Introduction
Acute ischemic stroke (AIS) is the most common type
of stroke, which may lead to severe brain injury and further induce
a considerable disease burden, such as disability and even death
(1). In China, it is reported that
~90% of strokes are categorized as AIS and 15.5 million AIS
incidents were recorded in 2020, which was reported to be a leading
cause of disability and death between 2018 and 2020 (2,3). The
neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio
and C-reactive protein (CRP) are recognized as the common hallmark
of inflammation, whose level is aberrant in diseases characterized
by inflammation. Specifically, the NLR is found abnormal in
inflammatory bowel disease, diabetes mellitus, gastrointestinal
conditions, cardiac conditions, thyroiditis and severe acute
respiratory virus coronavirus (Covid) 2 infection (4-8);
CRP is increased in diabetes mellitus, thyroiditis, diabetic
neuropathy, hepatitis and Covid 2019 (Covid-19) infection (9-13);
the platelet-to-lymphocyte ratio also reflects inflammatory burden
in thyroid conditions, gastrointestinal diseases, thyroiditis,
cancer, diabetes mellitus, irritable bowel disease and Covid-19
infection (14-19).
During the progression of AIS, the inflammation reflected by the
NLR, platelet-to-lymphocyte ratio and CRP has a crucial role in
promoting ischemic injury, endothelial cell dysregulation and
neural death (20-24).
Once these ischemic damages occur, they may cause aggravated
neuroinflammation by inducing the release of reactive oxygen
species and pro-inflammatory cytokines (25,26).
Eicosapentaenoic acid (EPA) and docosahexaenoic acid
(DHA) are omega-3 fatty acids that serve as protective factors in
cerebral diseases; the optimal concentration of EPA and DHA is
nearly 8-11% and a lower level correlates with reduced brain
volume, aggravated brain damage and elevated risk for total
mortality and ischemic stroke (27,28).
The resolvin family is a group of anti-inflammatory mediators
originating from EPA and DHA, which was reported to improve the
prognosis of stroke (29). Resolvin
D1 (RvD1), the main derivative, is a mediator during the metabolic
process of DHA, which is closely engaged in inhibiting vascular
chronic inflammation, neuroinflammation, and neuronal injury
(30-34).
For instance, a previous study reported that RvD1 inhibits the
production of inflammatory cytokines from macrophages and
peripheral blood mononuclear cells (30). In another study, RvD1 was shown to
repress the microglial pro-inflammatory response, neuronal
oxidative damage and apoptosis through several potential pathways
such as the MAPK pathway (31).
Even though a small number of current studies have preliminarily
explored the dysregulation of RvD1 in patients with stroke and its
association with short-term functional outcomes, these are limited
by small sample size, lack of an extended follow-up and single time
assessment; therefore, a definite exploration of the clinical value
of RvD1 measurement in assessing AIS prognosis is still warranted
(35,36).
In the current study, the serum RvD1 levels of
patients with AIS on admission and at discharge were detected,
aiming to evaluate the relationship of RvD1 at different
time-points with inflammation, neurological function recovery and
risk of recurrence and death in patients with AIS.
Materials and methods
Patients
A total of 261 patients with newly diagnosed AIS
were recruited between July 2021 and May 2023. The inclusion
criteria were as follows: i) Patients with newly diagnosed AIS
following the American Stroke Association Guidelines (37); ii) patients aged ≥18 years; iii)
patients who were admitted within 24 h since AIS symptoms occurred;
iv) either the patients or their guardians consented to the
patients undergoing serum collection and follow-up. The exclusion
criteria involved: i) Patients with intracranial hemorrhage; ii)
patients with other cancers or hematological malignant diseases;
iii) patients with severe infections. Besides, a total of 30
healthy people who were age-, gender- and body mass index
(BMI)-matched with the AIS group were enrolled as healthy controls
(HCs). The eligibility criteria were: i) No abnormalities in recent
physical examination; ii) no history of alcohol and drug abuse;
iii) consent to participate in the study. The present study was
approved by the Ethics Committee of Tongren Hospital, Shanghai Jiao
Tong University School of Medicine (Shanghai, China; approval no.
2020-082-01). Informed consent was obtained from the patients or
their guardians and HCs.
Data documentation
The clinical characteristics of the patients with
AIS enrolled in the current study were collected on admission and
included demographics, comorbidities, National Institutes of Health
Stroke Scale (NIHSS) score (38),
timeframe since first AIS symptom to admission, fasting plasma
glucose (FBG), serum creatinine (Scr), serum lipid indexes, NLR,
CRP and treatment information.
Serum RvD1 detection
Serum was collected within 24 h of admission and on
the day of discharge from the patients with AIS and within 24 h
after enrollment from the HCs. Serum RvD1 was detected using an
ELISA kit (cat. no. EK11723; SAB Biotherapeutics, Inc.). The
experimental procedure was briefly as follows: First, a microplate
pre-coated with an antibody specific to RvD1 was prepared, then the
standards or samples were pipetted into the wells and RvD1 was
bound through the immobilized antibody. After washing, a
biotin-conjugated antibody specific for RvD1 and
streptavidin-conjugated horseradish peroxidase were added. Finally,
a substrate solution was added for color rendering and the optical
intensity was detected at 450 nm using a microplate photometer
(Multiskan FC; Thermo Fisher Scientific, Inc.). The test was
conducted following the manufacturers' instructions and each serum
sample was tested in triplicate.
Assessments
Patients with AIS were regularly followed up until
July 2023. During the follow-up, recurrence and death were
recorded. The modified Rankin scale (mRS) score was assessed at 3
months after enrollment (M3) (39).
mRS≤2 indicated favorable prognosis, while mRS>2 indicated poor
prognosis (40).
Statistical analysis
SPSS (version 26.0; IBM, Corp.) was used for data
analysis. The normality distribution analysis was performed via the
Kolmogorov-Smirnov test. Continuous variables were expressed as the
mean ± standard deviation or median (interquartile range) and count
data as n (%). Correlation analyses were performed using Spearman's
correlation. Comparison between groups and among multiple groups
was performed by the Wilcoxon rank-sum test and the Kruskal-Wallis
test, respectively. The effect size ‘Z’ was calculated by
the Wilcoxon rank-sum test and the effect size ‘H’ was
calculated by the Kruskal-Wallis test. Comparison of RvD1 levels at
admission and discharge was performed using the Wilcoxon
signed-rank test. The distinguishing ability of RvD1 was determined
by receiver operator characteristic (ROC) curve analyses. P<0.05
was considered to indicate statistical significance.
