1. Introduction
Prolonged untreated gouty arthritis leads to
extensive deposition of serum-free monosodium urate (MSU) crystals
in peripheral joints, leading to inflammatory fibrous tissue
hyperplasia and tophus formation. Over time, this process causes
irreversible damage to the articular cartilage and synovium. In
advanced cases, the disease extends into bone tissue, producing
joint deformities and gout-related fractures. Current clinical
management strategies are insufficient to effectively address
gout-associated bone destruction. Conventional non-surgical
therapies fail to reverse the pathological progression of tophi
(1). Surgery carries risks such as
post-operative infection and a high risk of recurrence, and
numerous patients require additional operations (2). Therefore, an in-depth exploration of
the mechanisms underlying gout-induced bone destruction is
imperative.
A defining feature of bone destruction in gouty
arthritis is abnormal bone metabolism. MSU crystals impair bone
remodeling through direct and indirect mechanisms. Chhana et
al (3) reported a marked
reduction in osteoblast numbers adjacent to tophi in skeletal
samples from patients with gout. Dual-energy computed tomography
further revealed that MSU crystals drive bone erosion via an
‘outside-in’ mechanism, as MSU crystals are absent from the bone
marrow unless cortical fractures are present (4). Studies have indicated elevated serum
levels of receptor activator of NF-κB ligand (RANKL) and macrophage
colony-stimulating factor (M-CSF) in patients with severe erosive
gout. Serum RANKL concentrations showed a strong correlation with
radiographic erosion scores, while M-CSF levels were closely
associated with tophus burden (5,6). In
vitro experiments in which MSU crystals were added to ivory
slices for 14 days demonstrated no direct physicochemical erosion
of bone by MSU (6). Nevertheless,
the pathological processes of gouty arthritis have been shown to
clearly lead to severe joint destruction, with MSU crystal-induced
imbalance in bone metabolism playing a pivotal role.
2. Direct effects of MSU crystals on bone
tissue cells
Clinically, the accumulation of MSU crystals is
associated with the severity of cartilage and bone destruction.
Patients with severe erosive gout exhibit elevated circulating
levels of RANKL and M-CSF. However, whether MSU crystals directly
cause chondrocyte damage or disrupt the balance between osteoblast
and osteoclast remains debated. Extensive in vitro studies
indicate that MSU crystals indirectly inhibit the activity of
osteoblasts, osteocytes, and chondrocytes by inducing the
production of inflammatory mediators in fibroblast-like
synoviocytes, neutrophils, macrophages, and human monocytes. This
process promotes osteoclast activation and disrupts the
osteoprotegerin (OPG)/RANKL balance (7-11).
Chondrocytes
Under optical microscopy, cartilage adjacent to MSU
crystals exhibits a disorganized architecture, compromised hyaline
cartilage integrity, surface discontinuities, and altered
chondrocyte lacunae. Some researchers have proposed that MSU
crystals may induce chondrocyte death by disrupting nutrient supply
and enhancing cartilage matrix catabolism, although this hypothesis
remains unverified (12). In
vitro, however, MSU crystals have been demonstrated to directly
and rapidly reduce chondrocyte viability in a dose-dependent
manner, promote apoptosis, and suppress the mRNA expression of type
II collagen, aggrecan, and cytoplasmic proteins. Notably, this
inhibitory effect is independent of MSU crystal length.
Additionally, type II collagen in the chondrocyte extracellular
matrix forms complexes with MSU crystals. These complexes alter the
morphology and density of MSU crystals, promote their uptake by
macrophage phagocytosis, and amplify the release of
pro-inflammatory cytokines. As a result, they enhance the
recruitment of neutrophils and macrophages and intensify MSU-driven
inflammatory response (12).
Furthermore, another in vitro study indicated that MSU
directly stimulates chondrocytes to upregulate inflammatory
mediators associated with the NF-κB signaling pathway. Notably, MSU
has been found to enhance the synthesis of hydroxyproline and
glycosaminoglycans in chondrocytes, suggesting concurrent cartilage
damage and remodeling following MSU exposure (13). A previous study suggested that
MSU-induced chondrocyte death occurs primarily through autophagy
rather than apoptosis or endoplasmic reticulum stress (14).
Osteoblasts
In vitro investigations on osteoblasts have
revealed that MSU crystals inhibit osteocyte proliferation and
osteoblast activity. This effect is achieved by downregulating the
expression of osteogenic transcription factors, such as
runt-related transcription factor 2 (Runx2) and transcription
factor Sp7 (Sp7), and by inhibiting the synthesis of
integrin-binding sialoprotein (Ibsp) and bone bone
γ-carboxyglutamic acid-containing protein (Bglap) (3,10,15).
Furthermore, MSU has been shown to decrease alkaline phosphatase
(ALP) levels, thereby impairing bone matrix formation and
mineralization. By disrupting the OPG/RANKL balance, MSU has been
demonstrated to exacerbate the dysregulation of bone metabolism,
particularly in the presence of immune cells (7,9,16).
Osteoclasts
There is currently no consensus regarding the direct
role of MSU in promoting in vitro osteoclast differentiation
(6,17). Nonetheless, the involvement of
neutrophil extracellular traps (NETs), which are composed of
neutrophil antimicrobial proteins, was shown to enable MSU to
effectively induce osteoclast differentiation. This induction was
accompanied by the increased secretion of tartrate-resistant acid
phosphatase (TRAP), RANK, and cathepsin K (Ctsk), thereby enhancing
the bone resorption capacity of osteoclasts (9,18).
Further evidence revealed that MSU significantly promotes
osteoclast differentiation in the presence of RANKL and enhances
bone resorption through activation of the calcineurin-nuclear
factor of activated T-cells, cytoplasmic 1 (NFATc1) and c-Jun
N-terminal kinase (JNK) pathways (19).
In vitro experiments have also demonstrated
that MSU does not directly affect the expression of bone-related or
inflammatory genes in osteocytes; however, these effects can be
mediated by monocytes (11).
Additionally, MSU has been found to inhibit OPG expression in bone
marrow stromal cells (6). Notably,
the limited experimental evidence suggests that direct stimulation
of synovial mesenchymal stem cells by MSU crystals leads to a
marked increase in Runx2 protein expression (20). The effects of MSU crystals on
bone-related cells are summarized in Table I.
 | Table IDirect intervention effects of MSU on
the metabolism of bone-related cells. |
Table I
Direct intervention effects of MSU on
the metabolism of bone-related cells.
