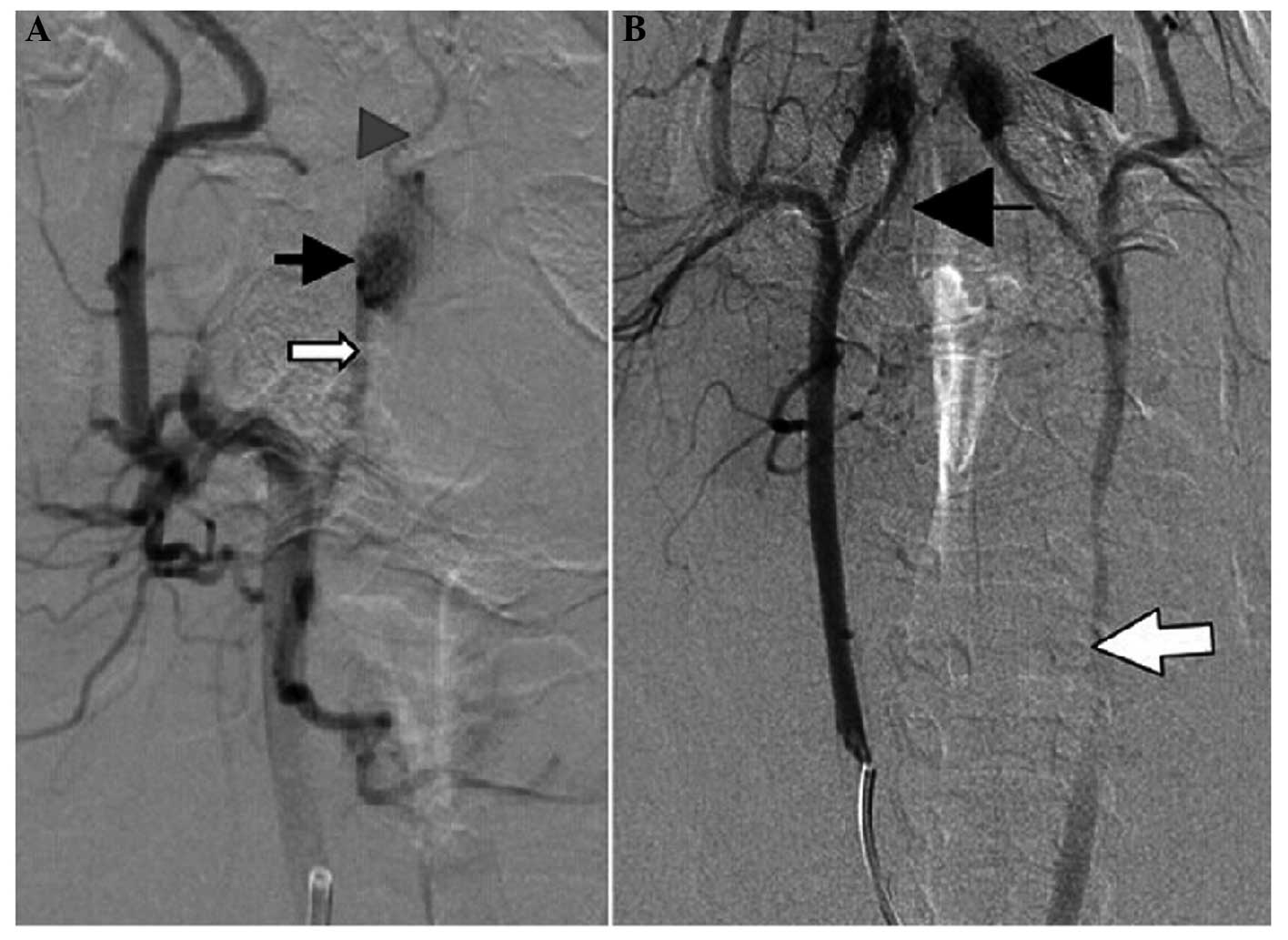
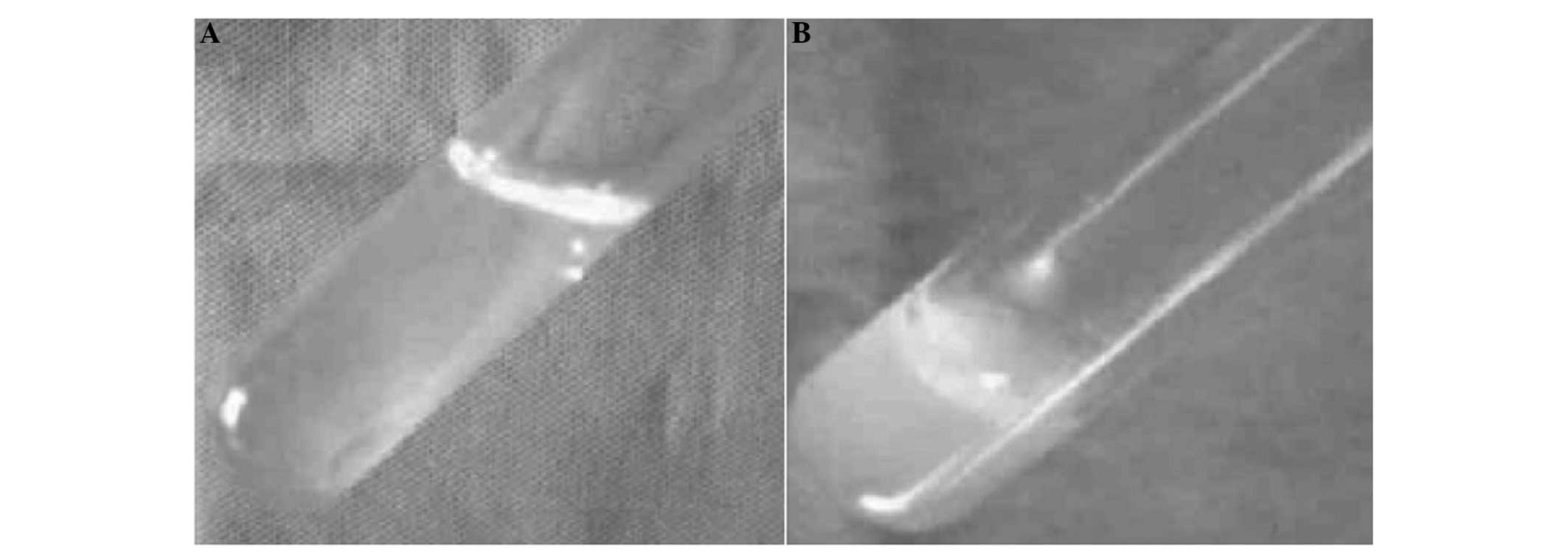
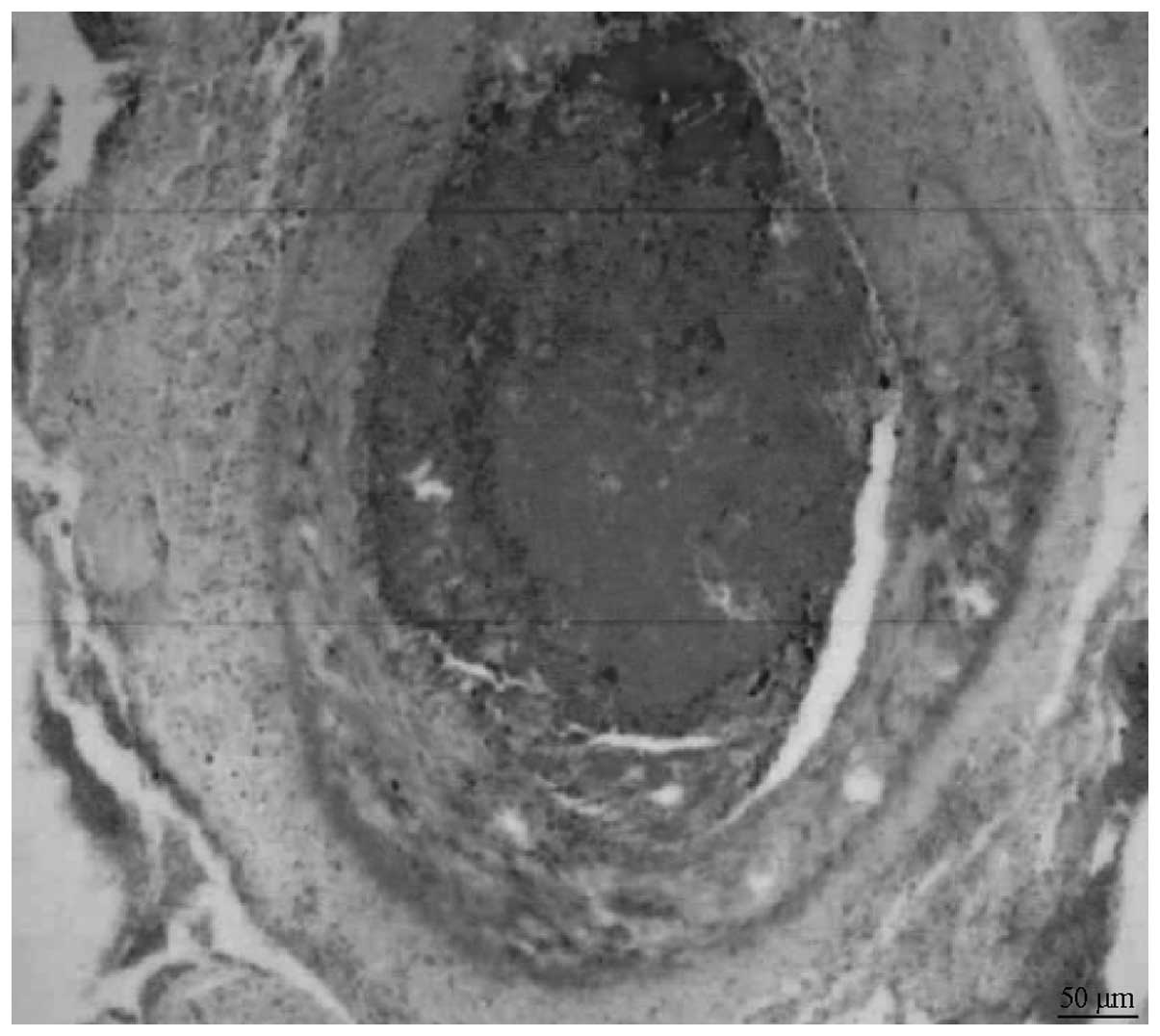
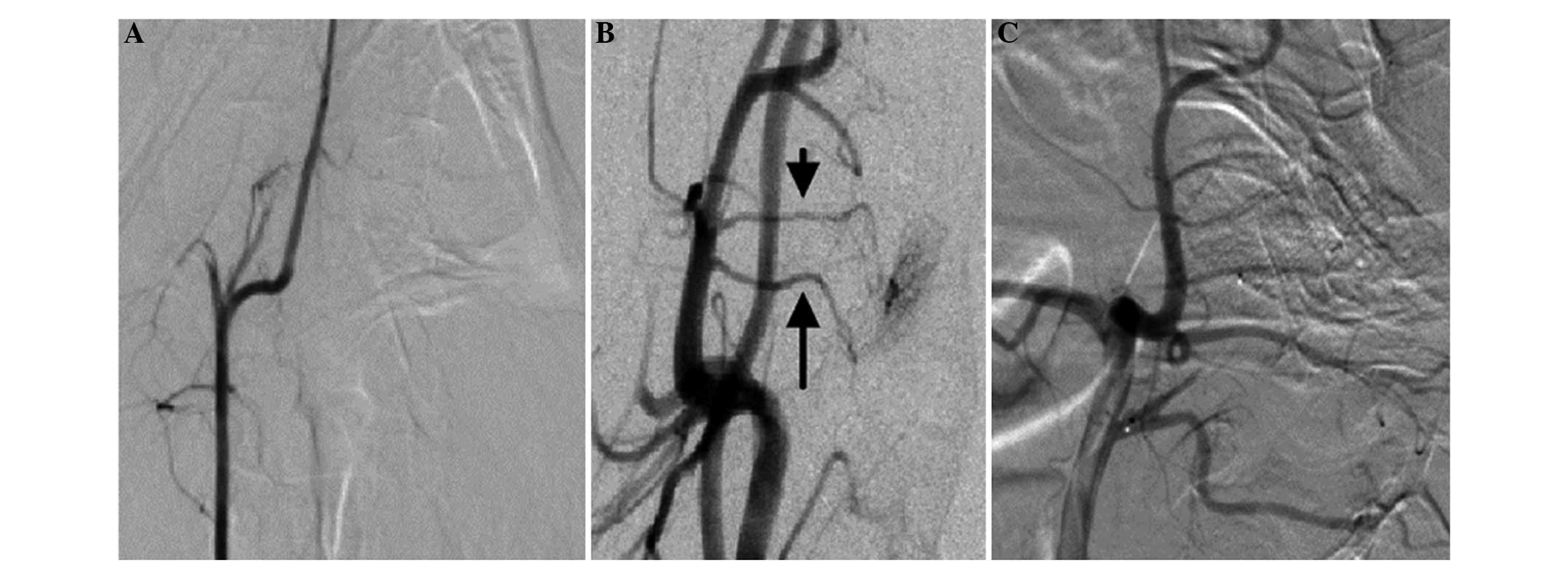
|
1
|
Aoun SG, Bendok BR and Batjer HH: Acute
management of ruptured arteriovenous malformations and dural
arteriovenous fistulas. Neurosurg Clin N Am. 23:87–103. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gross BA and Du R: Natural history of
cerebral arteriovenous malformations: a meta-analysis. J Neurosurg.
118:437–443. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jabbour MN, Elder JB, Samuelson CG, et al:
Aberrant angiogenic characteristics of human brain arteriovenous
malformation endothelial cells. Neurosurgery. 64:139–148. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chenite A, Chaput C, Wang D, et al: Novel
