Contents
Introduction
Molecular characterization
Genetic alterations
Specific molecular biomarkers
GBM subtypes
Clinical manifestations
Current standard of care
New targeted therapies
Conclusion
Introduction
Glioblastoma multiforme (GBM) is the most frequent
and aggressive primary brain tumor in humans, representing up to
50% of all primary brain gliomas, and the prognosis of patients
with GBM remains poor (1,2). A grading scheme that has been
proposed by the World Health Organization (WHO) distinguishes four
different grades of gliomas. One of these, GBM WHO IV with
predominant astrocytic differentiation, accounts for approximately
12–15% of all brain tumors and 60–75% of astrocytic tumors, and is
the most malignant type (3).
Approximately 51,000 primary brain tumors are diagnosed in the
United States each year, 36% of which are gliomas. Half of these
are GBM, with approximately 3 in 100,000 individuals newly
diagnosed with GBM each year (4).
The treatment difficulty is due to the exceptionally infiltrative
nature of GBM and its proclivity to integrate into normal brain
tissue (5). Fortunately, it is
worth noting that, with the notable recent advances in therapy, an
increasing number of GBM patients are surviving for 36 months or
longer, so that they are referred to as long-term survivors (LTS,
≥36 months) (6). To date, the
management of patients with GBM continues to harbor significant
challenges, and comprehensive genetic screens of tumor tissues and
signaling pathways have been explored to develop molecular-based
targeted therapies (7).
Molecular characterization
Although numerous genetic alterations have been
described in GBM (8,9), such markers have proven to be of
marginal utility in predicting outcome or guiding decisions
regarding disease management. In general, the molecular
characterization of GBM should provide a better understanding of
the genomic landscape of GBMs and more efficacious means for rapid,
high-throughput analyses of tumor cells and tissues.
Despite common clinical presentations and histology,
it has been clearly demonstrated that GBM is a highly anaplastic
and morphologically highly heterogeneous tumor. The presence of
microvascular proliferation and/or necrosis is an essential
criterion for the diagnosis of GBM (10). The diagnosis of GBM has been based
on a complete clinicopathological assessment and this has been an
extremely valuable approach. The pathognomonic features that
characterize GBM at the tissue level are the presence of areas of
necrosis with surrounding pseudopalisades and microvascular
hyperplasia, which are believed to be instrumental to its
accelerated growth (11).
Genetic alterations
Previous studies of the GBM genome and signaling
pathways have provided a more complete view of the landscape of
such alterations and their linked pathways. They indicate that
genetic loss is scattered across the entire genome, affecting
almost all chromosomes at frequencies ranging up to 80% of those of
GBMs. The common genetic alterations include epidermal growth
factor receptor (EGFR) amplification, mutations in TP53, P16, DCC
and RB, and deletions associated with chromosomes 19q and 22q,
chromosome 7 gain and chromosome 10 loss (12,13).
In particular, loss of chromosome 10q is a more frequent occurrence
in GBM than anaplastic astrocytoma and has been associated with
short GBM survival (14). Albarosa
et al analyzed 53 GBMs and found that the loss of
heterozygosity occurred in >90% of the tumors (15).
To identify the genetic alterations in GBMs, Parsons
et al sequenced 20,661 protein coding genes, determined the
presence of amplifications and deletions, and performed gene
expression analyses in 22 human tumor samples. They found recurrent
mutations in the active site of isocitrate dehydrogenase 1 (IDH1,
an enzyme that catalyzes the oxidative carboxylation of isocitrate
to α-ketoglutarate) (16). The
patients with mutated IDH1 have distinct clinical characteristics,
including a considerably improved clinical prognosis. IDH1 is
localized within the cytoplasm and peroxisomes and its activity
leads to nicotinamide adenine dinucleotide phosphate production,
and is thought to play a role in the cellular control of oxidative
damage (17). Mutations in IDH1
occur predominantly in a large proportion of young patients, which
is a hallmark of early cancerigenesis, and are typically associated
with low-grade tumors, in accordance with the research results of
Bleeker et al (18). The
patients who have IDH1 mutations have a high frequency (>70%) of
TP53 mutations and a very low frequency of mutations in other
commonly altered GBM genes (19).
These studies support the evidence that IDH1 is a pivotal GBM
cancer gene that has mutated and identify a potentially useful
genetic alteration for the classification and targeted therapy of
GBMs (17,20). It is conceivable that new
treatments may be designed to take advantage of IDH1 alterations in
these patients; for example, inhibition of a different IDH enzyme
(IDH2) may increase sensitivity of tumor tissues to a variety of
chemotherapeutic agents (21).
Pediatric GBMs have a pattern of chromosomal and
genetic modifications that are distinct from those in adults. The
overexpression of the EGFR protein is observed in 23–40% of
patients, and p53 gene mutations are very frequent, occurring in
approximately 33–63% of patients (22–24).