Results
Baseline characteristics of patients
with AIS and HCs
A total of 261 patients with AIS were included in
the present study with a mean age of 67.3±8.8 years, consisting of
170 males (65.1%) and 91 females (34.9%) (Table SI). The mean NIHSS score was
8.9±4.5. Furthermore, the NLR and CRP levels were 4.0 (2.9-6.0) and
4.5 (2.9-7.0) mg/l, respectively, in the patients with AIS. Other
detailed information is provided in Table I. In addition, the mean age of the
HCs was 66.6±6.9 years and the group comprised 10 (33.3%) females
and 20 (66.7%) males. The mean BMI of the HCs was 24.7±3.0
kg/m2. Of note, no difference was seen in age (P=0.661),
gender (P=0.867) or BMI (P=0.189) between patients with AIS and HCs
(Table SI).
 | Table IClinical characteristics of patients
with AIS (n=261). |
Table I
Clinical characteristics of patients
with AIS (n=261).
| Characteristic | Value |
|---|
| Age, years | 67.3±8.8 |
| Gender | |
|
Female | 91 (34.9) |
|
Male | 170 (65.1) |
| BMI,
kg/m2 | 25.4±2.7 |
| Current or former
smoker | 105 (40.2) |
| Hypertension | 213 (81.6) |
| Hyperlipidemia | 121 (46.4) |
| Diabetes | 74 (28.4) |
| Cardiovascular
disease | 111 (42.5) |
| Time since stroke
symptom to admission, h | 5.0 (3.0-7.0) |
| NIHSS score | 8.9±4.5 |
| FBG, mmol/l | 5.7 (4.9-6.8) |
| Scr, µmol/l | 84.5
(75.2-98.0) |
| TG, mmol/l | 1.8 (1.1-2.5) |
| TC, mmol/l | 4.8 (4.1-5.5) |
| LDL-C, mmol/l | 3.3 (2.7-4.2) |
| HDL-C, mmol/l | 1.0 (0.8-1.2) |
| NLR | 4.0 (2.9-6.0) |
| CRP, mg/l | 4.5 (2.9-7.0) |
| Treatment | |
|
IVT with
rtPA | 44 (16.9) |
|
IVT with
rtPA bridging to MT | 98 (37.5) |
|
IVT with
UK | 17 (6.5) |
|
IVT with UK
bridging to MT | 35 (13.4) |
|
MT | 67 (25.7) |
| Median follow-up
time, months | 11.4 |
| Follow-up range,
months | 1.1-21.0 |
| Recurrence | 17 (6.5) |
| Death | 6 (2.3) |
Comparison of serum RvD1 between
patients with AIS and HCs
The distribution of serum RvD1 in patients with AIS
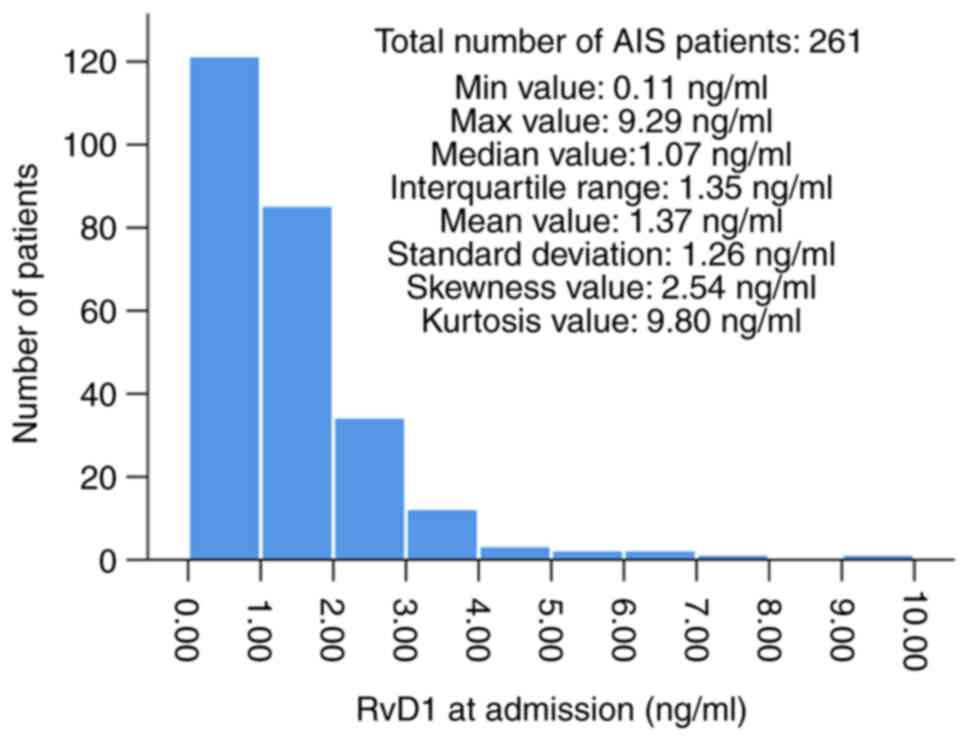
at admission is presented in Fig. 1
[median: 1.07 (range: 0.11-9.29) ng/ml]; specifically, serum RvD1
at admission was insufficient in most patients with AIS and only a
minority of patients had serum RvD1 at admission >4.00 ng/ml.
Besides, RvD1 was declined in patients with AIS compared to HCs
[median: 1.07 (range: 0.51-1.86) vs. 5.65 (range: 2.19-9.43) ng/ml,
P<0.001; Table SI].
Association of serum RvD1 with
comorbidities and common biochemical indexes in patients with
AIS
Serum RvD1 at admission was not significantly
associated with any comorbidities, including hypertension,
hyperlipidemia, diabetes and cardiovascular disease. Furthermore,
it was not correlated with any common biochemical indexes,
including FBG, Scr, triglyceride, total cholesterol, low- and
high-density lipoprotein cholesterol (all P>0.05; Table II).
 | Table IIComparison of RvD1 at admission in
patients with different characteristics. |
Table II
Comparison of RvD1 at admission in
patients with different characteristics.
