Introduction
The foramen ovale (FO) is one of the special
channels in the fetal period, which shunts blood reflowing from the
vena cava to the left cardiac system to allow regular circulation.
It is open prenatally, then it closes puerperally with the
increasing pressure of the left heart, which forces the FO valve to
move forward to the septum secundum (1–5).
When the size of the FO becomes excessively large, it produces a
foramen secundum atrial septal defect (ASD). This study discusses
the clinical value of FO size and the ratio of FO and aorta size
(FO/AO) in ROC curve analysis for the prediction of pASD.
Subjects and methods
Subjects
A total of 1,690 cases diagnosed using fetal
ultrasonic cardiogram (FUC) from September 2009 to March 2012 were
selected. The inclusion criteria were that the relative prenatal
and puerperal FUC data were accessible and the diagnosis of the
cardiogram was normal. The exclusion criteria were cardiac
abnormalities in the ultrasonic cardiograms, ratio imbalance of the
dextrocardia and levocardia and incomplete data for follow-up.
Informed consent was provided by the subjects themselves or their
relatives.
Instruments
FUC was performed using a GE Voluson E8 diasonogram
with an RAB4-8 probe at a frequency of 4–8 MHz in the ‘fetal
cardiac’ mode. Puerperal re-examination of ultrasonic cardiogram
(PUC) was performed using a Philips iE33 ultrasound apparatus with
a S5-1 probe at a frequency of 1–5 MHz in the ‘paediatrics’ mode
(Philips Ultrasound, Inc., Bothell, WA, USA).
Routine examination
The first examinations were biological indicator
investigations, including biparietal diameter (BPD), head
circumference, abdominal perimeter and femur length (FL) to verify
the fetal gestational weeks. Then FUC examinations were performed
according to the standard guide of FUC recommended by the United
States Association of Echocardiography (6). The scanning sequences of the standard
plane were: abdominal cross section, four chamber view, left
ventricular outflow tract (LVOT) section, right ventricular outflow
tract (RVOT) section, double ventricular short axis view, three
vessels and trachea section, long axis view of the aortic arch and
long axis view of the ductus arteriosus. Cardiac
abnormalities were excluded according to the above section
screening.
Measurement of FO and AO size
The best section for FO measurement was from the
transverse four chamber view. When the valve of the FO was observed
clearly and was opened extensively, the measurement was performed
under the condition of two dimensional ultrasound, three times for
the determination of the average value. The inner diameter of the
AO root was measured at the LVOT section in systole by processing
three times for the mean value.
Puerperal re-examination
FUC was rechecked no less than 12 months after
parturition, in accordance with the standard section, including the
left ventricular long axis view, short axis section of the large
artery, short axis section of the two ventricles, four chamber
view, five chamber view, sword double room section and suprasternal
fossa section. Then, the interatrial septum situation was observed
and cardiac abnormalities were excluded.
Statistical method
Statistical analysis was performed using SPSS 16.0
software (SPSS Inc., Chicago, IL, USA). Measurement data are
presented as the mean ± standard deviation. One-factor analysis of
variance was used for the mean values of multiple groups and the
LSD method was used for pairwise comparison. Linear regression
analysis was used to analyze the correlations between FO size, AO
size and gestational age. An independent-sample t-test was used for
the comparisons between the diagnosis of puerperal ASD (pASD) in
the DGWs and the puerperal re-examination of normal FO size and the
FO/AO ratio. ROC curve and area under the curve were adopted for
the estimation of the demarcation point, sensitivity and
specificity of ASD predicted by FO size and FO/AO.
Results
General clinical data
The patients were aged 17–45 years with an average
age of 28.45±4.34 years. The gestational age was 18–40 weeks with
an average of 26.25±3.71 weeks.
Among the 1,690 cases, 958 (56.69%) cases were
selected to complete follow-up data, while the remainder (732
cases) were excluded due to fetal cardiac abnormality, imbalance of
dextrocardia and levocardia or incomplete relative data.