The genes overexpressed in GBM usually produce extracellular
proteins, thereby providing possible therapeutic targets. In
addition, loss of p16 and p27 expression is observed in 68 and 54%
of cases, respectively, which is similar to that observed in adult
GBMs (23). A similar study by
Rood and MacDonald detected overexpression of EGFR, mutations of
PTEN, deletions or epigenetic inactivation of p16, and
amplification of MDM2 in pediatric GBMs (25).
Specific molecular biomarkers
With our current understanding of the expression of
specific molecular biomarkers, the use of methylguanyl
methyltransferase (MGMT) promoter methylation status for routine
diagnostic or therapeutic purposes is considered to be a promising
molecular factor that is predictive of the response of GBM patients
to treatment (26). It is
associated with prolonged progression-free and longer overall
survival in patients with GBM who receive alkylating chemotherapy
with carmustine, lomustine or temozolomide (TMZ). The MGMT gene is
located on chromosome 10q26 and encodes a DNA-repair protein that
removes alkyl groups from the O6 position of guanine, a significant
site of DNA alkylation (27). For
chemotherapy in patients with GBM, as a standard alkylating agent,
TMZ-induced injury is repaired by the DNA repair enzymes, including
MGMT, which is a unique DNA repair enzyme in the context of
alkylating chemotherapy that removes the DNA methylation that is
produced by TMZ. It is believed that alkylating agents cause cell
death by forming cross-links between adjacent strands of DNA, owing
to alkylation at the position of O6-guanine in DNA. Epigenetic
silencing of this gene by promoter methylation is associated with
loss of its expression and diminished DNA-repair activity. It
becomes permanently inactivated and depleted in the process.
Hypermethylation of the MGMT promoter decreases the expression of
the protein and, as a result, DNA damage from alkylating agents is
not repaired, leading to tumor cell death (27). The epigenetic silencing of the MGMT
gene through promoter methylation is correlated with a median
survival in patients who receive radiotherapy (RT) with TMZ for the
treatment of GBM. It is now commonly recognized that silencing of
the MGMT gene promoter by methylation is associated with improved
GBM response to combination treatment with radiation and TMZ
(28).
GBM subtypes
A number of studies have investigated molecular
subclasses in GBM. Significantly, expression profiling studies have
revealed that molecular classification of gliomas may be of
prognostic value (29–31). From a histopathological point of
view, the majority of GBMs (accounting for approximately 90% of
tumors) are diagnosed as de novo or primary tumors, are more
common in males and manifest a very rapid development of clinical
symptoms. Secondary GBM (occurring in approximately 10% of tumors)
progresses from lower-grade tumors (WHO grade II/III) with a mean
progression time of approximately 55 months (32). Secondary GBM is observed in younger
patients, is more evenly distributed between the genders, and
exhibits longer survival times (33,34).
Secondary GBM may be diagnosed by clinical (neuroimaging) or
histological evidence of evolution from a less malignant
astrocytoma (35). Noushmehr et
al have identified and characterized a distinct molecular
subgroup in GBM tumors (13).
Based on the context of The Cancer Genome Atlas (TCGA), they found
that a distinct subtype of samples display concerted
hypermethylation at a large number of loci, indicating the
existence of a glioma-CpG island methylator phenotype (G-CIMP).
G-CIMP-positive samples are associated with secondary or recurrent
tumors, and are tightly associated with IDH1 somatic mutation.
G-CIMP tumors also showed a relative lack of copy-number variation
commonly observed in GBM. These findings identify G-CIMP as a
distinct marker of human GBM using molecular and clinical features.
Van Meir et al suggested a new subtype in the classification
of GBM. They uncovered new genetic alterations and provided
preliminary evidence that GBM may be subdivided into four subtypes
(36). Verhaak et al
proposed a robust gene expression-based molecular classification of
GBM into proneural, neural, classical and mesenchymal subtypes, and
integrated multidimensional genomic data to establish patterns of
somatic mutations and DNA copy number (37).
Clinical manifestations
Clinical manifestations of GBM depend on the age of
the patient, location, size and rate of growth of the tumor.