| A, Categorical
variables |
|---|
| Item | RvD1 at admission,
ng/ml | Z | P-value |
|---|
| Hypertension | | -0.859 | 0.390 |
|
No | 1.14
(0.58-2.11) | | |
|
Yes | 1.05
(0.51-1.81) | | |
| Hyperlipidemia | | -1.378 | 0.168 |
|
No | 1.09
(0.56-1.87) | | |
|
Yes | 1.01
(0.45-1.82) | | |
| Diabetes | | -1.285 | 0.199 |
|
No | 1.09
(0.56-1.87) | | |
|
Yes | 0.91
(0.45-1.78) | | |
| Cardiovascular
disease | | -1.218 | 0.223 |
|
No | 1.08
(0.57-1.94) | | |
|
Yes | 1.05
(0.46-1.62) | | |
| B, Continuous
variables |
| Item | RvD1 at admission,
ng/ml | r | P-value |
| FBG | 1.07
(0.51-1.86) | -0.073 | 0.240 |
| Scr | 1.07
(0.51-1.86) | -0.031 | 0.618 |
| TG | 1.07
(0.51-1.86) | -0.108 | 0.083 |
| TC | 1.07
(0.51-1.86) | -0.073 | 0.241 |
| LDL-C | 1.07
(0.51-1.86) | -0.044 | 0.480 |
| HDL-C | 1.07
(0.51-1.86) | -0.059 | 0.340 |
Association of serum RvD1 at admission
with inflammation in patients with AIS
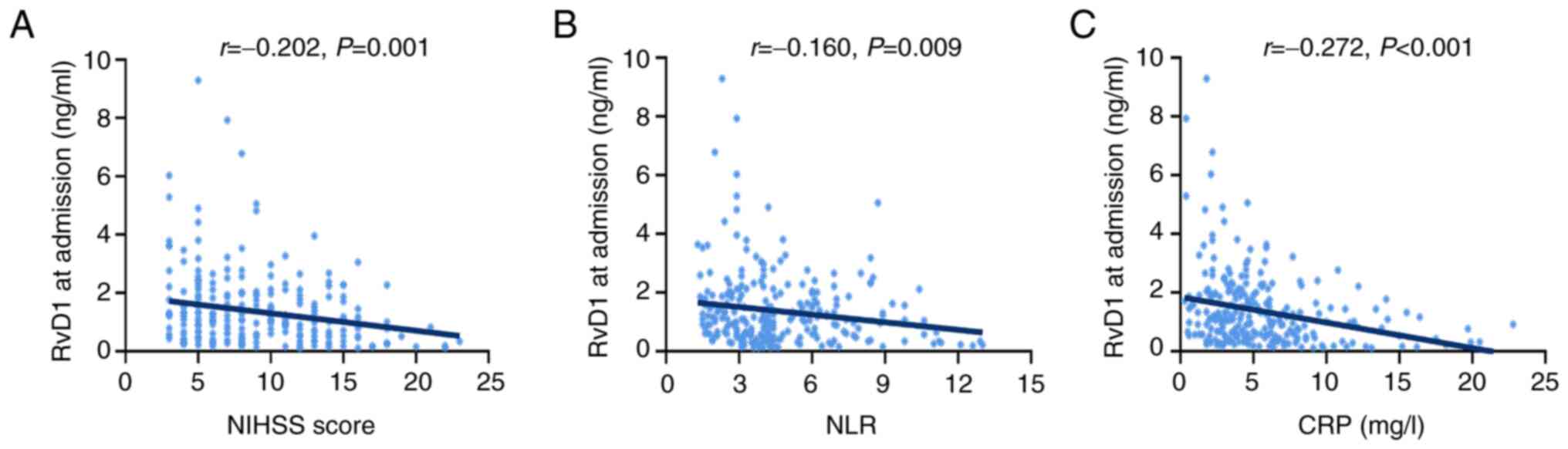
Serum RvD1 at admission was negatively correlated
with the neurofunctional status reflected by the NIHSS score
(r=-0.202; P=0.001; Fig. 2A). Of
note, serum RvD1 at admission was negatively correlated with the
NLR index (r=-0.160; P=0.009; Fig.
2B) and the CRP level (r=-0.272; P<0.001; Fig. 2C).
Relationship of serum RvD1 at
admission with neurofunctional rehabilitation, recurrence and death
in patients with AIS
During a median follow-up of 11.4 months (range,
1.1-21.0), there were 17 recurrence cases (6.5%) and six deaths
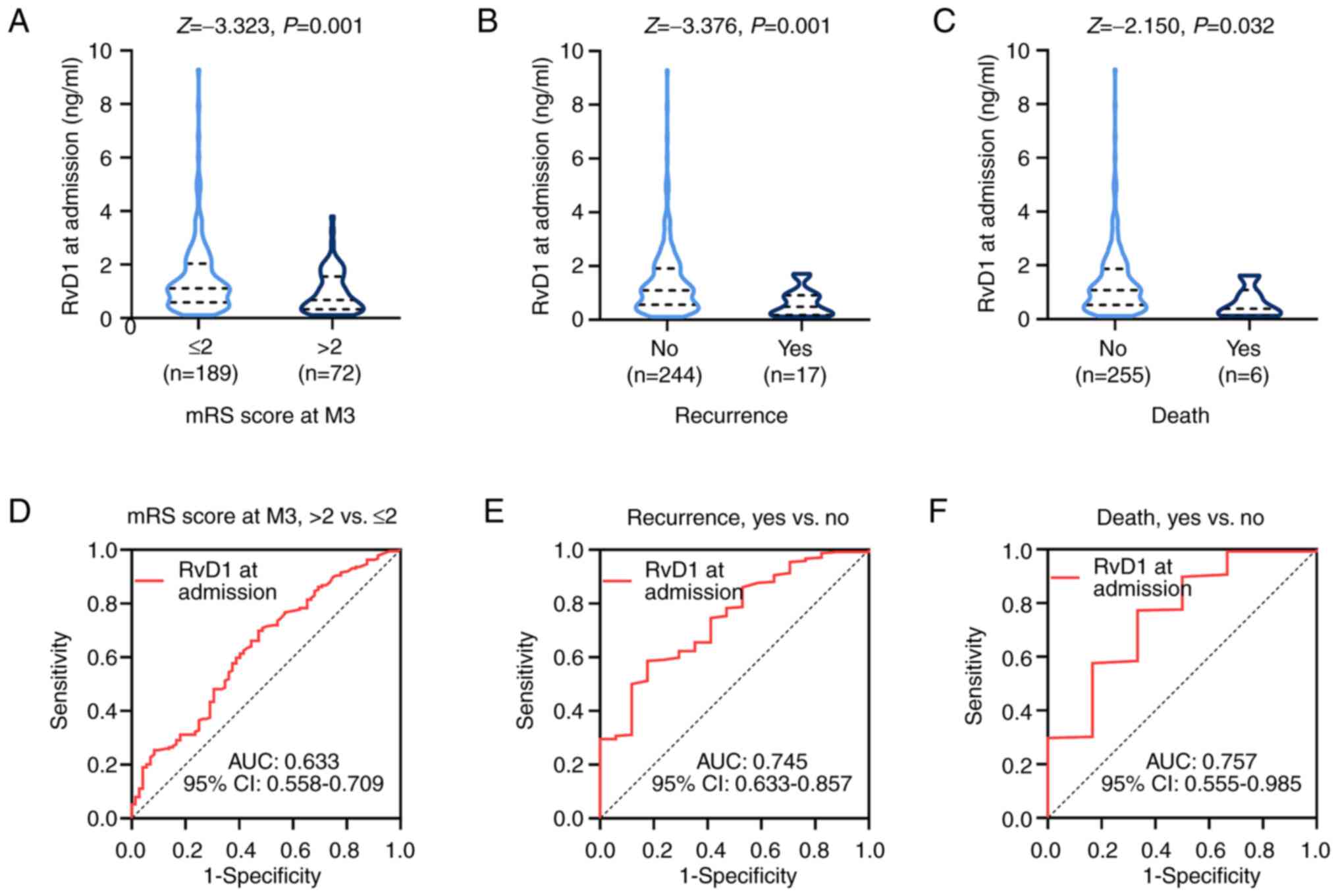
(2.3%) in the cohort of patients with AIS (Table I). Serum RvD1 at admission was lower
in patients with AIS and mRS score >2 compared with that in
patients with mRS score ≤2 at M3 (P=0.001; Fig. 3A). Furthermore, it was shown that
serum RvD1 at admission was also lower in patients who experienced
recurrence of AIS (P=0.001; Fig.
3B) or death (P=0.032; Fig. 3C)
during follow-up compared with those who did not.
Subsequent ROC curve analysis indicated that serum
RvD1 at admission may be used to estimate neurofunctional recovery
reflected by the mRS score at M3 [area under curve (AUC), 0.633;
95% CI, 0.558-0.709; Fig. 3D].
Serum RvD1 at admission also revealed acceptable values in
predicting risk of recurrence (AUC, 0.745; 95% CI, 0.633-0.857;
Fig. 3E) and death (AUC, 0.757; 95%
CI, 0.555-0.985; Fig. 3F).
