Introduction
Atherosclerosis of the intracranial arteries is
frequent and may account for almost one-third of ischemic strokes
in the Chinese population (1,2). The
underlying mechanisms of cerebral infarction include
artery-to-artery embolism, hemodynamic compromise, local branch
occlusion or a combination of those conditions (3). Clique histological study of the
middle cerebral artery (MCA) has demonstrated that luminal stenosis
is frequently caused by ruptured vulnerable plaques, which are
characterized by their specific morphology and composition, which
comprises a large lipid/necrotic core covered by a thin fibrous cap
infiltrated by macrophages and intraplaque hemorrhage (4). Each clique corresponds to a pair of
neighboring pixels, and the clique potential is designed to favor
similar classes in neighboring pixels. Progression and a greater
extent of intracranial atherosclerosis imply a higher risk of
recurrence (5).
High-resolution magnetic resonance imaging (HRMRI)
has a unique ability to provide information on plaque composition
comparable to that obtained by histology (6). Several studies have confirmed the
feasibility of using HRMRI to evaluate the intracranial artery wall
and have identified the presence of arterial plaques using HRMRI,
in cases where magnetic resonance angiography (MRA) showed no lumen
abnormality (7–9). In the present study, HRMRI was used
to identify the ischemic stroke subtypes of patients with
intracranial atherosclerosis and to investigate the possible
mechanisms.
Material and methods
Study population
A single-center, prospective trial was conducted in
the Neurology Department of Beijing Anzhen Hospital, Capital
Medical University (Beijing, China) between January 2010 and
January 2013. A total of 55 patients with acute cerebral infarction
were screened and were subsequently tested for ≥50% MCA and basilar
artery (BA) stenosis by cranial MRI and MRA. The ischemic stroke in
these patients was presumed to be caused by atherosclerotic
disease. Inclusion criteria were as follows: i) unilateral middle
cerebral artery stenosis (≥70%) or occlusion due to atherosclerosis
were observed while no ipsilateral internal carotid artery stenosis
or occlusion could be found; ii) there was infarcted focus within
corresponding stenosed artery on MRI. iii) Patients must have at
least one of the risk factors for atherosclerosis, including
hypertension, diabetes, hyperlipidemia, homocysteine and smoking.
Exclusion criteria included: i) patients with ipsilateral internal
carotid artery stenosis or occlusion. ii) Non-atherosclerotic
cerebral artery stenosis, such as fibromuscular dysplasia,
arteritis and dissecting aneurysm. iii) Patients suspected to have
symptoms of cardiogenic embolism, including recent myocardial
infarction, atrial fibrillation with or without mural thrombus,
mitral stenosis or prosthetic valve, dilated cardiomyopathy, sick
sinus syndrome, acute bacterial endocarditis and patent foramen
ovale. Written informed consent was obtained from all patients. The
Ethical Committee of Beijing Anzhen Hospital approved this study
(Beijing, China).
Clinical assessment
All patients underwent a detailed medical history
assessment and a physical examination at baseline that included
routine blood biochemistry tests, coagulation testing,
transthoracic or transesophageal echocardiography, Holter
electrocardiography, transcranial Doppler sonography, carotid
ultrasound, and computed tomography, MRI and MRA of the brain. Data
collected from patients included the following baseline
characteristics: Age, gender, vascular risk factors, such as
hypertension or history of hypertension or its complications,
history of diabetes mellitus (DM) or currently diagnosed DM,
hyperlipidemia, history of smoking, previous coronary artery
disease and previous cerebrovascular disease.
Ischemic stroke subclassification
According to the Chinese ischemic stroke
subclassification (10), the
patients were grouped into two mechanism-based categories:
Penetrating artery disease (PAD; no evidence of atherosclerotic
plaques or any degree of stenosis in the parent artery) and
large-artery atherosclerosis (LAA; plaques in the parent artery
occluding a penetrating artery, artery-to-artery embolism or
hypoperfusion/impaired emboli clearance).