Clinical manifestations include progressive headaches, dizziness,
seizures, increased intracranial pressure, focal neurological
deficits or changes in mental status (38). Aside from symptoms and signs, the
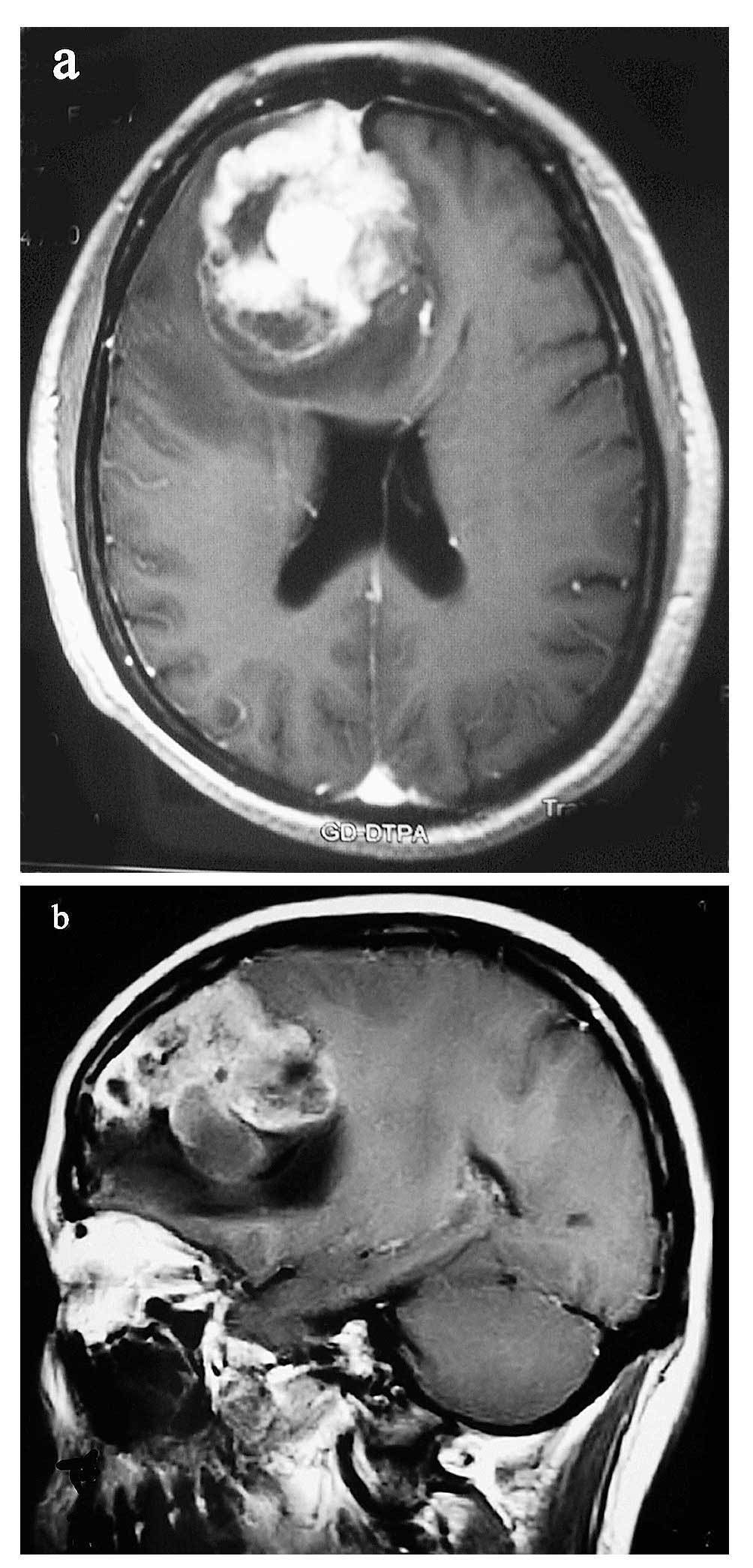
best imaging diagnostic method is a T1-weighted gadolinium-enhanced
MRI, particularly the three-planar images (Fig. 1A and B) and diffusion tensor
imaging. Radiologically, GBMs present with irregular contours and a
peripheral zone with strong contrast enhancement around a darker,
hypodense, necrotic area and with the non-enhancing tumor extending
outside the area of enhancement (39). Magnetic resonance spectroscopy
identifies tumor masses with a marked increase in the choline to
creatine ratio, reduction of N-acetylaspartate and the presence of
a lactate-lipid peak greatly assists neurological oncologists in
diagnosis and surgery. PET and SPECT imaging may be useful tools in
screening occult systemic disease. If MRI is contraindicated, a
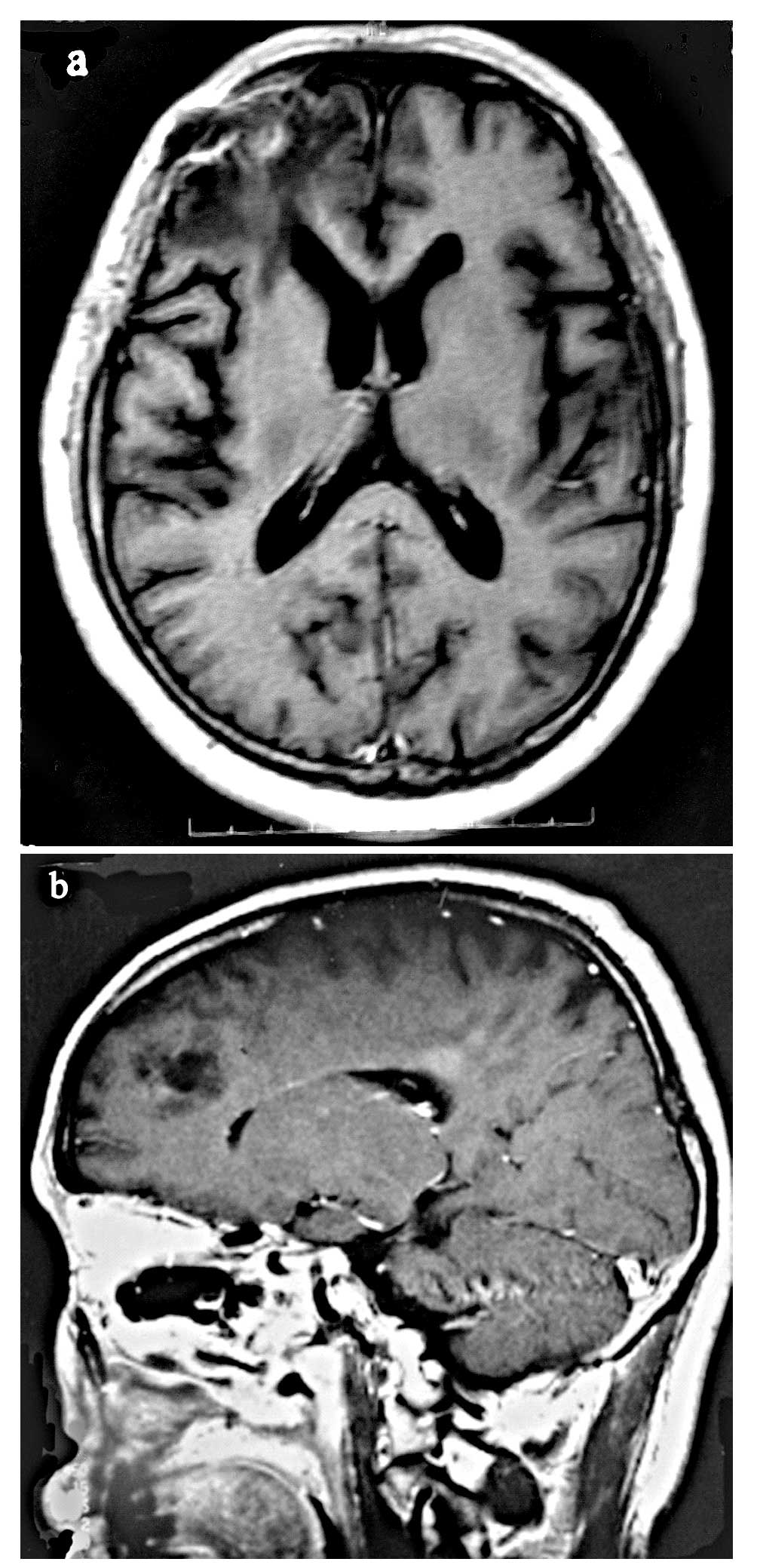
contrast-enhanced CT scan should be used. Relevant responses to
treatment may be assessed by neurological examination and by MRI
(Fig. 2A and B) or
contrast-enhanced CT scan following therapy, or during
follow-up.
With regards to gender, there is a higher incidence
of GBM in males, adjusted to the World Standard Population, of 3.32
in males and 2.24 in females (1,40).
GBM occurs in individuals of any age, although the median age is
>60 years, according to population-based studies. By contrast,
the median age of LTS is 51 years. This is in accordance with
numerous clinical studies that indicate that young age at the time
of diagnosis is a significant parameter associated with LTS. At the
same time, the median age in data that were collected from all 281