Change of serum RvD1 from admission to
discharge in patients with AIS
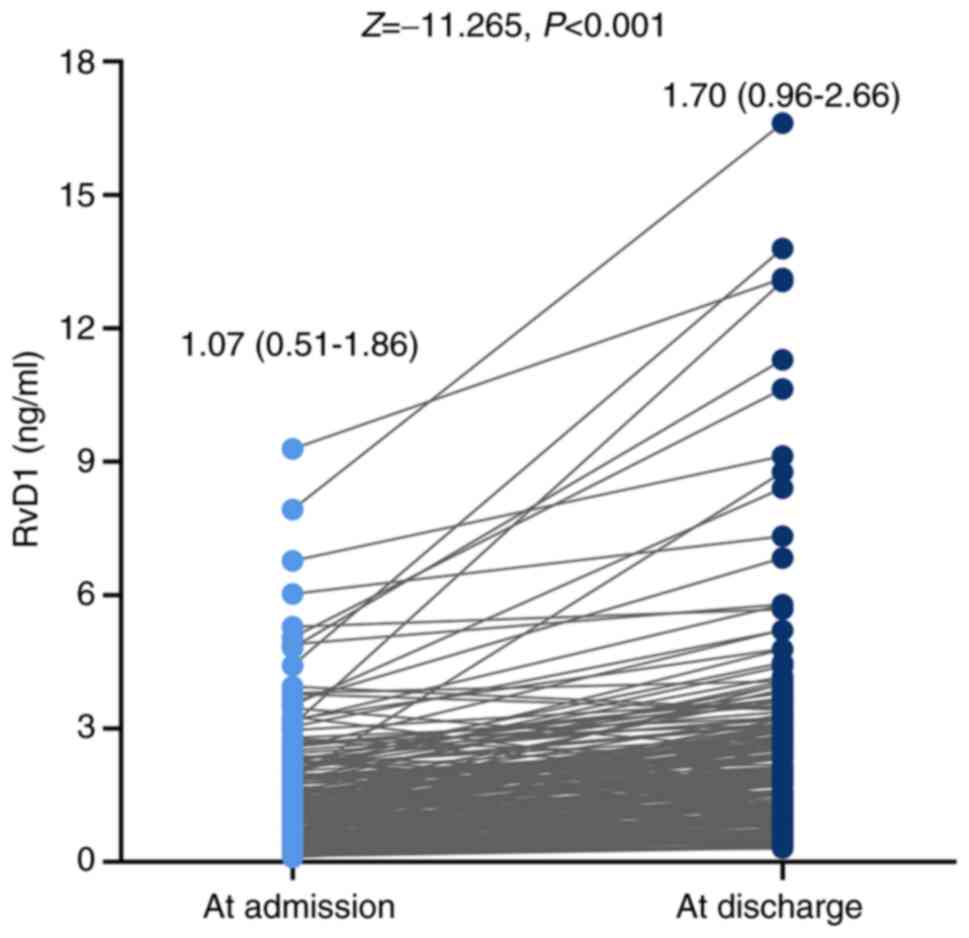
The median level of serum RvD1 was 1.07 (0.51-1.86)
ng/ml at admission, while it was 1.70 (0.96-2.66) ng/ml at
discharge. It was shown that the serum RvD1 was increased from
admission to discharge (P<0.001; Fig. 4). The median hospital stay of
patients with AIS was 15 days, ranging from 8 to 29 days. Besides,
serum RvD1 at admission (r=-0.086; P=0.165; Fig. S1A) and RvD1 at discharge (r=-0.097;
P=0.131; Fig. S1B) were not
associated with hospital stay in patients with AIS.
Relationship of serum RvD1 at
discharge with neurofunctional rehabilitation, recurrence and death
in patients with AIS
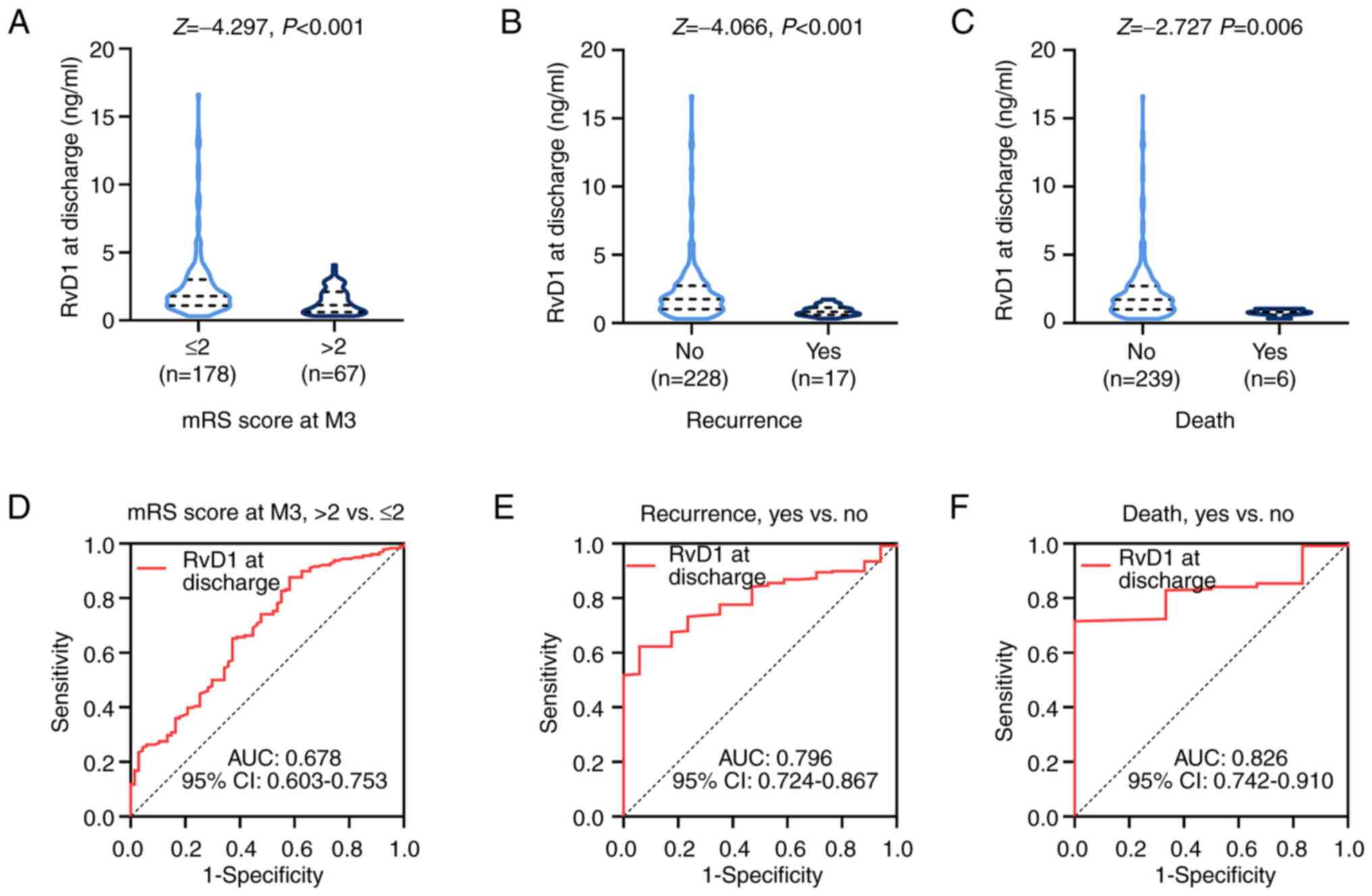
Serum RvD1 at discharge was decreased in patients
with AIS and an mRS score >2 compared with that in patients with
an mRS score ≤2 at M3 (P<0.001; Fig.