injectable neutral solutions of chitosan form biodegradable gels in
situ. Biomaterials. 21:2155–2161. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
National Research Council (USA) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals, . Guide for the Care and Use of Laboratory Animals. 8th.
National Academies Press; Washington D.C., USA: 2011
|
|
6
|
Lee DH, Wriedt CH, Kaufmann JC, Pelz DM,
Fox AJ and Vinuela F: Evaluation of three embolic agents in pig
rete. AJNR Am J Neuroradiol. 10:773–776. 1989.PubMed/NCBI
|
|
7
|
Massoud TF, Vinters HV, Chao KH, Viñuela F
and Jahan R: Histopathologic characteristics of a chronic
arteriovenous malformation in a swine model: preliminary study.
AJNR Am J Neuroradiol. 21:1268–1276. 2000.PubMed/NCBI
|
|
8
|
da Costa L, Wallace MC, Ter Brugge KG,
OKelly C, Willinsky RA and Tymianski M: The natural history and
predictive features of hemorrhage from brain arteriovenous
malformations. Stroke. 40:100–105. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Choi JH, Mast H, Hartmann A, Marshall RS,
Pile-Spellman J, Mohr JP and Stapf C: Clinical and morphological
determinants of focal neurological deficits in patients with
unruptured brain arteriovenous malformation. J Neurol Sci.
287:126–130. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gabriel RA, Kim H, Sidney S, et al:
Ten-year detection rate of brain arteriovenous malformations in a
large, multiethnic, defined population. Stroke. 41:21–26. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim H, Pawlikowska L, Chen Y, Su H, Yang
GY and Young WL: Brain arteriovenous malformation biology relevant
to hemorrhage and implication for therapeutic development. Stroke.
40:(Suppl). S95–S97. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Colombo F, Cavedon C, Casentini L,
Francescon P, Causin F and Pinna V: Early results of CyberKnife
radiosurgery for arteriovenous malformations. J Neurosurg.
111:807–819. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Natarajan SK, Ghodke B, Britz GW, Born DE
and Sekhar LN: Multimodality treatment of brain arteriovenous
malformations with microsurgery after embolization with onyx:
single-center experience and technical nuances. Neurosurgery.