published GBM LTS cases during the period from 1950 to 2006 is 36.9
years. These findings underline the association of GBM LTS with
prognostically favourable clinical factors, in particular young age
and good initial performance score, as well as MGMT promoter
hypermethylation (41). The
investigators also found that primary GBMs commonly develop in
older individuals (mean, 55 years), whereas secondary GBMs are
found in middle-aged individuals (approximately 39 years of age)
(42). In contrast to adult
populations, GBM is rarely observed in children compared with
adults, comprising only 5–10% of childhood intracranial neoplasms
(24).
Current standard of care
The current standard of care to improve the survival
of patients who have GBM begins with surgical removal as the
initial treatment of choice, followed by radiation and then
chemotherapy. Besides establishing a definitive histopathological
diagnosis, gross or near-total resection, if feasible, may lead to
rapid improvement of clinical symptoms and a reduction of steroid
doses or dehydrating agents. In turn, this will dictate subsequent
therapy options and significantly improve survival (43). Following surgery, a combination of
RT and TMZ followed by adjuvant therapy continues to be the most
effective therapy available for patients with GBM. Mineo et
al showed that 80% of patients received RT prior to
chemotherapy. The median survival rate was longer when RT and
chemotherapy were combined versus chemotherapy alone (16 months
versus 11 months) (44). Glas
et al reported a survival rate of 15.4% (6/39) after 5
years. The rate of patients who are surviving long term and who are
potentially cured is increased in this study, compared with the
rate of 4 to 5% that is historically reported for GBM (45, 46).