5A). It was also shown that serum RvD1 at discharge was
decreased in patients with AIS who experienced either recurrence
(P<0.001; Fig. 5B) or death
(P=0.006; Fig. 5C) compared with
that in those patients with AIS who did not.
ROC analysis indicated that the ability of serum
RvD1 at discharge to predict neurofunctional recovery reflected by
the mRS score at M3 was fair (AUC, 0.678; 95% CI, 0.603-0.753;
Fig. 5D). Of note, serum RvD1 at
discharge had a potential utility in predicting risk of recurrence
(AUC, 0.796; 95% CI, 0.724-0.867; Fig.
5E) and death (AUC, 0.826; 95% CI, 0.742-0.910; Fig. 5F). Referring to the AUC values, it
was shown that compared with the serum RvD1 level at admission,
serum RvD1 at discharge may be used for making predictions
regarding the prognosis of patients with AIS.
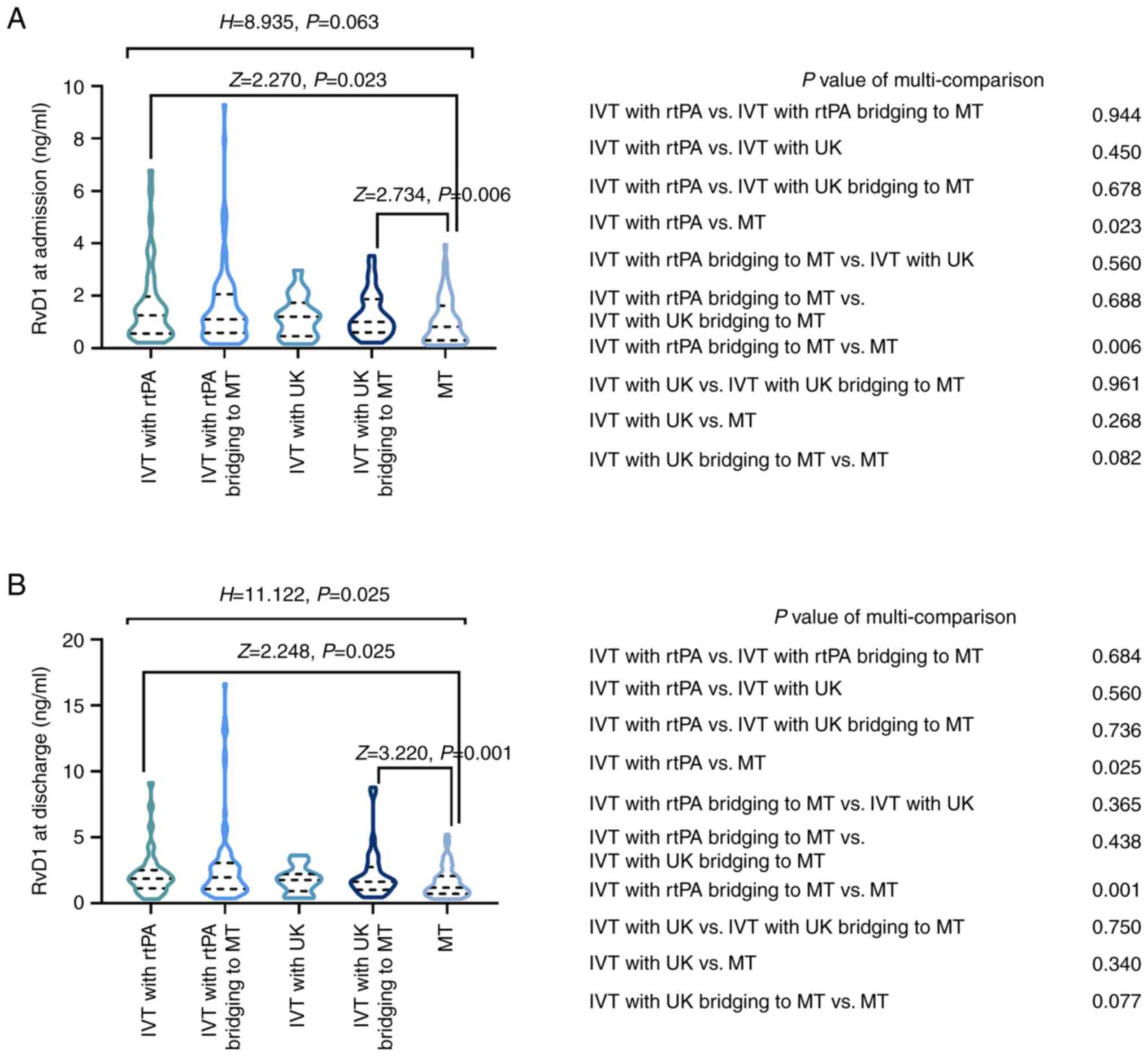
Comparison of serum RvD1 at admission
and discharge among patients with AIS receiving different
treatments
Serum RvD1 at admission did not differ among
patients with AIS receiving different regimens (P=0.063; Fig. 6A). However, subsequent
muti-comparison indicated that serum RvD1 at admission was higher
in patients with AIS receiving intravenous thrombolysis (IVT) with
recombinant tissue plasminogen activator (rtPA) compared with that
in patients receiving mechanical thrombectomy (MT) monotherapy
(P=0.023). In addition, serum RvD1 at admission was higher in
patients with AIS receiving IVT with rtPA bridging to MT compared
with that in patients receiving MT alone (P=0.006).
Serum RvD1 at discharge also differed among patients
with AIS receiving different regimens (P=0.025; Fig. 6B). Further muti-group comparison
indicated that serum RvD1 at discharge in patients with AIS
receiving IVT with rtPA was higher than that in patients receiving
MT monotherapy (P=0.025). Serum RvD1 was also higher in those
patients receiving IVT with rtPA bridging to MT compared with that
in patients receiving MT alone (P=0.001).
Discussion
RvD1 is a well-recognized endogenous
anti-inflammatory lipid mediator involved in acute inflammation,
chronic inflammation and neuroinflammation (33,41-44).
A previous study showed that RvD1 supports the resolution of acute
inflammation in mice with post-myocardial infarction (41). Another study indicated that RvD1
limits vascular chronic inflammation, thereby serving as a
potential strategy for atherosclerotic inflammation (33). Furthermore, a previous study
indicated that RvD1 was responsible for inflammation resolution in
early neuroinflammatory changes (42). However, the available clinical
evidence that supports the association of RvD1 with inflammation in
patients with AIS is currently insufficient. The present study
revealed that serum RvD1 was negatively correlated with CRP and NLR
in patients with AIS. The possible reasons behind this finding
include the following: i) RvD1 represses leukocyte recruitment and
infiltration in the ischemic core (32); and ii) RvD1 enhances the chemotaxis
of anti-inflammatory macrophages (M2-polarized macrophages) for
them to exert their anti-inflammatory properties (45,46).
