1. Introduction
Cancer remains a leading cause of premature death
worldwide, with 9.7 million cancer-related deaths and an estimated
19.3 million new cancer cases in 2022(1). Africa and Asia experience
disproportionate mortality rates compared to incidence rates
relative to other regions of the world which can be attributed to
multiple factors including delayed diagnosis, unequitable access to
quality health care and higher prevalence of infections with
cancer-associated pathogens. The majority of patients with cancer
choose conventional therapies which include surgery, chemotherapy,
and radiotherapy, and for some cancers, immunotherapy, hormone
therapy, and other precision-based therapies (2). Most patients with cancer receive a
combination of these treatment modalities, for instance, surgery
followed by radiotherapy and/or chemotherapy. The effectiveness of
the therapy is dependent on a number of interrelated factors, with
the type and stage of cancer, and the overall health of the patient
being major determinants.
Complementary and Alternative medicines (CAMs)
include healthcare practices which are not part of standard and
approved medical care, and an estimated 25-84% of patients with
cancer use CAM for reasons ranging from personal and cultural
beliefs, distrust of modern medicine, or in an attempt to manage
the adverse side-effects of conventional treatments (3). The proportion of patients with cancer
who primarily use CAMs is higher in developing countries, and this
is thought to result from cultural beliefs and practices within
specific communities. WHO estimates show that >80% of the
population in certain developing countries primarily use CAMs for
their health care needs (4). CAM
therapies, unlike conventional Western medicine, are not regulated
or controlled by any governmental health agency such as the Food
and Drug Administration, and although often categorized as
‘natural’, their use may prove harmful to users due to toxicity
and/or incompatibility with conventional treatments if taken
concurrently (5).
Nevertheless, natural products have played a crucial
role in the development of modern-day cancer therapies and remain
an essential source for the discovery and development of new drugs.
Due to their scaffold diversity and structural complexity, natural
products are structurally optimized by evolution to achieve highly
adapted and specific biological functions and as such provide an
important foundation to serve as lead molecules in the development
of novel and more effective pharmaceuticals (6). With the continued battle of drug
resistance and worsening of side effects, there have been renewed
efforts in the inclusion of naturally occurring compounds within
drug discovery platforms (7). These
include derivatives from medicinal plants, which, for centuries,
have been utilized by communities to treat an array of illnesses
(8).
2. Plant-derived therapeutics
The oldest evidence of the use of plants for
therapeutic purposes was found on a Sumerian clay slab from Nagpur,
which dates back 5000 years (9).
Historical records on the use of plants as powders, teas, tinctures
and poultices can be found in Chinese, Indian, Arab and Egyptian
cultures (8,10). Some currently in-use
chemotherapeutic drugs of herbal origin include: i) Paclitaxel, a
terpenoid isolated from the bark and needles of the Pacific yew
tree, used in the treatment of several cancers including breast,
ovarian, and lung cancer (11,12);
ii) etoposide, a non-alkaloid lignan derivative isolated from the
rhizomes and roots of the Mayapple, Podophyllum
peltatum/emodi used in the treatment testicular cancer,
non-Hodgkin lymphoma and several other cancers (13,14);
and iii) vincristine, a vinca alkaloid isolated from the leaves of
the Madagascar periwinkle, Catharanthus roseus, used to
treat non-Hodgkin lymphoma, breast cancer and leukemia (15,16).
Plant-derived bioactive compounds continue to be a valuable source
of anticancer drugs and documenting of current knowledge around the
use and properties of medicinal plants is therefore useful in the
developmental pipeline of these naturally derived drugs.
The evergreen shrub Dodonaea viscosa (DV) is
widely reported for its medicinal use in complementary medicine,
prescribed by ‘traditional healers’ or alternative medicine
practitioners in several countries within the tropical and
sub-tropical regions, including Australia, India, Southern African
countries, Mexico, Pacific Islands, the Caribbean, Southeast Asia,
and parts of the Middle East (Fig.
1A) (17). The bark and leaves
are the most commonly used parts of the plant and used as tea
infusions for a range of ailments including colds, influenza,
digestive disorders, thrush, and measles (18,19).
Leaf preparations are indicated for external use to treat skin
rashes, topical infections and wounds. To date, the therapeutic
properties of the plant and its derivatives, as well as the
mechanisms of action, remain largely undefined scientifically, but
reports have associated antimicrobial, anti-inflammatory,
anti-diarrheal, and anti-proliferative properties to both aqueous
and organic extracts of the plant, with biochemical analyses
revealing a broad but typical array of phytochemicals including
phenols, saponins, tannins and flavonoids (20-28).
Among the most abundant phytochemicals found in DV leaves, six are
shown in Fig. 1B, purified using
various extraction methods, and numerous additional chemical
isolates have been reported (20,21).
Of those, only few have been investigated for their pharmacological
activities to support potential health benefits. Some of the more
comprehensive studies are described further in the sections which
follow.
