT, each with their
own advantages and disadvantages. The most well recognized
classification system remains that proposed by Yerdel et al
(14) in 2000, which represents an
anatomical grading system (Fig. 1).
A subsequent classification system was proposed by the Baveno VI
Consensus, which has been widely adopted, and includes underlying
liver disease but not liver functional status (Table II) (11). More recently, Sarin et al
(15) introduced a novel
classification system that included functional aspects of PVT, such
as symptomatology at presentation and duration of symptoms. The
challenge in establishing a widely accepted classification system
reflects the complexity in defining and describing this clinical
entity and the continuous effort in creating a communication
formula, which can be applied worldwide and can provide effective
screening and treatment protocols for these high-risk patients.
 | Table IAdvantages and disadvantages of
imaging modalities. |
Table I
Advantages and disadvantages of
imaging modalities.
| Imaging mode | Advantages | Disadvantages |
|---|
|
Ultrasound-Doppler | Direction of
flow | Radiologist's
expertise |
| | Exact velocity
measurement | |
| | Least expensive
method | |
| Co-axial tomography
angiography | Extension of
thrombus | X-ray exposure |
| | Neighboring vessels
imaging | Nephroxicity |
| | Bland vs. tumor
thrombus | |
| | Shunts imaging | |
| Magnetic resonance
imaging angiography | Extension of
thrombus | Low sensitivity in
ascites presence |
| | Neighboring vessels
imaging | Most expensive
method |
| | Superior in
detecting partial thrombosis | Claustrophobia |
| | Resectability of
neoplasm | |
 | Table IIBaveno VI-classification of PVT. |
Table II
Baveno VI-classification of PVT.
| Class | Description |
|---|
| Site of PVT | |
|
Type 1 | Only trunk |
|
Type 2a | Only branch |
|
Type 2b | Both branches |
|
Type 3 | Trunk and
branches |
| Presentation | |
|
Recent
(R) | Clinical
presentation and presence of hyperdense thrombus on imaging |
|
Chronic
(Ch) | With portal
cavernoma and clinical features of portal hypertension, no
hyperdense thrombus |
| Type of underlying
liver disease | |
|
C | Cirrhotic |
|
N | Non-cirrhotic liver
disease |
|
H | Hepatocellular
carcinoma and other local malignancies |
|
L | Post-liver
transplant |
|
A | Absence of
underlying liver disease |
| Degree of portal
venous system occlusion | |
|
I | Incomplete: Flow
visible in PV lumen through imaging |
|
T | Total: No flow
visible in PV lumen on imaging |
| Extent of PV system
occlusion | |
|
S | Splenic vein |
|
M | Mesenteric |
|
SM | Both splenic vein
and mesenteric |
However, it should be highlighted that the use of
this novel classification proposal by the Baveno IV Consensus or
Sarin consensus rather than Yerdel's classification, seems to offer
no clinical advantage or improvement in decision-making during OLT
of patients with PVT. It is noteworthy that classifications
incorporating functional aspects of PVT do not actually influence
the selection of portal inflow to the graft, since surgical
techniques are anatomically dependent and highly individualized
(16,17).
4. Management of PVT
The management of PVT in the setting of OLT is
initiated preoperatively with three basic options (4). Initially, systematic anticoagulation
agents, such as vitamin K antagonists and low molecular weight
heparin (LMWH) are administered particularly in acute PVT or in
cases that have a suspected extension of a chronic thrombus
(18). Despite the low rate of
recanalization in patients with complete thrombosis, the efficacy
of these agents is well demonstrated in partial thrombosis cases
with reported success rates of 75% (4,5). The
second approach involves the placement of TIPS in order to
re-establish PV flow and decrease portal hypertension symptoms by
producing a low resistance shunt between the PV and the hepatic
vein connection (6). Despite the
lack of robust evidence supporting either anticoagulation or TIPS
as a first line approach to treat PVT, TIPS appears to be more
effective in patients with extended thrombosis and cavernoma
(6). Luca et al (19) reported complete recanalization in
57% of patients and partial recanalization in 30% of patients with
PVT treated with TIPS. The TIPS dysfunction rate after 12 and 24
months was 38 and 85% for the bare stent, and 21 and 29% for the
covered stent, respectively (Table
III) (19).
 | Table IIIAnticoagulation vs. TIPS. |
Table III
Anticoagulation vs. TIPS.
