Introduction
Inflammatory bowel disease (IBD) encompasses two
main disorders: Crohn's disease and ulcerative colitis. These
chronic conditions cause gastrointestinal (GI) tract inflammation.
Although the exact cause of IBD is unknown, it involves a
combination of genetic (1),
environmental (2), and immune
factors (3). In Crohn's disease,
inflammation can occur anywhere along the GI tract, from the mouth
to the anus, and it often affects the deeper layers of the bowel
(4). By contrast, ulcerative
colitis is confined to the colon and rectum, and the associated
inflammation is typically restricted to the innermost lining of the
colon (5). The symptoms of IBD vary
depending on the severity and location of inflammation; however,
they commonly include abdominal pain, diarrhea, rectal bleeding,
weight loss, and fatigue. Besides, the disease can lead to
complications such as strictures and fistulas and an increased risk
of colorectal cancer. Diagnosis typically involves a combination of
endoscopic procedures, imaging studies, and laboratory tests to
assess inflammation and exclude other conditions. The management of
IBD focuses on reducing inflammation, controlling symptoms, and
achieving and maintaining remission. Treatment options include
anti-inflammatory drugs, immune system suppressors, biologics, and
lifestyle modifications such as diet changes and stress management
(6). In severe cases, surgery may
be necessary to remove the damaged portions of the GI tract. Recent
experimental studies have reported anti-inflammatory effects of
several compounds in colitis models. Isoliquiritin, a flavonoid
glycoside, attenuated TNBS-induced colitis through suppression of
the caspase-3/HMGB1/TLR4-dependent pathway (7). A novel bipyrazole derivative, TMNB,
reduced disease activity and inflammatory mediators in experimental
colitis (8). In addition,
Artepillin C has been proposed as a candidate intestinal
anti-inflammatory and antitumor compound, potentially through
modulation of PAK1/NF-κB and PPAR-γ-related signaling (9). With proper treatment and monitoring,
many patients with IBD can lead active and fulfilling lives.
Ezoukogi (Acanthopanax senticosus Harms,
ASH), also known as Siberian ginseng or Eleuthero, is a deciduous
shrub native to the cold northern regions of Japan, China, and
Hokkaido (10). ASH is widely used
as a traditional herbal medicine and has recently been marketed as
a dietary supplement in Japan and Western countries. It contains
several active ingredients, including phytochemicals, which have
shown therapeutic effects in diabetes (11), allergies (12), gastric ulcers (13), neurodegenerative diseases (14), and cancers (15). In addition, ASH roots are known to
inhibit inflammation and oxidative stress (16) in a mouse model of arthritis, and the
aforementioned effects on IBD have been suggested to be
beneficial.
Among experimental models of colitis, the
DSS-induced model is one of the most widely used because of its
simplicity and reproducibility. Acute DSS-induced colitis is
generally considered to more closely resemble UC than Crohn's
disease, particularly with respect to colon-restricted epithelial
injury, diarrhea, rectal bleeding, crypt damage, and superficial
mucosal inflammation. In the present study, we investigated whether
ASH pretreatment attenuates acute DSS-induced experimental colitis
in mice.
Materials and methods
Animals
Six-week-old C57BL6/6J female mice were purchased
from HOKUDO (Hokkaido, Japan). The mice were housed in groups of
four per cage under controlled conditions (23˚C temperature, a
12:12 h light-dark cycle, and a relative humidity of 50-55%), with
free access to food and water. The study protocol was approved by
the Animal Experiments Committee of Health Sciences University of
Hokkaido (approval number: 21-016) and performed in compliance with
the guidelines of the Committee of Animal Care and Use of Health
