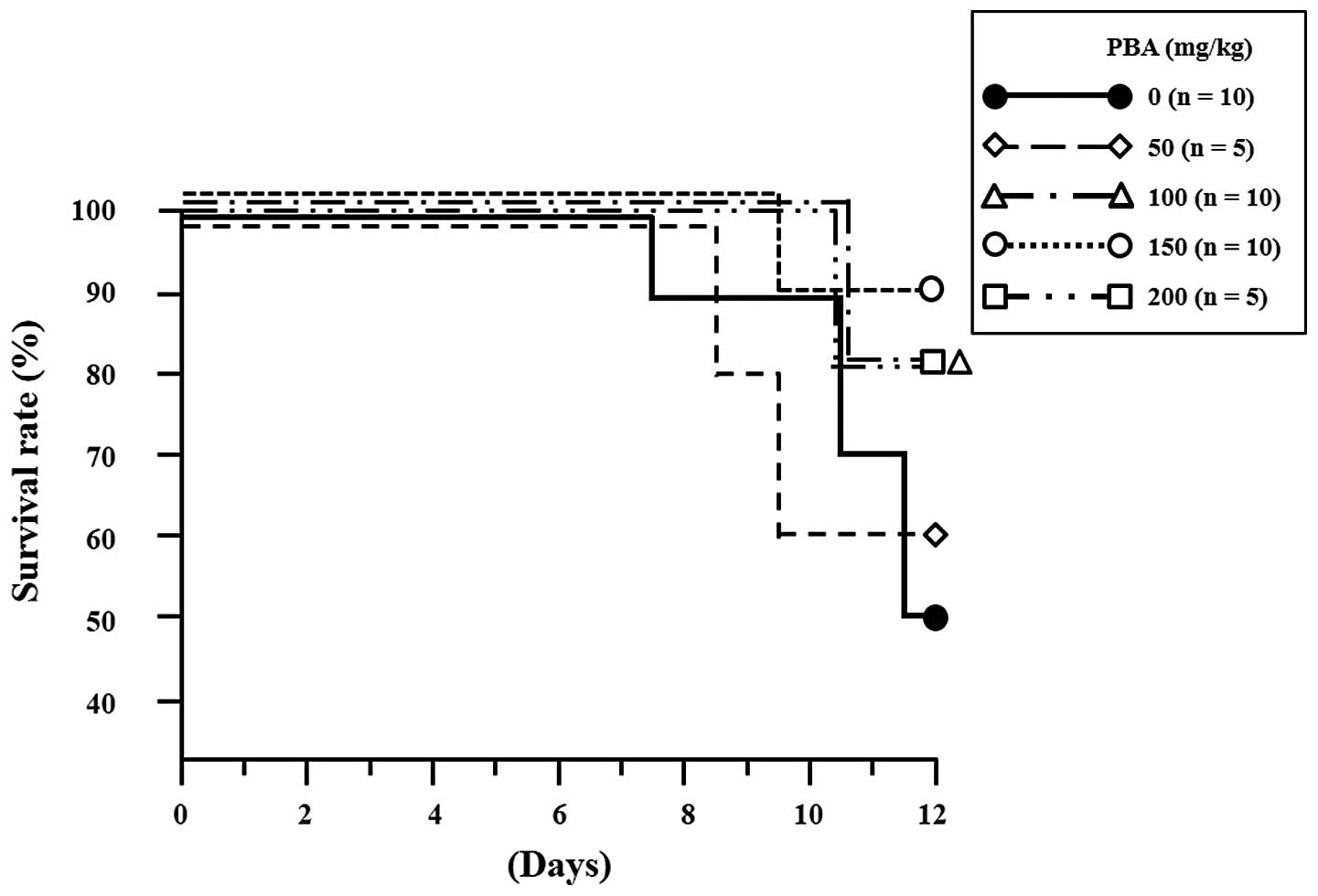
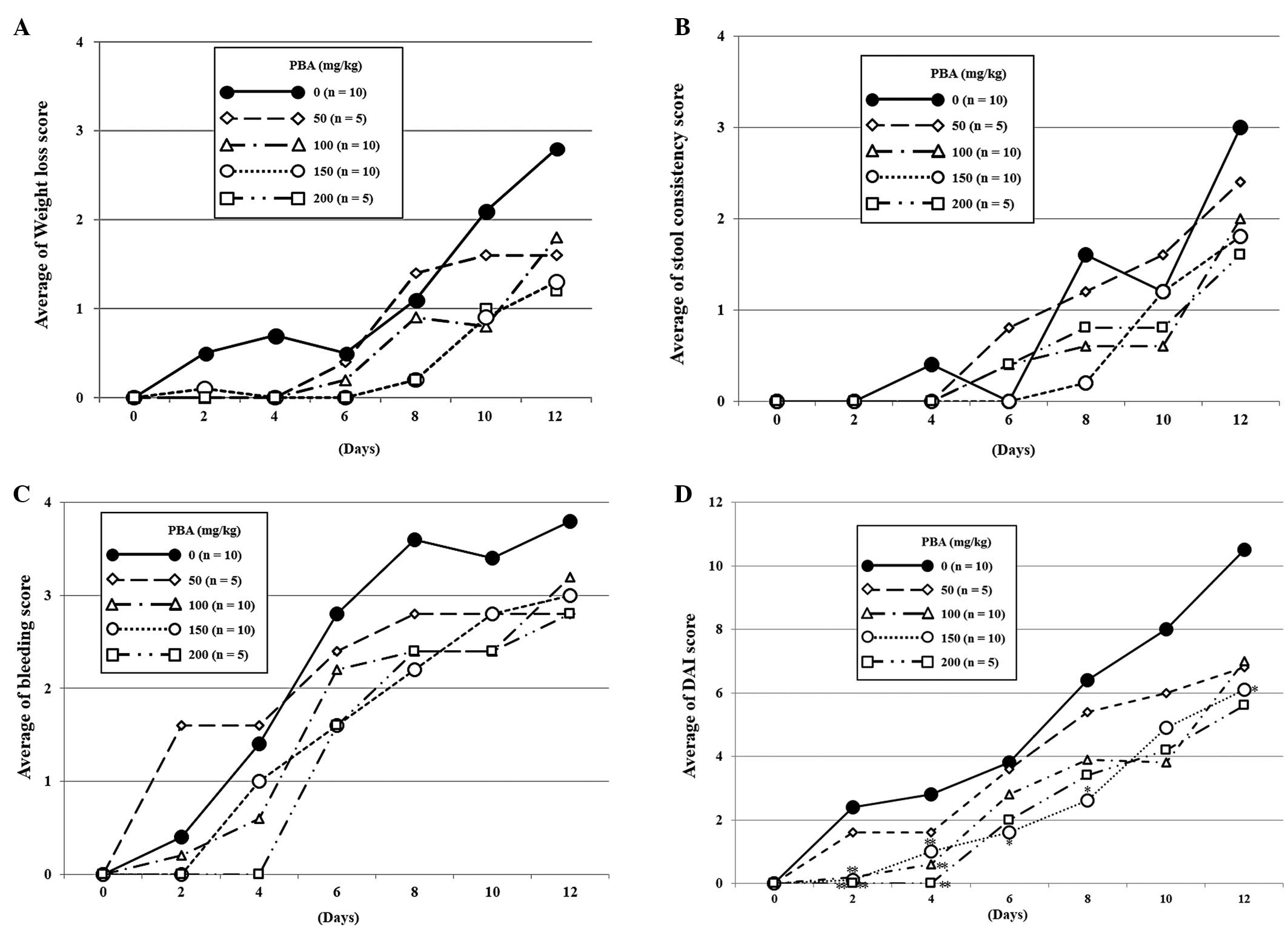
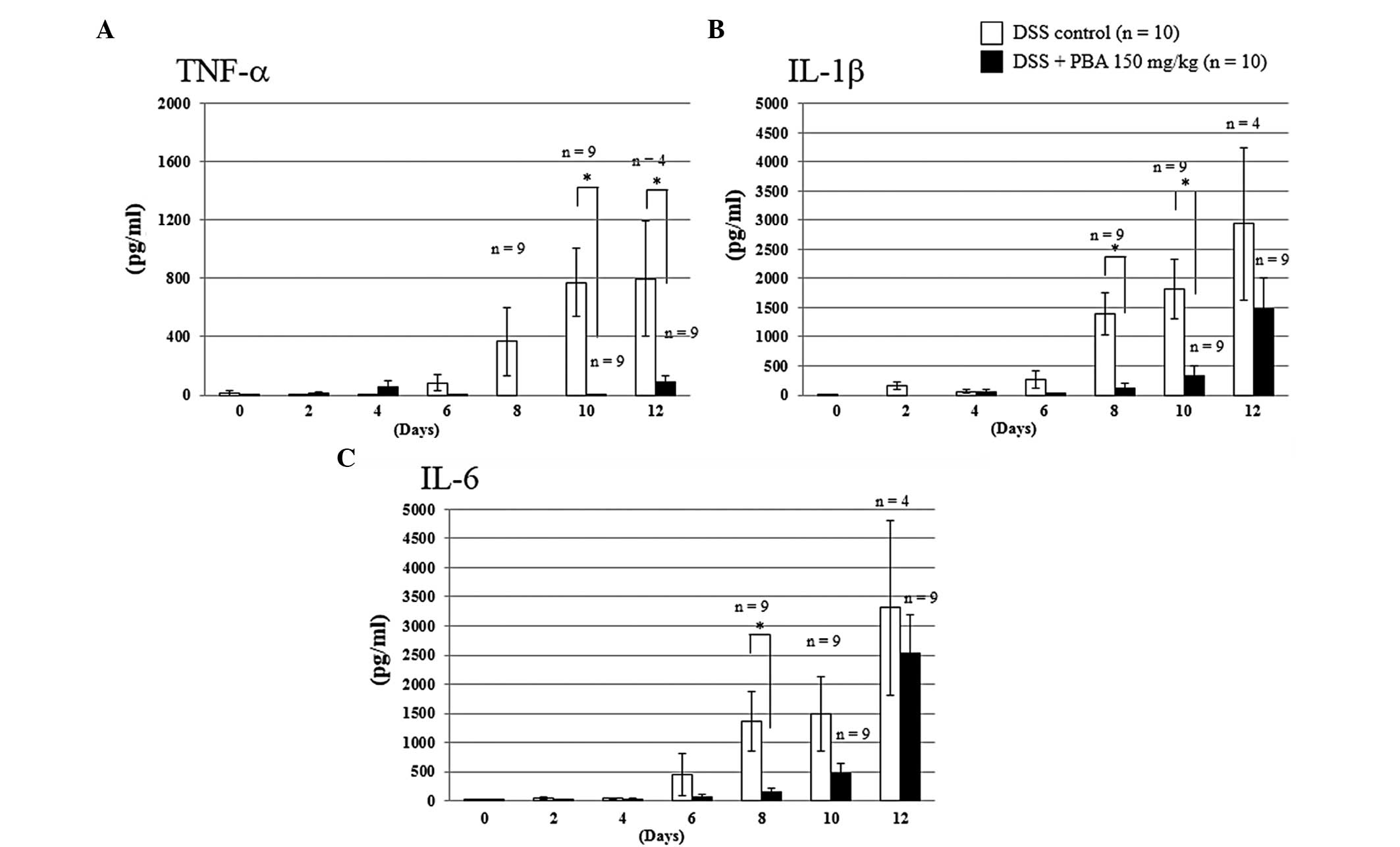
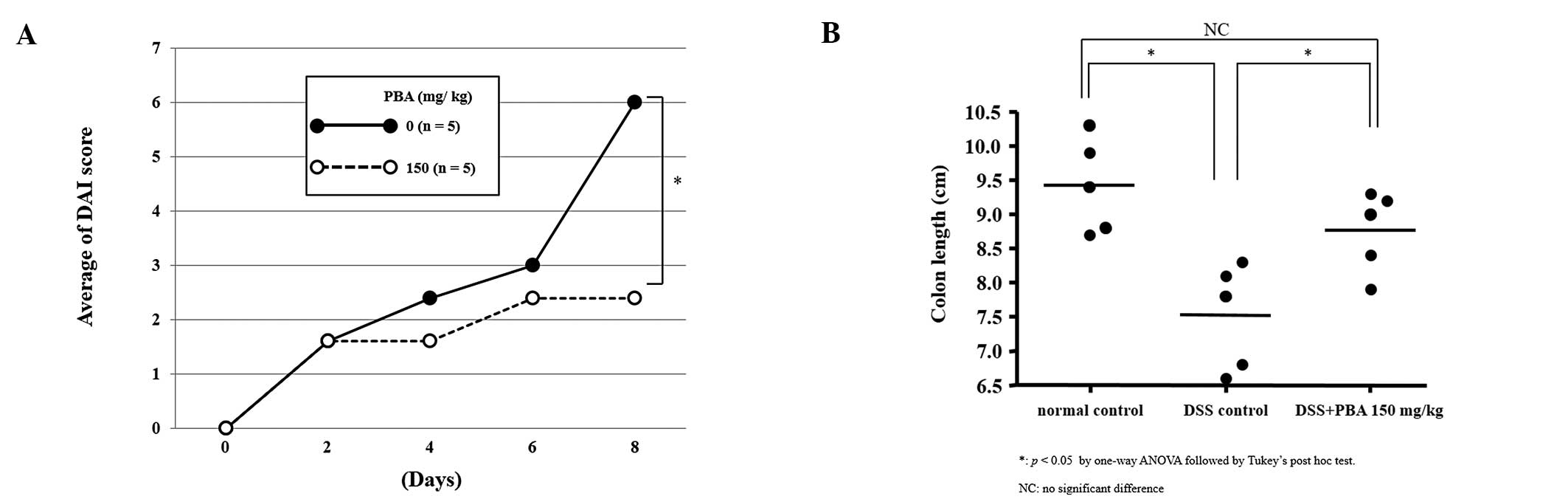
|
1
|
Fiocchi C: Inflammatory bowel disease:
etiology and pathogenesis. Gastroenterology. 115:182–205. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Podolsky DK: Inflammatory bowel disease. N
Engl J Med. 347:417–429. 2002. View Article : Google Scholar
|
|
3
|
Kaser A, Zeissig S and Blumberg RS:
Inflammatory bowel disease. Annu Rev Immunol. 28:573–621. 2010.
View Article : Google Scholar
|
|
4
|
Pullman WE, Elsbury S, Kobayashi M, Hapel
AJ and Doe WF: Enhanced mucosal cytokine production in inflammatory