In the National Cancer Institute population
databases, the five-year survival rates are approximately 13% for
15–45 year olds, but only 1% for those aged 75 and older (39). Younger patients survive
significantly longer than patients in all other age groups. The
median survival ranges from 8.8 months (<50 years) to 1.6 months
(>80 years). Liu et al compared differences in genetic
variation between short-term survivors (STS, <12 months) and
LTS, and explored the classification for survival data. They
revealed that patients >50 years old with LIG4 rs7325927 had the
worst survival (median survival time of 1.2 years) and exhibited
the highest risk of mortality compared to younger patients with
combined RTEL1 rs2297440 and HMGA2 rs1563834 genotypes (median
survival time of 7.8 years) (47).
Nevertheless, favorable prognostic factors were closely related to
young age, good Karnofsky performance status (KPS) score, tumors
located in the white matter of the frontal, temporal or occipital
lobe, and no tumors in the deep structures of the brain, such as
the basal ganglia, diencephalic structures or the brain stem,
histology and the extent of tumor resection (48,49).
Interstitial brachytherapy has been used as an
adjuvant treatment for malignant brain tumors as a component of the
standard therapy. In analyzing the results of this treatment,
numerous publications indicate an improvement in median survival
for highly selected patients. While iodine-125 (I-125) remains the
most popular radionuclide for brachytherapy, there is a move away
from temporary high-activity implants to permanent low-activity
implants (50). This is a
technique that is used to treat small (<4 cm), radiographically
well-defined lesions with a single high-dose fraction of ionizing
radiation in stereotactically directed narrow beams. This may
provide added improvement to the survival outcome (51). An advantage is that brachytherapy
is noninvasive, allowing treatment of patients with tumors in
surgically inaccessible or eloquent areas of the brain or in those
with serious coexisting medical illnesses. Stereotactic
brachytherapy (SB) is used in selected patients who have
supratentorial tumors that do not involve the corpus callosum,
brain stem, thalamus, intraventricular or ependymal surfaces, and
are unifocal, well-defined and 1–4 cm in diameter. SB uses
stereotactic techniques to accurately place inflating balloon
catheters that contain radioactive isotopes or interstitial
diffusion-based drug delivery systems within brain tumors, without
a cytotoxic effect on normal brain structures. Typically, SB
delivers an additional 50–60 Gy of radiation, bringing the total
dose of radiation up to 110–120 Gy, and a mean dose of 50 Gy. A
high and heterogeneous drug concentration via chronic low-flow
microinfusion (controlled-release polymer implants, i.e, Gliadel
wafers) may be subtherapeutic or toxic. An intratumoral
microdialysis catheter is used for a small volume to be distributed
within the tumor and the brain surrounding it (52).
New targeted therapies
Although the available standard of therapy for GBM
has evolved into multimodality measures, the majority of patients
still experience tumor progression due to the diffuse infiltration
of malignant tumor cells into the brain tissues following this
treatment. Thus, new ideas and more sensitive methods for treatment
have been proposed to target therapies with the goal of increasing
the specific efficacy for these patients (53). Additionally, radiation has been
promoted in the form of stereotactic radiosurgery for newly
diagnosed GBM or tumor recurrence.
The clinical literature confirms that GBM is a
highly vascularized tumor that relies on angiogenesis, the
formation of new blood vessels. The vascular structure of GBM is
disorganized, tortuous and functionally abnormal, which leads to
hypoxia, acidosis, increased interstitial pressure, blood brain
barrier disruption and cerebral edema or tissue necrosis (28,54).
VEGF-A is the predominant growth factor that is expressed by GBM
cells and signal transduction is mediated through its receptor,
VEGFR-2, which is highly expressed in the glioma-associated
endothelial cells. A number of targeted anti-angiogenesis agents
are currently being explored, which may normalize blood vessels and
enhance delivery of oxygen and cytotoxic agents to prevent tumor
progression and resistance to therapy (55). Bevacizumab (BV), a recombinant
humanized IgG1 monoclonal antibody (MAb) that targets VEGF (a key
regulator of tumor angiogenesis) is the first antiangiogenic
therapy approved for use in cancer, and received FDA approved for
the treatment of rGBM following primary therapy in 2009 (54). BV produces response rates of
approximately 20 to 40% in GBM and increases 6-month
progression-free survival (PFS6) to approximately 30 to 50%
(56), which is superior to the
21% PFS6 rate reported for TMZ (57). In a study using BV monotherapy for
rGBM, Moustakas et al reported that the majority of patients
showed a stable performance on a variety of tests at the six-week
follow-up and 18 to 25% had improved performance (58). These reports demonstrated that BV
therapy leads to rapid reductions in peritumoral edema, often
permitting a decrease in dose or even cessation of corticosteroid
use. The MAb study proved that multivalent proteins are engineered
to have high selectivity and affinity to antigenic epitopes, and
are capable of functioning on the eliminable side of blood vessels
without a need to traverse the blood-brain barrier. This may be
effective in the treatment of brain tumors (59). Luther et al found that MAb
has the potential to target GBMs. MAb-8H9 is specific for membrane
protein B7H3 and is reactive with the majority of human malignant
gliomas. The investigators tested the 8H9scFv-PE38 recombinant
pseudomonas immunotoxin in a preclinical model for malignant
glioma. For rats harboring intracranial U87 xenografts, an infusion
of 8H9scFv-PE38 increased the mean survival (60). Tumors also showed a volumetric
response to an infusion of 8H9scFv-PE38 by MRI. An interstitial
infusion of 8H9scFv-PE38 has shown potential for the treatment of
hemispherical and brain stem glioma. Recently, Iwatade et al
(61) evaluated the effectiveness
of various anticancer drugs for MGMT-positive GBM. They found that
individuals expressing MGMT-positive tumors, platinum agents and
taxanes had a more significant efficacy than other categories of
anticancer agents. The median survival following therapy was 20.1
months for their 74 patients with MGMT-positive GBM.
