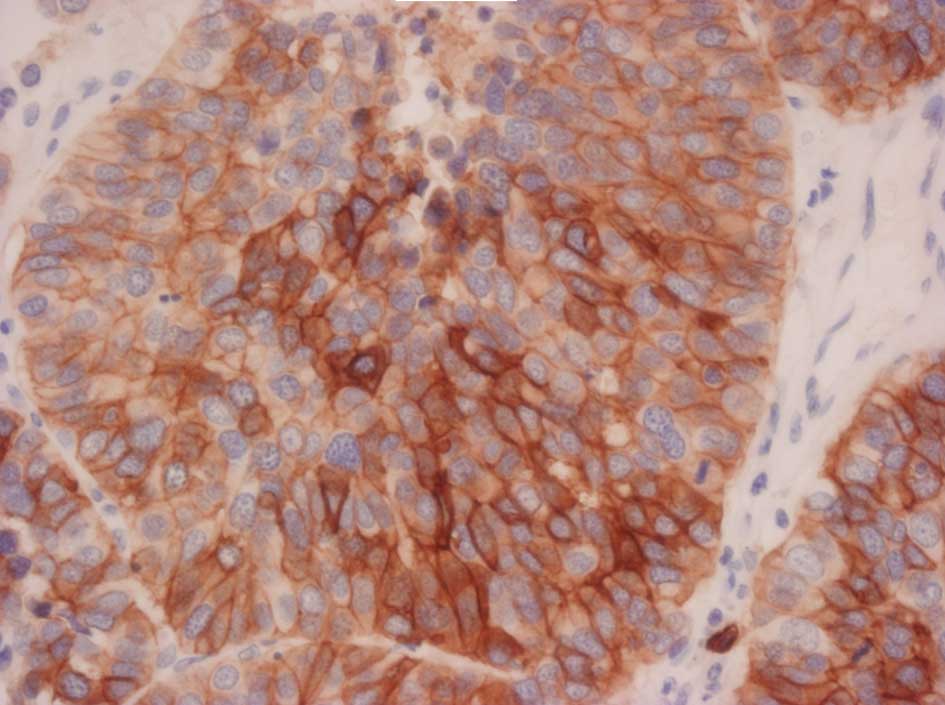
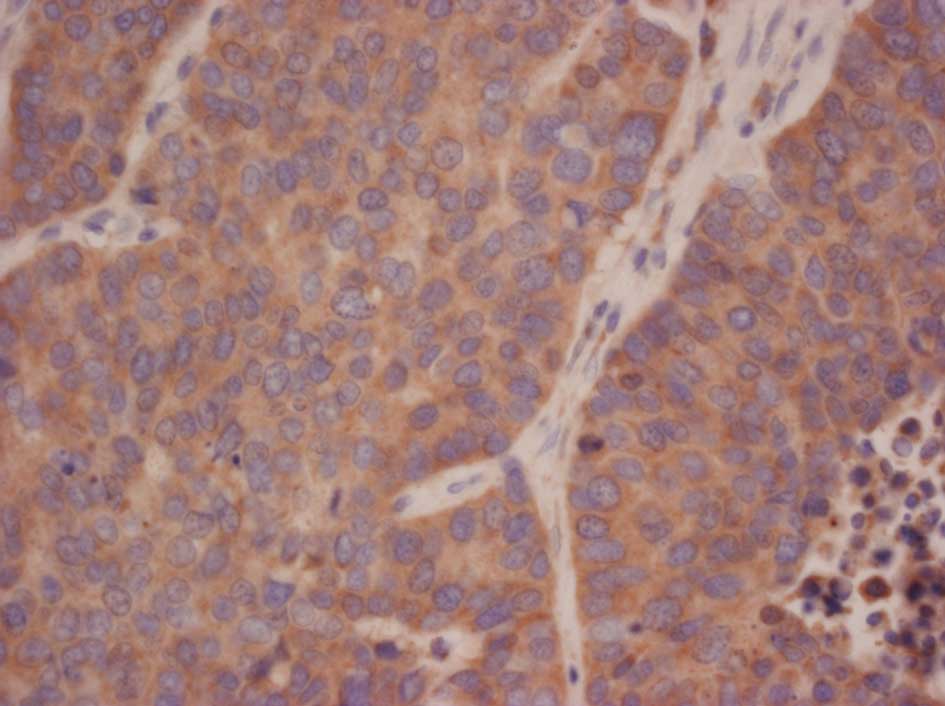
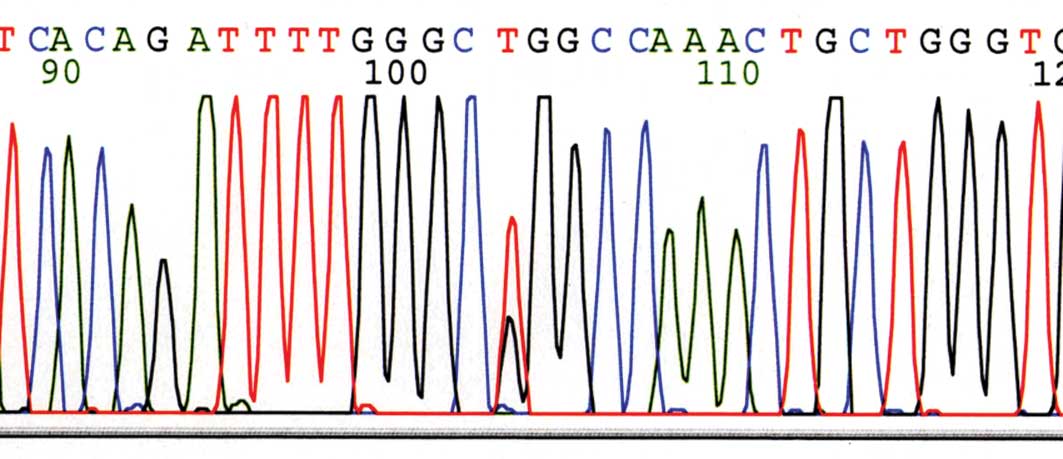
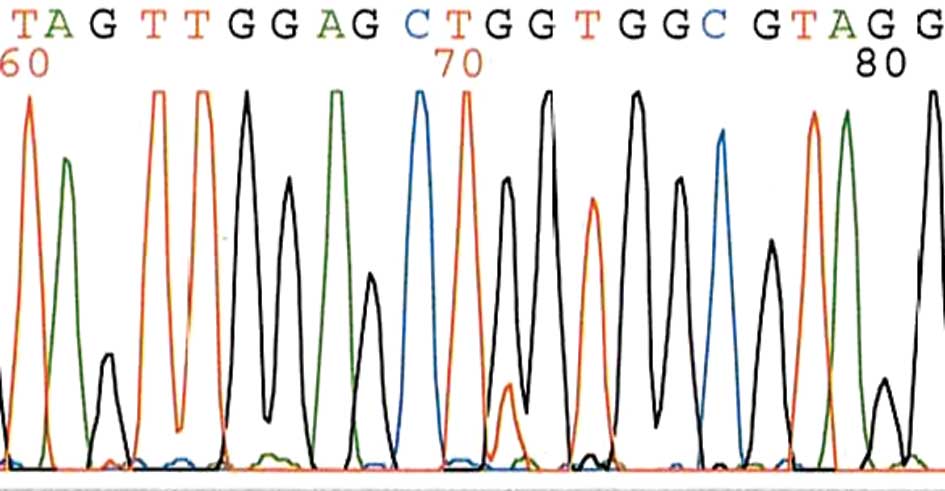
|
1
|
Travis WD, Colby TV, Corrin B, Shimosato Y
and Brambilla E: Histological Typing of Lung and Pleural Tumours.
World Health Organization International Histological Classification
of Tumors, XIII; 3rd edition. Springer-Verlag; Berlin/Heidelberg:
1999, View Article : Google Scholar
|
|
2
|
Iyoda A, Hiroshima K, Nakatani Y and
Fujisawa T: Pulmonary large cell neuroendocrine carcinoma: its
place in the spectrum of pulmonary carcinoma. Ann Thorac Surg.
84:702–707. 2007.PubMed/NCBI
|
|
3
|
Iyoda A, Hiroshima K, Toyozaki T, Haga Y,
Fujisawa T and Ohwada H: Clinical characterization of pulmonary
large cell neuroendocrine carcinoma and large cell carcinoma with
neuroendocrine morphology. Cancer. 91:1992–2000. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Takano T, Fukui T, Ohe Y, Tsuta K,
Yamamoto S, Nokihara H, Yamamoto N, Sekine I, Kunitoh H, Furuta K
and Tamura T: EGFR mutations predict survival benefit from
gefitinib in patients with advanced lung adenocarcinoma: a
historical comparison of patients treated before and after
gefitinib approval in Japan. J Clin Oncol. 26:5589–5595. 2008.