Sciences University of Hokkaido and ARRIVE guidelines.
Experimental design of DSS-induced
colitis
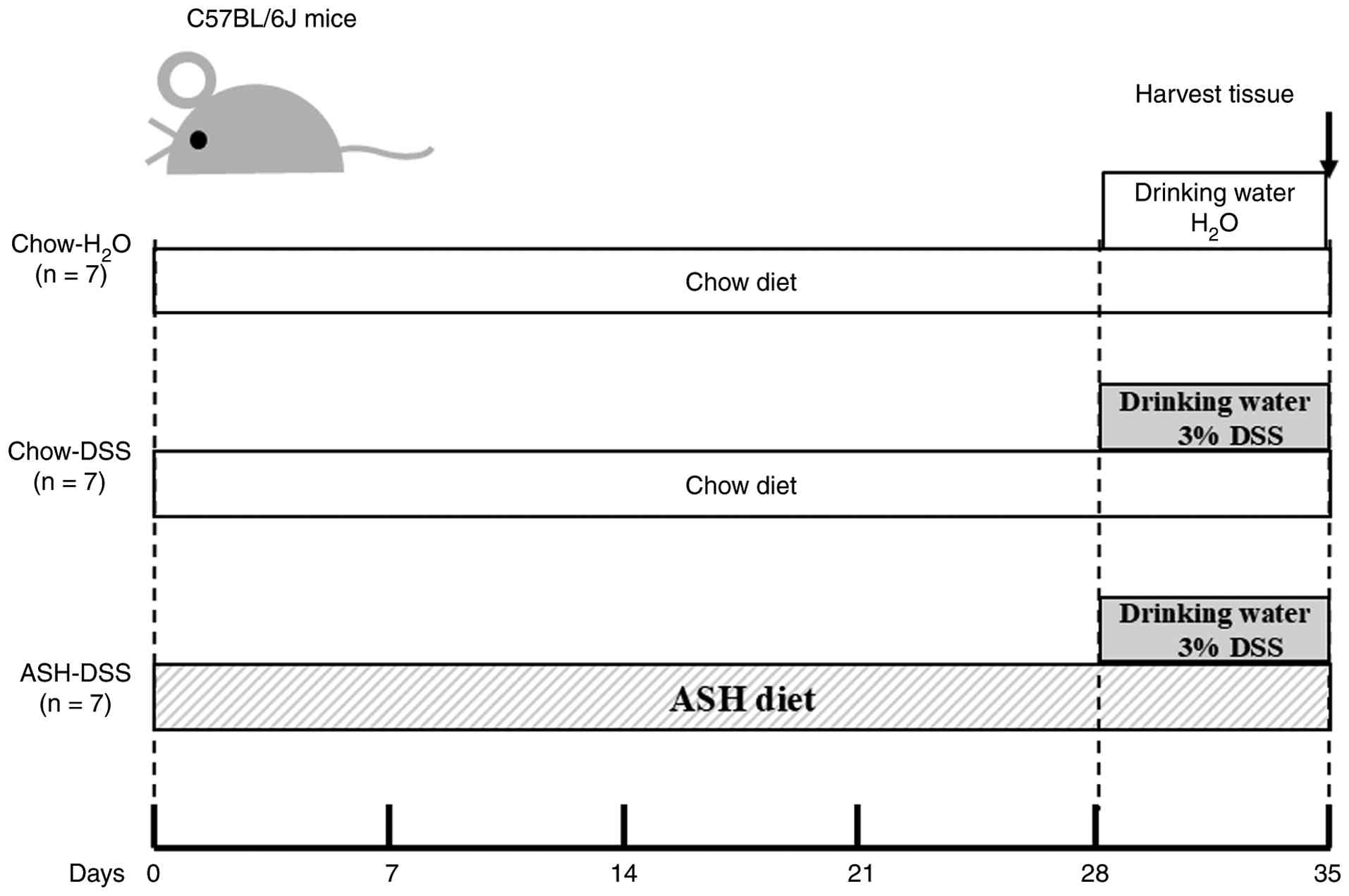
The experiments were designed as shown in Fig. 1. The mice were fed with either chow
diet alone (chow diet) or chow diet supplemented with 5% ASH (ASH
diet). The ASH used in this study was prepared and supplied by Sun
Chlorella Corp. (lot no. 5152). The processed diets were prepared
by Orient Yeast Corporation (Tokyo, Japan). After 4 weeks feeding,
the mice were administered with either H2O water or 3%
(w/v) DSS (MW 36,000-50,000; MP Biomedicals, Santa Ana. CA, USA) in
autoclaved drinking water for additional 7 days. Under deep
anesthesia with 5% isoflurane (VITAS Pharmaceuticals, Inc. Tokyo,
Japan), blood was collected from the inferior vena cava. Mice were
then euthanized by cervical dislocation, and death was confirmed by
cessation of respiration and heartbeat. Colon tissues were
collected immediately thereafter. During DSS administration, all
mice were monitored daily for body weight and clinical signs.
Predefined humane endpoints were as follows: >20% loss of
baseline body weight, inability to access food or water, and
moribund appearance. Mice that met any humane endpoint criterion
were humanely euthanized.
Assessment of colitis in mice
During DSS treatment, the severity of colitis was
evaluated using the disease activity index (DAI), as previously
described by Cooper et al (17). The DAI includes body weight loss (0,
none; 1, 1-5%; 2, 5-10%; 3, 10-20%; 4, >20%), stool consistency
(0, normal; 2, loose stool; 4, diarrhea), and rectal bleeding (0,
normal; 2, hemoccult; 4, gross bleeding). After DSS treatment for 7
days, the length of the colon was measured following separation of
the colon and cecum from the small intestine at the ileocecal
junction to the anus. Thereafter, the colon was fixed in 10%
buffered neutral formalin solution (MUTO PURE CHEMICALS, Tokyo,
Japan), processed for paraffin embedding, and sectioned into 4 µm
sections. Each section was deparaffinized, rehydrated, and stained
with H&E. The histological degree of colitis was evaluated
using the scoring system previously described by Williams et
al (18), which included
inflammation severity (0, none; 1, mild; 2, moderate; 3, severe),
extent of inflammation (0, none; 1, mucosa; 2, mucosa and
submucosa; 3, transmural), and crypt damage (0, none; 1, basal
one-third damaged; 2, basal two-thirds damaged; 3, crypt lost and
surface epithelium present; 4, loss of crypt and surface
epithelium). The histological score means the sum of these three
parameters.
Measurement of serum cytokine
Mice blood samples were collected from the inferior
vena cava, and their sera were separated via centrifugation at
3,000 rpm at 4˚C for 10 min and stored at -80˚C until measurement.
Cytokine concentrations were measured using Bio-Plex Pro Mouse
Cytokine 23-Plex Assay kits (Bio-Rad) according to the
manufacturer's protocol. This kit measures the concentration of 23
cytokines (IL-1α, IL-1β, IL-2, IL-3, IL-4, IL-5, IL-6, IL-9, IL-10,
IL-12p40, IL-12p70, IL-13, IL-17A, Eotaxin, G-CSF, GM-CSF, IFN-γ,
KC, MCP-1, MCP-1α, MCP-1β, RANTES, and TNF-α).
dROM and BAP measurements
The degree of oxidation and antioxidant defense
capacity, dROMs and BAP, in mice sera were measured by Wismerll
Co., Ltd. (Tokyo, Japan).
Immunostaining of NOX2
Mice colon sections were incubated with anti-NOX2
antibody (19013-1-AP, 1:1,000 dilution, Proteintech, Wuhan, China)
or anti-rabbit IgG (30,000-0-AP, 1:1,000 dilution, Proteintech,
Wuhan, China) overnight at 4˚C. The following day, the sections
were visualized with diaminobenzidine chromogen and counterstained
with Mayer's hematoxylin under a microscope (NIKON CORPORATION,
Tokyo, Japan). NOX2 expression was evaluated by their staining
pattern: weak (1), moderate
(2), diffuse (3) and intense (4) according to previously reported
(19).