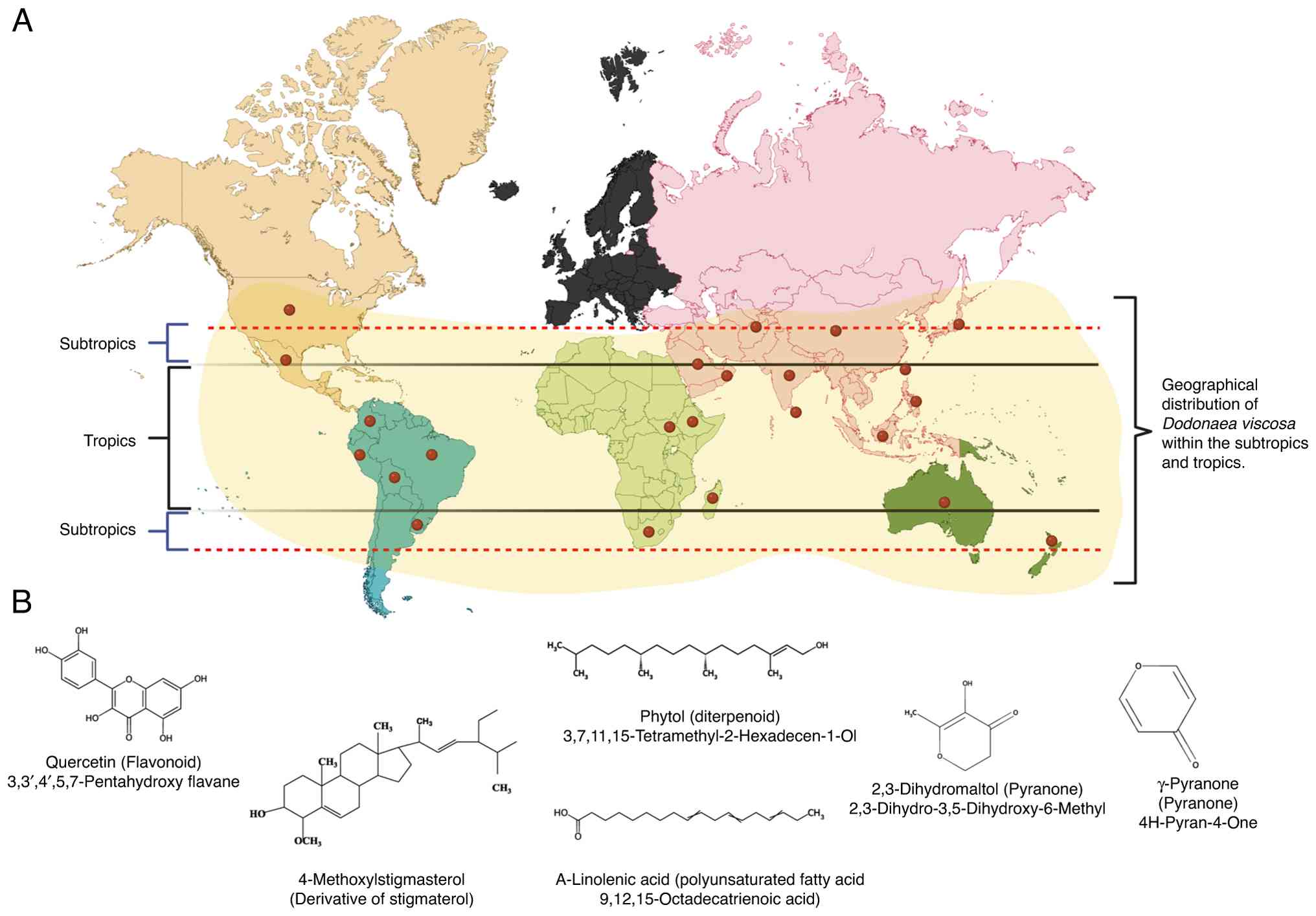 | Figure 1Geographical distribution and major
phytocomponents of DV. (A) Worldwide view showing reported
geographical distribution of DV in the tropics and subtropics.
Alphabetical list of countries where DV is used in traditional
medicine: Afghanistan, Australasia and Pacific, Australia, Bolivia,
Brazil, China, Colombia, Ethiopia, India, Indonesia, Japan,
Madagascar, Mexico, New Zealand, Oman (Arabian Peninsula), Peru,
Philippines, South Africa, Sri Lanka, Sudan, Taiwan, Uruguay,
Hawaii. (B) Names and structural representation of six of the
prevailing compounds isolated from DV leaves using gas
chromatography-mass spectroscopy (20,21).
DV, Dodonaea viscosa. |
3. Laboratory investigations involving DV
extracts
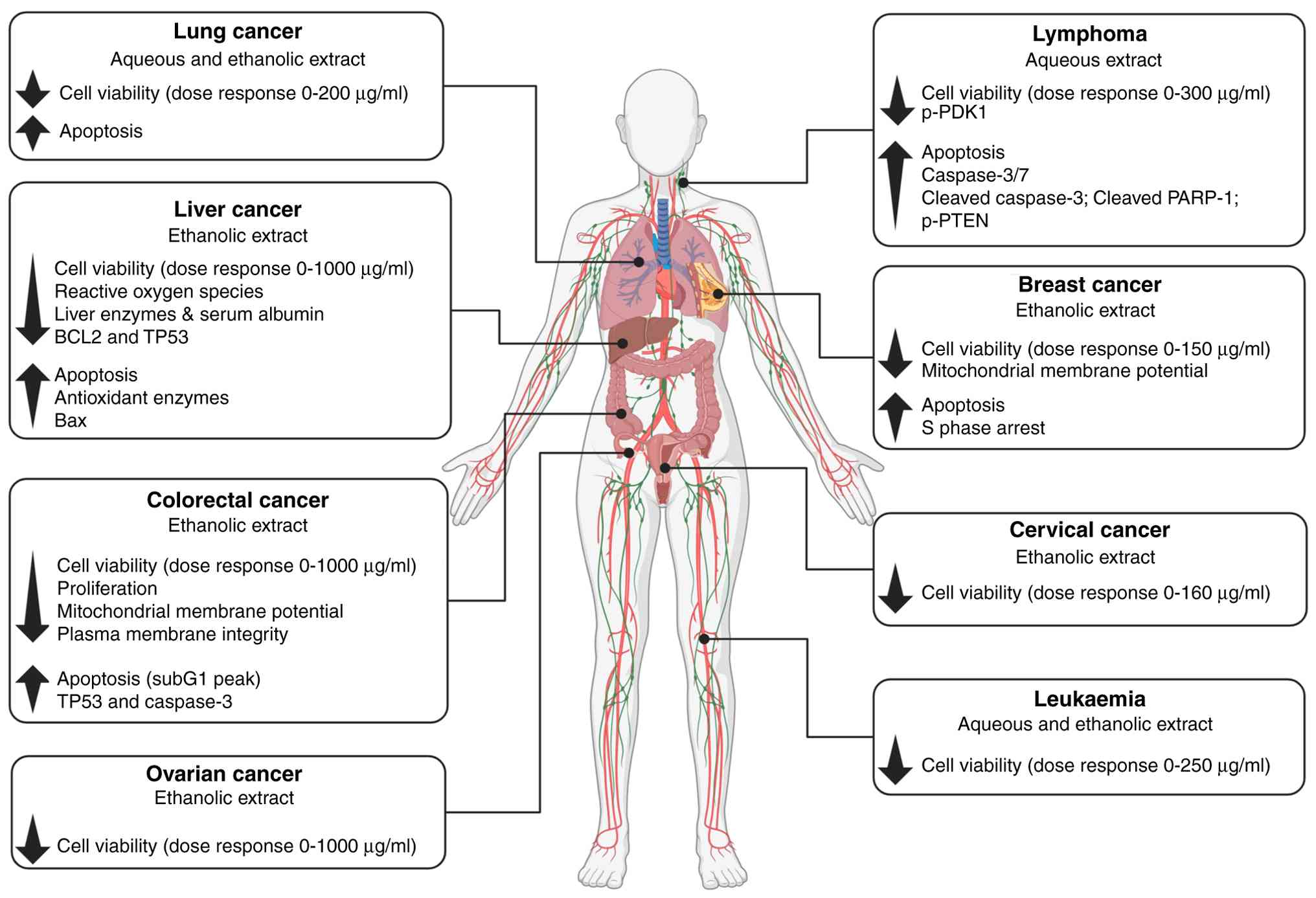
Anticancer properties
The antiproliferative effects of DV extracts, most
often demonstrated using in vitro cell viability assays,
have been reported on a range of cancer cell types, primarily of
epithelial origin, namely colon, cervical, ovarian, and breast
cancers (23-28)
(Table I). Studies using the breast
cancer cell lines MCF-7 and MDA-MB231 found that DV leaf extracts,
prepared using dried leaves ground to a powder and thereafter
suspended in solvent, potently inhibited proliferation, via S phase
arrest, with an IC50 of 75 µg/ml (23,24).