| Cell Type | Intervention | Results | (Refs.) |
|---|
| HC | MSU | Reduced cell
viability, decreased mRNA expression of Type II collagen, cartilage
matrix protein-aggregating protein and cytoplasmic proteins | (12) |
| Rat
chondrocytes | MSU | Significant
reduction in cell activity, increased expression of IL-1, IL-6,
TNF-α, hydroxyproline and glycosaminoglycans | (13) |
| HC | MSU | Decreased cell
viability, elevated expression of autophagy-related protein
LC3-II. | (14) |
| | MSU | Increased
expression of IL-6 and IL-8 | |
| HC | MSU-stimulated
synovial cell medium | Significant
increase in IL-6, IL-8, ROS and RNS expression | (8) |
| hFOB | MSU-stimulated
neutrophil-derived NETs | Reduced cell
viability, decreased ALP and OPG expression, increased RANKL
expression. | (9) |
| hFOB | MSU-stimulated
neutrophil-derived exosomes | Reduced cell
viability, decreased OPG and ALP expression, increased RANKL
expression. | (7) |
| Mouse
pre-osteoblasts (MC3T3-E1) | MSU | Reduced cell
viability, decreased expression of Runx2, Sp7, Ibsp and Bglap | (3) |
| Mouse
pre-osteoblasts (MC3T3-E1) Human osteoblasts | MSU-stimulated
RAW264.7 cell medium MSU-stimulated THP-1 cell medium | Normal cell
viability, inhibited osteogenic differentiation, suppressed
expression of Type I collagen α1 chain, Runx2, Sp7, Bglap, Ibsp and
Dmp1, no detectable RANKL expression Type I collagen α1 chain and
Ibsp expression similar to control, reduced Bglap expression,
transient increase in OPG, RANKL first inhibited then elevated,
significant increase in IL-1β, IL-6 and PTGS2 | (10) |
| SD rat-derived
osteoblasts | MSU | Reduced cell
viability, decreased expression of Runx2,Sp7, Ibsp, Bglap and
Dmp1 | (15) |
| Human osteosarcoma
cells (Saos-2) | MSU | Reduced cell count
and activity, decreased ALP expression | (16) |
| THP-1 cells | MSU-stimulated NETs
treated with osteoblast medium | Induced osteoclast
differentiation, significant increase in TRAP, RANK and Ctsk
expression | (9) |
| RAW 264.7
cells | MSU | No direct promotion
of osteoclast formation, no change in RANKL expression | (6) |
| Peripheral blood
mononuclear cells | MSU | Promoted osteoclast
differentiation, increased RANKL mRNA expression, suppressed OPG
expression | (17) |
| Osteoclast
precursors | MSU | Increased number of
TRAP+ multinucleated cells | (18) |
| Mouse osteocyte
line (MLO-Y4) | MSU | Reduced cell
viability, no difference in bone-related or inflammatory gene
expression | (11) |
| RAW264.7 cell
medium | MSU-stimulated
RAW264.7 cell medium | Normal cell
viability, increased the expression of RANKL, decreased OPG
expression | |
| Synovial
mesenchymal stem cells | MSU | Significant
increased the expression of Runx2, enhanced osteogenic
differentiation | (20) |
| Bone marrow stromal
cells (ST2) | MSU | Decreased the
expression of OPG | (6) |
3. Oxidative stress-driven bone destruction
by MSU crystals
During purine metabolism, the formation of MSU is
accompanied by redox reactions that lead to excessive production of
reactive oxygen species (ROS). Concurrently, MSU activates immune
cells to generate ROS, which interferes with bone remodeling by
impairing osteogenesis and promoting bone resorption, ultimately
resulting in joint destruction (21).
ROS generation
MSU is the primary end product of purine metabolism,
whereas ROS are byproducts generated during the catalytic activity
of xanthine oxidase (XO) in this pathway. XO facilitates the
breakdown of purine nucleotides by oxidizing hypoxanthine to
xanthine and subsequently producing uric acid. During this process,
oxygen molecules involved in metabolism are reduced to form
superoxide anion radicals and hydrogen peroxide (22). When serum uric acid levels reach
saturation, urate crystals precipitate in the joints, where they
combine with surrounding Na+ to form MSU. These crystals
subsequently deposit within the joints, leading to the formation of
gouty tophi. In vivo, MSU has been shown to impair nuclear
factor erythroid 2-related factor 2 (Nrf2)-mediated antioxidant
signaling and enhance ROS production (23). Upon MSU stimulation, inflamed and
infiltrating macrophages were shown to release substantial amounts
of IL-33, which promotes neutrophil influx into the joints and
initiates neutrophil-dependent ROS production during gout attacks
(24). López-Reyes et al
(8) suggested that during gout
attacks, in addition to activating the NOD-like receptor thermal
protein domain-associated protein 3 (NLRP3) inflammasome in
monocytes, MSU crystals also induce the release of significant
quantities of ROS, including superoxide anions.
Effects of ROS on bone metabolism
In the pathological process of gouty arthritis,
elevated levels of ROS have been found to reduce the osteogenic
differentiation potential of bone mesenchymal stem cells (BMSCs)
(25), induce apoptosis in
osteoblasts, and enhance osteoclast-mediated bone resorption
(26,27). However, the effect of ROS on
chondrogenic differentiation remains poorly understood. In
vitro studies have revealed that hydrogen peroxide
(H2O2) induces methylation of Kruppel-like
factor 5 (KLF5), leading to decreased β-catenin expression and
impaired osteogenic differentiation of BMSCs (28). Additionally,
H2O2 and hydroxyl radicals (OH-) were shown
to compete for binding to the transcription factor Nrf2, thereby
attenuating WNT/β-catenin signaling. This classical pathway is
essential for ALP expression and mineralization, and its inhibition
was demonstrated to disrupt the osteogenic differentiation of BMSCs
(29).
Additionally, research has shown that ROS can
stimulate NFATc1 expression through activation of NF-κB or via
calmodulin recruitment mediated by changes in membrane potential.
NFATc1, in turn, was revealed to regulate osteoclastogenesis and
the expression of bone resorption markers, such as TRAP and Ctsk
(30). Experimental evidence
indicates that NADPH oxidase 2, a key member of the NADPH oxidase
family, activates extracellular signal-regulated kinase 1/2 in
osteoclast precursors (31).
Moreover, ROS have been reported to upregulate tumor necrosis
factor receptor-associated factor 6 (TRAF6), which interacts with
RANKL and its receptor (RANK) to promote osteoclastogenesis
(32). Inhibition of
TRAF6/ROS-dependent activation of the mitogen-activated protein
kinase (MAPK) and NF-κB pathways was shown to effectively suppress
osteoclast formation (33).
Oxidative stress also impaired phosphorylation of
phosphatidylinositol 3-kinase (PI3K) and protein kinase B (PKB),
thereby disrupting PKB signaling. Zoledronic acid exhibited
anti-osteoclastic effects by modulating the ROS-PI3K/PKB signaling
pathway (34).
Research indicates a bidirectional relationship
between oxidative stress and inflammation: Inflammation can
increase ROS production, while ROS, in turn, can amplify
inflammatory responses. Experimental evidence revealed that ROS
directly activate the NF-κB pathway, promoting the production of
pro-inflammatory cytokines (35).
By activating the MAPK pathway, ROS was shown to upregulate NF-κB
and the NLRP3 inflammasome, thereby enhancing osteoclast-mediated
bone resorption in vitro (36). Additionally, it has been reported
that RANKL acts as an upstream signal that triggers the
ROS/MAPK/NF-κB/NLRP axis (37)
(Fig. 1).