Reverse transcription-quantitative PCR
(RT-qPCR) analysis
Total RNA was isolated from mice colons using RNeasy
Plus Mini Kit (Qiagen, Hilden, Germany). Total RNA concentration
was measured using NanoDrop spectrophotometer (NanoDrop
Technologies, Wilmington, DE, USA). cDNA was synthesized from 500
ng of total RNA using SuperScript IV First-Strand Synthesis System
(Invitrogen) according to the manufacturer's instructions. RT-qPCR
was performed in triplicate using the KAPA SYBR FAST qPCR Kit (Kapa
Biosystems, USA). PCR amplification protocol was as follows: 95˚C
for 3 min; 40 cycles of amplification (at 95˚C for 10 sec, 60˚C for
30 sec, and 72˚C for 5 sec). Changes in the NOX2 gene between cDNA
samples were determined using the 2-ΔΔCq method
(20). The primers were as follows:
NOX2 Forward primer; 5'-TCGAAAACTCCTTGGGTCAGC-3', Reverse primer;
5'-GTGCAATTGTGTGGATGGCG-3', Cu/Zn-superoxide dismutase (SOD)
Forward primer; 5'-AACCAGTTGTGTTGTCAGGAC-3', Reverse primer;
5'-CCACCATGTTTCTTAGAGTGAGG-3', nuclear factor erythroid 2-related
factor 2 (Nrf2) Forward primer; 5'-TCTTGGAGTAAGTCGAGAAGTGT-3',
Reverse primer; 5'-GTTGAAACTGAGCGAAAAAGGC-3', and GAPDH Forward
primer; 5'-GGCTGCCCAGAACATCAT-3', Reverse primer;
5'-CGGACACATTGGGGGTAG-3', and 18S ribosome Forward primer;
5'-AGTCCCTGCCCTTTGTACACA-3', Reverse primer;
5'-CGATCCGAGGGCCTCACTA-3'.
Statistical analysis
Statistical analyses were performed using
statistical analysis software (GraphPad Prism version 9.4.1).
Comparisons among the Chow-H2O, Chow-DSS and ASH-DSS
groups were conducted using one-way ANOVA followed by Tukey's post
hoc test. For ordinal data, including histological scores and
clinical scores, statistical analyses were performed using the
Kruskal-Wallis test followed by Dunn's multiple comparisons test.
For body weight, statistical comparisons were performed among
groups at each timepoint, and no within-group comparisons across
different timepoints were conducted. Statistical significance was
defined as P<0.05.
Results
Inhibitory effect of ASH diet on body
weight loss caused by dextran sulfate sodium (DSS)-induced
colitis
To examine the effect of ASH on mice weight, we
measured body weight of mice fed both the chow and the ASH diet for
28 days respectively. No significant change between mice fed the
chow and ASH diet was observed (Fig.
2A). After administration of 3% DSS, body weight loss was
gradually observed in mice fed the chow diet (Fig. 2B). However, the mice fed the ASH
diet showed the attenuation of body weight loss, which was similar
to the mice fed the chow diet and H2O water only (Day
35; Chow-H2O: 19.9±0.5 g, ASH-DSS: 19.3±1.4 g, Chow-DSS:
17.4±1.0 g).
Inhibition of colon length shortening
and exacerbation of the disease activity index (DAI) in mice fed
the ASH diet
After 3% DSS treatment for 7 days, the colon length
in mice fed the chow diet was significantly shortened
[Chow-H2O: 7.0±0.6 cm, Chow-DSS: 4.8±0.3 cm; P<0.01]
(Fig. 3A and B). Meanwhile, the mice fed the ASH diet
with DSS treatment showed a significant inhibition of colon length
shortening [ASH-DSS: 6.4±0.3 cm, P<0.01 vs. Chow-DSS]. The feces
of mice fed the chow diet with DSS (Chow-DSS) treatment showed
loose and gross bleeding (Fig. 3C).
The color of feces in mice fed the ASH diet with DSS treatment
(ASH-DSS) was dark brownish black, reflecting the original color of
ASH. Similar to the feces in mice fed the chow diet without DSS
treatment (Chow-H2O), fewer bloody and solid feces were
observed in mice fed the ASH diet with DSS treatment. Afterward, we
assessed the DAI score of colitis for 7 consecutive days (Day
28-35) during DSS treatment (Fig.
3D). The DAI score in mice fed the chow diet with DSS treatment
(Chow-DSS) gradually increased from day 31, whereas the increase in
DAI score in mice fed the ASH diet with DSS treatment (ASH-DSS)
tended to be attenuated compared with that in the Chow-DSS group,
suggesting that the ASH diet may mitigate the symptoms of
DSS-induced colitis.