Correlation between FO size, AO size and
gestational age
Table I presents
the measured values of AFO size, AO size and FO/AO in 958 cases
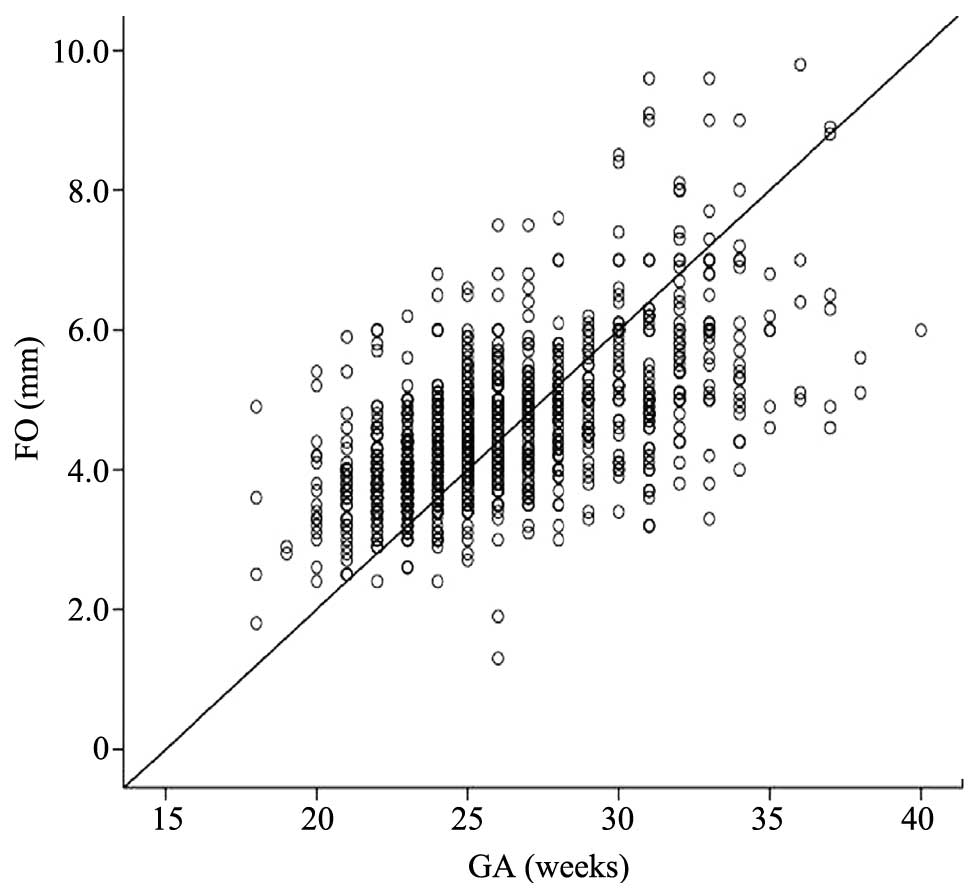
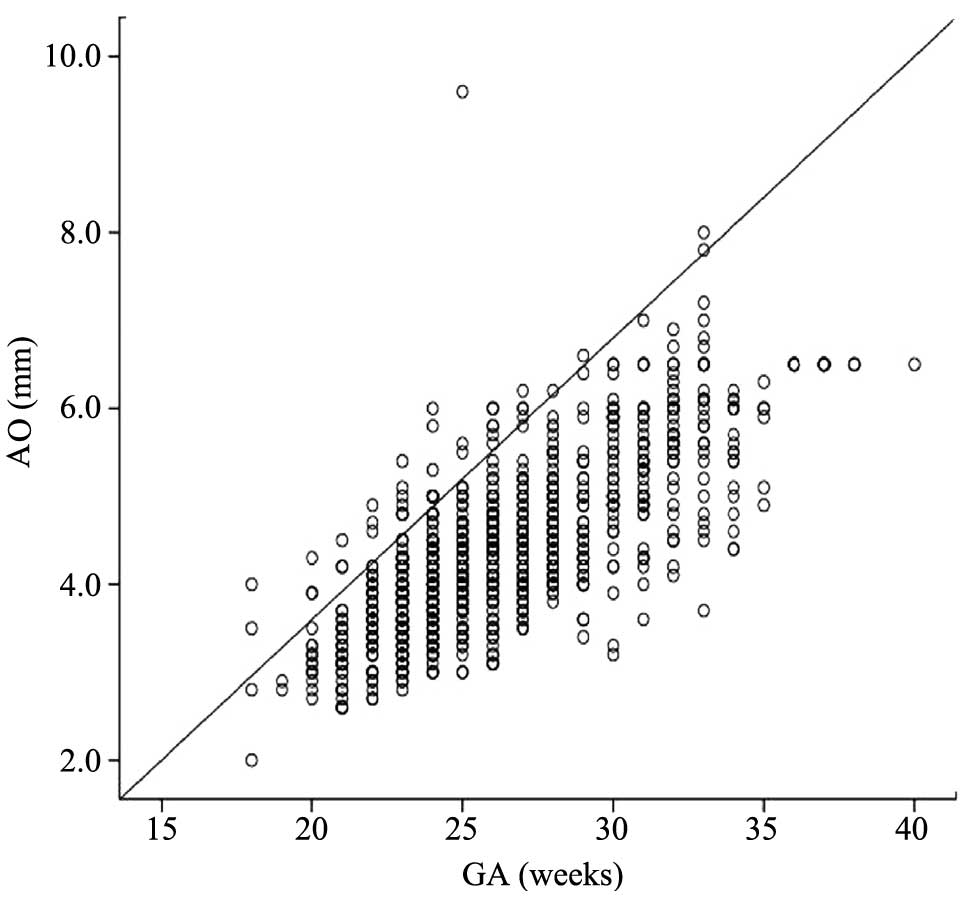
from 18 to 40 weeks. The size of the FO at 18–40 weeks increased
along with the increasing gestational age (Fig. 1), as well as AO (Fig. 2). The sizes of the FO and AO of all
groups were significantly different (P=0.000), while the
differences in FO/AO were not significant (P>0.05).
 | Table IComparisons between antepartum FO
size, AO size and FO/AO in DGWs. |
Table I
Comparisons between antepartum FO
size, AO size and FO/AO in DGWs.
| Gestational age
(weeks) | Number of cases | FO (mm) | AO (mm) | FO/AO |
|---|
| 18–22 | 118 | 3.82±0.79a | 3.42±0.50a | 1.13±0.22 |