MRI protocol and review
All patients were imaged at the Beijing Anzhen
Hospital using a Magnetom Verio 3T MRI scanner (Siemens AG,
Erlangen, Germany) and an eight-channel brain-array coil. A
standardized protocol was used to perform conventional brain T1-
and T2-weighted MRI and three-dimensional (3D) time-of-flight
(TOF)-MRA. 3D TOF-MRA data were obtained using an axial plane with
a repetition time (TR)/echo time (TE) of 21 msec/3.6 msec; flip
angle of 18°; field-of-view (FOV) of 220×220 mm; slice thickness of
0.5 mm; and a matrix size of 320×380 pixels. The TOF-MRA scan time
was 4 min. MRA data were reconstructed using a dedicated online
post-processing tool [multiplanner reconstruction (MPR), maximum
intensity (MIP), volume rendering (VR)] to determine the blood
vessel architecture.
HRMRI data were acquired from the patients with MCA
and BA steno-occlusive lesions along the short axes of the stenotic
segments on TOF-MRA images. The lesion site for the evaluation of
unilateral MCA stenosis was determined by the interpreting
neuroradiologist as the ipsilateral MCA in the symptomatic patients
and the side of severe MCA stenosis in the asymptomatic patients.
T1- and T2-weighted MRI was centered at the stenosis of the MCA-M1
segment, vertebral artery, BA and their confluences. The MRI
parameters were: T1-weighted, double inversion recovery, black
blood, two-dimensional turbo spin echo (TSE), TR/TE = 920 msec/27
msec, FOV = 120×120 mm, matrix size = 270×320 pixels, slice
thickness = 2.0 mm and 2NEX. For the T2-weighted HRMRI scans, the
TSE sequence used a TR/TE of 2,350 msec/78 msec, FOV of 120×120 mm,
matrix size of 270×320 pixels, slice thickness of 2.0 mm and 2NEX.
The black blood technique with pre-regional saturation pulses of
80- mm thickness to saturate incoming arterial flow was used for
the scans. The longitudinal coverage of each artery was 16 mm
(eight slices) for the two types of scans, with a scan time of ~3–4
min/scan. The total scan time was ~20 min and the patients remained
in the MRI machine for ~30 min.
Statistical analysis
A χ2 test was used to compare
frequencies. One-way analysis of variance and Student’s t-test were
used for normally distributed variables, whereas the Mann-Whitney U
test was used for non-normally distributed variables. All tests of
statistical significance were two-sided, with P<0.05 considered
to indicate a statistically significant difference. All statistical
analyses were performed using SPSS software, version 17.0 (SPSS,
Inc., Chicago, IL, USA).
Results
A total of 55 patients who met the eligibility
criteria were recruited to this study. There were 38 males and 17
females with a median age of 58.9 years (standard deviation, ±13.4
years; range, 35–81 years). Among the 55 patients, 17 had
vertebrobasilar stenosis (31%) and 38 had MCA stenosis (69%). PAD
was diagnosed in 20 of the patients (36%) and LAA was diagnosed in
35 of the patients, which included 19 with a parent artery plaque
occluding a penetrating artery (POPA) (35%) and 16 with
artery-to-artery embolism and/or hypoperfusion (29%). The baseline
characteristics in the two groups were comparable, with the
exceptions that the patients with PAD had a higher frequency of
hypertension compared with that of the patients with LAA (80 versus
29%; P<0.001) and the patients with LAA had a higher frequency
of DM than that of the patients with PAD (40 versus 15%; P=0.054)
(Table I). POPA occurred more
frequently in the patients with mild to moderate artery stenosis
(63%; P<0.05) than in the patients with severe artery stenosis
or occlusion (37%). However, the stroke mechanisms of
artery-to-artery embolism and/or hypoperfusion were mainly observed
in the patients with severe artery stenosis or occlusion (68%)
compared with in the patients with mild to moderate artery stenosis
(13.3%; P=0.060).
 | Table IBaseline characteristics of patients
with PAD and LAA. |
Table I
Baseline characteristics of patients
with PAD and LAA.