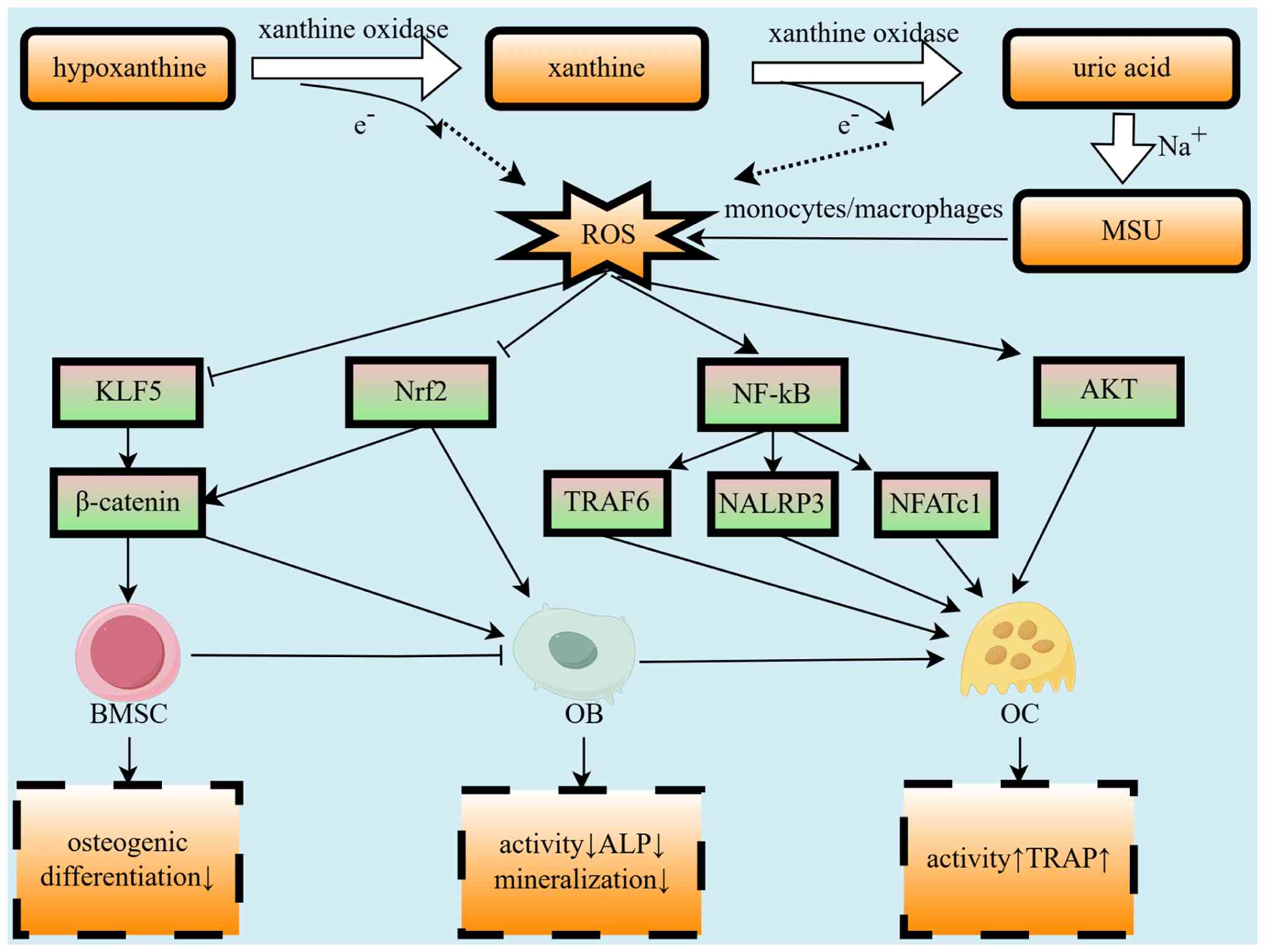 | Figure 1Impact of oxidative stress during MSU
formation on bone metabolism. MSU, monosodium urate; ROS, reactive
oxygen species; KLF5, Kruppel-like factor 5; Nrf2, nuclear factor
erythroid 2-related factor 2; TRAF6, tumor necrosis factor
receptor-associated factor 6; NLRP3, NOD-like receptor family,
pyrin domain containing 3; NFATc1, nuclear factor of activated
T-cells, cytoplasmic 1; BMSC, bone mesenchymal stem cell; OB,
osteoblast, OC, osteoclast; ALP, alkaline phosphatase; TRAP,
tartrate-resistant acid phosphatase. |
4. MSU stimulates inflammatory bone
destruction
The central mechanism of gout-induced bone
destruction involves the interplay between MSU crystals, which
trigger inflammatory cascades and bone metabolism. MSU influences
bone metabolism through dual-signal activation of the NLRP3
inflammasome, promotion of NET release, and regulation of
macrophage polarization (38).
NLRP3 inflammasome and bone
metabolism
Research indicates that a key step in gouty
arthritis flare-ups is the activation of leukocytes by MSU
crystals, which act as a danger signal to initiate inflammatory
cascades. As a damage-associated molecular pattern (DAMP), MSU has
been shown to activate the innate immune system. Evidence has
revealed that activation of the NLRP3 inflammasome in macrophages
and monocytes occurs through a dual-signal mechanism. The first
signal involves Toll-like receptor (TLR)4, TLR2, and transient
receptor potential vanilloid 4 (TRPV4) receptors activating the
NF-κB pathway, leading to the synthesis of pro-IL-1β, pro-IL-18,
and NLRP3 protein (39). MSU
crystals serve as the second signal, inducing NLRP3
oligomerization, recruitment of apoptosis-associated speck-like
protein containing a CARD, and activation of caspase-1. This
process has been shown to catalyze the cleavage of pro-IL-1β and
pro-IL-18 into their mature forms (40). IL-1β and IL-18 then bind to their
receptors to trigger downstream signaling cascades, activating
pro-inflammatory cytokines and chemokines, and promoting the
recruitment of immune cells, including neutrophils and macrophages,
to sites of crystal deposition (41).
Research has demonstrated that the NLRP3
inflammasome, a key inflammatory mediator in gouty arthritis,
exacerbates bone resorption under conditions of estrogen deficiency
or prolonged parathyroid hormone exposure. Notably, NLRP3 deletion
has been reported to mitigate bone loss in models with high bone
turnover (42). Experimental
evidence suggests that the NLRP3 inflammasome impairs osteogenic
differentiation by inhibiting deacetylase 1, thereby promoting
adipogenesis in BMSCs, a process activated by lipopolysaccharides
and palmitic acid (43).
Furthermore, studies indicate that the downstream NF-κB and MAPK
signaling pathways, activated by the NLRP3 inflammasome and IL-1β,
upregulate RANKL and M-CSF production. This cascade has been
reported to induce osteoclastogenesis, reduce OPG expression in
osteoblasts, and disrupt bone remodeling (44,45).
IL-1β, a major inflammatory cytokine, has been shown to facilitate
the sustained secretion of matrix-degrading enzymes, contributing
to cartilage and bone degradation (46). Mature IL-1β was demonstrated to
enhance the expression of RANKL in osteoblasts and BMSCs, promoting
osteoclastogenesis (47). It can
also act on T cells, B cells, and macrophages to increase RANKL
production (48), enhance
osteoclastogenesis through insulin-like growth factors and
chemokines in non-osteoclast cells (49), and inhibit TGF-β signaling and Runx2
activation, thereby blocking osteogenesis (50). IL-18, a downstream mediator in
MSU-induced inflammatory processes, has been reported to play a
critical role in stimulating Th17 cells to secrete IL-17, which
subsequently upregulates RANKL. IL-18 inhibition was shown to
reduce the Th17/Treg ratio in vitro, contributing to the
restoration of trabecular bone microarchitecture (51). Targeting the
NLRP3/caspase-1/IL-1β/IL-18 signaling axis can also ameliorate
estrogen deficiency-induced osteoporosis (52). Additionally, a Mendelian
randomization analysis using genome-wide data has identified a
correlation between IL-18 levels and osteoporosis risk (53).
NETs and bone metabolism
Research has shown that following their recruitment
to sites of inflammation, neutrophils release cytokines such as
IL-1, IL-6, and TNF-α, along with other mediators, including matrix
metalloproteinases (MMPs), prostaglandins, leukotrienes, ROS, and
lysosomal enzymes (54). These
substances have been reported to further promote the accumulation
of mononuclear phagocytes, neutrophils, and other immune cells at
inflammatory sites, initiating inflammatory cascades. Additionally,
neutrophils can release DNA, histones, and NETs through exocytosis.
The formation of large amounts of NETs has been shown to lead to
the development of aggregated NETs (aggNETs), which trap and
encapsulate MSU crystals, thereby preventing their accumulation and
limiting inflammation (55).