| 23–26 | 455 | 4.27±0.77a | 4.06±0.64a | 1.06±0.22 |
| 27–30 | 232 | 4.90±0.93a | 4.71±0.70a | 1.05±0.21 |
| 31–34 | 133 | 5.70±1.31a | 5.57±0.78a | 1.04±0.26 |
| 35–40 | 20 | 6.22±1.47a | 6.26±0.47a | 0.99±0.23 |
Comparisons of pASD in DGWs with
puerperal normal FO size and FO/AO
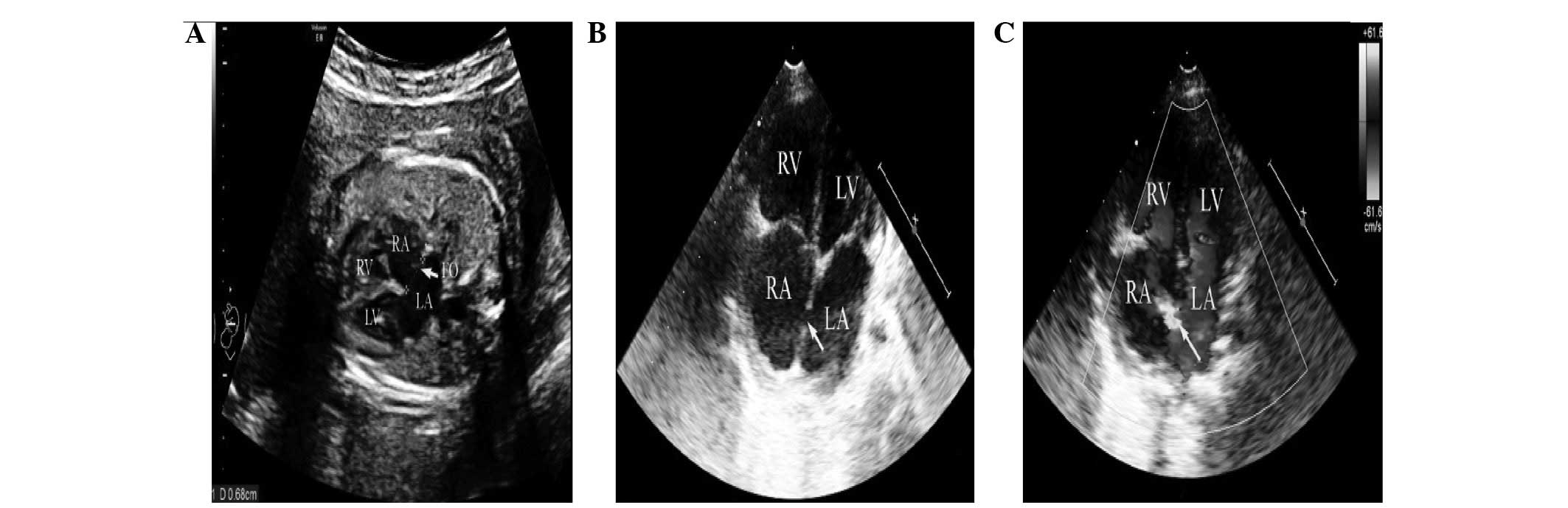
Among the 958 cases, there were 31 cases of pASD
from 18 to 40 weeks. The measured values of pASD, normal antepartum
FO size and FO/AO of the heart are presented in Tables II and III, respectively. These demonstrated
significant differences (P=0.000). The size of the puerperal FO at
12 months, re-examined using ultrasonic cardiography, resulted in a
diagnosis of ASD in certain patients (Fig. 3).
 | Table IIComparison of pASD in DGWs with
puerperal re-examined normal antepartum and puerperal FO sizes. |
Table II
Comparison of pASD in DGWs with
puerperal re-examined normal antepartum and puerperal FO sizes.
| pASD
| Puerperal normal
|
|---|
| Gestational age
(weeks) | Number of cases | Antepartum FO
(mm) | Number of cases | Antepartum FO
(mm) |
|---|
| 18–22 | 5 | 5.76±0.23a | 113 | 3.73±0.69 |
| 23–26 | 12 | 6.23±0.62a | 443 | 4.22±0.70 |
| 27–30 | 6 | 7.53±0.79a | 226 | 4.83±0.82 |
| 31–34 | 5 | 9.26±0.31a | 128 | 5.56±1.13 |
| 35–40 | 3 | 9.17±0.55a | 17 | 5.71±0.78 |
 | Table IIIComparisons of pASD in DGWs with
puerperal re-examined normal antepartum and puerperal FO/AO. |
Table III
Comparisons of pASD in DGWs with
puerperal re-examined normal antepartum and puerperal FO/AO.