| Characteristic | PAD (n=20) | LAA (n=35) | P-value |
|---|
| Mean age (years; SD,
range) | 62 (14.5, 35–81) | 58 (12.1, 47–75) | 0.569 |
| Gender
(male/female) | 13/7 | 25/10 | 0.619 |
| Hypertension | 16 (80%) | 10 (29%) | <0.001 |
| DM | 3 (15%) | 14 (40%) | 0.054 |
| Hyperlipidemia | 4 (20%) | 6 (17%) | 0.792 |
| History of
smoking | 8 (40%) | 13 (37%) | 0.834 |
| History of CAD | 5 (25%) | 9 (26%) | 0.953 |
| History of CVD | 3 (15%) | 7 (20%) | 0.644 |
HRMRI enabled the detection of the lumen wall, and
the MCA and BA were clearly observed in all cases. The
cross-sectional imaging findings of patients who were diagnosed
with LAA indicated that the presence of focal arterial wall
thickening was consistent with a plaque on the level of the MCA and
BA stenotic area on MRA images. The plaque appeared as a
crescent-shaped or eccentric thickening surrounding a circular
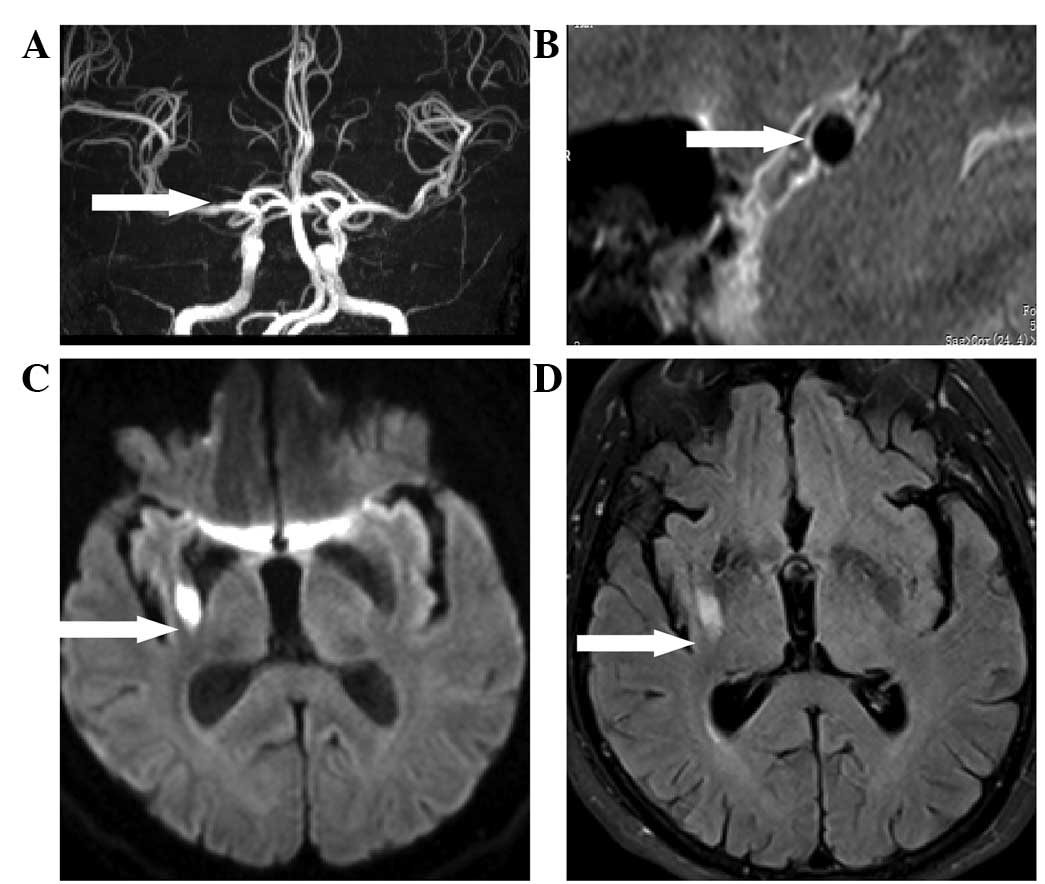
lumen. As shown in Fig. 1, in a
72-year-old male patient, MRA revealed relatively mild stenosis in
the MCA-M1 segment, while HRMRI showed no lumen abnormality. A
high-intensity infarct lesion revealed by diffusion-weighted