| Treatment | Advantages | Disadvantages |
|---|
|
Anticoagulation | Non invasive | Difficulties in
dosage impairment at patients with renal failure |
| | Proven efficacy in
portal vein thrombosis | Increased
hemorrhage risk |
| | Reversibility prior
to OLT | Low percentages of
recanalization in cases of complete thrombosis |
| TIPS | Feasible in 70-80%
of patients | Technical
difficulties during OLT |
| | Recanalization
achieved in cases of complete thrombosis | Contraindication at
patients with high MELD score |
| | Low percentage of
dysfunction | Increased hepatic
encephalopathy risk |
Finally, thrombolysis represents a third approach of
preoperative PVT management. The supporting literature is weak with
only a limited number of cases reported (20). The surgical technique of OLT in the
setting of PVT is tailored based on the extent of the thrombus, the
presence of pre-existing portosystemic shunts (surgical or
spontaneous) and the expertise of the surgeon and center in
performing LT. For practical reasons, all possible reconstruction
techniques are primarily described according to the classical
classification proposed by Yerdel et al (14).
5. OLT techniques in cases with PVT
classified as YerdelI and II
Patients with PVT classified as Yerdel I and II can
be adequately treated by thrombectomy (simple, with eversion or
with the use of a Fogarty catheter), suggesting that a complicated
reconstruction is not required; if the thrombus extents to the
superior mesenteric or the splenic vein, a complete exposure of the
vessels may be required. However, this procedure entails the risk
of retro pancreatic hemorrhages (21). In certain cases,
thromboendovenectomy (removing of the intima) is performed in order
to achieve adequate inflow to the allograft (21). If none of the aforementioned methods
are efficient, the affected portion of the vein is removed.
Reconstruction with end-to-end portal anastomosis, or
reconstruction using an interposition vein graft is performed
(22). An additional anastomosis
can be performed just underneath the primary end-to-end portal
anastomosis, with or without an interposition graft of a nearby
portal branch, such as the coronary gastric vein or of an arterial
vessel. This aims to reinforce the blood flow (22).
6. OLT techniques in cases with PVT
classified as Yerdel III
In patients with total PV obstruction and thrombus
extension in the proximal superior mesenteric vein (SMV), a
complete thrombectomy is usually inadequate or not even feasible.
In these cases, a jump vein graft can be anastomosed directly to
the donor PV cephalad and at the distal free part of the SMV
caudal. A free segment of donor iliac vein, which is placed
anterior to the pancreas and posterior to the stomach, may serve as
a suitable vein graft for this type of reconstruction (4,22). An
artificial graft can also be used, originating either from the SMV
or the inferior mesenteric vein (23). Anastomosis can be made in a
side-to-end or an end-to-end manner in cases where the distal end
of the graft (synthetic or not) is anastomosed with the SMV. A
reasonable alternative option described by Magistri et al
(24) involves the placement of an
extra-anatomic jump graft from the right colic vein with donor
iliac vein interposition.
7. OLT techniques in cases with PVT
classified as Yerdel IV
In patients with diffuse splanchnic thrombosis, the
indicated technique depends on the presence of pre-existing
surgical or spontaneous portosystemic shunts. Cases with
pre-existing portosystemic shunts, adequate flow and vessel
diameter can be often used for portal anastomosis. The
reconstruction alternatives include the use of enlarged veins
(coronary, right/middle colic, ileocolic and right gastroepiploic),
or even pericholedochal varices for anastomosis to graft PV.
Moreover, in case of a patent splenorenal shunt, the left renal
vein can be disconnected at the junction with the vena cava
inferior (IVC) and anastomosed (directly or even with an
interposition graft) to the PV of the graft. A sufficient
mesenteriocaval shunt may also be used in same manner (16,25-28).
All these types of reconstruction are actually considered as
physiological since the graft inflow is supplied by the blood of
the PV system. The patients with an existing portosystemic shunt
that drains directly in the IVC may be treated with cavoportal
hemitransposition, which represents an alternative option (25,26).
In the absence of pre-existing shunts, alternative
options are limited. The three feasible techniques are: Cavoportal
hemitransposition, renoportal anastomosis (in end-to-end manner
with the left renal vein) and PV arterializations (22,29-31).
Multivisceral transplantation (MVT) including the liver, small
bowel, stomach and the pancreas has also been proposed as another
alternative. With the exception of MVT, all other reconstructions
are non-physiological since the inflow to the liver graft is
provided by the systemic venous or arterial systems (32).