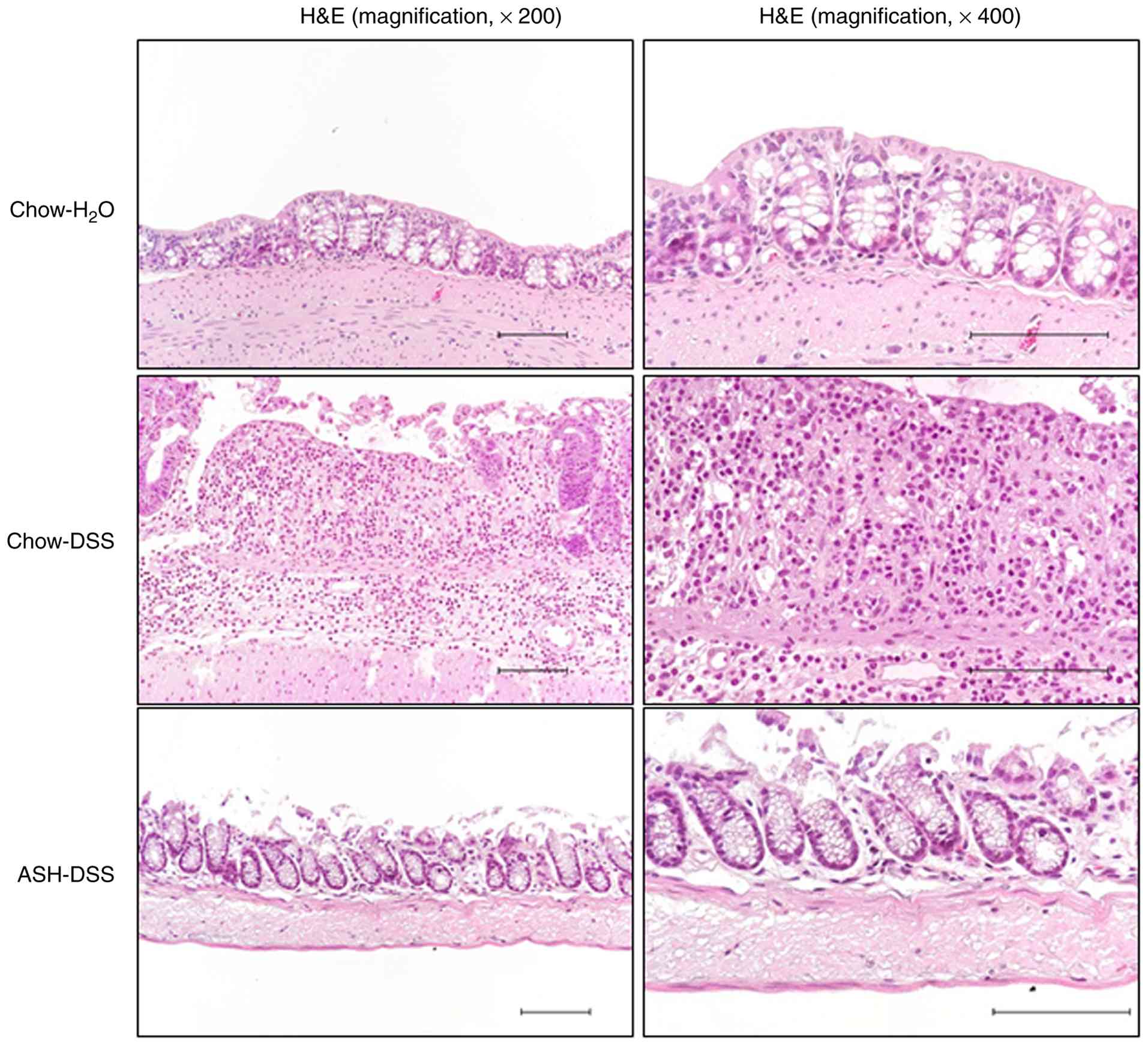
Histological improvement in colitis in
mice fed the ASH diet
Representative hematoxylin and eosin
(H&E)-stained images of mice colons are shown in Fig. 4. The colon tissues of DSS-treated
mice fed the chow diet (Chow-DSS) showed strong inflammatory cell
infiltration, crypt damage, and a thickened edematous submucosal
layer. However, these findings were less observed in the colon
tissues of mice fed the ASH diet with DSS treatment (ASH-DSS).
According to these morphological findings, the histological score
in mice fed the ASH diet with DSS treatment tended to be lower than
that in mice fed the chow diet [Chow-DSS: 7.57±1.72, ASH-DSS diet:
3.57±2.76; Table I]. These results
indicated that the ASH diet had a preventive effect against
DSS-induced colitis in mouse models.
 | Table IHistological assessment of
DSS-induced colitis. |
Table I
Histological assessment of
DSS-induced colitis.
| Parameter |
Chow-H2O | Chow-DSS | ASH-DSS |
|---|
| Score |
0.00±0.00a | 7.57±1.72 | 3.57±2.76 |
| Range |
(0,0,0,0,0,0,0) |
(5,6,7,8,8,9,10) |
(0,0,3,4,5,6,7) |
Decreased inflammatory cytokine
production and oxidative stress in mice fed the ASH diet
To elucidate the mechanism of the effect of ASH on
improvement in colitis, the concentrations of 23 inflammatory
cytokines were measured in mice sera. Among them, five cytokines,
IL-2, IL-9, Eotaxin, GM-CSF, and MCP-1α, were undetectable in all
three mice groups (Table II). All
measurable cytokines, except IL-4, and RANTES, showed decreased
concentrations in mice fed the ASH diet (ASH-DSS) compared with
that in DSS-treated mice fed the chow diet (Chow-DSS). Among these
cytokines, IL-1α, IL-6, IL-12p40, IL-12p70, G-CSF, MCP-1, and TNF-α
concentrations was significantly decreased in mice fed the ASH diet
(ASH-DSS) (Fig. 5A-G). Thereafter,
we measured the degree of oxidation and antioxidant defense
capacity. Decreased diacron-reactive oxygen metabolites (dROMs) and
increased biological antioxidant potential (BAP) were observed in
the sera of mice fed the ASH diet (ASS-DSS) (dROMs; Chow-DSS:
137.4±25.16 U.CARR, ASH-DSS: 95.43±6.97 U.CARR; P<0.05, BAP;
Chow-DSS: 3,572±333 µmol/l, ASH-DSS: 4,175±550 µmol/l; P<0.01;
Fig. 5H and I). These results suggest that reduced
inflammatory cytokines and oxidative stress resulting from an ASH
diet contributes to the mitigation of DSS-induced colitis.
 | Figure 5ASH diet decreases inflammatory
cytokine production and oxidative stress. Measurement of cytokines
in mouse sera: (A) IL-1α, (B) IL-6, (C) IL-12 p40, (D) IL-12 p70,
(E) G-CSF, (F) MCP-1 and (G) TNF-α. The degree of oxidation and
antioxidant defense capacity: (H) dROMs and (I) BAP (n=7). All
units (y-axis), except for dROMs and BAP, are pg/ml.