| pASD
| Puerperal normal
|
|---|
| Gestational age
(weeks) | Number of cases | Antepartum FO/AO
(mm) | Number of cases | Antepartum FO/AO |
|---|
| 18–22 | 5 | 1.62±0.27a | 113 | 1.10±0.19 |
| 23–26 | 12 | 1.50±0.21a | 443 | 1.05±0.20 |
| 27–30 | 6 | 1.54±0.18a | 226 | 1.04±0.19 |
| 31–34 | 5 | 1.60±0.28a | 128 | 1.01±0.23 |
| 35–40 | 3 | 1.41±0.87a | 17 | 0.92±0.16 |
Prediction of pASD by ROC curve
analysis
Related indicatrix of the ROC curve of pASD
predicted by FO size and FO/AO in DGWs are presented in Table IV and V. The areas under the curve of pASD
predicted by FO size at 18–22, 23–26, 27–30, 31–34 and 35–40 weeks
were 0.991, 0.986, 0.991, 0.998 and 1.000, with demarcation points
as 5.02, 5.15, 6.55, 8.55 and 7.90 mm, respectively. Their
specificities were 98.2, 90.5, 97.3, 99.2 and 100%, while the
sensitivities were all 100%. The areas under the curve of pASD
predicted by FO/AO at 18–22, 23–26, 27–30, 31–34 and 35–40 weeks
were 0.958, 0.937, 0.974, 0.967 and 0.961, with demarcation points
as 1.28, 1.40, 1.32, 1.33 and 1.22, respectively. The sensitivities
were 100, 83.3, 100, 100 and 100% and their specificities were
83.0, 78.4, 92.9, 92.2 and 94.1%, respectively. The above results
demonstrate that ROC curve analysis of AFO and FO/AO for prediction
of pASD is highly accurate.
 | Table IVRelative indices of the ROC curve for
prediction of pASD by FO size at different gestational ages. |
Table IV
Relative indices of the ROC curve for
prediction of pASD by FO size at different gestational ages.
| GA (weeks) | Area under curve | 95% confidence
interval | DP (mm) | Sensitivity (%) | Specificity (%) |
|---|
| 18–22 | 0.991 | 0.975–1.000 | 5.02 | 100 | 98.2 |
| 23–26 | 0.986 | 0.972–1.000 | 5.15 | 100 | 90.5 |
| 27–30 | 0.991 | 0.979–1.000 | 6.55 | 100 | 97.3 |
| 31–34 | 0.998 | 0.994–1.000 | 8.55 | 100 | 99.2 |
| 35–40 | 1.000 | 1.000–1.000 | 7.90 | 100 | 100 |
 | Table VRelative indexes of ROC curve for
prediction of pASD by FO/AO at different gestational ages. |
Table V
Relative indexes of ROC curve for
prediction of pASD by FO/AO at different gestational ages.
| GA (weeks) | Area under curve | 95% confidence
interval | DP (mm) | Sensitivity (%) | Specificity (%) |
|---|
| 18–22 | 0.958 | 0.898–1.000 | 1.28 | 100 | 83.0 |
| 23–26 | 0.937 | 0.879–0.994 | 1.40 | 83.3 | 78.4 |
| 27–30 | 0.974 | 0.947–1.000 | 1.32 | 100 | 92.9 |
| 31–34 | 0.967 | 0.933–1.000 | 1.33 | 100 | 92.2 |
| 35–40 | 0.961 | 0.874–1.000 | 1.22 | 100 | 94.1 |
Discussion
At the end of the fourth week of fetal development,
a diaphragm-like tissue erupts downward on the centre line of the
top of the common atrium, which is the septum primum. This
diaphragm grows up in the direction of the endocardial cushion,
with the formation of an ostiole called the foramen primum before
the cushion merges with the diaphragm. With the coalition of the
inferior margin of the septum primum and the endocardial cushion,
the foramen primum is sealed off and the heart atrium is divided
into the left and right atrium. Then, the upper local tissues are
dissolved and absorbed gradually, forming a tunnel gallery called
the foramen secundum. Additionally, on the top of the right atrium
of the septum primum, a diaphragm-like tissue erupts, which is the
septum secundum. With the complete anastomosis of the inferior
margin of the septum secundum and the endocardial cushion, the
diaphragm spreads downward and covers the upper foramen secundum,
behind which there is an ostiole called the FO. The thinner
valve-like tissues block the FO from the left atrium, performing a
valve-like function on the blood circulation of the fetus. The ‘FO
valve’ prevents the blood that has entered the left atrium from the
right atrium from reversing. Following birth, the FO valve is
pressed toward the septum secundum as the pressure of the left
atrium is higher than that of the right, leading to the closure of
the FO. If the FO is oversized, ASD of the foramen secundum occurs
as the septum primum is not able to provide a shade for the FO.