Research has shown that NETs are secreted by
activated neutrophils to capture and eliminate extracellular
pathogens, a process that relies on ROS. However, the structure and
function of NETs are nonspecific, with DAMPs serving as the primary
triggers for NET formation. Activation of NETs can lead to
uncontrolled inflammation. NETs induced by MSU crystals have been
found to contribute to the degradation and remodeling of local
cartilage and peripheral tissues and play a significant role in
bone erosion. NETs have been reported to inhibit the expression of
ALP and OPG in chondrocytes while promoting increased expression of
RANKL (9). Concurrently, NETs have
been shown to reduce osteoblast viability and stimulate RANKL
release from osteoblasts, thereby enhancing osteoclast activity and
inhibiting OPG formation (9,56).
Furthermore, NETs have been reported to facilitate
osteoclastogenesis through mechanisms involving the activation of
TLR4, histones, and neutrophil elastase, with carbamylated NETs
accelerating this process (57).
Cytokines such as IL-1β, IL-6, and TNF-α have been demonstrated to
directly promote osteoclastogenesis and bone resorption, while also
stimulating osteoblasts to produce RANKL and acting synergistically
with RANKL (58). IL-6 has been
shown to inhibit WNT/β-catenin signaling by upregulating TNF-α in
osteoblasts; suppression of IL-6 enhances the osteogenic capacity
of BMSCs and increases the expression of Runx2, ALP, osteopontin,
and Bglap (59). Notably, low
levels of TNF-α were demonstrated to promote osteoblast
proliferation, whereas high levels inhibited it (60).
Macrophage polarization and bone
metabolism
The interplay between macrophage polarization and
bone metabolism has been the focus of extensive research in recent
years. Macrophages, a phagocytic subset of leukocytes, are integral
to the innate immune system. Their polarization state is considered
‘dynamic’ rather than ‘fixed’, allowing rapid shifts between M1 and
M2 phenotypes in response to changes in the microenvironment.
Research indicates that macrophages in patients with acute gout
initially polarize towards the M1 phenotype and subsequently
transition to the M2 phenotype in later stages, whereas macrophages
in patients with chronic gout predominantly exhibit M2 polarization
(61). Nevertheless, an elevated
M1/M2 macrophage ratio may represent a critical phenotype
contributing to bone destruction in the context of prolonged
chronic inflammation, partially contradicting previous findings. In
MSU-induced gouty arthritis, research has shown that neutrophils
release TNF-α to promote M1 macrophage polarization, and these
macrophages in turn produce endogenous TNF-α (62). Furthermore, it has been demonstrated
that MSU facilitates M1 polarization through the miR-449a/NLRP3
axis and the STAT3/NF-κB signaling pathways, thereby disrupting the
M1/M2 balance (63). Such an
imbalance in M1/M2 ratios may result in bone metabolic disorders.
Experimental evidence indicates that activated M1 macrophages
produce substantial amounts of pro-inflammatory cytokines and ROS,
and can even transfer mitochondria to BMSCs, thereby inhibiting
their osteogenic differentiation (64). By contrast, M2 macrophages have been
reported to secrete significantly higher levels of bone
morphogenetic protein-2 than M0 and M1 macrophages, thereby
promoting the osteogenic differentiation of BMSCs (Fig. 2) (65).
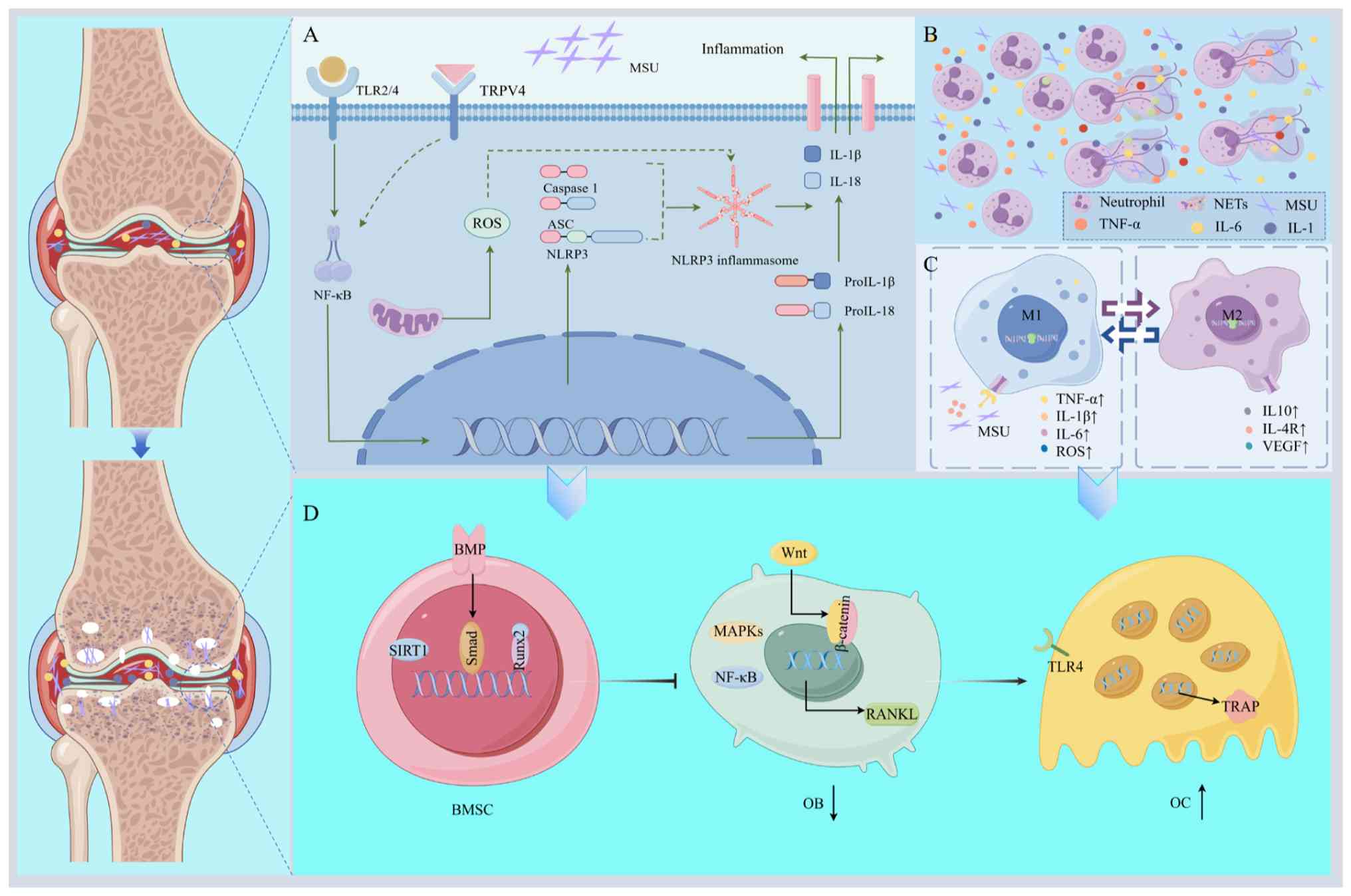 | Figure 2Mechanisms of bone destruction caused
by MSU-induced inflammation. (A) MSU-induced activation of the
NLRP3 inflammasome. (B) MSU-induced NETosis in neutrophils. (C)
MSU-induced macrophage polarization. (D) Inflammatory
cytokine-mediated modulation of BMSCs, osteoblasts and osteoclasts
in gouty arthritis-associated bone erosion. MSU, monosodium urate;
NLRP3, NOD-like receptor thermal protein domain-associated protein
3; BMSCs, bone mesenchymal stem cells; TRL, Toll-like receptor;
TRPV4, transient receptor potential vanilloid 4; ASC,
apoptosis-associated speck-like protein containing a CARD; ROS,
reactive oxygen species; Il-, interleukin; TNF-α, tumor necrosis
factor-α; NETs, neutrophil extracellular traps; VEGF, vascular
endothelial growth factor; BMPs, bone morphogenic proteins; SIRT1,
sirtuin 1; MAPK, mitogen-activated protein kinase; Runx2,
runt-related transcription factor 2; RANKL, receptor activator of
NF-κB ligand; TRAP, tartrate-resistant acid phosphatase; OB,
osteoblasts; OC, osteoclasts. |
5. Other influencing factors (vitamin D and
estrogen)
An observational study involving Han Chinese women
found a significant correlation between hyperuricemia and reduced
vitamin D levels, particularly among patients with chronic kidney
disease (66). MSU crystals have
been found to suppress 1α-hydroxylase protein and mRNA expression
in proximal renal tubular cells, thereby impairing the conversion
of 25-hydroxyvitamin D (25(OH)D) to its active form,
1,25-dihydroxyvitamin D (1,25(OH)2D), and ultimately decreasing the
levels of 1,25(OH)2D. Treatment with the urate-lowering agent
allopurinol has been observed to increase vitamin D levels in
affected patients (67). Vitamin D
is critical for regulating osteoblast function, extracellular
matrix mineralization, and osteoclast differentiation through the
induction of RANKL expression in osteoblasts. Notably,
physiological doses of 1,25-dihydroxyvitamin D3 have been shown to
enhance osteoblast activity and number, whereas supraphysiological
doses tend to increase osteoclast activity (68). Consequently, uric acid may influence
bone resorption and formation by inhibiting vitamin D activation.