*P<0.05 vs. chow-DSS. **P<0.01 vs.
chow-DSS. DSS, dextran sulfate sodium; ASH, Acanthopanax
senticosus Harms; G-CSF, granulocyte colony-stimulating factor;
MCP-1, monocyte chemoattractant protein-1; dROMs, diacron-reactive
oxygen metabolites; BAP, biological antioxidant potential. |
 | Table IIMeasurement of cytokines in mouse
sera. |
Table II
Measurement of cytokines in mouse
sera.
| Cytokine | Chow-H2O
(pg/ml) | Chow-DSS
(pg/ml) | ASH-DSS
(pg/ml) |
|---|
| IL-1α |
0.50±1.73a | 7.81±2.34 |
4.58±0.46a |
| IL-1β | n.d. | 3.16±1.16 | 2.53±0.37 |
| IL-2 | n.d. | n.d. | n.d. |
| IL-3 | 3.17±1.61 | 1.95±0.89 | 1.40±0.22 |
| IL-4 | 0.82±1.00 | 2.03±1.34 | 2.04±0.75 |
| IL-5 | n.d. | 6.00±4.43 | 3.63±0.57 |
| IL-6 |
5.90±1.15a | 54.58±20.66 |
20.20±5.88a |
| IL-9 | n.d. | n.d. | n.d. |
| IL-10 |
0.46±1.00a | 20.81±4.68 | 18.85±1.27 |
| IL-12p40 |
972.45±2.93a |
3,400.23±1,916.89 |
1,545.61±244.91b |
| IL-12p70 |
23.11±1.04a | 39.49±15.59 |
26.70±2.31b |
| IL-13 |
0.50±15.62a | 45.48±25.10 | 33.60±11.05 |
| IL-17A |
15.62±2.08a | 36.30±12.70 | 25.01±6.59 |
| Eotaxin | n.d. | n.d. | n.d. |
| G-CSF |
84.22±1.15a |
3,814.11±1,052.78 |
2,411.41±243.65a |
| GM-CSF | n.d. | n.d. | n.d. |
| IFN-γ |
0.84±2.02a | 5.95±4.28 | 4.21±0.90 |
| KC |
32.72±2.52a | 339.14±235.79 | 186.66±49.26 |
| MCP-1 |
85.13±1.00a | 351.22±156.13 |
171.48±24.44a |
| MCP-1α | n.d. | n.d. | n.d. |
| MCP-1β |
36.05±1.61a | 60.97±21.78 | 44.32±8.60 |
| RANTES | n.d. | 65.97±10.65 | 78.51±11.98 |
| TNF-α | 29.20±1.73 | 43.42±18.61 |
28.03±4.93b |
ASH restored antioxidant capacity by
suppressing NOX2 expression in mice colon
We examined the expression of NOX2, which produced
oxidants and contributed to the development of experimental models
of IBD, in mouse colon (Fig. 6A-C).
The results of immunostaining and RT-qPCR showed that NOX2
expression at the protein and mRNA levels were lower in mice fed
the ASH diet (ASH-DSS) than in those fed the chow diet (Chow-DSS).
In mice fed the ASH diet (ASH-DSS), the reduction in Nrf2
expression was mitigated (Fig. 6D),
while the expression of Cu/Zn-SOD, an intracellular superoxide
dismutase, was restored (Fig. 6E).
The RT-qPCR results of NOX2, Nrf2, and Cu/Zn-SOD genes were
verified using two housekeeping genes, GAPDH and 18S ribosome
(Fig. S1). These results suggest
that the restoration of antioxidant capacity by ASH intervention
suppressed inflammation and inflammatory cell infiltration in the
intestinal mucosa.
Discussion
The present study examined the effects of ASH on
DSS-induced colitis in mouse models. Mice fed the ASH diet showed a
tendency toward reduced severity of colitis compared with mice fed
the chow diet. In addition, inflammatory cytokine production and
oxidative stress tended to be lower in ASH-fed mice. Since ASH was
administered prior to DSS exposure, our results support only its
prophylactic effects and do not demonstrate its efficacy against
colitis that has already developed. If ASH were administered after
disease onset, its effects might be weaker than those observed with
prophylactic administration, as epithelial colon damage and the
inflammation would already begin. Nevertheless, the reductions in
oxidative stress and inflammatory cytokines observed in this study
suggest that a partial therapeutic effect is biologically
plausible, and it is worthwhile to conduct direct evaluations in
future post-onset treatment designs.
NOX2, also known as GP91phox, is a member of the NOX
superfamily that catalyzes the formation of oxidants; it is mainly
expressed in leukocytes such as neutrophils and macrophages
(21). When activated, NOX2 forms a
complex with p22phox and p67phox, followed by the production of a
superoxide, which includes reactive oxygen species (ROS). NOX2 also
contributes to the development of experimental animal models of IBD
(22,23). Furthermore, Nrf2-deficient mice was
reported to increase susceptibility to DSS-induced colitis
(24). Our results suggest that the
ASH diet inhibits oxidative stress by partially affecting Nrf2-NOX2
axis. The other findings that Cu/Zn-SOD, the antioxidant enzyme,
was restored in ASH-fed mice during DSS treatment, followed by the
mitigation of oxidative stress by decreased degree of oxidation
(dROMs) and increased antioxidant defense capacity (BAP). These
findings suggest an association between ASH pretreatment and
modulation of oxidative stress-related pathways. However, direct
activation of Nrf2 signaling at the protein level was not
demonstrated in this study.