Fetal FO has a growth tendency, similar to other
organs. In 1990, Wilson et al(7) determined that the FO of gestational
aged fetuses are almost equal size to the aortic root inner
diameter, or the d-value was <1.0 mm, by means of observing the
FO and AO roots of 48 fetuses. In 1994, Phillipos et
al(3) demonstrated, by
analyzing the correlation between fetal FO size and gestational age
(GA) of 100 cases from 20 to 38 gestational weeks, that the fetal
FO enlarges with increasing gestational age. By analyzing 958 cases
divided into five groups according to the number of gestational
weeks, we identified that FO size and AO size among all the groups
demonstrated statistical differences, FO/AO did not increase with
increasing gestational age and FO/AO presented no significant
difference among all the groups. In the present study, we
demonstrated that the pASD demarcation point (DP) predicted by the
FO size at 18–34 gestational weeks increases with the augmentation
of gestational age. However, the DP decreases at 35–40 gestational
weeks, which may be related to the gradual closure of the FO in the
late stage of pregnancy. A number of scholars have noted that fetal
hemodynamics may be altered when the FO size is smaller than normal
(8–16). In the current study, we observed
the FO of DGWs in detail, by dividing the 958 cases into different
groups, which provided referred evidence for certain fetuses
accompanied with congenital heart growth restriction or early
closure of the FO.
The FO is one of the special channels involved in
sustaining normal blood circulation (17–19).
As the FO closes gradually after birth, infants aged >1 year
generally present echo interruption in the middle of the atrial
septum and color Doppler reveals that the atrial level shunts from
left to right, which may be diagnosed as ASD. Certain researchers
suggested that when the size of the FO in the fetal period is
>8.0 mm, it should be considered as ASD, while the puerperal
follow-up periods identified that not all FO >8 mm led to ASD.
Additionally, certain FO <8 mm resulted in ASD. Thus, it is
difficult to predict pASD via the observation of the size of the
FO. Theoretically speaking, the size of the FO in DGWs during the
fetal period grows with the increase of gestational age. Therefore,
the prediction of pASD using only one value is not accurate. In the
present study, we divided 958 cases into groups according to the
number of gestational weeks. Then, relative referenced data were
obtained by utilizing ROC curve analysis for the evaluation of the
size of the FO to predict the value of pASD in DGWs and to analyze
the correlations among fetal FO size, AO size and FO/AO in DGWs.
The false positive rate of ASD when the FO closes naturally within
a year of birth may be reduced by rechecking the ultrasound
cardiogram of patients when they are aged >12 months. The
puerperal diagnosed FO size and AO size, and the rechecked normal
FO size and AO size were significantly different, indicating that
measured values of FO size and FO/AO of pASD were larger than
puerperal re-examined values. The results of pASD predicted by FO
size and FO/AO in DGWs revealed that the size of the FO in DGWs
produced different DPs, specifically, the DPs of pASD predicted by
FO size also increased with increasing gestational age, which
contributed to the growth tendency of the FO; the 35–40 week
gestational age was an exception, when the DPs decreased instead.
This study demonstrated that FO/AO is not correlated with
gestational age. The DP of pASD predicted by FO/AO was 1.22–1.40.
Therefore, there is clinical value in utilizing the size of the FO
combined with FO/AO for the prediction of ASD. In this study, the
area under the ROC curve, for the prediction of pASD via AFO size
and FO/AO in DGWs, was >0.9, illustrating the high accuracy of
diagnosis with this method.
In conclusion, the AFO size and FO/AO of pASD in
DGWs were greater than those of a normal heart. Prediction of pASD
was more accurate when combining the above two indices (AFO size
and FO/AO in DGWs). Due to the lack of sample capacity in this
study, an increase in sample numbers is required for a more indepth
investigation.