bowel disease. Gastroenterology. 102:529–537. 1992.PubMed/NCBI
|
|
5
|
Bouma G and Strober W: The immunological
and genetic basis of inflammatory bowel disease. Nat Rev Immunol.
3:521–533. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Strober W, Fuss I and Mannon P: The
fundamental basis of inflammatory bowel disease. J Clin Invest.
117:514–521. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sartor RB: Mechanisms of disease:
pathogenesis of Crohn’s disease and ulcerative colitis. Nat Clin
Pract Gastroenterol Hepatol. 3:390–407. 2006.
|
|
8
|
Maloy KJ and Powrie F: Intestinal
homeostasis and its breakdown in inflammatory bowel disease.
Nature. 474:298–306. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Scharl M and Rogler G: Inflammatory bowel
disease pathogenesis: what is new? Curr Opin Gastroenterol.
28:301–309. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sands BE: Therapy of inflammatory bowel
disease. Gastroenterology. 118:S68–S82. 2000. View Article : Google Scholar
|
|
11
|
Shteyer E and Wilschanski M: Novel
therapeutic modalities in pediatric inflammatory bowel disease. Isr
Med Assoc J. 10:816–820. 2008.PubMed/NCBI
|
|
12
|
Reddy JG and Loftus EV Jr: Safety of
infliximab and other biologic agents in the inflammatory bowel
disease. Gastroenterol Clin North Am. 35:837–855. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Katz JA: Management of inflammatory bowel
disease in adults. J Dig Dis. 8:65–71. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yamamoto T, Umegae S and Matsumoto K:
Mucosal healing in patients with ulcerative colitis during a course
of selective leukocytapheresis therapy: a prospective cohort study.
Inflamm Bowel Dis. 16:1905–1911. 2010. View Article : Google Scholar
|
|
15
|
Maestri NE, Brusilow SW, Clissold DB and
Bassett SS: Long-term treatment of girls with ornithine
transcarbamylase deficiency. N Engl J Med. 335:855–859. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Collins AF, Pearson HA, Giardina P,
McDonagh KT, Brusilow SW and Dover GJ: Oral sodium phenylbutyrate
therapy in homozygous beta thalassemia: a clinical trial. Blood.