DSS binds to medium-chain fatty acids in the colon
and induces inflammation, leading to colitis in mice and rats
(25). These experimental animal
models of IBD show increased production of numerous inflammatory
cytokines, followed by exacerbation in ulcerative colitis. Previous
studies on DSS-induced colitis suggest that targeting inflammatory
cytokines such as IL-6(26), IL-1α
(27), IL-12p40/p70(28), and TNF-α (29) is promising treatment with IBD.
Furthermore, MCP-1 and G-CSF are potential biomarkers for patients
with IBD (30). In the present
study, ASH treatment significantly decreased the concentration of
these cytokines in the serum, suggesting that the anti-inflammatory
effect of ASH plays an important role in a DSS-induced colitis
model, resulting in the prevention of colitis exacerbation.
ASH is a widely used component of Chinese
traditional medicine, and it is prescribed for the treatment of
several clinical diseases such as heart disease, hypertension, and
allergies, which are related to inflammation and oxidative stress
(31). Recent studies have
elucidated the mechanisms underlying the clinical effects of ASH.
Takahashi et al (16)
investigated the effects of ASH on collagen-induced arthritis in
mice and demonstrated that ASH delayed the onset of arthritis and
reduced its severity. Moreover, ASH exhibited antioxidant
properties, suppressing the production of ROS and inflammatory
cytokines such as TNF-α and IL-6. Other studies have shown that the
anti-inflammatory effects of ASH can be attributed to its
inhibition of the expression of inducible nitric oxide synthase and
cyclooxygenase-2 in activated macrophages (32). Regarding antioxidative stress, a
study on the effect of ASH on patients with cancer experiencing
cancer-related fatigue demonstrated an increased ratio of BAP to
dROMs (33). Both the
anti-inflammatory and antioxidant effects of ASH were exerted in
DSS-induced colitis models, showing improved clinical
manifestations. Several bioactive constituents of ASH have been
reported to exert colon-protective effects in DSS-induced colitis
models. Isofraxidin attenuated DSS-induced colitis by reducing
reactive oxidative species and promoting Nrf2 activation (34). Cotreatment with Syringin and DSS
also mitigated colitis by suppressing IL-1β, IL-6, TNF-α, iNOS, and
COX-2 expression, accompanied by inhibition of NF-κB and activation
of Nrf2 signaling (35).
Pretreatment with Chlorogenic acid before DSS exposure reduced
disease activity, colon shortening, and TNF-α levels, and dysbiosis
(36).
The limitations of this study were as follows.
First, the present model represents acute DSS-induced experimental
colitis rather than chronic IBD. For future studies, chronic models
such as repeated-cycle DSS colitis, chronic TNBS colitis,
oxazolone-based chronic colitis, and genetically engineered models
including IL-10-deficient mice may provide complementary
information regarding persistent inflammation, fibrosis, and
long-term disease mechanisms (37).
Second, the main bioactive compound in ASHE is not determined.
There are several reports for the effect of ASHE compounds,
isofraxidin (34), syringin
(35), and chlorogenic acid
(36), on the experimental colitis
model. We are not sure whether these compounds could
synergistically ameliorate colitis, but these reports support that
ASHE has a prophylactic role as a dietary supplement. Another
limitation is that we did not perform gain-of-function or
loss-of-function experiments targeting NOX2. Therefore, although
ASH pretreatment was associated with reduced NOX2 expression and
improved colitis severity, a direct causal role of NOX2 suppression
in mediating the protective effect of ASH cannot be concluded from
the present data alone. Finally, female mice were used in the
present proof-of-concept study to minimize biological variability
associated with a mixed-sex design. This choice was also informed
by previous reports showing that DSS-induced colitis severity
differs by sex and is influenced by estrogen-related signaling
(38,39). However, because only female mice
were studied, the present findings should not be generalized across
sexes, and future studies in male mice will be required.
In conclusion, ASH prophylaxis prevented the
exacerbation of colitis in experimental mice models. Decreased
inflammatory cytokine levels and oxidative stress caused by the
inhibition of NOX2 expression would lead to improvements in the
clinical and pathological features of IBD. As ASH is not only safe
but also widely available for purchase as a commercial food
product, regular intake may be considered for IBD prophylaxis.
Supplementary Material
mRNA expression levels of (A) NOX2,
(B) Nrf2 and (C) Cu/Zn-SOD in mouse colons. The 18S ribosome gene
was used as an internal control. *P<0.05 vs.
chow-DSS. **P<0.01 vs. chow-DSS. DSS, dextran sulfate
sodium; ASH, Acanthopanax senticosus Harms; NOX2, NADPH
oxidase 2; Nrf2, nuclear factor erythroid 2-related factor 2;
Cu/Zn-SOD, Cu/Zn-superoxide dismutase.
Acknowledgements
The authors would like to thank Dr Tamaki Tamaki
(Department of Clinical Laboratory Science, School of Medical
Technology, Health Sciences University of Hokkaido, Sapporo,
Hokkaido 002-8072, Japan) for technical assistance.
Funding
Funding: The present study was funded by Sun Chlorella Co., Ltd.
(grant no. 201701).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YKa, DT, HF, STI and MT performed the experiments.
TM and HTakek provided and prepared the Acanthopanax
senticosus Harms extracts. YS, YT, KM, YKu, MS and HTaked
participated in the design of the study. YKa and MT wrote the main
manuscript, and YKa prepared Figs.