If a cavoportal hemitransposition is recommended,
the anastomosis can be performed in an end-to-end or in
aside-to-end manner. The latter enables the IVC to be ligated or
partially narrowed (maintaining a bloodstream to right ventricle)
at a higher level, in order to enhance the blood inflow towards
graft PV. However, if a porto-portal anastomosis (with inadequate
flow) can be performed, an additional side-to-end
porto-cavalanastomosis, with or without venous interposition graft,
(above the level of the primary anastomosis) may also support the
graft supply (29,30).
A cavoportal hemitransposition may be accompanied by
several complications, primarily due to untreated portal
hypertension. Moreover, the occurrence of refractory ascites and
the risk of variceal bleeding can cause congestion of the systemic
venous system, potentially leading to gradual impairment of kidney
function and excessive peripheral edema. Finally, postoperative
gastrointestinal bleeding or development of vena cava thrombosis
may also occur (33).
The PV arterialization is mainly considered a
salvage procedure in cases of complete absence of portal flow. It
has been also proposed in revision operations following OLT
complicated with extensive postoperative portal thrombosis. In
selected cases, an arterial branch of graft celiac trunk may be
used to establish an additional anastomosis to the PV in order to
support its primarily insufficient flow (31). The first referrals to partial or
even complete PV arterialization involved usage of a donor's
splenic artery stump and preparation of an end-to-side anastomosis
between the subrenal aorta and the PV of the liver graft with
interposition of the donor's iliac artery graft (22,31).
It should be noted that in cases of arterialization, the portal
hypertension and its associated complications cannot be overcome.
Furthermore, the liver graft lacks nutritional factors derived from
the splanchnic venous system. Finally, due to increased
intraparenchymal portal pressure, arterialization is associated
with aneurysmatic malformations of intrahepatic portal branches and
progressive liver fibrosis (31,34).
The complex procedure of MVT causes re-establishment
of the blood flow by anastomosing the donor's celiac trunk with the
recipient's infrarenal aorta (35).
The first successful case of MVT was described in 2002 by Florman
et al (34) in a patient
with diffuse visceral splanchnic thrombosis and protein C
deficiency. A total of 25 cases with MVT for diffuse PVT have been
previously reported. The actual patient survival was 80, 72 and 72%
at 1-, 3- and 5 years, respectively (36).
In the early postoperative period, patients should
be treated with LMWH. Long-term anticoagulation therapy is not
indicated in patients with anatomical end-to end portal anastomosis
and adequate portal flow, given the absence of a thrombophilic
condition. The available data regarding the long-term use of
anticoagulation agents in cases with non-anatomical anastomosis for
extended thrombosis with a significantly higher risk of
re-thrombosis are limited (6).
8. Outcome of LT in the presence of PVT
Patients with PVT undergoing more complex operations
are at high-risk for various complications, affecting both graft
and patient survival. Recurrence of thrombosis ranges from 4-39%
amongst published studies during the early post-operative period
(37-39).
It should be noted that PVT recurrence within the early
postoperative period usually leads to graft loss, whereas a delayed
occurrence is mostly associated with re-development of portal
hypertension. These patients have a two-fold increased overall risk
of post-LT vascular thrombosis (in general) compared with those
transplanted without PVT. Furthermore, PVT is considered an
independent risk factor for thrombosis of the hepatic artery
(38). Higher rates of PV
re-thrombosis are primarily observed in cases of primary PVT
initially staged as Yerdel IV. Furthermore, the use of vascular
grafts during reconstruction is accompanied by significant risk of
re-thrombosis (~17% of cases) (38,39).
However the risk associated with the presence of vein grafts
appears to affect only the early postoperative period (37).
In addition to PVT stage Yerdel IV and the use of
vascular grafts, several additional predisposing factors for
re-thrombosis have been described. More specifically, incomplete
thrombectomy, low postoperative portal flow, the presence of
ascites and non-anatomical anastomoses are associated with higher
re-thrombosis rates (40). Several
types of vessel grafts can be used during OLT in cases of severe
PVT. Specifically, cadaveric-cryopreserved veins and arteries,
autologous veins or artificial grafts, such as
polytetrafluoroethylene, may all serve as potential grafts in cases
of reconstruction. Intima injury of cryo-preserved vessels, graft
kinking or graft length excess are risk factors predisposing an
individual to re-thrombosis (40).
Several studies have shown that the aforementioned grafts all have
similar efficacy (23,41,42).
Although autologous vessels are not immunogenic, cadaveric vessels
are much easier to obtain. Moreover, artificial grafts are
considered foreign bodies (40).