imaging (DWI) was present in the lateral striate arterial
territory, indicating that this was a case of the PAD stroke
subtype. HRMRI findings of the lumen elucidated the underlying
mechanisms in which lipohyalinosis or microatheroma may be the
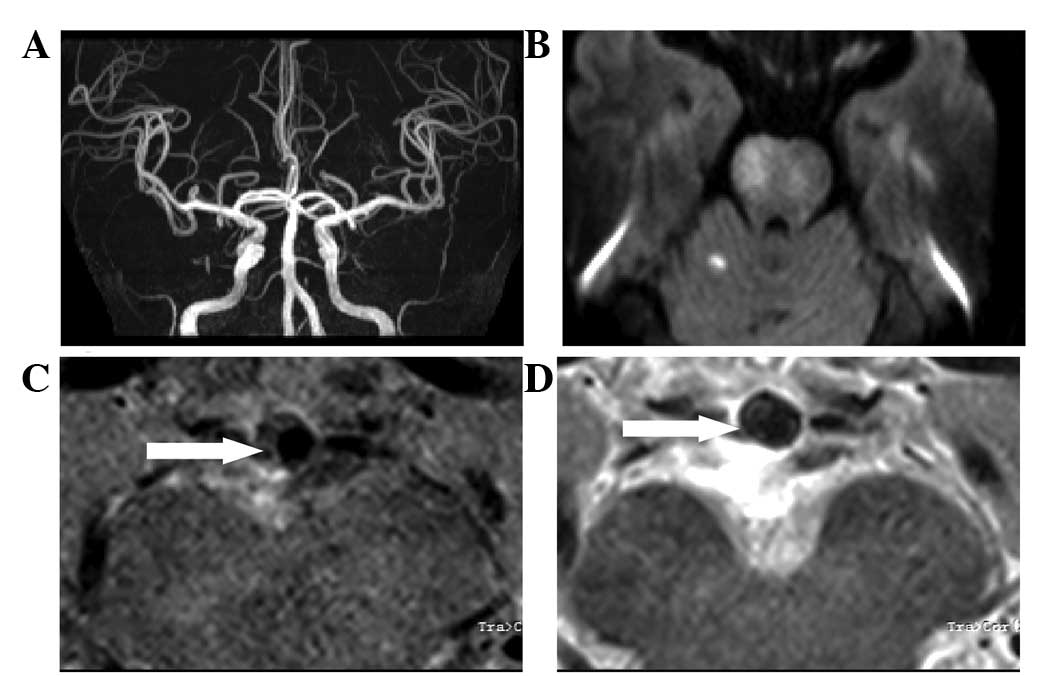
etiology for PAD. Furthermore, in a 60-year-old female patient, MRA
showed relatively mild to moderate stenosis in the BA where an
eccentric plaque located in the lumen wall was shown to occlude the
paramedian pontine arteries by HRMRI. An acute ischemic infarction
was verified by DWI (Fig. 2). This
was indicated to be a typical case, where the infarction was
confined to the territory of a single branch artery or a few
penetrating branches that were occluded by the plaques of their
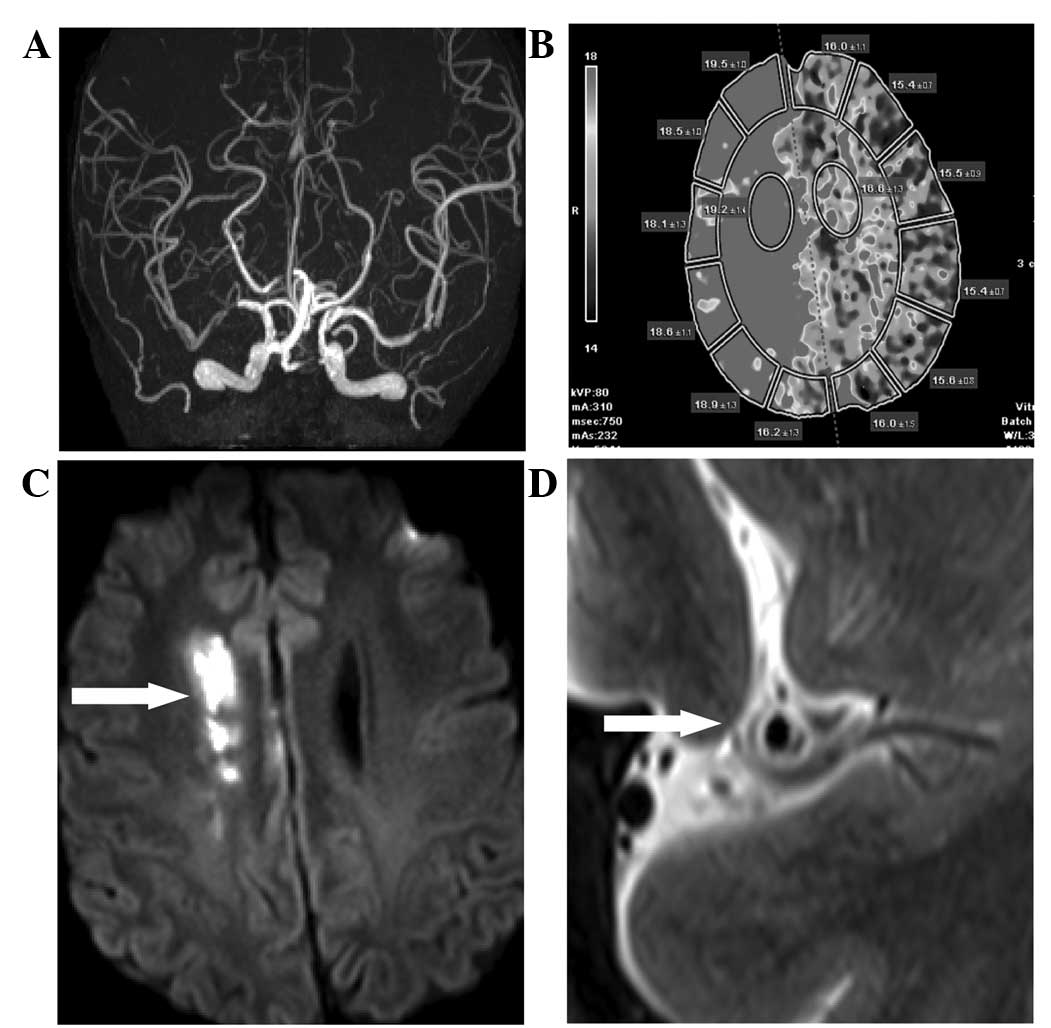
parent arteries. Borderzone infarction and artery-to-artery
embolism occurred in the most severe stenosis and occlusion of the
MCA and BA. Computed tomography perfusion imaging of a 71-year-old
female patient demonstrated relatively low cerebral blood flow and
elevated time-to-peak in a region of the MCA, indicating the
potential mechanism of hypoperfusion (Fig. 3).
Discussion
Intracranial atherosclerotic disease causes ischemic
strokes, and the rates of recurrent vascular ischemic events and
vascular mortalities are very high (11). However, intracranial
atherosclerosis, which affects cerebral arteries such as the BA and
MCA, remains an infradiagnosed and understudied disease (12). The diagnosis of intracranial
stenosis is traditionally dependent on conventional angiography and
several reliable noninvasive diagnostic methods, including
transcranial color-coded duplex sonography, MRA and computed
tomography angiography (13,14).
Cross-sectional HRMRI is a promising technique for imaging carotid
plaques, with a sensitivity of 85% and a specificity of 92% for
identifying soft plaques (necrotic core or hemorrhage) (15–17).
HRMRI has consistently emerged as a potential technique for imaging
atherosclerotic plaques in the intracranial arteries (7). This imaging technique may provide
information about the histopathological nature of the intracranial
atherosclerotic lesion responsible for arterial narrowing (9).