In a study assessing crude ethanolic extracts of DV leaves, as well
as purified fractions (hexane, chloroform, ethyl acetate, butanol
and aqueous fractions), the growth of the established human
colorectal adenocarcinoma cell line HT29 was found to be inhibited
(25). The mouse 3T3 embryonic
non-malignant cell line was also included for comparison. The crude
ethanol extract and the chloroform fraction in particular showed
significant selectivity towards the cancer cell line, with the
chloroform fraction displaying an impressive 12-fold reduced
IC50 value relative to the 3T3 cells. However, the two
major limitations of the study were inclusion of only one
representative cancer cell line, and the lack of directly
comparable non-malignant cell lines as controls (25). In a subsequent study, the same
researchers fractionated and analysed hydroethanolic extracts of DV
leaves and identified over 50 individual chemical constituents, a
majority of which were flavonoids and diterpenoids (26). They found the hydroethanolic DV
extract to be slightly more selective towards the two human
colorectal cancer cell lines SW480 and SW620, compared with Chinese
hamster ovary cells (CHO-1) and the human benign keratinocyte cell
line HaCaT. The effects on cell proliferation and cell death were
demonstrated using several standard assays, including analysis of
cell morphology using microscopy, induction of apoptosis using
Annexin V-PI labelling, and assessment of mitochondrial membrane
potential disruptions (26).
Extractions using a range of solvents, as well as combinations of
solvents, found the flowers and leaves of the plant to yield the
highest percentage extract, relative to the stems and roots
(27). Notably, the yield varied
depending on the solvent type, indicating a richness of
phytoconstituents with diverse chemical composition. Except for the
flower-derived extract, fractions showed potent cytotoxicity
against human leukemic (THP-1) and liver (Hep-G2) cell lines,
although selectivity towards cancer cells remains undetermined due
to the absence of non-malignant control cells (27). In yet another study, the
antiproliferative activity of two purified root extracts, both
triterpenoid saponins, was demonstrated against the human ovarian
cancer cell line A2780(28), while
fractions from ethanolic crude extracts prepared from leaves showed
growth inhibition in the human lung adenocarcinoma cell line
A549(29). These studies are
summarized in Fig. 2, and provide
strong evidence for the DV plant as a potential lead source of
anticancer bioactive phytochemicals, although limitations remain
including the robustness of some of the data especially related to
selectivity towards malignant cells. To date, few studies have
reported on the cellular pathways mediating the cytotoxic mechanism
of action of DV extracts. A recent study revealed potent and
selective killing of Burkitt lymphoma cells, mediated, in part,
through inhibition of the oncogenic P13K/Akt pathway (30).
 | Table IReported cytotoxicity (in
vitro and in vivo) of Dodonaea viscosa extracts
on cancer cells. |
Table I
Reported cytotoxicity (in
vitro and in vivo) of Dodonaea viscosa extracts
on cancer cells.
| Cancer type | Experimental
approach | Findings | (Refs.) |
|---|
| Breast cancer | MDA-MB232 cell
line: | | (23) |
| | • MTT assays | • Inhibited cell
viability | |
| | • Annexin V
assay | • Induced apoptosis
(increase in late and early apoptotic cells) | |
| | • Microscopic
analysis (cell morphology) | • Decrease in cell
density | |
| | • Mitochondrial
membrane potential (Rhodamine 123) | • Decrease in
mitochondrial membrane potential | |
| | • Cell cycle
profiling | • Cell cycle arrest
in the S phase | |
| | MCF-7 cell
line: | | (24) |
| | • MTT assay | • Inhibited cell
viability | |
| | • Tryphan Blue
assay | • Inhibited cell
viability | |
| | • Microscopic
analysis (cell density) | • Decrease in cell
density | |
| Cervical
cancer | HeLa cell
line: | | (24) |
| | • MTT assay | • Inhibited cell
viability | |
| Colorectal
cancer | SW480 and SW620
cell lines: | | (26) |
| | • Sulforhodamine B
(SRB) colorimetric assay | • Decrease in cell
density and proliferation | |
| | • Annexin V
assay | • Induced apoptosis
(increase in necrotic cells) | |
| | • Cell cycle
profiling | • Cell cycle arrest
in the sub G0/G1 phase (SW620) | |
| | • Mitochondrial
membrane potential | • Decrease in
mitochondrial membrane potential [Propidium iodide and
DiOC6(3)] | |
| | • Protein
extraction and quantification | • Increase in
caspase 3 and p53 expression (SW620) | |
| | HT29 cell
line: | | (26) |
| | • Sulforhodamine B
(SRB) colorimetric assay | • Decrease in cell
density | |
| | HCT116 cell
line: | | (24,35) |
| | • MTT assay | • Inhibited cell
viability | |
| Liver | HepG2 cell
line: | | (27,35) |
| | • Sulforhodamine B
(SRB) colorimetric assay | • Decrease in cell
density | |
| | • MTT assay | • Inhibited cell
viability | |
| | In vivo
model-adult Wistar male rats: | In
hepatocellular-induced carcinoma rats treated with DV AgNPs: | (36) |
| | • Blood biochemical
analysis (ALT, AST, serum albumin, GPx) | • Decrease in ALT,
AST and serum albumin, and increase in GPx | |
| | • Comet assay | • Reduced formation
of DNA adducts and minimised damage to DNA structures | |
| | • Annexin V
assay | • Increase in
apoptotic cells | |
| | • Flow
cytometry | • Decrease in ROS
production | |
| | • RT-qPCR | • Decrease in
BCL2 and p53, and increase in Bax gene
expression | |
| Leukaemia | THP-1 cell
line: | | (27) |
| | • MTT assay | • Inhibited cell
viability | |
| Ovarian | A2780 cell
line: | | (28) |
| | • MTT assay | • Inhibited cell
viability | |
| | SKOV-3 cell
line: | | (33) |
| | • MTT assay | • Inhibited cell
viability | |
| Lung | A549 cell
line: | | (33,34) |
| | • MTT assay | • Inhibited cell
viability | |
| | • Microscopic
analysis | • Decrease in cell
density, membrane blebbing, degradation of cell membrane | |
| | • Live/dead cells
assay by high content screening | • Induced
apoptosis | |
| Burkitt
lymphoma | Ramos and BL41 cell
lines: | | (30) |
| | • WST-1 viability
assay | • Decrease in cell
viability | |
| | • Microscopic
analysis | • Typical features
of apoptosis | |
| | • Annexin V
assay | • Increased
apoptosis | |
| | • Caspase activity
assay | • Increased caspase
3/7 enzyme activities | |
| | • Western blot
analyses of apoptotic markers | • Increased cleaved
caspase and cleaved PARP-1 expression | |
| | • Western blot
analyses of PI3K/Akt pathway markers | • Reduced p-PDK1
and increased p-PTEN levels expression | |
The use of nanotechnology in the delivery of
DV-derived compounds has shown promising results. Advances in
nanotechnology research have allowed for improved drug delivery,
demonstrating increased efficacy and specificity of treatment, and
reduced adverse effects (31,32).