View Article : Google Scholar
|
|
5
|
Zhu CQ, da Cunha Santos G, Ding K,
Sakurada A, Cutz JC, Liu N, Zhang T, Marrano P, Whitehead M, Squire
JA, Kamel-Reid S, Seymour L, Shepherd FA and Tsao MS; National
Cancer Institute of Canada Clinical Trials Group Study BR.21: Role
of KRAS and EGFR as biomarkers of response to erlotinib in National
Cancer Institute of Canada Clinical Trials Group Study BR.21. J
Clin Oncol. 26:4268–4275. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mitsudomi T, Kosaka T, Endoh H, Horio Y,
Hida T, Mori S, Hatooka S, Shinoda M, Takahashi T and Yatabe Y:
Mutations of the epidermal growth factor receptor gene predict
prolonged survival after gefitinib treatment in patients with
non-small-cell lung cancer with postoperative recurrence. J Clin
Oncol. 23:2513–2520. 2005. View Article : Google Scholar
|
|
7
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000. View Article : Google Scholar
|
|
8
|
Hurwitz H, Fehrenbacher L, Novotny W,
Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S,
Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R and Kabbinavar F:
Bevacizumab plus irinotecan, fluorouracil, and leucovorin for
metastatic colorectal cancer. N Engl J Med. 350:2335–2342. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Seto T, Higashiyama M, Funai H, Imamura F,
Uematsu K, Seki N, Eguchi K, Yamanaka T and Ichinose Y: Prognostic
value of expression of vascular endothelial growth factor and its
flt-1 and KDR receptors in stage I non-small-cell lung cancer. Lung
Cancer. 53:91–96. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sandler A, Gray R, Perry MC, Brahmer J,
Schiller JH, Dowlati A, Lilenbaum R and Johnson DH:
Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell
lung cancer. N Engl J Med. 355:2542–2550. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lennon S, Barton C, Banken L, Gianni L,
Marty M, Baselga J and Leyland-Jones B: Utility of serum HER2
extracellular domain assessment in clinical decision making: pooled
analysis of four trials of trastuzumab in metastatic breast cancer.
J Clin Oncol. 27:1685–1693. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hirota S, Isozaki K, Moriyama Y, Hashimoto
K, Nishida T, Ishiguro S, Kawano K, Hanada M, Kurata A, Takeda M,
Muhammad Tunio G, Matsuzawa Y, Kanakura Y, Shinomura Y and Kitamura
Y: Gain-of-function mutations of c-kit in human gastrointestinal
stromal tumors. Science. 279:577–580. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Demetri GD, von Mehren M, Blanke CD, van
den Abbeele AD, Eisenberg B, Roberts PJ, Heinrich MC, Tuveson DA,
Singer S, Janicek M, Fletcher JA, Silverman SG, Silberman SL,
Capdeville R, Kiese B, Peng B, Dimitrijevic S, Druker BJ, Corless
C, Fletcher CD and Joensuu H: Efficacy and safety of imatinib
mesylate in advanced gastrointestinal stromal tumors. N Engl J Med.
347:472–480. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Verweij J, Casali PG, Zalcberg J, LeCesne
A, Reichardt P, Blay JY, Issels R, van Oosterom A, Hogendoorn PC,
van Glabbeke M, Bertulli R and Judson I: Progression-free survival
in gastrointestinal stromal tumours with high-dose imatinib:
randomised trial. Lancet. 364:1127–1134. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsuda H, Akiyama F, Terasaki H, Hasegawa
T, Kurosumi M, Shimadzu M, Yamamori S and Sakamoto G: Detection of
HER-2/neu (c-erb B-2) DNA amplification in primary breast
carcinoma. Interobserver reproducibility and correlation with
immunohistochemical HER-2 overexpression. Cancer. 92:2965–2974.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hirota S, Nishida T, Isozaki K, Taniguchi
M, Nakamura J, Okazaki T and Kitamura Y: Gain-of-function mutation
at the extracellular domain of KIT in gastrointestinal stromal
tumours. J Pathol. 193:505–510. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yamazaki S, Sekine I, Matsuno Y, Takei H,
Yamamoto N, Kunitoh H, Ohe Y, Tamura T, Kodama T, Asamura H,
Tsuchiya R and Saijo N: Clinical responses of large cell
neuroendocrine carcinoma of the lung to cisplatin-based
chemotherapy. Lung Cancer. 49:217–223. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hirsch FR, Herbst RS, Olsen C, Chansky K,
Crowley J, Kelly K, Franklin WA, Bunn PA Jr, Varella-Garcia M and
Gandara DR: Increased EGFR gene copy number detected by fluorescent
in situ hybridization predicts outcome in non-small-cell lung
cancer patients treated with cetuximab and chemotherapy. J Clin
Oncol. 26:3351–3357. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heinrich MC, Owzar K, Corless CL, Hollis
D, Borden EC, Fletcher CD, Ryan CW, von Mehren M, Blanke CD, Rankin
C, Benjamin RS, Bramwell VH, Demetri GD, Bertagnolli MM and
Fletcher JA: Correlation of kinase genotype and clinical outcome in
the North American Intergroup Phase III Trial of imatinib mesylate
for treatment of advanced gastrointestinal stromal tumor: CALGB
150105 Study by Cancer and Leukemia Group B and Southwest Oncology
Group. J Clin Oncol. 26:5360–5367. 2008.
|