62:1213–1226. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sorimachi T, Koike T, Takeuchi S, et al:
Embolization of cerebral arteriovenous malformations achieved with
polyvinyl alcohol particles: Angiographic reappearance and
complications. AJNR Am J Neuroradiol. 20:1323–1328. 1999.PubMed/NCBI
|
|
15
|
Lieber BB, Wakhloo AK, Siekmann R and
Gounis MJ: Acute and chronic swine rete arteriovenous malformation
models: effect of ethiodol and glacial acetic acid on penetration,
dispersion and injection force of N-butyl 2-cyanoacrylate. AJNR Am
J Neuroradiol. 26:1707–1714. 2005.PubMed/NCBI
|
|
16
|
Natarajan SK, Born D, Ghodke B, Britz GW
and Sekhar LN: Histopathological changes in brain arteriovenous
malformations after embolization using Onyx or N-butyl
cyanoacrylate. Laboratory investigation. J Neurosurg. 111:105–113.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Starke RM, Komotar RJ, Otten ML, et al:
Adjuvant embolization with N-butyl cyanoacrylate in the treatment
of cerebral arteriovenous malformations: outcomes, complications
and predictors of neurologic deficits. Stroke. 40:2783–2790. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Strozyk D, Nogueira RG and Lavine SD:
Endovascular treatment of intracranial arteriovenous malformation.
Neurosurg Clin N Am. 20:399–418. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dudeck O, Jordan O, Hoffmann KT, et al:
Intrinsically radiopaque iodine-containing polyvinyl alcohol as a
liquid embolic agent: evaluation in experimental wide-necked
aneurysms. J Neurosurg. 104:290–297. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Panagiotopoulos V, Gizewski E, Asgari S,
Regel J, Forsting M and Wanke I: Embolization of intracranial
arteriovenous malformations with ethylene-vinyl alcohol copolymer
(Onyx). AJNR Am J Neuroadiol. 30:99–106. 2009. View Article : Google Scholar
|
|
21
|
Murayama Y, Viñuela F, Ulhoa A, et al:
Nonadhesive liquid embolic agent for cerebral arteriovenous
malformations: preliminary histopathological studies in swine rete
mirabile. Neurosurg. 1998.43:1164–1175. View Article : Google Scholar
|
|
22
|
Chenite A, Chaput C, Wang D, et al: Novel
injectable neutral solutions of chitosan form biodegradable gels in
situ. Biomaterials. 21:2155–2161. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Casettari L, Cespi M, Palmieri GF and
Bonacucina G: Characterization of the interaction between chitosan
and inorganic sodium phosphates by means of rheological and optical
microscopy studies. Carbohydr Polym. 91:597–602. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hsiao MH, Larsson M, Larsson A, Evenbratt
H, Chen YY, Chen YY and Liu DM: Design and characterization of a
novel amphiphilic chitosan nanocapsule-based thermo-gelling biogel
with sustained in vivo release of the hydrophilic anti-epilepsy
drug ethosuximide. J Control Release. 161:942–948. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang Y, Xu N, Luo Q, et al: In vivo
assessment of chitosan/β-glycerophosphate as a new liquid embolic
agent. Interv Neuroradiol. 17:87–92. 2011.PubMed/NCBI
|
|
26
|
Lajud SA, Han Z, Chi FL, et al: A
regulated delivery system for inner ear drug application. J Control
Release. 166:268–276. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Khodaverdi E, Tafaghodi M, Ganji F, Abnoos
K and Naghizadeh H: In vitro insulin release from thermosensitive
chitosan hydrogel. AAPS PharmSciTech. 13:460–466. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu Z, Wang H, Wang Y, et al: The
influence of chitosan hydrogel on stem cell engraftment, survival
and homing in the ischemic myocardial microenvironment.
Biomaterials. 33:3093–3106. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Alinaghi A, Rouini MR, Johari Daha F and
Moghimi HR: Hydrogel-embeded vesicles, as a novel approach for
prolonged release and delivery of liposome, in vitro and in vivo. J
Liposome Res. 23:235–243. 2013. View Article : Google Scholar : PubMed/NCBI
|