Furthermore, the interaction between serum uric acid and vitamin D
warrants further investigation (69,70).
Epidemiological studies have revealed that gout is
3-10 times more prevalent in males than in females and is strongly
linked to estrogen levels (71). A
nationwide Chinese health survey identified significant differences
in the prevalence of hyperuricemia and gout between pre- and
post-menopausal women (72).
Another cross-sectional study found that endogenous estrogen
exposure reduces the risk of gout, whereas exogenous hormone
exposure increases it (73), likely
due to the role of estrogen in promoting renal urate excretion.
However, no evidence suggests that uric acid or MSU inversely
regulates estrogen. Clinical evidence indicates that combining
estrogen-progestogen therapy with antihypertensive treatment in
post-menopausal women prevents hyperuricemia (74).
6. Conclusion
Oxidative stress is widely acknowledged as an
independent risk factor for osteoporosis. Although MSU exists in
ionic form (Na+ and urate ions) dissolved in body fluids
and exhibits antioxidant properties at low serum uric acid
concentration, long-term elevation of serum uric acid causes
soluble MSU to promote oxidative stress and precipitate as solid
crystals that deposit in joints. Meanwhile, ROS generated during
uric acid production plays a pivotal role in local bone destruction
associated with gouty arthritis. Notably, the influence of
inflammation on bone metabolism varies across diseases. For
example, local inflammatory responses during fracture repair
facilitate angiogenesis and stimulate the proliferation and
differentiation of BMSCs and osteoprogenitor cells (75). In heterotopic ossification,
inflammatory mediators such as TNF-α and IL-17 induce aberrant
expression of bone morphogenic proteins (76). Clinically, tophi contribute not only
to joint erosion but also affect tendons, skin, and even the spinal
cord (77-79),
likely due to MSU-induced inflammatory infiltration and
MMP-mediated matrix degradation (80), underscoring the widespread nature of
inflammatory damage triggered by MSU.
MSU crystals significantly disrupt the function of
bone-related cells. In chondrocytes, MSU reduces cell viability in
a dose-dependent manner, triggers autophagic cell death, and
suppresses the production of type II collagen and other cartilage
matrix proteins, resulting in structural disorganization. MSU also
forms complexes with type II collagen, which enhances macrophage
phagocytosis and further amplifies inflammatory responses. In
osteoblasts, MSU directly suppresses key transcription factors
(Runx2 and Sp7) as well as osteogenic markers such as Ibsp, Bglap,
while reducing ALP activity, thereby impairing bone matrix
mineralization. Additionally, MSU disrupts the OPG/RANKL balance,
indirectly favoring osteoclast differentiation. Although the direct
role of MSU in osteoclastogenesis remains controversial, its
pro-osteoclastic effects are markedly enhanced in inflammatory
microenvironments, such as those involving neutrophil extracellular
traps. This is reflected by increased secretion of TRAP and Ctsk,
and along with heightened bone resorption mediated through the
calcineurin-NFATc1 and JNK signaling pathways.
MSU crystals generate substantial amounts of ROS
during purine metabolism and inhibit Nrf2-mediated antioxidant
pathways, creating a pro-oxidative milieu. Elevated ROS levels
impair BMSC osteogenic differentiation by disrupting WNT/β-catenin
signaling (for instance, KLF5 methylation and Nrf2 competitive
binding) and activate NF-κB and MAPK pathways to upregulate RANKL
expression, thereby further driving osteoclastogenesis. MSU
activates the NLRP3 inflammasome through a dual-signal mechanism:
TLR/TRPV4-mediated NF-κB signaling initiates the synthesis of
pro-inflammatory cytokines (IL-1β and IL-18), while crystal
deposition triggers NLRP3 oligomerization, caspase-1 activation,
and cytokine maturation. IL-1β and IL-18 exacerbate bone remodeling
imbalance by stimulating RANKL production, suppressing TGF-β/Runx2
pathways, and promoting osteoclast activity. NETs encapsulate MSU
crystals to form aggNETs, which suppress osteoblast OPG expression
and enhance osteoclast function. Macrophage polarization,
characterized by an M1/M2 imbalance, further aggravates bone
destruction through the release of TNF-α, IL-6, and ROS.
In this review, the mechanisms by which MSU crystals
contribute to bone destruction were elucidated, highlighting both
their direct effects on bone cell metabolism and indirect pathways
involving oxidative stress and inflammatory responses. An overview
of the clinical implications of bone damage associated with gouty
arthritis was first provided, emphasizing the strong
epidemiological and radiological associations between MSU
deposition and osteolysis. Experimental evidence that demonstrates
the direct inhibitory effects of MSU on bone-related cells in
vitro was then synthesized. Subsequently, the molecular
mechanisms by which oxidative stress and inflammation, arising
during MSU formation and metabolism, disrupt bone homeostasis were
analyzed, while their interactions with vitamin D and estrogen were
also considered. Overall, the mechanisms underlying MSU-induced
bone destruction are complex and multifaceted, occasionally
exhibiting bidirectional regulatory effects.
With the improvement in living standards and changes
in lifestyle habits, the incidence of gouty arthritis is likely to
continue rising and remain associated with a high burden of
disability. Currently, specific clinical therapies targeting bone
destruction are lacking. Consequently, an in-depth understanding of
the mechanisms underlying MSU-induced bone destruction is
essential. Exploring therapeutic strategies that target ROS, NLRP3,
NETs, or macrophage polarization may provide valuable guidance for
future clinical drug development.
Acknowledgements
Not applicable.
Funding
Funding: The present review was supported by grants from the
National Natural Science Foundation of China (grant no. 82560945),
the Jiangxi Province 2024 Science and Technology Special Funds
(grant no. 20243BCE51009) and the Jiangxi University of Chinese
Medicine University-level Graduate Student Innovation Special Fund
Project (grant no. XJ-B202406).