As no underlying cause is found in >30% of stroke
cases, HRMRI detection of intracranial atherosclerotic lesions may
have significant clinical implications (3). Detection of the vessel wall of
arteries such as the MCA and BA may improve the ability to identify
advanced but unrecognized intracranial atherosclerotic disease
(18). In the present study, the
features of the MCA and BA luminal wall in patients who had
suffered cerebral infarction were explored. Recognizing the
characteristics of LAA and PAD may contribute to improved risk
stratification and allow aggressive interventions to be targeted at
patients with plaques that are prone to rupture (19–21).
A previous study of carotid artery plaques has demonstrated a
significant correlation between plaque characteristics identified
by HRMRI and subsequent stroke patterns (19). In the present study, HRMRI clearly
confirmed the presence of a reduced arterial lumen associated with
a focal wall thickening and plaques at the level of MCA and BA
stenosis, suggesting that the HRMRI technique was useful in
determining the etiology of PAD and POPA.
In a comparison of the patients with PAD and those
with LAA, it was observed that patients with PAD had a higher
frequency of hypertension whereas patients with LAA had a higher
frequency of DM. Aging and chronic hypertension are risk factors
for large-artery atherosclerosis (22). Such changes include replacement of
the smooth muscle cells by fibro-hyaline material with thickening
of the wall and narrowing of the vascular lumen. Arteriolosclerosis
may be one of the reasons that the blood supply to the white matter
is altered, and this vascular change may lead to localized ischemic
areas of necrosis and cavitations (14). Previous studies have shown a high
frequency of intracranial stenosis in diabetic Caucasian patients,
an independent association of type II DM to a greater extent of
intracranial LAA, and a significantly higher number of diseased
vessels in diabetic patients compared with that in nondiabetic
patients (13,23). The association between diabetes and
more diffuse and advanced intracranial atherosclerosis is unclear.
Consequently, among the traditional vascular risk factors, diabetes
appears to play a preeminent role in intracranial macroangiopathy
in the Chinese population (24).
Additionally, the presence of intracranial LAA disease contributes
to a poorer outcome for patients with LAA disease, which may be
stratified as very high risk in secondary prevention (25).
In conclusion, stroke patterns of intracranial
atherosclerotic arteries are complicated and mainly include LAA and
PAD. HRMRI has the ability to identify the mechanisms behind
intracranial atherosclerotic ischemic stroke by showing the luminal
wall. It may also provide a useful tool in risk stratification and
the selection of candidates for invasive therapies.
References
|
1
|
Pu Y, Liu L, Wang Y, et al; Chinese
IntraCranial AtheroSclerosis (CICAS) Study Group. Geographic and
sex difference in the distribution of intracranial atherosclerosis
in China. Stroke. 44:2109–2114. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rincon F, Sacco RL, Kranwinkel G, et al:
Incidence and risk factors of intracranial atherosclerotic stroke:
the Northern Manhattan Stroke Study. Cerebrovasc Dis. 28:65–71.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shi MC, Wang SC, Zhou HW, et al:
Compensatory remodeling in symptomatic middle cerebral artery
atherosclerotic stenosis: a high-resolution MRI and microemboli
monitoring study. Neurol Res. 34:153–158. 2012.PubMed/NCBI
|
|
4
|
Naghavi M, Libby P, Falk E, et al: From
vulnerable plaque to vulnerable patient: a call for new definitions
and risk assessment strategies: Part I. Circulation. 108:1664–1672.
2003. View Article : Google Scholar
|
|
5
|
Xu WH, Li ML, Gao S, et al: In vivo
high-resolution MR imaging of symptomatic and asymptomatic middle
cerebral artery atherosclerotic stenosis. Atherosclerosis.