Elevated serum RvD1 was associated with reduced CRP and NLR in
patients with AIS.
Sustained neuroinflammation and microglia activation
was observed to lead to neuronal damage and degeneration (47). Given that RvD1 ameliorates
neuroinflammation and inactivates inflammatory signaling in
microglial cells, its engagement in cerebral neurological
dysfunction has been revealed in several studies (36,48-50).
A previous study suggested that RvD1 triggers functional recovery
and neuroprotection after focal brain injury (49). Another study showed that RvD1 serves
as a beneficial factor for cognitive impairment following traumatic
brain injury by protecting astrocytic mitochondria (50). The present study revealed that
elevated serum RvD1 was associated with a reduced NIHSS score and
an mRS score ≤2 at M3 in patients with AIS. A likely explanation
could be that RvD1 suppressed microglia activation, inhibited
neuronal cell death and restrained neuroinflammation in remote
regions to improve neurological function (49,51).
As a result, increased serum RvD1 was related to improved
neurological rehabilitation in patients with AIS.
The present study also revealed elevation of serum
RvD1 from admission to discharge in patients with AIS, likely due
to the level of inflammation in patients with AIS being
correspondingly ameliorated by in-hospital treatment (52,53).
In addition, serum RvD1 is a widely endorsed anti-inflammatory
factor, the elevation of which represents resolution of acute
inflammation (54). Thus, serum
RvD1 in patients with AIS was increased at discharge compared with
that at admission. Furthermore, the present study also showed that
serum RvD1 was elevated in patients undergoing IVT with rtPA
compared with that in patients treated with MT alone. Also, RvD1
was increased in patients receiving IVT with rtPA bridging to MT
compared with that in patients treated with MT alone. A relevant
explanation may be that the selection of either IVT or MT was
mainly based on the time window as well as assessment of the
infarct core and salvageable penumbra; therefore, patients with AIS
who were ineligible for IVT tended to experience a higher level of
AIS severity, manifested by an elevated inflammatory level
(55). Combined with the finding
that serum RvD1 was negatively associated with inflammation as
mentioned above, patients with AIS receiving MT therefore had
reduced serum RvD1 compared with that in patients who received IVT
with rtPA or IVT with rtPA bridging to MT.
The value of RvD1 for estimating clinical outcomes
of certain cardiovascular and cerebrovascular events has been
previously reported (35,56,57).
According to a recent study, RvD1 has prognostic potency for its
association with left ventricular ejection fraction in patients
with ST-segment elevation myocardial infarction (56). In addition, another study indicated
that RvD1 may be used to predict early neurological deterioration
and worse outcomes in patients with acute supratentorial
intracerebral hemorrhage (57). The
current study showed that high serum RvD1 was associated with lower
risks of recurrence and death in patients with AIS. The likely
reasons for this finding include the following: i) RvD1 restrained
atherosclerotic plaque rupture and necrosis by modulating
macrophage-mediated clearance of necrotic cells; meanwhile, the
enhanced stability of atherosclerosis resulted in a reduced risk of
recurrence in patients with AIS (33,58);
and ii) RvD1 suppressed pro-fibrotic genes and collagen deposition
to protect against fibrosis, which alleviated central nervous
system injury and improved survival in patients with AIS (39,59).
Combining the aforementioned aspects, high serum RvD1 may reflect a
reduced risk of recurrence and death in patients with AIS. In
addition, it is noteworthy that the ability of serum RvD1 to
estimate the risks of recurrence and death was increased at
discharge compared with that at admission, which may be explained
by the fact that after treatment, the difference in every patient
with AIS was enhanced. Consequently, the clinical value of serum
RvD1 at discharge was greater than that of RvD1 at admission in
patients with AIS; however, further validation is necessary.
Some inevitable limitations should be mentioned: i)
In the current study, the mRS score was only analyzed within 3
months of patient enrolment, while the predictive role of RvD1 for
more extended neurological rehabilitation in patients with AIS
requires further exploration; ii) serum RvD1 was only determined at
admission and at discharge, and its longitudinal change in patients
with AIS remains elusive; iii) this study only enrolled patients
with AIS, but lacked a control group, which may cause confounders;
and iv) RvD1 levels were not determined in the cerebrospinal fluid
of patients with AIS, which requires further investigation.
Collectively, the present study indicated that
elevated serum RvD1 reflects improved inflammation status and is a
predictor of better neurological recovery and lower risks of
recurrence and death in patients with AIS. More specifically,
compared with the level of RvD1 on admission, the level of RvD1 at
discharge exhibits a higher predictive value in estimating
prognosis of AIS. The findings of the present study suggest that
patients with AIS with abnormally reduced RvD1 levels tend to have
inferior outcomes and require more intensive and specific treatment
in clinical practice.
Supplementary Material
Serum RvD1 at admission and discharge
was not related to hospital stay in patients with AIS. Correlation
of serum RvD1 at (A) admission and (B) discharge with hospital stay
in patients with AIS. RvD1, resolvin D1; AIS, acute ischemic
stroke.
Comparison of characteristics and RvD1
between patients with AIS and HCs.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
EC was responsible for study conception and design.
EC and DZ interpreted the data and drafted the manuscript. DZ and
RD helped with the data analysis and edited the manuscript. All
authors have read and approved the final version of the manuscript.
EC and DZ confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
The study gained the approval of the Ethics
Committee of Tongren Hospital, Shanghai Jiao Tong University School
of Medicine (Shanghai, China; approval no. 2020-082-01). Informed
consent was obtained from the patients or their guardians and the
HCs.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bathla G, Ajmera P, Mehta PM, Benson JC,
Derdeyn CP, Lanzino G, Agarwal A and Brinjikji W: Advances in acute
ischemic stroke treatment: Current status and future directions.
AJNR Am J Neuroradiol. 44:750–758. 2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Tu WJ, Zhao Z, Yin P, Cao L, Zeng J, Chen
H, Fan D, Fang Q, Gao P, Gu Y, et al: Estimated Burden of stroke in
China in 2020. JAMA Netw Open. 6(e231455)2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wu S, Wu B, Liu M, Chen Z, Wang W,
Anderson CS, Sandercock P, Wang Y, Huang Y, Cui L, et al: Stroke in
China: Advances and challenges in epidemiology, prevention, and
management. Lancet Neurol. 18:394–405. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Posul E, Yilmaz B, Aktas G and Kurt M:
Does neutrophil-to-lymphocyte ratio predict active ulcerative
colitis? Wien Klin Wochenschr. 127:262–265. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Duman TT, Aktas G, Atak BM, Kocak MZ,
Erkus E and Savli H: Neutrophil to lymphocyte ratio as an
indicative of diabetic control level in type 2 diabetes mellitus.
Afr Health Sci. 19:1602–1606. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Sahin S, Sarikaya S, Alcelik A, Erdem A
and Karaman KJAMM: Neutrophil to lymphocyte ratio is a useful
predictor of atrial fibrillation in patients with diabetes
mellitus. Acta Medica Mediterranea. 29:847–851. 2013.
|
|
7
|
Aktas G, Sit M, Dikbas O, Erkol H,
Altinordu R, Erkus E and Savli H: Elevated neutrophil-to-lymphocyte
ratio in the diagnosis of Hashimoto's thyroiditis. Rev Assoc Med
Bras (1992). 63:1065–1068. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Khalid A, Ali Jaffar M, Khan T, Abbas Lail
R, Ali S, Aktas G, Waris A, Javaid A, Ijaz N and Muhammad N:
Hematological and biochemical parameters as diagnostic and
prognostic markers in SARS-COV-2 infected patients of Pakistan: A
retrospective comparative analysis. Hematology. 26:529–542.