Availability of data and materials
Not applicable.
Authors' contributions
LH and HuaL conceived and designed the review. LH
and HuiL wrote major sections of the manuscript. HX, WS and QY
performed literature searches. HuaL provided funding. All authors
read and approved the final manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen L, Gamble GD, Horne A, Drake J, Doyle
AJ, Uhlig T, Stamp LK and Dalbeth N: Changes in tophus composition
during Urate-lowering therapy: A dual-energy computed tomography
study. Arthritis Care Res (Hoboken). 75:1949–1954. 2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Espinel DA, Martínez DC, Gómez MÁ, Duque
DF, Torres PA and Rincón JV: Surgical management of tophaceous gout
in the upper limb. J Hand Surg Am. 49:1275.e1–1275.e10.
2024.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Chhana A, Callon KE, Pool B, Naot D,
Watson M, Gamble GD, Mcqueen FM, Cornish J and Dalbeth N:
Monosodium urate monohydrate crystals inhibit osteoblast viability
and function: Implications for development of bone erosion in gout.
Ann Rheum Dis. 70:1684–1691. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Towiwat P, Doyle AJ, Gamble GD, Tan P,
Aati O, Horne A, Stamp LK and Dalbeth N: Urate crystal deposition
and bone erosion in gout: ‘Inside-out’ or ‘outside-in’? A
dual-energy computed tomography study. Arthritis Res Ther.
18(208)2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zou Y, Fei Y, Gao H, Xie LF, Zhong YC,
Yang QZ, Wang DL and Zhang XW: Association between musculoskeletal
ultrasonography and bone remodelling markers and its role in
disease monitoring of gout and hyperuricaemia. Clin Exp Rheumatol.
38:896–902. 2020.PubMed/NCBI
|
|
6
|
Dalbeth N, Smith T, Nicolson B, Clark B,
Callon K, Naot D, Haskard DO, Mcqueen FM, Reid IR and Cornish J:
Enhanced osteoclastogenesis in patients with tophaceous gout: Urate
crystals promote osteoclast development through interactions with
stromal cells. Arthritis Rheum. 58:1854–1865. 2008.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jia E, Zhu H, Geng H, Zhong L, Qiu X, Xie
J, Xiao Y, Jiang Y, Xiao M, Zhang Y, et al: The inhibition of
osteoblast viability by monosodium Urate Crystal-stimulated
neutrophil-derived exosomes. Front Immunol.
13(809586)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lopez-Reyes A, Medina-Luna D,
Santamaria-Olmedo M, Martinez-Flores K, Zamudio-Cuevas Y,
Fernandez-Torres J, Martinez-Nava GA, Olivos-Meza A, Camacho-Rea C,
Fernandez-Moreno M, et al: Soluble inflammatory mediators of
synoviocytes stimulated by monosodium urate crystals induce the
production of oxidative stress, pain, and inflammation mediators in
chondrocytes: Secretome of synoviocytes induces chondrocyte damage.
Clin Rheumatol. 40:3265–3271. 2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Jia E, Li Z, Geng H, Zhu H, Wang Y, Lin F,
Jiang Y and Zhang J: Neutrophil extracellular traps induce the bone
erosion of gout. BMC Musculoskelet Disord. 23(1128)2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Naot D, Pool B, Chhana A, Gao R, Munro JT,
Cornish J and Dalbeth N: Factors secreted by monosodium urate
crystal-stimulated macrophages promote a proinflammatory state in
osteoblasts: A potential indirect mechanism of bone erosion in
gout. Arthritis Res Ther. 24(212)2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chhana A, Pool B, Callon KE, Tay ML,
Musson D, Naot D, Mccarthy G, Mcglashan S, Cornish J and Dalbeth N:
Monosodium urate crystals reduce osteocyte viability and indirectly
promote a shift in osteocyte function towards a proinflammatory and
proresorptive state. Arthritis Res Ther. 20(208)2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chhana A, Callon KE, Pool B, Naot D,
Gamble GD, Dray M, Pitto R, Bentley J, Mcqueen FM, Cornish J and
Dalbeth N: The effects of monosodium urate monohydrate crystals on
chondrocyte viability and function: Implications for development of
cartilage damage in gout. J Rheumatol. 40:2067–2074.
2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Cao Y: Icariin alleviates MSU-induced rat
GA models through NF-kappaB/NALP3 pathway. Cell Biochem Funct.
39:357–366. 2021.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Hwang HS, Yang CM, Park SJ and Kim HA:
Monosodium Urate Crystal-induced chondrocyte death via autophagic
process. Int J Mol Sci. 16:29265–29277. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yan B, Liu D, Zhu J and Pang X: The
effects of hyperuricemia on the differentiation and proliferation
of osteoblasts and vascular smooth muscle cells are implicated in
the elevated risk of osteopenia and vascular calcification in gout:
An in vivo and in vitro analysis. J Cell Biochem. 120:19660–19672.
2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Nam JS, Jagga S, Sharma AR, Lee JH, Park
JB, Jung JS and Lee SS: Anti-inflammatory effects of traditional
mixed extract of medicinal herbs (MEMH) on monosodium urate
crystal-induced gouty arthritis. Chin J Nat Med. 15:561–575.
2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Lee SJ, Nam KI, Jin HM, Cho YN, Lee SE,
Kim TJ, Lee SS, Kee SJ, Lee KB, Kim N and Park YW: Bone destruction
by receptor activator of nuclear factor κB ligand-expressing T
cells in chronic gouty arthritis. Arthritis Res Ther.
13(R164)2011.PubMed/NCBI View
Article : Google Scholar
|
|
18
|
Cunningham CC, Corr EM, Mccarthy GM and
Dunne A: Intra-articular basic calcium phosphate and monosodium
urate crystals inhibit anti-osteoclastogenic cytokine signalling.
Osteoarthritis Cartilage. 24:2141–2152. 2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kim SK, Choe JY, Kim JW and Park KY:
Histone Deacetylase 6 inhibitor CKD-WID suppressed monosodium
urate-induced osteoclast formation by blocking Calcineurin-NFAT
pathway in RAW 264.7 cells. Pharmaceuticals (Basel).
16(446)2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Martinez-Flores K, Plata-Rodriguez R,
Olivos-Meza A, Lopez-Macay A, Fernandez-Torres J, Landa-Solis C and
Zamudio-Cuevas Y: Osteogenic potential of monosodium urate crystals
in synovial mesenchymal stem cells. Medicina (Kaunas).
58(1724)2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yang K, Li J and Tao L: Purine metabolism
in the development of osteoporosis. Biomed Pharmacother.
155(113784)2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Bortolotti M, Polito L, Battelli MG and
Bolognesi A: Xanthine oxidoreductase: One enzyme for multiple
physiological tasks. Redox Biol. 41(101882)2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yin C, Lyu Q, Dong Z, Liu B, Zhang K, Liu
Z, Yu Q, Li P, Wei Z, Tai Y, et al: Well-defined alginate
oligosaccharides ameliorate joint pain and inflammation in a mouse
model of gouty arthritis. Theranostics. 14:3082–3103.
2024.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Yin C, Liu B, Li Y, Li X, Wang J, Chen R,
Tai Y, Shou Q, Wang P, Shao X, et al: IL-33/ST2 induces
neutrophil-dependent reactive oxygen species production and
mediates gout pain. Theranostics. 10:12189–12203. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Li Q, Gao Z, Chen Y and Guan MX: The role
of mitochondria in osteogenic, adipogenic and chondrogenic
differentiation of mesenchymal stem cells. Protein Cell. 8:439–445.