As a result, several nano-based treatment modalities have been
translated into clinical trials, including silver nanoparticles
(AgNPs), which are widely used in clinical applications due to
their antimicrobial, antioxidant, antiviral, and antidiabetic
properties, and as conjugates with chemotherapeutic drugs (33). Notably, AgNps synthesized from DV
leaf extracts have exhibited potent anti-proliferative properties
in vitro against ovarian (SKOV-3) and lung (A549) cancer
cell lines (34,35). In both of these studies, DVE-AgNPs
were significantly more toxic to the cancer cells compared with
non-cancerous control cells or cancer cells treated with the crude
extract alone. Furthermore, cancer cells were markedly more
sensitive to AgNPs synthesized from solvents such as petroleum
ether, acetone, methanol, and acetonitrile compared with those
synthesized from an aqueous solvent (34,35).
The use of zinc oxide nanoparticle (ZnO NP) preparations from
ethanol-, petroleum ether-, chloroform-, and methanol-derived DV
leaf extracts have also yielded promising results, demonstrating
significant inhibition of cell viability towards liver (HepG2) and
colorectal (HCT-116) cancer cell lines compared with fibroblast
cells (3T3), showing superior cytotoxicity relative to
Tamoxifen-treated cancer cells (36). ZnO NPs exhibited superior
cytotoxicity relative to chloroform-derived DV fractions
(IC50 values of ZnO NPs: HepG2, 16.4±4 µg/ml and HCT116,
29.07±2.7 µg/ml vs. IC50 values of chloroform DV
fractions: HepG2, 26.4±3.3 µg/ml and HCT116, 39.8±13 µg/ml)
(36).
To date, only a few studies have made use of
preclinical models to evaluate the efficacy and safety of DV
extracts. Nevertheless, these have yielded promising outlooks on
anticancer efficacy, bioavailability and toxicity. For instance,
AgNPs synthesized from ethanolic DV leaf extracts inhibited the
development of tumours induced by N-nitrosodiethylamine in the
liver of Wistar rats (37). Serum
levels of enzymatic markers indicative of liver damage, alanine
transaminase and aspartate aminotransferase, were significantly
reduced, while albumin levels, a marker of less aggressive tumour
progression, were reduced. While DNA damage was reduced in hepatic
tissues of rats which received the DV-AgNPs, the apoptosis rate was
increased. Overall, DV AgNPs exhibited protective effects against
liver damage and significantly inhibited hepatocellular carcinoma
progression in rats receiving DV extracts compared with controls
(37).
Antioxidant activity
Oxidative damage to proteins and DNA resulting from
disrupted reactive oxygen species (ROS) homeostasis is a major
contributor to genomic alterations that enhance oncogenic
phenotypes in cancer cells. Numerous natural products are rich
sources of antioxidants able to act as effective scavengers of free
radicals and ROS (38). The
administration of extracts from DV, both organic and aqueous,
proved protective towards carbon tetrachloride (CCL4)-induced
hepatotoxicity in rats (39). CCL4
is known to cause liver damage through the formation of ROS, and in
that particular study, hautriwaic acid was suggested as the main
DV-derived protective compound identified via HPLC fingerprinting
(39). In a more recent study
(40), through the use of the DPPH
(2,2-diphenyl-1-picrylhydrazyl) free radical scavenging in
vitro assay, stem and leaf extracts of DV were found to have
strong antioxidant activities, which corroborated an earlier study
where stem extracts of the plant showed strong scavenging
activities (27).
Anti-inflammatory activity
The innate inflammatory response is the first line
of defense against damaging agents and invading pathogens, but its
effects are mitigated by the use anti-inflammatory medicines to
mitigate tissue damage caused by prolonged or excessive responses.
Naturally occurring agents with anti-inflammatory properties offer
potentially safer and more effective alternatives to synthetic
options for managing inflammation. The carrageenan rat paw edema
model was used to demonstrate that hydroalcoholic (DVHA) and
n-hexane (DVH) extracts from DV leaves were more effective compared
with the anti-inflammatory drug indomethacin, in reducing
inflammation (41). The study
demonstrated that the DVHA and DVH extracts effectively suppressed
carrageenan-induced inflammation in rats compared with control rats
which either did not receive DV or received indomethacin. In
another study, this time using mice ear edema as a model to measure
inflammation, a 64% reduction in ear edema was observed in mice
receiving DV dichloromethane extracts, compared with a 40%
reduction in mice receiving indomethacin (42). A potential mechanism mediating these
anti-inflammatory effects was suggested to be through the action of
the phytochemical viscosine. Using molecular docking simulations,
viscosine was found to impair the activity of lipoxygenase, an
enzyme responsible for generating pro-inflammatory mediators and
implicated in inflammatory diseases (43). Another notable observation was the
potent reduction of nitric oxide production, prostaglandin E2 and
tumor necrosis factor-α in the culture medium of
lipopolysaccharide-induced murine macrophages (RAW264.7), once
again suggesting that extracts from DV possess significant
anti-inflammatory activity (43).
Antimicrobial activity
Extracts from the DV plants have been reported to
inhibit the growth and biofilm formation of several bacterial and
fungal pathogens including Staphylococcus aureus,
Streptococcus mutans, Candida albicans, Salmonella typhi,
Shigella flexneri, Escherichia coli, Vibrio cholera, Mycobacterium
tuberculosis and Pseudomonas fluorescens (18,27,29,35,44-46).
In rats infected with S. aureus, those receiving oral
administration of ethanolic DV extracts for 30 days had improved
kidney histopathology with normal glomeruli and convoluted tubules
compared with untreated mice who developed thickening of the wall
of renal vessels, infiltrating lymphocytes in the kidneys, and
damaged glomeruli (44). Notably,
extracts from the DV plant have been shown to display inhibitory
effects against strains of Mycobacterium tuberculosis (M.
tuberculosis), the organism responsible for multidrug-resistant
tuberculosis which remains a public health concern worldwide. Using
the resazurin microtiter assay the anti-mycobacterial efficacy of
methyl alcohol and chloroform extracts of DV was assessed against
three distinct strains of M. tuberculosis namely bg 1972, bg
206, and H37Rv (47). A
dose-dependent decrease in the growth of the bacteria was observed,
with the most pronounced effect exerted against the H37Rv
strain.