Taking into account the low blood flow in the portal venous system,
the long-term patency of artificial grafts remains questionable. In
patients with obesity, refractory ascites or increased
intra-abdominal pressure, such as in cases of portorenal shunts,
artificial or arterial grafts are suggested, since vein grafts are
more compressible (40,43).
The clinical impact of PVT on patient and graft
survival following OLT was assessed in a US-based retrospective
cohort study (Organ Procurement and Transplant Network database)
including all adult patients that underwent a primary OLT between
2002 and 2014(44). Data analysis
revealed that the presence of PVT either at listing or during OLT
exhibited a significantly negative effect on both patient and graft
survival following OLT. Moreover, cases with long standing PVT
exhibited significantly worse survival rates compared with that of
new cases of PVT (7). A possible
explanation of these findings is the fact that complete removal of
a new thrombus during OLT is more likely to be successful compared
with removal of an old and firmly organized clot.
A recent meta-analysis of 44 studies focused on the
mortality of OLT recipients with PVT (45). The 30-day and 1-year mortality rates
were significantly higher in recipients with PVT than in those
without PVT [odds ratio (OR) 2.29; P<0.0001 and OR 1.38;
P<0.0001, respectively]. In addition, the parameters 30-day
mortality (four relevant studies) and 1-year mortality (two
relevant studies) were higher in patients with complete PVT
compared with those noted in patients with partial PVT (OR 5.65;
P=0.001 and OR 2.48; P=0.05, respectively).
9. Conclusion
The prevalence of PVT amongst patients on waiting
lists is considerably high. Close follow-up of enlisted patients is
of cardinal importance in order to detect and initiate the
recommended medical or even radiological management regimes in
cases with PVT. PVT is no longer considered an absolute
contraindication for OLT. Due to the development of modern surgical
techniques combined with anticoagulation therapy, the postoperative
results of patients with partial or low grade PVT are comparable to
those noted in patients without PVT. Patients with PVT undergoing
OLT should be referred to highly experienced centers due to the
complexity of venous reconstructions that are occasionally
essential to be performed.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
EK, SK and NN conceived and designed the study. EK,
NM and GCS drafted the manuscript. EK, NM, GCS and NN revised the
manuscript for important intellectual content. All authors have
read and approved the final manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Basit SA, Stone CD and Gish R: Portal vein
thrombosis. Clin Liver Dis. 19:199–221. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Rodriguez-Castro KI, Porte RJ, Nadal E,
Germani G, Burra P and Senzolo M: Management of nonneoplastic
portal vein thrombosis in the setting of liver transplantation: A
systematic review. Transplantation. 94:1145–1153. 2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Qi X, Dai J, Jia J, Ren W, Yang M, Li H,
Fan D and Guo X: Association between portal vein thrombosis and
survival of liver transplant recipients: A systematic review and
meta-analysis of observational studies. J Gastrointestin Liver Dis.
24:51–59. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Francoz C, Valla D and Durand F: Portal
vein thrombosis, cirrhosis, and liver transplantation. J Hepatol.
57:203–212. 2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Conzen KD and Pomfret EA: Liver transplant
in patients with portal vein thrombosis: Medical and surgical
requirements. Liver Transpl. 23:S59–S63. 2017.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Mantaka A, Augoustaki A, Kouroumalis EA
and Samonakis DN: Portal vein thrombosis in cirrhosis: Diagnosis,
natural history, and therapeutic challenges. Ann Gastroenterol.
31:315–329. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Montenovo M, Rahnemai-Azar A, Reyes J and
Perkins J: Clinical impact and risk factors of portal vein
thrombosis for patients on wait list for liver transplant. Exp Clin
Transplant. 16:166–171. 2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Intagliata NM, Caldwell SH and Tripodi A:
Diagnosis, development, and treatment of portal vein thrombosis in
patients with and without cirrhosis. Gastroenterology.
156:1582–1599. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zocco MA, Di Stasio E, De Cristofaro R,
Novi M, Ainora ME, Ponziani F, Riccardi L, Lancellotti S,
Santoliquido A, Flore R, et al: Thrombotic risk factors in patients
with liver cirrhosis: Correlation with MELD scoring system and
portal vein thrombosis development. J Hepatol. 51:682–689.
2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chawla Y, Duseja A and Dhiman RK: Review
article: The modern management of portal vein thrombosis. Aliment
Pharmacol Ther. 30:881–894. 2009.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Faccia M, Ainora ME, Ponziani FR, Riccardi
L, Garcovich M, Gasbarrini A, Pompili M and Zocco MA: Portal vein
thrombosis in cirrhosis: Why a well-known complication is still
matter of debate. World J Gastroenterol. 25:4437–4451.