2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Tao H, Ge G, Liang X, Zhang W, Sun H, Li M
and Geng D: ROS signaling cascades: Dual regulations for osteoclast
and osteoblast. Acta Biochim Biophys Sin (Shanghai). 52:1055–1062.
2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Agidigbi TS and Kim C: Reactive oxygen
species in osteoclast differentiation and possible pharmaceutical
targets of ROS-mediated osteoclast diseases. Int J Mol Sci.
20(3576)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Li L, Wang H, Chen X, Li X, Wang G, Jie Z,
Zhao X, Sun X, Huang H, Fan S, et al: Oxidative stress-induced
hypermethylation of KLF5 promoter mediated by DNMT3B impairs
osteogenesis by diminishing the interaction with beta-Catenin.
Antioxid Redox Signal. 35:1–20. 2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Rai D, Tripathi AK, Sardar A, Pandey AR,
Sinha S, Chutani K, Dhaniya G, Kothari P, Sashidhara KV and Trivedi
R: A novel BMP2 secretagogue ameliorates glucocorticoid induced
oxidative stress in osteoblasts by activating NRF2 dependent
survival while promoting Wnt/β-catenin mediated osteogenesis. Free
Radic Biol Med. 190:124–147. 2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Liu M, Liu S and Zhang Q, Fang Y, Yu Y,
Zhu L, Liu Y, Gong W, Zhao L, Qin L and Zhang Q: Curculigoside
attenuates oxidative stress and osteoclastogenesis via modulating
Nrf2/NF-κB signaling pathway in RAW264.7 cells. J Ethnopharmacol.
275(114129)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Nakanishi A, Hie M, Iitsuka N and
Tsukamoto I: A crucial role for reactive oxygen species in
macrophage colony-stimulating factor-induced RANK expression in
osteoclastic differentiation. Int J Mol Med. 31:874–880.
2013.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kim B, Lee KY and Park B: Icariin
abrogates osteoclast formation through the regulation of the
RANKL-mediated TRAF6/NF-κB/ERK signaling pathway in Raw264.7 cells.
Phytomedicine. 51:181–190. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Xiao L, Zhong M, Huang Y, Zhu J, Tang W,
Li D, Shi J, Lu A, Yang H, Geng D, et al: Puerarin alleviates
osteoporosis in the ovariectomy-induced mice by suppressing
osteoclastogenesis via inhibition of TRAF6/ROS-dependent MAPK/NF-κB
signaling pathways. Aging (Albany NY). 12:21706–21729.
2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Liu L, Geng H, Mei C and Chen L:
Zoledronic acid enhanced the antitumor effect of cisplatin on
orthotopic osteosarcoma by ROS-PI3K/AKT signaling and attenuated
osteolysis. Oxid Med Cell Longev. 2021(6661534)2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Rea IM, Gibson DS, Mcgilligan V, Mcnerlan
SE, Alexander HD and Ross OA: Age and Age-related diseases: Role of
inflammation triggers and cytokines. Front Immunol.
9(586)2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
An Y, Zhang H, Wang C, Jiao F, Xu H, Wang
X, Luan W, Ma F, Ni L, Tang X, et al: Activation of
ROS/MAPKs/NF-κB/NLRP3 and inhibition of efferocytosis in
osteoclast-mediated diabetic osteoporosis. FASEB J. 33:12515–12527.
2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Wang Y, Li X, Zhou S, Li J, Zhu Y, Wang Q
and Zhao F: MCU inhibitor ruthenium red alleviates the
osteoclastogenesis and ovariectomized osteoporosis via suppressing
RANKL-induced ROS production and NFATc1 activation through P38 MAPK
signaling pathway. Oxid Med Cell Longev.
2022(7727006)2022.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Tan H, Zhang S, Zhang Z, Zhang J, Wang Z,
Liao J, Qiu X and Jia E: Neutrophil extracellular traps promote M1
macrophage polarization in gouty inflammation via targeting
Hexokinase-2. Free Radic Biol Med. 224:540–553. 2024.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Lan Z, Chen L, Feng J, Xie Z, Liu Z, Wang
F, Liu P, Yue X, Du L, Zhao Y, et al: Mechanosensitive TRPV4 is
required for crystal-induced inflammation. Ann Rheum Dis.
80:1604–1614. 2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Martinon F, Petrilli V, Mayor A, Tardivel
A and Tschopp J: Gout-associated uric acid crystals activate the
NALP3 inflammasome. Nature. 440:237–241. 2006.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Galozzi P, Bindoli S, Doria A, Oliviero F
and Sfriso P: Auto-inflammatory features in gouty arthritis. J Clin
Med. 10(1880)2021.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Alippe Y, Wang C, Ricci B, Xiao J, Qu C,
Zou W, Novack DV, Abu-Amer Y, Civitelli R and Mbalaviele G: Bone
matrix components activate the NLRP3 inflammasome and promote
osteoclast differentiation. Sci Rep. 7(6630)2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Wang L, Chen K, Wan X, Wang F, Guo Z and
Mo Z: NLRP3 inflammasome activation in mesenchymal stem cells
inhibits osteogenic differentiation and enhances adipogenic
differentiation. Biochem Biophys Res Commun. 484:871–877.
2017.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Liang S, Nian Z and Shi K: Inhibition of
RIPK1/RIPK3 ameliorates osteoclastogenesis through regulating
NLRP3-dependent NF-κB and MAPKs signaling pathways. Biochem Biophys
Res Commun. 526:1028–1035. 2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Mcdonald MM, Khoo WH, Ng PY, Xiao Y,
Zamerli J, Thatcher P, Kyaw W, Pathmanandavel K, Grootveld AK,
Moran I, et al: Osteoclasts recycle via osteomorphs during
RANKL-stimulated bone resorption. Cell. 184:1330–1347.e13.
2021.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Schlesinger N and Thiele RG: The
pathogenesis of bone erosions in gouty arthritis. Ann Rheum Dis.
69:1907–1912. 2010.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Pietschmann P, Mechtcheriakova D,
Meshcheryakova A, Foger-Samwald U and Ellinger I: Immunology of
osteoporosis: A mini-review. Gerontology. 62:128–137.
2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Ruscitti P, Cipriani P, Carubbi F,
Liakouli V, Zazzeroni F, Di Benedetto P, Berardicurti O, Alesse E
and Giacomelli R: The role of IL-1β in the bone loss during
rheumatic diseases. Mediators Inflamm. 2015(782382)2015.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Otsuka Y, Kondo T, Aoki H, Goto Y,
Kawaguchi Y, Waguri-Nagaya Y, Miyazawa K, Goto S and Aoyama M:
IL-1β promotes osteoclastogenesis by increasing the expression of
IGF2 and chemokines in non-osteoclastic cells. J Pharmacol Sci.
151:1–8. 2023.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Mao CY, Wang YG, Zhang X, Zheng XY, Tang
TT and Lu EY: Double-edged-sword effect of IL-1β on the
osteogenesis of periodontal ligament stem cells via crosstalk
between the NF-κB, MAPK and BMP/Smad signaling pathways. Cell Death
Dis. 7(e2296)2016.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Mansoori MN, Shukla P, Kakaji M, Tyagi AM,
Srivastava K, Shukla M, Dixit M, Kureel J, Gupta S and Singh D:
IL-18BP is decreased in osteoporotic women: Prevents Inflammasome
mediated IL-18 activation and reduces Th17 differentiation. Sci
Rep. 6(33680)2016.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Qiao S, Zhang X, Chen Z, Zhao Y and Tzeng
CM: Alloferon-1 ameliorates estrogen deficiency-induced
osteoporosis through dampening the NLRP3/caspase-1/IL-1β/IL-18
signaling pathway. Int Immunopharmacol. 124(110954)2023.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Kou N, Zhou W, He Y, Ying X, Chai S, Fei
T, Fu W, Huang J and Liu H: A Mendelian randomization analysis to
expose the causal effect of IL-18 on osteoporosis based on
genome-wide association study data. Front Bioeng Biotechnol.