The antifungal activity of the acetone-derived DV
extract was evaluated against 40 Candida albicans (C.
albicans) strains, 20 of which were isolated from individuals
living with HIV and 20 from individuals without HIV (48). Although no discernible difference in
effect was observed between the C. albicans isolates from
the two groups, all strains were potently inhibited by the
extracts. Notably, another study demonstrated that planktonic cells
of C. albicans exposed to acetone DV extracts and DV
bioactive compound (5, 6, 8-trihydroxy-7, 4' dimethoxy flavone)
could not form germ tubes (49).
These findings lend credence to the use of DV extracts by
traditional medical practitioners to treat oral thrush and related
infections, and suggest that DV may serve as a natural source of
antifungal agents. Collectively, these results indicated that DV is
a promising source of phytochemicals possessing antibacterial and
antimycobacterial properties.
Viral diseases such as Zika, Ebola, AIDS, SARS,
MERS, influenza, and pneumonia are major contributors to mortality
and disability around the world. In low-income countries, chronic
viral infections are attributable to up to 26% of cancer cases,
where cancer incidence and related deaths are expected to increase
significantly by 2050, highlighting the urgent need for effective
prevention and treatment of viral infections (50). In a study by Rashed et al
(51), the human CD4+
lymphoid cell line C8166 was used to demonstrate DV-induced
inhibition of HIV-1 infection. Chromatographic separation of the
extract with the highest anti-HIV-1 activity (petroleum
ether-derived) identified two compounds, β-sitosterol and
stigmasterol, as active antiviral agents (51). In yet another study, the effects of
five different extracts from DV leaves were examined against
coxsackievirus B3 and rotavirus SA-11 viral infections (52).
Antidiabetic effects
The hypoglycaemic activity of DV leaf extracts
(derived from chloroform, methanol, butanol, and aqueous) was
analysed in normal and alloxan-induced diabetic rabbits in several
independent studies, all of which reported hypoglycaemic effects
within 1-2 h after oral administration (53-56).
A significant reduction in the blood glucose levels was observed in
diabetic rabbits receiving DV compared with those treated with
glibenclamide or untreated normal rabbits (53). Prolonged treatment, for 10/15/30
days, potently and consistently reduced blood glucose levels in
alloxan-induced diabetic rabbits compared with normal and untreated
controls, and a significant increase in plasma insulin levels and a
reduction in urea, total cholesterol and triglycerides were
observed during the treatment period (54,55).
In a different animal model, that of streptozotocin-induced
diabetic rats, a significant reduction in blood glucose, pyruvic
transaminase, glutamic oxaloacetic transaminase, creatine, and urea
was observed in DV-treated animals compared with untreated ones,
accompanied by reduced levels of total cholesterol, triglycerides,
low-density lipoprotein-cholesterol and pro-inflammatory biomarkers
in serum, while no significant change was observed in high-density
lipoprotein-cholesterol serum levels (56). Histopathological analysis of the
liver and renal tissues from the DV-treated animals showed
significant liver protection as evidenced by the regeneration of
hepatocytes with a lack of fatty lobulation and necrosis, and lack
of renal tubular necrosis which was evident in animals that did not
receive the plant extracts (56).
The cumulative findings from these studies provide strong support
for the consideration of DV extract as a viable antidiabetic
treatment.
Wound healing effects
The application of DV-derived products to assist in
wound healing has been reported in alternative therapy, prompting
investigation of this potential therapeutic benefit in
laboratory-based in vivo studies using rats. Ethyl acetate
flavonoid-rich fractions extracted from DV leaves were shown to
significantly increase levels of hydroxyproline and hexosamine,
important constituents of the extracellular matrix, improve
collagen formation, and fasten epithelialization and
vascularization of wounds, relative to control groups (57). In a separate but similar study using
the same in vivo model and approach, DV extracts were found
to perform similarly to the known and approved antibiotic
nitrofurazone in preventing infection and accelerating wound
healing (58). These findings
warrant further investigations aimed at isolating the bioactive
constituents with wound healing and antibiotic properties from the
crude extracts of DV.
Other reported therapeutic
properties
Extracts from DV leaves were shown to have
gastroprotective effects and protect against the development of
ulcers in a rodent model (59).
Pretreatment with DV hexane extracts blocked the formation of
ethanol/indomethacin-induced gastric ulcer lesions in the Charles
Wister rats which displayed reduced gastric glutathione levels,
inhibition in the accumulation of alkaline phosphatase and
increased gastric pH. These effects were similar to those observed
in rats treated with the proton-pump inhibitor drug, omeprazole. In
a separate study, the anti-diarrheal activity of DV root extracts
(alcohol and aqueous) was analysed in male albino mice fed castor
oil to induce diarrhea (60). Both
alcohol and aqueous DV extracts significantly reduced diarrhea
episodes and stool weight in the mice in a dose-dependent manner,
similar to what was observed in the groups receiving the
anti-diarrheal drug, loperamide. Additionally, the DV extracts were
found to be non-toxic to the mice at a dose of up to 2,000 mg/kg.
No clinical signs of weakness or other adverse events were observed
in the animals after DV treatment (60).
4. Conclusion and future perspectives
The present review provides a comprehensive and
thorough overview of the updated published literature reporting on
scientific investigations into the therapeutic potential of
extracts derived from the DV plant, the latter being traditionally
used in ethnomedicine around the world. Collectively, studies
assessing anticancer potential (primarily anti-proliferative and
antioxidant) provide a strong foundation warranting more
comprehensive investigations, with emphasis on obtaining the
anticancer bioactive components of DV extracts, as well as the
application of systems pharmacology and cheminformatics. Compared
with more established medicinal plants such as Artemisia
annua (source of artemisinin, an antimalarial agent),
Catharanthus roseus (source of vinblastine and vincristine,
used in cancer therapy) and Taxus brevifolia (source of
paclitaxel, an anticancer drug), only a moderate number of
laboratory-based scientific studies have been performed on
DV-derived extracts and phytochemicals, and even fewer in
vivo investigations using animal models. Although numerous
DV-derived chemicals have been identified, detailed mechanistic
studies aimed at elucidating bioactivities remain sparse, with no
reported clinical trials. Nevertheless, the current existing
evidence shows considerable promise.
The great diversity in the phytochemical composition
of the DV plant extracts as well as the yield thereof appear to be
influenced by the choice of extraction solvent. In addition, this
diversity is shaped by the specific plant part utilized
(leaf/stem/root/flowers). A general trend of decreasing extract
yield is observed with a shift in the polarity of the solvent from
polar to non-polar, likely due to both the bioavailability and
composition of the phytochemicals.
To date, the majority of investigations have
employed in vitro assays, making use of established cancer
cell lines (Table I). Most studies
apply crude extracts or fractionated compounds directly onto cells,
however where more sophisticated delivery methods have been
employed, particularly AgNPs, greater biological impact is
observed. This is expected, given the well described advantages of
AgNP technology, including improved stability, increased
surface-area-to-volume ratio and enhanced uptake. Significantly
more preclinical studies are needed, using relevant animal models,
to demonstrate the desired anticancer biological effects, but
crucially, to gather data on the safety of the novel compounds.