2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bilgin S, Kurtkulagi O, Atak BM, Duman TT,
Kahveci G, Khalid A and Aktas G: Does C-reactive protein to serum
Albumin Ratio correlate with diabEtic nephropathy in patients with
Type 2 dIabetes MEllitus? The CARE TIME study. Prim Care Diabetes.
15:1071–1074. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Demirkol ME and Aktas G: C-reactive
protein to LymphocytE count ratio could be a reliable mArkeR of
thyroiditis; the CLEAR-T study. Precision Med Sci. 11:31–34.
2022.
|
|
11
|
Mojahedi MJ, Bonakdaran S, Hami M,
Sheikhian MR, Shakeri MT and Aiatollahi H: Elevated serum
C-reactive protein level and microalbuminuria in patients with type
2 diabetes mellitus. Iran J Kidney Dis. 3:12–6. 2009.PubMed/NCBI
|
|
12
|
Demirkol ME, Aktas G, Bilgin S, Kahveci G,
Kurtkulagi O, Atak BM and Duman TT: C-reactive protein to
lymphocyte count ratio is a promising novel marker in hepatitis C
infection: The clear hep-c study. Rev Assoc Med Bras (1992).
68:838–841. 2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Demirkol ME, Bilgin S, Kahveci G,
Kurtkulagi O, Atak BM, Duman TT and Aktas G: C-reactive
protein-to-lymphocyte ratio is a reliable marker in patients with
COVID-19 infection: The CLEAR COVID study. Cir Cir. 90:596–601.
2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Afsin H and Aktas G: Platelet to
Lymphocyte and Neutrophil to Lymphocyte Ratios are useful in
differentiation of thyroid conditions with normal and increased
uptake. Ethiop J Health Dev. 35:149–153. 2021.
|
|
15
|
Erge E, Kiziltunc C, Balci SB, Atak BM,
Bilgin S, Duman TT and Aktas G: A novel inflammatory marker for the
diagnosis of hashimoto’s thyroiditis:
Platelet-count-to-lymphocyte-count ratio. Diseases.
11(15)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Atak BM, Kahveci G, Bilgin S, Kurtkulagi O
and Kosekli MA: Platelet to lymphocyte ratio in differentiation of
benign and malignant thyroid nodules. Exp Biomed Res. 4:148–153.
2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Atak B, Aktas G, Duman TT, Erkus E, Kocak
MZ and Savli H: Diabetes control could through
platelet-to-lymphocyte ratio in hemograms. Rev Assoc Med Bras
(1992). 5:38–42. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Aktas G, Duman TT, Atak BM, Kurtkulagi O,
Bilgin S, Basaran E, Demirkol ME and Kosekli MA: Irritable bowel
syndrome is associated with novel inflammatory markers derived from
hemogram parameters. Fam Med Prim Care Rev. 22:107–110. 2020.
|
|
19
|
Aktas G: Hematological predictors of novel
Coronavirus infection. Rev Assoc Med Bras (1992). 67 (Suppl
1):S1–S2. 2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Liu YL, Wu ZQ, Qu JF, Qiu DH, Luo GP, Yin
HP, Fang XW, Wang F and Chen YK: High neutrophil-to-lymphocyte
ratio is a predictor of poor short-term outcome in patients with
mild acute ischemic stroke receiving intravenous thrombolysis.
Brain Behav. 10(e01857)2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yang Y, Xie D and Zhang Y: Increased
Platelet-to-Lymphocyte Ratio is an Independent Predictor of
Hemorrhagic Transformation and In-Hospital Mortality among acute
ischemic stroke with large-artery atherosclerosis patients. Int J
Gen Med. 14:7545–7555. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Wang H, Zhang S, Xie L, Zhong Z and Yan F:
Neuroinflammation and peripheral immunity: Focus on ischemic
stroke. Int Immunopharmacol. 120(110332)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cheng Y, Zhang W, Cao W, Shao M, Lin Y,
Shao B, Yu H and Deng B: 2-BFI attenuates ischemic injury by
modulating mTOR signaling and neuroinflammation in rats. Neurosci
Lett. 750(135766)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhu H, Zhang Y, Zhong Y, Ye Y, Hu X, Gu L
and Xiong X: Inflammation-mediated angiogenesis in ischemic stroke.
Front Cell Neurosci. 15(652647)2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Charabati M, Zandee S, Fournier AP, Tastet
O, Thai K, Zaminpeyma R, Lecuyer MA, Bourbonniere L, Larouche S,
Klement W, et al: MCAM+ brain endothelial cells contribute to
neuroinflammation by recruiting pathogenic CD4+ T lymphocytes.
Brain. 146:1483–1495. 2023.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Weiner HL and Selkoe DJ: Inflammation and
therapeutic vaccination in CNS diseases. Nature. 420:879–884.
2002.PubMed/NCBI View Article : Google Scholar
|
|
27
|
von Schacky C: Importance of EPA and DHA
Blood Levels in Brain Structure and Function. Nutrients.
13(1074)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Sun GY, Simonyi A, Fritsche KL, Chuang DY,
Hannink M, Gu Z, Greenlief CM, Yao JK, Lee JC and Beversdorf DQ:
Docosahexaenoic acid (DHA): An essential nutrient and a
nutraceutical for brain health and diseases. Prostaglandins Leukot
Essent Fatty Acids. 136:3–13. 2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Tulowiecka N, Kotlega D, Prowans P and
Szczuko M: The role of resolvins: EPA and DHA derivatives can be
useful in the prevention and treatment of ischemic stroke. Int J
Mol Sci. 21(7628)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Liu G, Fiala M, Mizwicki MT, Sayre J,
Magpantay L, Siani A, Mahanian M, Chattopadhyay M, La Cava A and
Wiedau-Pazos M: Neuronal phagocytosis by inflammatory macrophages
in ALS spinal cord: Inhibition of inflammation by resolvin D1. Am J
Neurodegener Dis. 1:60–74. 2012.PubMed/NCBI
|
|
31
|
Liu GJ, Tao T, Wang H, Zhou Y, Gao X, Gao
YY, Hang CH and Li W: Functions of resolvin D1-ALX/FPR2 receptor
interaction in the hemoglobin-induced microglial inflammatory
response and neuronal injury. J Neuroinflammation.
17(239)2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Li L, Cheng SQ, Sun YQ, Yu JB, Huang XX,
Dong YF, Ji J, Zhang XY, Hu G and Sun XL: Resolvin D1 reprograms
energy metabolism to promote microglia to phagocytize neutrophils
after ischemic stroke. Cell Rep. 42(112617)2023.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Arnardottir H, Thul S, Pawelzik SC,
Karadimou G, Artiach G, Gallina AL, Mysdotter V, Carracedo M,
Tarnawski L, Caravaca AS, et al: The resolvin D1 receptor GPR32
transduces inflammation resolution and atheroprotection. J Clin
Invest. 131(e142883)2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Mena HA and Spite M: Proresolving receptor
tames inflammation in atherosclerosis. J Clin Invest.