Conclusion
GBM is the most common and most malignant brain
tumor in adults and carries the poorest prognosis. Recent progress
in molecular biology, neuroimaging and neurosurgical care, has led
to the increasing use of new targeted therapies on a multi-modality
standard treatment basis in the management of GBM. The median
survival time for patients with GBM has improved from an average of
12 months. The molecular-based targeted therapies being tested in
clinical trials represent a new era in GBM therapeutics that bring
hope to those individuals who are afflicted with this refractory
disease, which may have a significant impact on quality of life for
patients with GBM.
Acknowledgements
We thank Dr Junli Huo for preparing
the materials of the patients, and Dr Yijun Chen for his editorial
review (Stanford Comprehensive Cancer Center, Stanford University
School of Medicine, Stanford, CA, USA).
References
|
1.
|
H OhgakiP DessenB JourdeGenetic pathways
to glioblastoma: a population-based studyCancer
Res6468926899200410.1158/0008-5472.CAN-04-133715466178
|
|
2.
|
R StuppWP MasonMJ van den BentRadiotherapy
plus concomitant and adjuvant temozolomide for glioblastomaN Engl J
Med352987996200510.1056/NEJMoa04333015758009
|
|
3.
|
DN LouisH OhgakiOD WiestlerThe 2007 WHO
classification of tumors of the central nervous systemActa
Neuropathol11497109200710.1007/s00401-007-0243-417618441
|
|
4.
|
CBTRUSStatistical report: primary brain
tumors in the United States, 2000–2004Central brain tumor registry
of the United StatesChicago2008
|
|
5.
|
J ClarkeN ButowskiSR ChangRecent advances
in therapy for glioblastomaArch
Neurol67279283201010.1001/archneurol.2010.5
|
|
6.
|
M SchmidingerL LinzmayerA
BechererPsychometric-and quality-of-life assessment in long-term
glioblastoma survivorsJ
Neurooncol635561200310.1023/A:102374030316212814255
|
|
7.
|
The Cancer Genome Atlas (TCGA) Research
NetworkComprehensive genomic characterization defines human
glioblastoma genes and core
pathwaysNature45510611068200810.1038/nature0738518772890
|
|
8.
|
A Von DeimlingDN LouisOD WiestlerMolecular
pathways in the formation of gliomasGlia1532833819958586467
|
|
9.
|
K WatanabeO TachibanaK SataY YonekawaP
KleihuesH OhgakiOverexpression of the EGF receptor and p53
mutations are mutually exclusive in the evolution of primary and
secondary glioblastomasBrain
Pathol6217223199610.1111/j.1750-3639.1996.tb00848.x8864278
|
|
10.
|
PA SteckH LinLA LangfordFunctional and
molecular analyses of 10q deletions in human gliomasGenes
Chromosomes
Cancer24135143199610.1002/(SICI)1098-2264(199902)24:2%3C135::AID-GCC6%3E3.0.CO;2-A9885980
|
|
11.
|
DJ BratEG van MeirVaso-occlusive and
prothrombotic mechanisms associated with tumor hypoxia, necrosis,
and accelerated growth in glioblastomaLab
Invest84397405200410.1038/labinvest.370007014990981
|
|
12.
|
H FujisawaRM ReisM NakamuraLoss of
heterozygosity on chromosome 10 is more extensive in primary (de
novo) than in secondary glioblastomasLab
Invest806572200010.1038/labinvest.378000910653004
|
|
13.
|
H NoushmehrDJ WeisenbergerK
DiefesIdentification of a CpG island methylator phenotype that
defines a distinct subgroup of gliomaCancer
Cell17510522201010.1016/j.ccr.2010.03.01720399149
|
|
14.
|
MC SchmidtS AntweilerN UrbanImpact of
genotype and morphology on the prognosis of glioblastomaJ
Neuropathol Exp Neurol61321328200211939587
|
|
15.
|
R AlbarosaBM ColomboL RozDeletion mapping
of gliomas suggest the presence of two small regions for candidate
tumor-suppressor genes in a 17-cM interval on chromosome 10qAm J
Hum Genet581260126719968651304
|
|
16.