Notably, investigations have primarily involved epithelial-derived
cancer cells and reveal a gap regarding the potential of DV
extracts for the treatment of sarcomas, melanomas, and
haematological malignancies.
Since plant extracts represent a mixture of
secondary metabolites, it is unsurprising that studies show DV
extracts to have antimicrobial, antiviral, and antidiabetic
effects, among others (Table II).
Such is the case for other plants that have been widely
investigated, such as curcumin, cannabis, and galanga. Therefore,
the separation of active ingredients and analysis of bioactivity of
each compound to identify those with therapeutic promise is
essential, however this can be an arduous, costly and lengthy
process. While the rich biodiversity enhances the value of such
investigations, there can be significant batch-to-batch variability
which represents a regulatory hurdle. To minimise this, cultivation
and harvest protocols need to be strictly standardized, while
maintaining stable processing conditions (61). These factors can
translate into quite extended safety evaluations during the drug
development process, and once approved, may require ongoing safety
monitoring. Another important and often neglected aspect of
ethnobotanical studies is the preservation of traditional knowledge
and sustainable use of natural products. Nevertheless,
plant-derived compounds remain an important source for the
development of novel and effective therapeutics, and it is
essential to adopt a multidisciplinary framework that brings
together conservation programmes, environmentally responsible
harvesting strategies, comprehensive scientific and clinical
investigations, stringent quality-management standards, regulatory
coherence, and ethical governance.
 | Table IIReported (non-cancer related)
therapeutic biological potential of DV. |
Table II
Reported (non-cancer related)
therapeutic biological potential of DV.
| Biological
activity | Experimental
approach | Findings | (Refs.) |
|---|
| Antioxidant | Chloroform and
methanol DV extract tested in Wistar rats with liver damage induced
by carbon tetrachloride | Reduced
CCL4-induced liver injury as evidenced by reduction of serum
markers; histopathology and immunochemistry | (39) |
| | In vitro
free radical scavenging assay (DDPH) measuring total antioxidant
capacity and reducing power of DV | Increased free
radical scavenging ability in the presence of DV extracts | (27) |
|
Anti-inflammatory | Carrageenan rat paw
edema model in rats fed hydroalcoholic and n-hexane DV
extracts | Extracts suppressed
inflammation relative to controls | (41) |
| | Used a mice ear
edema model, inducing inflammation using tetradecanoyl phorbol
13-acetate, and assessed DV dichloromethane-derived extracts | 64% reduction in
ear edema in mice relative to control mice treated with
indomethacin (40%) | (42) |
| | Molecular docking
simulation | Identified
viscosine, which impaired the activity of lipoxygenase | (43) |
| | Measurement of
production of nitric oxide and of proinflammatory cytokines in
culture medium of lipopolysaccharide-induced murine macrophages
(RAW264.7), in the presence of DV extract | Potently reduced
nitric oxide production, prostaglandin E2, and tumor necrosis
factor-α | (24) |
| Antimicrobial | Rats infected with
S. aureus and orally fed with ethanolic DV extract | Improved kidney
histopathology with normal glomeruli and convoluted tubules
compared with controls | (44) |
| | Used resazurin
microtiter assay to measure anti-mycobacterial efficacy of methyl
alcohol and chloroform extracts of DV in three Mtb strains | Dose-dependent
decrease in the growth of all three Mtb strains | (47) |
| | Tested efficacy of
HIV-1 infection of the human CD4+ lymphoid cell line
C8166 | DV isolated active
compounds β-sitosterol and stigmasterol as active antiviral
agents | (51) |
| | Infection by
coxsackievirus B3 and rotavirus SA-11 viral stocks of MA 104 and
GMK cells in the presence of 5 different DV extracts | Methanol crude
extract showed the strongest antiviral effect against both
viruses | (52) |
| Antidiabetic | Assessed blood
glucose levels in alloxan-induced diabetic rabbits | Rabbits receiving
DV had significantly reduced blood glucose levels within 1-2 h.
Prolonged treatment produced a potent and consistent increase in
plasma insulin levels; significant reduction in urea, total
cholesterol, and triglycerides | (53-55) |
| | Used
streptozotocin-induced diabetic rats treated with DV extracts | Significant
reduction in blood glucose, serum insulin, and other markers
including total cholesterol, triglycerides and low-density
lipoprotein-cholesterol | (56) |
| Wound healing | Used rats with
induced excision and incision wounds, applied various DV extracts
to wounds | DV-treated animals
had improved skin architecture and faster epithelialization and
vascularization of wounds | (57) |
| Anti-ulcer | Rats were
pretreated with DV extracts and were thereafter induced to develop
gastric lesions | Pretreated rats had
reduced gastric glutathione levels, lower accumulation of alkaline
phosphatase and increased gastric pH | (59) |
| Anti-diarrhea | Assessed castor-oil
induced diarrhea in mice | Mice fed with DV
extracts had significantly reduced diarrhea episodes and stool
weight | (60) |
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
AS and BY proposed the review topic and wrote the
original draft of this review article. AS and SM contributed to the
manuscript revision and editing. SM substantially edited the final
version. All authors have reviewed and revised the manuscript, and
provided feedback. In addition, all authors read and approved the
final manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Cavalcanti IDL and Soares JCS:
Conventional Cancer Treatment. In: Advances in Cancer Treatment.
Springer, Cham, pp29-56, 2021.
|
|
3
|
Hulya E, Burcu C, Birol O, Sibel OO, Erdem
C and Ozkan K: Evaluation of complementary and alternative medicine
use in cancer patients: A survey study in Turkey. AIMED. 12:99–103.
2025.
|
|
4
|
Keene MR, Heslop IM, Sabesan SS and Glass
BD: Complementary and alternative medicine use in cancer: A
systematic review. Complement Ther Clin Pract. 35:33–47.
2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Knecht K, Kinder D and Stockert A:
Biologically-Based complementary and alternative medicine (CAM) use
in cancer patients: The good, the bad, the misunderstood. Front
Nutr. 6(196)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Atanasov AG, Zotchev SB and Dirsch VM:
International Natural Product Sciences Taskforce. Supuran CT:
Natural products in drug discovery: advances and opportunities. Nat
Rev Drug Discov. 20:200–216. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Basmadjian C, Zhao Q, Bentouhami E, Ajehal
A, Nebigil CG, Johnson RA, Serova M, de Gramont A, Faivre S,
Raymond E and Désaubry LG: Cancer wars: Natural products strike
back. Front Chem. 2(20)2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Pattanayak S: Plants in healthcare: Past,
present and future. Explor Anim Med Res. 11:140–144. 2021.
|
|
9
|
Kelly K: History of Medicine. Oxford
University Press, New York, 2009.
|
|
10
|
Petrovska BB: Historical review of
medicinal plants' usage. Pharmacogn Rev. 6:1–5. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yang YH, Mao JW and Tan XL: Research
progress on the source, production, and anti-cancer mechanisms of
paclitaxel. Chin J Nat Med. 18:890–897. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhu L and Chen L: Progress in research on
paclitaxel and tumour immunotherapy. Cell Mol Biol Lett.