2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Harding DJ, Perera MT, Chen F, Olliff S
and Tripathi D: Portal vein thrombosis in cirrhosis: Controversies
and latest developments. World J Gastroenterol. 21:6769–6784.
2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
de Franchis R and Faculty BVI: Expanding
consensus in portal hypertension: Report of the baveno VI consensus
workshop: Stratifying risk and individualizing care for portal
hypertension. J Hepatol. 63:743–752. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yerdel MA, Gunson B, Mirza D, Karayalçin
K, Olliff S, Buckels J, Mayer D, McMaster P and Pirenne J: Portal
vein thrombosis in adults undergoing liver transplantation: Risk
factors, screening, management, and outcome. Transplantation.
69:1873–1881. 2000.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sarin SK, Philips CA, Kamath PS, Choudhury
A, Maruyama H, Nery FG and Valla DC: Toward a comprehensive new
classification of portal vein thrombosis in patients with
cirrhosis. Gastroenterology. 151:574–577. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bhangui P, Lim C, Levesque E, Salloum C,
Lahat E, Feray C and Azoulay D: Novel classification of
non-malignant portal vein thrombosis: A guide to surgical
decision-making during liver transplantation. J Hepatol.
71:1038–1050. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Mancuso A: Classification of portal vein
thrombosis in cirrhosis. Gastroenterology. 152(1247)2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Loudin M and Ahn J: Portal vein thrombosis
in cirrhosis. J Clin Gastroenterol. 51:579–585. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Luca A, Miraglia R, Caruso S, Milazzo M,
Sapere C, Maruzzelli L, Vizzini G, Tuzzolino F, Gridelli B and
Bosch J: Short- and long-term effects of the transjugular
intrahepatic portosystemic shunt on portal vein thrombosis in
patients with cirrhosis. Gut. 60:846–852. 2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
De Santis A, Moscatelli R, Catalano C,
Iannetti A, Gigliotti F, Cristofari F, Trapani S and Attili AF:
Systemic thrombolysis of portal vein thrombosis in cirrhotic
patients: A pilot study. Dig Liver Dis. 42:451–455. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Molmenti EP, Roodhouse TW, Molmenti H,
Jaiswal K, Jung G, Marubashi S, Sanchez EQ, Gogel B, Levy MF,
Goldstein RM, et al: Thrombendvenectomy for organized portal vein
thrombosis at the time of liver transplantation. Ann Surg.
235:292–296. 2002.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Stieber AC, Zetti G, Todo S, Tzakis AG,
Fung JJ, Marino I, Casavilla A, Selby RR and Starzl TE: The
spectrum of portal vein thrombosis in liver transplantation. Ann
Surg. 213:199–206. 1991.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Hwang HP, Yang JD, Bae SI, Hwang SE, Cho
BH and Yu HC: Usefulness of artificial jump graft to portal vein
thrombosis in deceased donor liver transplantation. Yonsei Med J.
56:586–590. 2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Magistri P, Tarantino G, Olivieri T,
Pecchi A, Ballarin R and Di Benedetto F: Extra-anatomic jump graft
from the right colic vein: A novel technique to manage portal vein
thrombosis in liver transplantation. Case Rep Surg.
2018(4671828)2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Davidson BR, Gibson M, Dick R, Burroughs A
and Rolles K: Incidence, risk factors, management, and outcome of
portal vein abnormalities at orthotopic liver transplantation.
Transplantation. 57:1174–1177. 1994.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Rudroff C and Scheele J: The middle colic
vein: An alternative source of portal inflow in orthotopic liver
transplantation complicated by portal vein thrombosis. Clin
Transplant. 12:538–542. 1998.PubMed/NCBI
|
|
27
|
Kato T, Levi DM, DeFaria W, Nishida S,
Pinna A, Nery J and Tzakis AG: A new approach to portal vein
reconstruction in liver transplantation in patients with distal
splenorenal shunts. Transplant Proc. 33(1326)2001.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Manzia TM, Fazzolari L, Manuelli M,
Pellicciaro M, Baiocchi L and Tisone G: Liver transplantation in a
patient with complete portal vein thrombosis, is there a surgical
way out? A case report. Ann Med Surg (Lond). 11:5–8.