212:507–511. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ma N, Jiang WJ, Lou X, et al: Arterial
remodeling of advanced basilar atherosclerosis: a 3-tesla MRI
study. Neurology. 75:253–258. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bodle JD, Feldmann E, Swartz RH, Rumboldt
Z, Brown T and Turan TN: High-resolution magnetic resonance
imaging: an emerging tool for evaluating intracranial arterial
disease. Stroke. 44:287–292. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Niizuma K, Shimizu H, Takada S and
Tominaga T: Middle cerebral artery plaque imaging using 3-Tesla
high-resolution MRI. J Clin Neurosci. 15:1137–1141. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ryu CW, Jahng GH, Kim EJ, Choi WS and Yang
DM: High resolution wall and lumen MRI of the middle cerebral
arteries at 3 tesla. Cerebrovasc Dis. 27:433–442. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gao S, Wang YJ, Xu AD, et al: Chinese
ischemic stroke subclassification. Front Neurol. 2:62011.
|
|
11
|
Turan TN, Maidan L, Cotsonis G, et al;
Warfarin-Aspirin Symptomatic Intracranial Disease Investigators.
Failure of antithrombotic therapy and risk of stroke in patients
with symptomatic intracranial stenosis. Stroke. 40:505–509. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim JM, Jung KH, Sohn CH, Moon J, Han MH
and Roh JK: Middle cerebral artery plaque and prediction of the
infarction pattern. Arch Neurol. 69:1470–1475. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Arenillas JF and Alvarez-Sabin J: Basic
mechanisms in intracranial large-artery atherosclerosis: advances
and challenges. Cerebrovasc Dis. 20(Suppl 2): 75–83. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yazdani SK, Vorpahl M, Ladich E and
Virmani R: Pathology and vulnerability of atherosclerotic plaque:
identification, treatment options, and individual patient
differences for prevention of stroke. Curr Treat Options Cardiovasc
Med. 12:297–314. 2010. View Article : Google Scholar
|
|
15
|
Cai JM, Hatsukami TS, Ferguson MS, Small
R, Polissar NL and Yuan C: Classification of human carotid
atherosclerotic lesions with in vivo multicontrast magnetic
resonance imaging. Circulation. 106:1368–1373. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kampschulte A, Ferguson MS, Kerwin WS, et
al: Differentiation of intraplaque versus juxtaluminal
hemorrhage/thrombus in advanced human carotid atherosclerotic
lesions by in vivo magnetic resonance imaging. Circulation.
110:3239–3244. 2004. View Article : Google Scholar
|
|
17
|
Larose E, Yeghiazarians Y, Libby P, et al:
Characterization of human atherosclerotic plaques by intravascular
magnetic resonance imaging. Circulation. 112:2324–2331. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lam WW, Wong KS, So NM, Yeung TK and Gao
S: Plaque volume measurement by magnetic resonance imaging as an
index of remodeling of middle cerebral artery: correlation with
transcranial color Doppler and magnetic resonance angiography.
Cerebrovasc Dis. 17:166–169. 2004.
|
|
19
|
Gao T, Zhang Z, Yu W, Zhang Z and Wang Y:
Atherosclerotic carotid vulnerable plaque and subsequent stroke: a
high-resolution MRI study. Cerebrovasc Dis. 27:345–352. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mono ML, Karameshev A, Slotboom J, et al:
Plaque characteristics of asymptomatic carotid stenosis and risk of
stroke. Cerebrovasc Dis. 34:343–350. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lindsay AC, Biasiolli L, Lee JM, et al:
Plaque features associated with increased cerebral infarction after
minor stroke and TIA: a prospective, case-control, 3-T carotid
artery MR imaging study. JACC Cardiovasc Imaging. 5:388–396. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pantoni L and Garcia JH: Pathogenesis of
leukoaraiosis: a review. Stroke. 28:652–659. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Arenillas JF, Molina CA, Chacón P, et al:
High lipoprotein (a), diabetes, and the extent of symptomatic
intracranial atherosclerosis. Neurology. 63:27–32. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Thomas GN, Lin JW, Lam WW, et al:
Albuminuria is a marker of increasing intracranial and extracranial
vascular involvement in Type 2 diabetic Chinese patients.
Diabetologia. 47:1528–1534. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kwan MW, Mak W, Cheung RT and Ho SL:
Ischemic stroke related to intracranial branch atheromatous disease
and comparison with large and small artery diseases. J Neurol Sci.
303:80–84. 2011. View Article : Google Scholar : PubMed/NCBI
|