24(40)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Keller-Juslen C, Kuhn M, Staehelin H and
Von Wartburg A: Synthesis and antimitotic activity of glycosidic
lignan derivatives related to podophyllotoxin. J Med Chem.
14:936–940. 1971.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Reyhanoglu G and Tadi P: Etoposide.
StatPearls Publishing, Treasure Island, FL, 2025.
|
|
15
|
Noble RL: The discovery of the vinca
alkaloids-chemotherapeutic agents against cancer. Biochem Cell
Biol. 68:1344–1351. 1990.PubMed/NCBI
|
|
16
|
Škubník J, Pavlíčková VS, Ruml T and
Rimpelová S: Vincristine in combination therapy of cancer: Emerging
trends in clinics. Biology (Basel). 10(849)2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Harris S: Dodonaea viscosa var.
angustifolia. Karoo Desert National Botanical Garden. In:
PlantZAfrica. Available from: https://pza.sanbi.org/dodonaea-viscosa-var-angustifolia,
2012.
|
|
18
|
Ngabaza T, Johnson MM, Moeno S and Patel
M: Identification of
5,6,8-Trihydroxy-7-methoxy-2-(4-methoxyphenyl)-4H-chromen-4-one
with antimicrobial activity from Dodonaea viscosa var.
angustifolia. S Afr J Bot. 112:48–53. 2017.
|
|
19
|
Al-Snafi AE: A review on Dodonaea viscosa:
A potential medicinal plant. IOSR J Pharm. 7:10–21. 2017.
|
|
20
|
Hossain MA: Biological and phytochemicals
review of Omani medicinal plant Dodonaea viscosa. J King Saud Univ
Sci. 31:1089–1094. 2019.
|
|
21
|
Saranya K and Divyabharathi U: Gas
chromatography and mass spectroscopic analysis of phyto-compounds
in Dodonaea viscosa leaf extract. Pramana Res J. 9:26–35.
2019.https://www.pramanaresearch.org/gallery/prj-p1302.pdf.
|
|
22
|
Hoffman M, Marx J, Kaigongi MM, Masondo
NA, Lukhoba CW, Yenesew A and Makunga NP: Reflecting on the
interesting phytopharmacology of the cosmopolitan Dodonaea genus:
Past, present and future. Phytochem Rev: Aug 1, 2025 (Epub ahead of
print).
|
|
23
|
Mossa GD and Al-Shawi AA: Induction of
apoptosis through S-phase in human breast cancer MDA-MB231 cells by
ethanolic extract of Dodonaea viscosa L. - an Iraqi medicine plant.
J Basrah Res. 41:119–128. 2015.
|
|
24
|
Ramkumar R, Periyasamy SK, Venkatraman BR
and Sekar KG: In-vitro anti-cancer and anti-inflammatory screening
of dodonaea viscosa. J Pharm Res Int. 33:186–192. 2021.
|
|
25
|
Herrera-Calderon O, Rahman MH, Pena-Rojas
G and Andia-Ayme V: (2020) Dodonaea viscosa Jacq: A medicinal plant
with cytotoxic effect on colon cancer cell line (HT-29). J Pure
Appl Microbiol. 14:1927–1934. 2020.
|
|
26
|
Herrera-Calderon O, Herrera-Ramírez A,
Cardona-G W, Melgar-Merino EJ, Chávez H, Pari-Olarte JB,
Loyola-Gonzales E, Kong-Chirinos JF, Almeida-Galindo JS, Peña-Rojas
G and Andía-Ayme V: Dodonaea viscosa Jacq. induces cytotoxicity,
antiproliferative activity, and cell death in colorectal cancer
cells via regulation of caspase 3 and p53. Front Pharmacol.
14(1197569)2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Malik MN, Haq I, Fatima H, Ahmad M, Naz I,
Mirza B and Kanwal N: Bioprospecting Dodonaea viscosa Jacq.; a
traditional medicinal plant for antioxidant, cytotoxic,
antidiabetic and antimicrobial potential. Arab J Chem.
15(103688)2022.
|
|
28
|
Cao S, Brodie P, Callmander M,
Randrianaivo R, Razafitsalama J, Rakotobe E, Rasamison VE, TenDyke
K, Shen Y, Suh EM and Kingston DG: Antiproliferative triterpenoid
saponins of Dodonaea viscosa from the Madagascar dry forest. J Nat
Prod. 72:1705–1707. 2009.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Priya VT, Balasubramanian N, Shanmugaiah
V, Sathishkumar P, Kannan ND, Karunakaran C, Alfarhan A and
Antonisamy P: Partially purified lead molecules from Dodonaea
viscosa and their antimicrobial efficacy against infectious human
pathogens. J Infect Public Health. 14:1822–1830. 2021.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Saferdien A: Anti-cancer effects of
aqueous extracts of Dodonaea viscosa on Burkitt lymphoma. PhD
dissertation, University of Cape Town, 2023. http://hdl.handle.net/11427/42528.
|
|
31
|
Jin C, Wang K, Oppong-Gyebi A and Hu J:
Application of nanotechnology in cancer diagnosis and therapy-A
mini-review. Int J Med Sci. 17:2964–2973. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Mosleh-Shirazi S, Abbasi M, Moaddeli MR,
Vaez A, Shafiee M, Kasaee SR, Amani AM and Hatam S: Nanotechnology
advances in the detection and treatment of cancer: An overview.
Nanotheranostics. 6:400–423. 2022.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Gomes HIO, Martins CSM and Prior JAV:
Silver nanoparticles as carriers of anticancer drugs for efficient
target treatment of cancer cells. Nanomaterials (Basel).
11(964)2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Al-Musawi ZFH and Al-Saadi NHM: Antitumor
activities of biosynthesized silver nanoparticles using Dodonaea
viscosa (L.) leaves extract. Basrah J Agri Sci. 34:42–59. 2021.
|
|
35
|
Anandan M, Poorani G, Boomi P, Varunkumar
K, Anand K, Chuturgoon AA, Saravanan M and Gurumallesh Prabu H:
Green synthesis of anisotropic silver nanoparticles from the
aqueous leaf extract of Dodonaea viscosa with their antibacterial
and anticancer activities. Process Biochem. 80:80–88. 2019.
|
|
36
|
Alghamdi MD, Nazreen S, Ali NM and Amna T:
ZnO Nanocomposites of Juniperus procera and Dodonaea viscosa
extracts as antiproliferative and antimicrobial agents.