2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Tzakis AG, Kirkegaard P, Pinna AD, Jovine
E, Misiakos EP, Maziotti A, Dodson F, Khan F, Nery J, Rasmussen A,
et al: Liver transplantation with cavoportal hemitransposition in
the presence of diffuse portal vein thrombosis. Transplantation.
65:619–624. 1998.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Azoulay D, Hargreaves GM, Castaing D and
Bismuth H: Caval inflow to the graft: A successful way to overcome
diffuse portal system thrombosis in liver transplantation. J Am
Coll Surg. 190:493–496. 2000.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Erhard J, Lange R, Giebler R, Rauen U, de
Groot H and Eigler FW: Arterialization of the portal vein in
orthotopic and auxiliary liver transplantation. A report of three
cases. Transplantation. 60:877–879. 1995.PubMed/NCBI
|
|
32
|
Vianna R and Beduschi T: Multivisceral
transplantation for diffuse splanchnic venous thrombosis. Curr Opin
Organ Transplant. 21:201–208. 2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Selvaggi G, Weppler D, Nishida S, Moon J,
Levi D, Kato T and Tzakis AG: Ten-year experience in porto-caval
hemitransposition for liver transplantation in the presence of
portal vein thrombosis. Am J Transplant. 7:454–460. 2007.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Florman SS, Fishbein TM, Schiano T,
Letizia A, Fennelly E and DeSancho M: Multivisceral transplantation
for portal hypertension and diffuse mesenteric thrombosis caused by
protein C deficiency. Transplantation. 74:406–407. 2002.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Tekin A, Beduschi T, Vianna R and Mangus
RS: Multivisceral transplant as an option to transplant cirrhotic
patients with severe portal vein thrombosis. Int J Surg.
82S:115–121. 2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Vianna RM, Mangus RS, Kubal C, Fridell JA,
Beduschi T and Tector AJ: Multivisceral transplantation for diffuse
portomesenteric thrombosis. Ann Surg. 255:1144–1150.
2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Nikitin D, Jennings LW, Khan T, Vasani S,
Ruiz R, Sanchez EQ, Chinnakotla S, Levy MF, Goldstein RM and
Klintmalm GB: Twenty years' follow-up of portal vein conduits in
liver transplantation. Liver Transpl. 15:400–406. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
38
|
Kim SJ, Yoon YC, Park JH, Oh DY, Yoo YK
and Kim DG: Hepatic artery reconstruction and successful management
of its complications in living donor liver transplantation using a
right lobe. Clin Transplant. 25:929–938. 2011.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Rhu J, Choi GS, Kwon CHD, Kim JM and Joh
JW: Portal vein thrombosis during liver transplantation: The risk
of extra-anatomical portal vein reconstruction. J Hepatobiliary
Pancreat Sci. 27:242–253. 2020.PubMed/NCBI View
Article : Google Scholar
|
|
40
|
Ozer A, Aktas H, Yilmaz TU, Can MG,
Gurluler E, Yildiz I and Emiroglu R: Liver transplant in patients
with portal vein thrombosis: The experience of 55 patients. Exp
Clin Transplant 8: doi: 10.6002, 2019.
|
|
41
|
Llado L, Fabregat J, Castellote J, Ramos
E, Torras J, Jorba R, Garcia-Borobia F, Busquets J, Figueras J and
Rafecas A: Management of portal vein thrombosis in liver
transplantation: Influence on morbidity and mortality. Clin
Transplant. 21:716–721. 2007.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Pomposelli JJ, Akoad M, Khwaja K, Lewis
WD, Cheah YL, Verbesey J, Jenkins RL and Pomfret EA: Evolution of
anterior segment reconstruction after live donor adult liver
transplantation: a single-center experience. Clin Transplant.
26:470–475. 2012.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Kim SJ, Kim DG, Park JH, Moon IS, Lee MD,
Kim JI, Yoon YC and Yoo YK: Clinical analysis of living donor liver
transplantation in patients with portal vein thrombosis. Clin
Transplant. 25:111–118. 2011.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Ponziani FR, Zocco MA, Senzolo M, Pompili
M, Gasbarrini A and Avolio AW: Portal vein thrombosis and liver
transplantation: Implications for waiting list period, surgical
approach, early and late follow-up. Transplant Rev (Orlando).
28:92–101. 2014.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Zanetto A, Rodriguez-Kastro KI, Germani G,
Ferrarese A, Cillo U, Burra P and Senzolo M: Mortality in liver
transplant recipients with portal vein thrombosis-an updated
meta-analysis. Transpl Int. 31:1318–1329. 2018.PubMed/NCBI View Article : Google Scholar
|