1, 2, 3, 4,
5 and 6 and Tables
I and II. All authors reviewed
the manuscript. MT and HF confirm the authenticity of all the raw
data. All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The study protocol was approved by the Animal
Experiments Committee of Health Sciences University of Hokkaido
(approval no. 21-016; Sapporo, Japan). The present study was
performed in compliance with the guidelines of the Committee of
Animal Care and Use of Health Sciences University of Hokkaido and
Animal Research: Reporting of In Vivo Experiments
guidelines.
Patient consent for publication
Not applicable.
Competing interests
YKa received research grants from Sun Chlorella Co.,
Ltd. MT received research grants from Sun Chlorella Co., Ltd. TM
and HTakek are employees of Sun Chlorella Co., Ltd. This company
had no role in interpreting, drafting or deciding whether to
publish the manuscript, although the company Sun Chlorella Co.,
Ltd. provided funding. All other authors declare that they have no
competing interests.
References
|
1
|
Kuhnen A: Genetic and environmental
considerations for inflammatory bowel disease. Surg Clin North Am.
99:1197–1207. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Vedamurthy A and Ananthakrishnan AN:
Influence of environmental factors in the development and outcomes
of inflammatory bowel disease. Gastroenterol Hepatol (N Y).
15:72–82. 2019.PubMed/NCBI
|
|
3
|
Scalavino V, Piccinno E, Giannelli G and
Serino G: Inflammasomes in intestinal disease: Mechanisms of
activation and therapeutic strategies. Int J Mol Sci.
25(13058)2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Centanni L, Bencardino S, D'Amico F, Zilli
A, Parigi TL, Allocca M, Danese S and Furfaro F: Targeting mucosal
healing in Crohn's disease: Efficacy of novel pathways and
therapeutic targets. Expert Opin Ther Targets. 28:963–978.
2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kohgo Y, Ashida T, Maemoto A and Ayabe T:
Leukocytapheresis for treatment of IBD. J Gastroenterol. 38 (Suppl
15):S51–S54. 2003.PubMed/NCBI
|
|
6
|
Deleu S, Becherucci G, Godny L, Mentella
MC, Petito V and Scaldaferri F: The key nutrients in the
mediterranean diet and their effects in inflammatory bowel disease:
A narrative review. Nutrients. 16(4201)2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Miao Z, Gu M, Raza F, Zafar H, Huang J,
Yang Y, Sulaiman M, Yan J and Xu Y: Isoliquiritin ameliorates
ulcerative colitis in rats through caspase 3/HMGB1/TLR4 dependent
signaling pathway. Curr Gene Ther. 24:73–92. 2024.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bseiso Y, Gammoh O, Alqudah M, Altaber S,
Qnais E, Wedyan M, Alqudah A and Alotaibi BS: Evaluating the
anti-inflammatory efficacy of a novel bipyrazole derivative in
alleviating symptoms of experimental colitis. Curr Mol Pharmacol.
17(e18761429333261)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
da Silva LM: Perspectives on the role of
P21-activated kinase 1 (PAK1) in the intestinal anti-inflammatory
and antitumor potential of artepillin C. Curr Mol Pharmacol.
17(e260423216212)2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Davydov M and Krikorian AD:
Eleutherococcus senticosus (Rupr. & Maxim.) Maxim. (Araliaceae)
as an adaptogen: A closer look. J Ethnopharmacol. 72:345–393.
2000.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liu KY, Wu YC, Liu IM, Yu WC and Cheng JT:
Release of acetylcholine by syringin, an active principle of
eleutherococcus senticosus, to raise insulin secretion in Wistar
rats. Neurosci Lett. 434:195–199. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yi JM, Hong SH, Kim JH, Kim HK, Song HJ
and Kim HM: Effect of Acanthopanax senticosus stem on mast
cell-dependent anaphylaxis. J Ethnopharmacol. 79:347–352.
2002.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Fujikawa T, Yamaguchi A, Morita I, Takeda
H and Nishibe S: Protective effects of Acanthopanax
senticosus Harms from Hokkaido and its components on gastric
ulcer in restrained cold water stressed rats. Biol Pharm Bull.
19:1227–1230. 1996.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Liu SM, Li XZ, Zhang SN, Yang ZM, Wang KX,
Lu F, Wang CZ and Yuan CS: Acanthopanax senticosus protects
structure and function of mesencephalic mitochondria in A mouse
model of Parkinson's disease. Chin J Integr Med. 24:835–843.
2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kawano Y, Tanaka M, Fujishima M, Okumura
E, Takekoshi H, Takada K, Uehara O, Abiko Y and Takeda H:
Acanthopanax senticosus Harms extract causes G0/G1 cell
cycle arrest and autophagy via inhibition of Rubicon in human liver
cancer cells. Oncol Rep. 45:1193–1201. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Takahashi Y, Tanaka M, Murai R,
Kuribayashi K, Kobayashi D, Yanagihara N and Watanabe N:
Prophylactic and therapeutic effects of Acanthopanax
senticosus Harms extract on murine collagen-induced arthritis.
Phytother Res. 28:1513–1519. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
17
|
Cooper HS, Murthy SN, Shah RS and
Sedergran DJ: Clinicopathologic study of dextran sulfate sodium
experimental murine colitis. Lab Invest. 69:238–249.
1993.PubMed/NCBI
|
|
18
|
Williams KL, Fuller CR, Dieleman LA,
DaCosta CM, Haldeman KM, Sartor RB and Lund PK: Enhanced survival
and mucosal repair after dextran sodium sulfate-induced colitis in
transgenic mice that overexpress growth hormone. Gastroenterology.