Nanomaterials (Basel). 12(664)2022.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Alasmari A: Hepatoprotective activity of
Dodonaea viscosa leaf extract nanoparticles against
N-nitrosodiethylamine induced tumor initiation in rat liver:
Modulation of apoptosis and signaling pathways. J Environ Biol.
43:177–187. 2022.
|
|
38
|
Sznarkowska A, Kostecka A, Meller K and
Bielawski KP: Inhibition of cancer antioxidant defense by natural
compounds. Oncotarget. 8:15996–16016. 2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ali H, Kabir N, Muhammad A, Shah MR,
Musharraf SG, Iqbal N and Nadeem S: Hautriwaic acid as one of the
hepatoprotective constituents of Dodonaea viscosa. Phytomedicine.
21:131–140. 2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Auta R, Waziri P, Oweh OT, Wayah S,
Tyoapine D, Bobai M, Solomon B and Onyemaobi U: Antibacterial,
anticancer and antioxidant properties of the stem and leaf extract
of Dodonaea viscosa. Sci World J. 20:331–335. 2025.
|
|
41
|
Ramkumar R and Periyasamy SK:
Anti-inflammatory activity of Dodonaea viscosa leaves. IJRAR.
6:223–225. 2019.
|
|
42
|
Salinas-Sánchez DO, Herrera-Ruiz M, Pérez
S, Jimenez-Ferre E and Zamilpa A: Anti-inflammatory activity of
hautriwaic acid isolated from Dodonaea viscosa leaves. Molecules.
17:4292–4299. 2012.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Khan AZ, Mohammad A, Iqbal Z, Anis I, Shah
MR, Nadeem S, Rabnawaz M, Shahidullah A, Khan H and Khan I:
Molecular docking of viscosine as a new lipoxygenase inhibitor
isolated from Dodonaea viscosa. Bangladesh J Pharmacol. 8:36–39.
2013.
|
|
44
|
Majeed ZH, Hasan SA and Ismail RM:
Evaluate the benefit effects of Dodonaea viscosa in kidney of
infected rats with Staphylococcus aureus. J Pharm Negat Results.
13:291–294. 2022.
|
|
45
|
Hamed Al Bimani BM and Hossain MA: A new
antimicrobial compound from the leaves of Dodonaea viscosa for
infectious diseases. Bioact Mater. 5:602–610. 2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Naidoo R, Patel M, Gulube Z and Fenyvesi
I: Inhibitory activity of Dodonaea viscosa var. angustifolia
extract against Streptococcus mutans and its biofilm. J
Ethnopharmacol. 144:171–174. 2012.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Tong ZW, Gul H, Awais M, Saddick S, Khan
FS, Gulfraz M, Afzal U, Nazir K, Malik MY, Khan SU and Khan MI:
Determination of in vivo biological activities of Dodonaea viscosa
flowers against CCL4 toxicity in albino mice with bioactive
compound detection. Sci Rep. 11(13336)2021.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Patel M and Coogan MM: Antifungal activity
of the plant Dodonaea viscosa var. angustifolia on Candida albicans
from HIV-infected patients. J Ethnopharmacol. 118:173–176.
2008.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Patel M, Srivastava V and Ahmad A:
Dodonaea viscosa var angustifolia derived 5, 6, 8-trihydroxy-7, 4'
dimethoxy flavone inhibits ergosterol synthesis and the production
of hyphae and biofilm in Candida albicans. J Ethnopharmacol.
259(112965)2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Bizuayehu HM, Ahmed KY, Kibret GD, Dadi
AF, Belachew SA, Bagade T, Tegegne TK, Venchiarutti RL, Kibret KT,
Hailegebireal AH, et al: Global disparities of cancer and its
projected Burden in 2050. JAMA Netw Open.
7(e2443198)2024.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Rashed K, Meng TL, Lin-Tao Z and Yong-Tang
Z: Dodonaea viscosa (L.) extracts as anti human immunodeficiency
virus type-1 (HIV-1) agents and phytoconstituents. Peak J of
Medicinal Plant Research. 1:19–25. 2013.
|
|
52
|
Shaheen M, Borsanyiova M, Mostafa S,
Chawla-Sarkar M, Bopegamage S and El-esnawy N: In vitro effect of
Dodonaea viscosa extracts on the replication of coxackievirus B3
(Nancy) and rotavirus (SA-11). JMAA. 1:47–54. 2015.
|
|
53
|
Akhtar MS, Ahmed M, Gulzar K and Adnan H:
Hypoglycemic activity of Dodonaea viscosa leaves in normal and
alloxan-induced diabetic rabbits. Diabetologia Croatica. 40:71–79.
2011.
|
|
54
|
Muthukumran P, Hazeena Begumand V and
Kalaiarasan P: Anti-diabetic activity of Dodonaea viscosa (L) leaf
extracts. Int J PharmTech Res. 3:136–139. 2011.
|
|
55
|
Sandhya Rani M, Venkatesh P, Pippalla RS
and Mohan GK: Biochemical and histological study of traditional
plant: Dodonaea viscosa Linn extracts in diabetic rats. J
Phytopharmacol. 2:13–21. 2013.
|
|
56
|
Alanazi AZ, Al-Rejaie SS, Ahmed MM,
Alhazzani K, Alhosaini K, As Sobeai HM, Alsanea S, Alam P,
Almarfadi OM, Alqahtani AS, et al: Protective role of Dodonaea
viscosa extract against streptozotocin-induced hepatotoxicity and
nephrotoxicity in rats. Saudi Pharm J. 31(101669)2023.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Subramanian S, Duraipandian C, Alsayari A,
Ramachawolran G, Wong LS, Sekar M, Gan SH, Subramaniyan V,
Seethalakshmi S, Jeyabalan S, et al: Wound healing properties of a
new formulated flavonoid-rich fraction from Dodonaea viscosa Jacq.
leaves extract. Front Pharmacol. 14(1096905)2023.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Nayeem N, Asdaq SMB, Alamri AS, Alsanie
WF, Alhomrani M, Mohzari Y, Alrashed AA, Alotaibi N, aalhathal AS,
Alharbi MA, et al: Wound healing potential of Dodonaea viscosa
extract formulation in experimental animals. J King Saud Univ Sci.
33(101476)2021.
|
|
59
|
Arun M and Asha VV: Gastroprotective
effect of Dodonaea viscosa on various experimental ulcer models. J
Ethnopharmacol. 118:460–465. 2008.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Rajamanickam V, Rajasekaran A,
Anandarajagopal K, Sridharan D, Selvakumar K and Benjamin SR:
Anti-diarrheal activity of Dodonaea viscosa root extracts. Int J
Pharma Bio Sci. 1:182–185. 2010.
|