Acknowledgements
This study was supported by the
Natural Science Foundation of China (81171350).
References
|
1.
|
Gardiner HM: Fetal echocardiography: 20
years of progress. Heart. 86(Suppl 2): S12–S22. 2001.PubMed/NCBI
|
|
2.
|
Wilson AD, Rao PS and Aeschlimann S:
Normal fetal foramen flap and transatrial Doppler velocity pattern.
J Am Soc Echocardiogr. 3:491–494. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Phillipos EZ, Robertson MA and Still KD:
The echocardiographic assessment of the human fetal foramen ovale.
J Am Soc Echocardiogr. 7:257–263. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Kiserud T and Rasmussen S: Ultrasound
assessment of the fetal foramen ovale. Ultrasound Obstet Gynecol.
17:119–124. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Wilhelm C, Prompeler H, Barth R and
Schillinger H: Ultrasound biometry of the fetal heart. Zentralbl
Gynakol. 114:279–286. 1992.(In German).
|
|
6.
|
Rychik J, Ayres N, Cuneo B, Gotteiner N,
Hornberger L, Spevak PJ and Van Der Veld M: American Society of
Echocardiography guidelines and standards for performance of the
fetal echocardiogram. J Am Soc Echocardiogr. 17:803–810. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Wilson AD, Rao PS and Aeschlimann S:
Normal fetal foramen flap and transatrial doppler velocity pattern.
J Am Soc Echocardiogr. 3:491–494. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Zimmer LP, Dillenburg RF, Dornelles AP,
Andrade A and Zielinsky P: Antepartum restriction of the foramen
ovale. Arq Bras Cardiol. 68:285–288. 1997.(In Portuguese).
|
|
9.
|
Salih M, Demirel LC and Kurtay G:
Antepartum diagnosis of ostium secundum atrial septal defect by
M-mode fetal echocardiography. Gynecol Obstet Invest. 45:68–70.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
van Eyck J, Stewart PA and Wladimiroff JW:
Human fetal foramen ovale flow velocity waveforms relative to
behavioral states in normal term pregnancy. Am J Obstet Gynecol.
163:1239–1242. 1990.
|
|
11.
|
Donofrio MT, Bremer YA and Moskowitz WB:
Diagnosis and management of restricted or closed foramen ovale in
fetuses with congenital heart disease. Am J Cardiol. 94:1348–1351.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Chaoui R and Bollmann R: Fetal color
Doppler echocardiography. Part 1: General principles and normal
findings. Ultraschall Med. 15:100–104. 1994.(In German).
|
|
13.
|
Chaoui R and Bollmann R: Fetal color
Doppler echocardiography. Part 2: Abnormalities of the heart and
great vessels. Ultraschall Med. 15:105–111. 1994.(In German).
|
|
14.
|
Chobot V, Hornberger LK, Hagen-Ansert S
and Sahn DJ: Prenatal detection of restrictive foramen ovale. J Am
Soc Echocardiogr. 3:15–19. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Phillipos EZ, Robertson MA and Still DK:
Antepartum detection of foramen ovale obstruction without hydrops
fetalis. J Am Soc Echocardiogr. 3:495–498. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Hagen A, Albig M, Schmitz L, Hopp H, van
Baalen A, Becker R and Entezami M: Antepartum diagnosis of isolated
foramen ovale obstruction. A report of two cases Fetal Diagn Ther.
20:70–73. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Feit LR, Copel JA and Kleinman CS: Foramen
ovale size in the normal and abnormal human fetal heart: An
indicator of transatrial flow physiology. Ultrasound Obstet
Gynecol. 1:313–319. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Kleinman CS and Copel JA: Fetal
cardiovascular physiology and therapy. Fetal Diagn Ther. 7:147–157.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Friedman AH and Fahey JT: The transition
from fetal to neonatal circulation: normal responses and
implications for infants with heart disease. Semin Perinatol.
17:106–121. 1993.PubMed/NCBI
|