|
DW ParsonsS JonesX ZhangAn integrated
genomic analysis of human glioblastoma
multiformeScience32118071812200810.1126/science.116438218772396
|
|
17.
|
SM LeeHJ KohDC ParkBJ SongTL HuhJW
ParkCytosolic NADP(+)-dependent isocitrate dehydrogenase status
modulates oxidative damage to cellsFree Radic Biol
Med32118511962002
|
|
18.
|
FE BleekerS LambaS LeenstraIDH1 mutations
at residue p.R132 (IDH1(R132)) occur frequently in high-grade
gliomas but not in other solid tumorsHum
Mutat30711200910.1002/humu.2093719117336
|
|
19.
|
A PeraudK WatanabeKH PlateY YonekawaP
KleihuesH Ohgakip53 mutations versus EGF receptor expression in
giant cell glioblastomasJ Neuropathol Exp
Neurol5612361241199710.1097/00005072-199711000-000089370234
|
|
20.
|
BK RasheedRE McLendonHS FriedmanChromosome
10 deletion mapping in human gliomas: a common deletion region in
10q25Oncogene102243224619957784070
|
|
21.
|
IS KilSY KimSJ LeeJW ParkSmall interfering
RNA-mediated silencing of mitochondrial NADP+-dependent
isocitrate dehydrogenase enhances the sensitivity of HeLa cells
toward tumor necrosis factor-alpha and anticancer drugsFree Radic
Biol Med4311971207200717854715
|
|
22.
|
GJ DohrmannJR FarwellJT
FlanneryGlioblastoma multiforme in childrenJ
Neurosurg44442448197610.3171/jns.1976.44.4.0442
|
|
23.
|
U SureD RüediO TachibanaDetermination of
p53 mutations, EGFR overexpression, and loss of p16 expression in
pediatric glioblastomasJ Neuropathol Exp
Neurol56782789199710.1097/00005072-199756070-000049210874
|
|
24.
|
V SuriP DasP PathakPediatric
glioblastomas: a histopathological and molecular genetic studyNeuro
Oncol11274280200910.1215/15228517-2008-09218981259
|
|
25.
|
BR RoodTJ MacDonaldPediatric high-grade
glioma: molecular genetic clues for innovative therapeutic
approachesJ
Neurooncol75267272200510.1007/s11060-005-6749-516195804
|
|
26.
|
N ShahB LinZ SibenallerComprehensive
analysis of MGMT promoter methylation: correlation with MGMT
expression and clinical response in GBMPLoS
One6e16146201110.1371/journal.pone.001614621249131
|
|
27.
|
ME HegiAC DiserensT GorliaMGMT gene
silencing and benefit from temozolomide in glioblastomaN Engl J
Med3529971003200510.1056/NEJMoa04333115758010
|
|
28.
|
K PalanichamyM ErkkinenA
ChakravartiPredictive and prognostic markers in human
glioblastomasCurr Treat Options
Oncol7490504200610.1007/s11864-006-0024-717032561
|
|
29.
|
MD PradosV LevinBiology and treatment of
malignant gliomaSemin Oncol271102000
|
|
30.
|
WA FreijeFE Castro-VargasZ FangGene
expression profiling of gliomas strongly predicts survivalCancer
Res6465036510200410.1158/0008-5472.CAN-04-045215374961
|
|
31.
|
HS PhillipsS KharbandaR ChenMolecular
subclasses of high-grade glioma predict prognosis, delineate a
pattern of disease progression, and resemble stages in
neurogenesisCancer Cell9157173200610.1016/j.ccr.2006.02.019
|
|
32.
|
K WatanabeK SatoW BiernatIncidence and
timing of p53 mutations during astrocytoma progression in patients
with multiple biopsiesClin Cancer Res352353019979815715
|
|
33.
|
C AdamsonOO KanuAI MehtaGlioblastoma
multiforme: a review of where we have been and where we are
goingExpert Opin Investig
Drugs1810611083200910.1517/1354378090305276419555299
|
|
34.
|
FB FurnariT FentonRM BachooMalignant
astrocytic glioma: genetics, biology, and paths to treatmentGenes
Dev2126832710200710.1101/gad.159670717974913
|
|
35.
|
H OhgakiP KleihuesGenetic pathways to
primary and secondary glioblastomaAm J
Pathol7014451453200710.2353/ajpath.2007.070011
|
|
36.
|
EG Van MeirCG HadjipanayisAD NordenHK
ShuPY WenJJ OlsonExciting new advances in neuro-oncology: the
avenue to a cure for malignant gliomaCA Cancer J
Clin60166193201020445000
|
|
37.
|
RG VerhaakKA HoadleyE PurdomIntegrated
genomic analysis identifies clinically relevant subtypes of
glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR
and NF1Cancer Cell1798110201010.1016/j.ccr.2009.12.02020129251
|
|
38.
|
SA GrossmanJF BataraCurrent management of
glioblastoma multiformeSemin
Oncol31635644200410.1053/j.seminoncol.2004.07.00515497116
|
|
39.