131(e155240)2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Hao J, Feng Y, Xu X, Li L, Yang K, Dai G,
Gao W, Zhang M, Fan Y, Yin T, et al: Plasma lipid mediators
associate with clinical outcome after successful endovascular
thrombectomy in patients with acute ischemic stroke. Front Immunol.
13(917974)2022.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kotlega D, Peda B, Drozd A, Zembron-Lacny
A, Stachowska E, Gramacki J and Szczuko M: Prostaglandin E2, 9S-,
13S-HODE and resolvin D1 are strongly associated with the
post-stroke cognitive impairment. Prostaglandins Other Lipid
Mediat. 156(106576)2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Jauch EC, Saver JL, Adams HP Jr, Bruno A,
Connors JJ, Demaerschalk BM, Khatri P, McMullan PW Jr, Qureshi AI,
Rosenfield K, et al: Guidelines for the early management of
patients with acute ischemic stroke: A guideline for healthcare
professionals from the American Heart Association/American Stroke
Association. Stroke. 44:870–947. 2013.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Brott T, Adams HP Jr, Olinger CP, Marler
JR, Barsan WG, Biller J, Spilker J, Holleran R, Eberle R, Hertzberg
V, et al: Measurements of acute cerebral infarction: A clinical
examination scale. Stroke. 20:864–870. 1989.PubMed/NCBI View Article : Google Scholar
|
|
39
|
van Swieten JC, Koudstaal PJ, Visser MC,
Schouten HJ and van Gijn J: Interobserver agreement for the
assessment of handicap in stroke patients. Stroke. 19:604–607.
1988.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Suzuki K, Matsumaru Y, Takeuchi M,
Morimoto M, Kanazawa R, Takayama Y, Kamiya Y, Shigeta K, Okubo S,
Hayakawa M, et al: Effect of mechanical thrombectomy without vs
with intravenous thrombolysis on functional outcome among patients
with acute ischemic stroke: The SKIP Randomized clinical trial.
JAMA. 325:244–253. 2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Kain V, Ingle KA, Colas RA, Dalli J,
Prabhu SD, Serhan CN, Joshi M and Halade GV: Resolvin D1 activates
the inflammation resolving response at splenic and ventricular site
following myocardial infarction leading to improved ventricular
function. J Mol Cell Cardiol. 84:24–35. 2015.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Krashia P, Cordella A, Nobili A, La
Barbera L, Federici M, Leuti A, Campanelli F, Natale G, Marino G,
Calabrese V, et al: Blunting neuroinflammation with resolvin D1
prevents early pathology in a rat model of Parkinson's disease. Nat
Commun. 10(3945)2019.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Salas-Hernandez A, Espinoza-Perez C, Vivar
R, Espitia-Corredor J, Lillo J, Parra-Flores P, Sanchez-Ferrer CF,
Peiro C and Diaz-Araya G: Resolvin D1 and E1 promote resolution of
inflammation in rat cardiac fibroblast in vitro. Mol Biol Rep.
48:57–66. 2021.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wei C, Guo S, Liu W, Jin F, Wei B, Fan H,
Su H, Liu J, Zhang N, Fang D, et al: Resolvin D1 ameliorates
Inflammation-Mediated Blood-Brain Barrier Disruption After
Subarachnoid Hemorrhage in rats by Modulating A20 and NLRP3
Inflammasome. Front Pharmacol. 11(610734)2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Gemperle C, Tran S, Schmid M, Rimann N,
Marti-Jaun J, Hartling I, Wawrzyniak P and Hersberger M: Resolvin
D1 reduces inflammation in co-cultures of primary human macrophages
and adipocytes by triggering macrophages. Prostaglandins Leukot
Essent Fatty Acids. 174(102363)2021.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Titos E, Rius B, Gonzalez-Periz A,
Lopez-Vicario C, Moran-Salvador E, Martinez-Clemente M, Arroyo V
and Claria J: Resolvin D1 and its precursor docosahexaenoic acid
promote resolution of adipose tissue inflammation by eliciting
macrophage polarization toward an M2-like phenotype. J Immunol.
187:5408–5418. 2011.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Stuckey SM, Ong LK, Collins-Praino LE and
Turner RJ: Neuroinflammation as a key driver of secondary
neurodegeneration following stroke? Int J Mol Sci.
22(13101)2021.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Roohbakhsh A, Etemad L and Karimi G:
Resolvin D1: A key endogenous inhibitor of neuroinflammation.
Biofactors. 48:1005–1026. 2022.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Bisicchia E, Sasso V, Catanzaro G, Leuti
A, Besharat ZM, Chiacchiarini M, Molinari M, Ferretti E, Viscomi MT
and Chiurchiu V: Resolvin D1 Halts Remote neuroinflammation and
improves functional recovery after focal brain damage Via ALX/FPR2
Receptor-Regulated MicroRNAs. Mol Neurobiol. 55:6894–6905.
2018.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Ren YZ, Zhang BZ, Zhao XJ and Zhang ZY:
Resolvin D1 ameliorates cognitive impairment following traumatic
brain injury via protecting astrocytic mitochondria. J Neurochem.
154:530–546. 2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Jurcau A and Simion A: Neuroinflammation
in cerebral ischemia and ischemia/reperfusion injuries: From
pathophysiology to therapeutic strategies. Int J Mol Sci.
23(14)2021.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Chen PY, Chen GC, Hsiao CL, Hsu PJ, Yang
FY, Liu CY, Tsou A, Chang WL, Liu HH and Lin SK: Comparison of
clinical features, immune-inflammatory markers, and outcomes
between patients with acute in-hospital and out-of-hospital
ischemic stroke. J Inflamm Res. 15:881–895. 2022.PubMed/NCBI View Article : Google Scholar
|
|
53
|
DeLong JH, Ohashi SN, O'Connor KC and
Sansing LH: Inflammatory responses after ischemic stroke. Semin
Immunopathol. 44:625–648. 2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Recchiuti A: Resolvin D1 and its GPCRs in
resolution circuits of inflammation. Prostaglandins Other Lipid
Mediat. 107:64–76. 2013.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Phipps MS and Cronin CA: Management of
acute ischemic stroke. BMJ. 368(l6983)2020.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Karayigit O, Nurkoc SG, Basyigit F and
Kiziltunc E: The role of serum resolvin D1 levels in determining
the presence and prognosis of ST-Segment elevation myocardial
infarction. Med Princ Pract. 31:548–554. 2022.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Li W, Shan H, Ma Y, Lv X and Zhu S:
Prognostic significance of serum resolvin D1 levels in patients
with acute supratentorial intracerebral hemorrhage: A prospective
longitudinal cohort study. Clin Chim Acta.
547(117446)2023.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Bazan HA, Lu Y, Jun B, Fang Z, Woods TC
and Hong S: Circulating inflammation-resolving lipid mediators RvD1
and DHA are decreased in patients with acutely symptomatic carotid
disease. Prostaglandins Leukot Essent Fatty Acids. 125:43–47.
2017.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Fehlberg CR and Lee JK: Fibrosis in the
central nervous system: From the meninges to the vasculature. Cell
Tissue Res. 387:351–360. 2022.PubMed/NCBI View Article : Google Scholar
|