120:925–937. 2001.PubMed/NCBI View Article : Google Scholar
|
|
19
|
García-Díez E, López-Oliva ME,
Caro-Vadillo A, Pérez-Vizcaíno F, Pérez-Jiménez J, Ramos S and
Martín MÁ: Supplementation with a cocoa-carob blend, alone or in
combination with metformin, attenuates diabetic cardiomyopathy,
cardiac oxidative stress and inflammation in zucker diabetic rats.
Antioxidants (Basel). 11(432)2022.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Nauseef WM: The phagocyte NOX2 NADPH
oxidase in microbial killing and cell signaling. Curr Opin Immunol.
60:130–140. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Regmi SC, Park SY, Ku SK and Kim JA:
Serotonin regulates innate immune responses of colon epithelial
cells through Nox2-derived reactive oxygen species. Free Radic Biol
Med. 69:377–389. 2014.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Hoffmann MH and Griffiths HR: The dual
role of reactive oxygen Species in autoimmune and inflammatory
diseases: Evidence from preclinical models. Free Radic Biol Med.
125:62–71. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Khor TO, Huang MT, Kwon KH, Chan JY, Reddy
BS and Kong AN: Nrf2-deficient mice have an increased
susceptibility to dextran sulfate sodium-induced colitis. Cancer
Res. 66:11580–11584. 2006.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Bamba S, Andoh A, Ban H, Imaeda H, Aomatsu
T, Kobori A, Mochizuki Y, Shioya M, Nishimura T, Inatomi O, et al:
The severity of dextran sodium sulfate-induced colitis can differ
between dextran sodium sulfate preparations of the same molecular
weight range. Dig Dis Sci. 57:327–334. 2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Xiao YT, Yan WH, Cao Y, Yan JK and Cai W:
Neutralization of IL-6 and TNF-α ameliorates intestinal
permeability in DSS-induced colitis. Cytokine. 83:189–192.
2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Menghini P, Corridoni D, Buttó LF, Osme A,
Shivaswamy S, Lam M, Bamias G, Pizarro TT, Rodriguez-Palacios A,
Dinarello CA and Cominelli F: Neutralization of IL-1α ameliorates
Crohn's disease-like ileitis by functional alterations of the gut
microbiome. Proc Natl Acad Sci USA. 116:26717–26726.
2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kim DJ, Kim KS, Song MY, Seo SH, Kim SJ,
Yang BG, Jang MH and Sung YC: Delivery of IL-12p40 ameliorates
DSS-induced colitis by suppressing IL-17A expression and
inflammation in the intestinal mucosa. Clin Immunol. 144:190–199.
2012.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zeng Z, Lin H, Jiang M, Yuan J, Li X, Jia
Y, Yang L and Zhang H: Anti-TNFα in inflammatory bowel disease:
From originators to biosimilars. Front Pharmacol.
15(1424606)2024.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ott A, Tutdibi E, Goedicke-Fritz S, Schöpe
J, Zemlin M and Nourkami-Tutdibi N: Serum cytokines MCP-1 and GCS-F
as potential biomarkers in pediatric inflammatory bowel disease.
PLoS One. 18(e0288147)2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Huang L, Zhao H, Huang B, Zheng C, Peng W
and Qin L: Acanthopanax senticosus: Review of botany,
chemistry and pharmacology. Pharmazie. 66:83–97. 2011.PubMed/NCBI
|
|
32
|
Jung CH, Jung H, Shin YC, Park JH, Jun CY,
Kim HM, Yim HS, Shin MG, Bae HS, Kim SH and Ko SG: Eleutherococcus
senticosus extract attenuates LPS-induced iNOS expression through
the inhibition of Akt and JNK pathways in murine macrophage. J
Ethnopharmacol. 113:183–187. 2007.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kawano Y, Watanabe N, Nishiyama M, Ohmura
T, Mihara H, Ono K, Tanaka M, Sato Y, Tomonari T, Takeda H and
Takayama T: Feasibility and safety of food containing
Acanthopanax senticosus for treating patients with
cancer-related fatigue. Palliat Med Rep. 5:381–386. 2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
He S, Zhang T, Wang YY, Yuan W, Li L, Li
J, Yang YY, Wu DM and Xu Y: Isofraxidin attenuates dextran sulfate
sodium-induced ulcerative colitis through inhibiting pyroptosis by
upregulating Nrf2 and reducing reactive oxidative species. Int
Immunopharmacol. 128(111570)2024.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang H, Gu H, Jia Q, Zhao Y, Li H, Shen
S, Liu X, Wang G and Shi Q: Syringin protects against colitis by
ameliorating inflammation. Arch Biochem Biophys.
680(108242)2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhang P, Jiao H, Wang C, Lin Y and You S:
Chlorogenic acid ameliorates colitis and alters colonic microbiota
in a mouse model of dextran sulfate sodium-induced colitis. Front
Physiol. 10(325)2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Lee CH, Koh SJ, Radi ZA and Habtezion A:
Animal models of inflammatory bowel disease: Novel experiments for
revealing pathogenesis of colitis, fibrosis, and colitis-associated
colon cancer. Intest Res. 21:295–305. 2023.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Bábíčková J, Tóthová Ľ, Lengyelová E,
Bartoňová A, Hodosy J, Gardlík R and Celec P: Sex differences in
experimentally induced colitis in mice: A role for estrogens.
Inflammation. 38:1996–2006. 2015.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Goodman WA, Havran HL, Quereshy HA, Kuang
S, De Salvo C and Pizarro TT: Estrogen receptor α loss-of-function
protects female mice from DSS-induced experimental colitis. Cell
Mol Gastroenterol Hepatol. 5:630–633.e1. 2017.PubMed/NCBI View Article : Google Scholar
|