8(201)2020.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Szekanecz Z, Szamosi S, Kovacs GE, Kocsis
E and Benko S: The NLRP3 inflammasome-interleukin 1 pathway as a
therapeutic target in gout. Arch Biochem Biophys. 670:82–93.
2019.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Chen T, Zhou J and Dang W: Mechanism of
neutrophil extracellular traps in the pathogenesis of gout. Clin
Exp Rheumatol. 42:2272–2279. 2024.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Schneider AH, Taira TM, Publio GA, Da
Silva Prado D, Donate Yabuta PB, Dos Santos JC, Machado CC, De
Souza FFL, Rodrigues Venturini LG, De Oliveira RDR, et al:
Neutrophil extracellular traps mediate bone erosion in rheumatoid
arthritis by enhancing RANKL-induced osteoclastogenesis. Br J
Pharmacol. 181:429–446. 2024.PubMed/NCBI View Article : Google Scholar
|
|
57
|
O'neil LJ, Oliveira CB, Wang X, Navarrete
M, Barrera-Vargas A, Merayo-Chalico J, Aljahdali R, Aguirre-Aguilar
E, Carlucci P, Kaplan MJ and Carmona-Rivera C: Neutrophil
extracellular trap-associated carbamylation and histones trigger
osteoclast formation in rheumatoid arthritis. Ann Rheum Dis.
82:630–638. 2023.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Kwan Tat S, Padrines M, Theoleyre S,
Heymann D and Fortun Y: IL-6, RANKL, TNF-alpha/IL-1: Interrelations
in bone resorption pathophysiology. Cytokine Growth Factor Rev.
15:49–60. 2004.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Malysheva K, De Rooij K, Lowik CW, Baeten
DL, Rose-John S, Stoika R and Korchynskyi O: Interleukin 6/Wnt
interactions in rheumatoid arthritis: Interleukin 6 inhibits Wnt
signaling in synovial fibroblasts and osteoblasts. Croat Med J.
57:89–98. 2016.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Karnes JM, Daffner SD and Watkins CM:
Multiple roles of tumor necrosis factor-alpha in fracture healing.
Bone. 78:87–93. 2015.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Zhao L, Ye W, Zhu Y, Chen F, Wang Q, Lv X,
Hua Y, Du Z, Zhu X, Yu Y, et al: Distinct macrophage polarization
in acute and chronic gout. Lab Invest. 102:1054–1063.
2022.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Shapouri-Moghaddam A, Mohammadian S,
Vazini H, Taghadosi M, Esmaeili SA, Mardani F, Seifi B, Mohammadi
A, Afshari JT and Sahebkar A: Macrophage plasticity, polarization,
and function in health and disease. J Cell Physiol. 233:6425–6440.
2018.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Wang Y: Tripterine ameliorates monosodium
urate crystal-induced gouty arthritis by altering macrophage
polarization via the miR-449a/NLRP3 axis. Inflamm Res. 70:323–341.
2021.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Cai W, Zhang J, Yu Y, Ni Y, Wei Y, Cheng
Y, Han L, Xiao L, Ma X, Wei H, et al: Mitochondrial transfer
regulates cell fate through metabolic remodeling in osteoporosis.
Adv Sci (Weinh). 10(e2204871)2023.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Zhang Y, Bose T, Unger RE, Jansen JA,
Kirkpatrick CJ and Van Den Beucken J: Macrophage type modulates
osteogenic differentiation of adipose tissue MSCs. Cell Tissue Res.
369:273–286. 2017.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Peng H, Li H, Li C, Chao X, Zhang Q and
Zhang Y: Association between vitamin D insufficiency and elevated
serum uric acid among middle-aged and elderly Chinese Han women.
PLoS One. 8(e61159)2013.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Chen W, Roncal-Jimenez C, Lanaspa M,
Gerard S, Chonchol M, Johnson RJ and Jalal D: Uric acid suppresses
1 alpha hydroxylase in vitro and in vivo. Metabolism. 63:150–160.
2014.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Christakos S, Dhawan P, Verstuyf A,
Verlinden L and Carmeliet G: Vitamin D: Metabolism, molecular
mechanism of action, and pleiotropic effects. Physiol Rev.
96:365–408. 2016.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Sugimoto R, Watanabe H, Ikegami K, Enoki
Y, Imafuku T, Sakaguchi Y, Murata M, Nishida K, Miyamura S, Ishima
Y, et al: Down-regulation of ABCG2, a urate exporter, by
parathyroid hormone enhances urate accumulation in secondary
hyperparathyroidism. Kidney Int. 91:658–670. 2017.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Copur S, Demiray A and Kanbay M: Uric acid
in metabolic syndrome: Does uric acid have a definitive role? Eur J
Intern Med. 103:4–12. 2022.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Singh JA and Gaffo A: Gout epidemiology
and comorbidities. Semin Arthritis Rheum. 50:S11–S16.
2020.PubMed/NCBI View Article : Google Scholar
|
|
72
|
He H, Pan L, Liu F, Ren X, Cui Z, Pa L,
Zhao J, Wang D, Du J, Wang H, et al: The mediation effect of body
composition on the association between menopause and hyperuricemia:
Evidence from china national health survey. Front Endocrinol
(Lausanne). 13(879384)2022.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Eun Y, Kim IY, Han K, Lee KN, Lee DY, Shin
DW, Kang S, Lee S, Cha HS, Koh EM, et al: Association between
female reproductive factors and gout: A nationwide population-based
cohort study of 1 million postmenopausal women. Arthritis Res Ther.
23(304)2021.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Posadzy-Malaczynska A, Rajpold K,
Woznicka-Leskiewicz L and Marcinkowska J: Reversal of an
unfavorable effect of hydrochlorothiazide compared to angiotensin
converting enzyme inhibitor on serum uric acid and oxypurine levels
by estrogen-progestin therapy in hypertensive postmenopausal women.
Curr Med Res Opin. 35:1687–1697. 2019.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Goodnough LH and Goodman SB: Relationship
of aging, inflammation, and skeletal stem cells and their effects
on fracture repair. Curr Osteoporos Rep. 20:320–325.
2022.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Van Der Kraan PM and Davidson EN:
Cross-talk between bone morphogenetic proteins and inflammatory
pathways. Arthritis Res Ther. 17(326)2015.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Goebel L, Schneider G, Bohle R, Veith C
and Orth P: Gouty tophus in the quadriceps tendon: exclude
malignancy. Lancet. 394(2197)2019.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Bao W, Xue Y, Cheng X, Wang P, Yin B, Su Y
and Jia C: Gout-associated uric acid crystals induce tophi
ulcerations and impair wound healing in a novel gouty ulcer model.
Wound Repair Regen. 30:132–139. 2022.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Si M, Cong M, Wang D and Ma H: Teaching
NeuroImages: Intraspinal gouty tophus. Neurology. 96:e159–e160.
2021.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Peng Z, Ding YM, Pei L, Yao HH, Zhang XW
and Tang SM: Clinical characteristics of crystal deposits in joints
and tendons in patients with gout. Beijing Da Xue Xue Bao Yi Xue
Ban. 53:1067–1071. 2021.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
