85:43–49. 1995.PubMed/NCBI
|
|
17
|
Mercuri E, Bertini E, Messina S,
Pelliccioni M, D’Amico A, Colitto F, Mirabella M, Tiziano FD,
Vitali T, Angelozzi C, et al: Pilot trial of phenylbutyrate in
spinal muscular atrophy. Neuromuscul Disord. 14:130–135. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Camacho LH, Olson J, Tong WP, Young CW,
Spriggs DR and Malkin MG: Phase I dose escalation clinical trial of
phenylbutyrate sodium administered twice daily to patients with
advanced solid tumors. Invest New Drugs. 25:131–138. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Roy A, Ghosh A, Jana A, Liu X, Brahmachari
S, Gendelman HE and Pahan K: Sodium phenylbutyrate controls
neuroinflammatory and antioxidant activities and protects
dopaminergic neurons in mouse models of Parkinson’s disease. PLoS
One. 7:e381132012.PubMed/NCBI
|
|
20
|
Luo ZF, Feng B, Mu J, Qi W, Zeng W, Guo
YH, Pang Q, Ye ZL, Liu L and Yuan FH: Effects of 4-phenylbutyric
acid on the process and development of diabetic nephropathy induced
in rats by streptozotocin: regulation of endoplasmic reticulum
stress-oxidative activation. Toxicol Appl Pharmacol. 246:49–57.
2010. View Article : Google Scholar
|
|
21
|
Park JS, Lee EJ, Lee JC, Kim WK and Kim
HS: Anti-inflammatory effects of short chain fatty acids in
IFN-gamma-stimulated RAW 264.7 murine macrophage cells: involvement
of NF-kappaB and ERK signaling pathways. Int Immunopharmacol.
7:70–77. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vij N, Fang S and Zeitlin PL: Selective
inhibition of endoplasmic reticulum-associated degradation rescues
DeltaF508-cystic fibrosis transmembrane regulator and suppresses
interleukin-8 levels: therapeutic implications. J Biol Chem.
281:17369–17378. 2006. View Article : Google Scholar
|
|
23
|
Ono K, Ikemoto M, Kawarabayashi T, Ikeda
M, Nishinakagawa T, Hosokawa M, Shoji M, Takahashi M and Nakashima
M: A chemical chaperone, sodium 4-phenylbutyric acid, attenuates
the pathogenic potency in human alpha-synuclein A30P + A53T
transgenic mice. Parkinsonism Relat Disord. 15:649–654.
2009.PubMed/NCBI
|
|
24
|
Wirtz S, Neufert C, Weigmann B and Neurath
MF: Chemically induced mouse models of intestinal inflammation. Nat
Protoc. 2:541–546. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cooper HS, Murthy SN, Shah RS and
Sedergran DJ: Clinicopathologic study of dextran sulfate sodium
experimental murine colitis. Lab Invest. 69:238–249.
1993.PubMed/NCBI
|
|
26
|
Dionne S, D’Agata ID, Hiscott J, Vanounou
T and Seidman EG: Colonic explant production of IL-1 and its
receptor antagonist is imbalanced in inflammatory bowel disease
(IBD). Clin Exp Immunol. 112:435–442. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tountas NA, Casini-Raggi V, Yang H, Di
Giovine FS, Vecchi M, Kam L, Melani L, Pizarro TT, Rotter JI and
Cominelli F: Functional and ethnic association of allele 2 of the
interleukin-1 receptor antagonist gene in ulcerative colitis.
Gastroenterology. 117:806–813. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kwon KH, Murakami A, Hayashi R and
Ohigashi H: Interleukin-1beta targets interleukin-6 in progressing
dextran sulfate sodium-induced experimental colitis. Biochem
Biophys Res Commun. 337:647–654. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rutgeerts P, Van Assche G and Vermeire S:
Optimizing anti-TNF treatment in inflammatory bowel disease.
Gastroenterology. 126:1593–1610. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yun L and Hanauer S: Selecting appropriate
anti-TNF agents in inflammatory bowel disease. Expert Rev
Gastroenterol Hepatol. 3:235–248. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bosani M, Ardizzone S and Porro GB:
Biologic targeting in the treatment of inflammatory bowel diseases.
Biologics. 3:77–97. 2009.PubMed/NCBI
|
|
32
|
Baert F, Noman M, Vermeire S, Van Assche
G, D’Haens G, Carbonez A and Rutgeerts P: Influence of
immunogenicity on the long-term efficacy of infliximab in Crohn’s
disease. N Engl J Med. 348:601–608. 2003.
|
|
33
|
Farrell RJ, Alsahli M, Jeen YT, Falchuk
KR, Peppercorn MA and Michetti P: Intravenous hydrocortisone
premedication reduces antibodies to infliximab in Crohn’s disease:
a randomized controlled trial. Gastroenterology. 124:917–924.
2003.PubMed/NCBI
|