|
OO KanuA MehtaC DiGlioblastoma multiforme:
a review of therapeutic targetsExpert Opin Ther
Targets13701718200910.1517/1472822090294234819409033
|
|
40.
|
H OhgakiGenetic pathways to
glioblastomasNeuropathology2517200510.1111/j.1440-1789.2004.00600.x
|
|
41.
|
D KrexB KlinkC HartmannLong-term survival
with glioblastoma
multiformeBrain13025962606200710.1093/brain/awm20417785346
|
|
42.
|
D XieYX ZengHJ WangAmplification and
overexpression of epidermal growth factor receptor gene in
glioblastomas of Chinese patients correlates with patient’s age but
not with tumor’s clinicopathological pathwayActa
Neuropathol110481489200516151725
|
|
43.
|
M LacroixD Abi-SaidDR FourneyA
multivariate analysis of 416 patients with glioblastoma multiforme:
prognosis, extent of resection, and survivalJ
Neurosurg95190198200110.3171/jns.2001.95.2.019011780887
|
|
44.
|
JF MineoA BordronM BaronciniPrognosis
factors of survival time in patients with glioblastoma multiforme:
a multivariate analysis of 340 patientsActa
Neurochir149245252200710.1007/s00701-006-1092-y17273889
|
|
45.
|
M GlasC HappoldJ RiegerLong-term survival
of patients with glioblastoma treated with radiotherapy and
lomustine plus temozolomideJ Clin
Oncol2712571261200910.1200/JCO.2008.19.219519188676
|
|
46.
|
RE McLendonEC HalperinIs the long-term
survival of patients with intracranial glioblastoma multiforme
overstated?Cancer9817451748200310.1002/cncr.1166614534892
|
|
47.
|
YH LiuS SheteCJ EtzelPolymorphisms of
LIG4, BTBD2, HMGA2, and RTEL1 genes involved in the double-strand
break repair pathway predict glioblastoma survivalJ Clin
Oncol2824672474201010.1200/JCO.2009.26.621320368557
|
|
48.
|
JP SteinbachHP BlaicherU
HerrlingerSurviving glioblastoma for more than 5 years: the
patient’s perspectiveNeurology66239242200616434662
|
|
49.
|
LM DeAngelisChemotherapy for brain
tumors-a new beginningN Engl J
Med35210361038200510.1056/NEJMe05801015758016
|
|
50.
|
MW McDermottPK SneedPH GutinInterstitial
brachytherapy for malignant brain tumorsSemin Surg
Oncol147987199810.1002/(SICI)1098-2388(199801/02)14:1%3C79::AID-SSU10%3E3.0.CO;2-49407634
|
|
51.
|
BL LiuJX ChengX ZhangWei
ZhangControversies concerning the application of brachytherapy in
central nervous system tumorsJ Cancer Res Clin
Oncol136173185201010.1007/s00432-009-0741-y19956971
|
|
52.
|
H BremS PiantadosiPC
BurgerPlacebo-controlled trial of safety and efficacy of
intraoperative controlled delivery by biodegradable polymers of
chemotherapy for recurrent gliomas. The Polymer-brain Tumor
Treatment
GroupLancet34510081012199510.1016/S0140-6736(95)90755-6
|
|
53.
|
S SathornsumeteeJN RichNew treatment
strategies for malignant gliomasExpert Rev Anticancer
Ther610871104200610.1586/14737140.6.7.1087
|
|
54.
|
AS ChiAG SorensenRK JainTT
BatchelorAngiogenesis as a therapeutic target in malignant
GliomasOncologist14621636200910.1634/theoncologist.2008-027219487335
|
|
55.
|
RK JainA new target for tumor therapyN
Engl J Med36026692671200910.1056/NEJMcibr090205419535806
|
|
56.
|
PY WenTherapy for recurrent high-grade
gliomas: does continuous dose-Intense temozolomide have a role?J
Clin Oncol2820512057201020308652
|
|
57.
|
WK YungRE AlbrightJ OlsonA phase II study
of temozolomide vs. procarbazine in patients with glioblastoma
multiforme at first relapseBr J
Cancer83588593200910.1054/bjoc.2000.131610944597
|
|
58.
|
A MoustakasTN KreislNew treatment options
in the management of glioblastoma multiforme: a focus on
bevacizumabOncol Targets Ther32738201020616955
|
|
59.
|
S SathornsumeteeJN RichDesigner therapies
for glioblastoma multiformeAnn NY Acad
Sci1142108132200810.1196/annals.1444.00918990124
|
|
60.
|
N LutherNK CheungEP
SouliopoulosInterstitial infusion of glioma-targeted recombinant
immunotoxin 8H9scFv-PE38Mol Cancer
Ther910391046201010.1158/1535-7163.MCT-09-099620371725
|
|
61.
|
Y IwadateT MatsutaniY HasegawaSelection of
chemotherapy for glioblastoma expressing
O6-methyltransferaseExp Ther Med15357201023136592
|