Introduction
Previous epidemiological studies have demonstrated
that there is a dose-response association between alcohol use and
the risk of several diseases and mortality. It has also been
reported that light to moderate alcohol consumption has beneficial
effects on many aspects of health, particularly on cardiovascular
outcomes (1,2). On the contrary, heavy alcohol
consumption is considered to be responsible for hundreds of
thousands of deaths annually worldwide. It is also known to be the
cause of a number of diseases and is a precursor to injury and
violence, and often leads to alcohol use disorders (AUDs) (alcohol
abuse and dependence) (3,4).
Oxidative stress results from an imbalance between
oxidants and antioxidants in favor of the oxidants, leading to
reversible redox modification of molecules involved in cellular
signaling pathways, and damage to biological molecules (lipids,
proteins and DNA) (5). Oxidative
stress is responsible for the development of several pathological
conditions, and it can be induced by numerous factors, including
alcohol (6). Excessive, chronic
alcohol consumption may lead to an impaired redox status, through
both the increased production of reactive oxygen species (ROS) and
impaired antioxidant defense mechanisms (7), and is associated with the pathogenesis
of alcohol-related diseases, such as alcoholic liver disease,
alcoholic cardiomyopathy and cancer (7,8). A
number of studies have demonstrated that acute exercise can
increase oxidative stress in humans (9–13).
Exercise-induced oxidative stress activates signaling pathways that
increase the expression of antioxidants and are also responsible
for the process of exercise-induced adaptation (11,13).
This adaptation is influenced by various factors, including
training volume, intensity, frequency and the mode of exercise
(13).
Although it is not yet well established, exercise is
a promising non-pharmaceutical intervention which may be used to
reduce alcohol intake or even to help heavy drinkers and
individuals with AUDs to stop excessive alcohol intake (14–16).
Over the past 40 years, a small number of studies have investigated
the effects of exercise on alcohol intake in individuals with AUDs
(15,16); however, only one recent study
investigated the physiological responses to acute exercise in
alcoholic patients. Jamurtas et al (17) examined the effects of low-intensity
exercise on the urge to drink alcohol, the levels of β-endorphin
(β-E) and lactic acid, as well as the hematological parameters
[complete blood count (CBC)]. Their results revealed that the
pre-exercise levels of β-E were significantly lower in alcoholic
patients, whereas exercise led to significantly (p<0.001)
increased β-Ε levels only in alcoholic patients. Lactic acid and
hematological parameters assessed through CBC did not differ
between the 2 groups; however, exercise led to significantly
increased levels of lactic acid, red blood cells, hemoglobin and
hematocrit in both groups. Moreover, there was a 17% decrease in
the urge to consume alcohol in the alcoholic patients. The results
from this study indicate that a bout of low-intensity exercise
affects endogenous opioids in alcoholic patients. Greater increases
in β-Ε levels as a response to exercise of a different type and/or
higher intensity has been observed in other specific populations
(18). Since chronic excessive
exposure to alcohol leads to decreased β-Ε production, which may be
responsible for negative reinforcement (19), a greater increase in β-Ε levels after
exercise may lead to a significant reduction in the urge to consume
alcohol. Therefore, exercise may be used as a healthy alternative
to alcohol intake. The effects of acute and chronic exercise of
different intensities and types on the urge to consume alcohol and
on the health status in individuals with AUDs, as well as the
physiological mechanisms involved should thus be investigated.
Since there is gap in the literature on the acute
effects of exercise on the metabolism and redox status in
individuals with AUDs, the aim of the present study was to
investigate the effects of acute exercise of moderate intensity on
the indices of liver function and redox status in heavy drinkers.
This is a preliminary step in describing the responses to exercise
of individuals who consume alcholol excessively, in order to
develop exercise training programmes that aim to halt alcohol abuse
and improve health.
Subjects and methods
Subjects
A total of 17 heavy drinkers [age, 31.6±3.2 years;
body mass index (BMI), 27.4±0.8 kg/m2; experimental
group (EG)] and 17 controls that did not exceed moderate alcohol
consumption [age, 33.5±1.3 years; BMI, 26.1±1.4 kg/m2;
control group (CG)] participated in this study. All subjects were
sedentary, and the level of physical activity was assessed with the
International Physical Activity Questionnaire. The subjects in the
2 groups were also matched in terms of the number of cigarettes
smoked per day.
Individuals with alcohol consumption levels
exceeding the limits set by the National Institute on Alcohol Abuse
and Alcoholism (>14 drinks per week or >4 drinks per occasion
for men, >7 drinks per week or >3 drinks per occasion for
women) were identified as heavy drinkers. According to the National
Institute on Alcohol Abuse and Alcoholism, exceeding these drinking
limits significantly increases the risk of developing an AUD
(4). Moreover, the Alcohol Use
Disorders Identification Test (AUDIT) (20) was also used in order to identify
individuals with AUDs. An AUDIT score between 8 and 15 indicates
hazardous alcohol drinking, a score between 16 and 19 indicates
harmful alcohol drinking, and a score of 20 or above indicates
alcohol dependence (21). A total of
6 heavy drinkers had a score between 8 and 15, 5 heavy drinkers had
a score between 16 and 19, and 6 heavy drinkers had a score of 20
or above (total AUDIT score, 17.65±1.25).
All subjects were informed about the study protocol,
and the associated risks and benefits, and they signed an informed
consent form prior to participation. Before proceeding with the
other measurements, the medical history of the participants was
reviewed and a resting electrocardiogram was performed in order to
detect any heart abnormalities and contraindications to exercise.
The procedures were in accordance with the 1975 Declaration of
Helsinki, and ethics approval was obtained from the University of
Thessaly Review Board (Larissa, Greece). Exclusion criteria
included serious health issues, physical disabilities, or any other
medical condition that contraindicated safe participation in
exercise (e.g., a history of drug abuse other than alcohol or being
over 60 years of age).
Experimental design
Subjects reported to our laboratory following an
overnight fast, which included abstaining from both alcohol and
smoking. The anthropometric and physiological characteristics of
the study subjects were measured prior to exercise, and thereafter
the subjects underwent one trial of acute exercise of moderate
intensity (50–60% of the heart rate reserve) for 30 min on a cycle
ergometer (Monark Ergomedic 874E; Monark Exercise AB, Vansbro,
Sweden). The heart rate (HR) was monitored during exercise by
short-range telemetry (Polar RC3 GPS HR; Polar Electro Oy, Kempele,
Finland). Blood samples were collected prior to and immediately
following exercise for later determination of the indices of liver
function [γ-glutamyl transferase (γ-GT), aspartate aminotransferase
(AST) and alanine aminotransferase (ALT) levels] and blood redox
status [reduced glutathione (GSH), catalase activity, uric acid
(UA), total antioxidant capacity (TAC), total bilirubin,
thiobarbituric acid-reactive substances (TBARS) and protein
carbonyl (PC) levels].
Blood sampling and handling
Blood samples (15 ml) were drawn from a vein in the
forearm and, in order to obtain plasma, a portion of the blood was
placed in separate tubes, mixed with EDTA (20 µl/ml of blood) and
centrifuged at 1,370 × g for 10 min at 4°C. The supernatant was
aliquoted and stored at −80°C for later determination of the TAC,
TBARS and PC levels. For red blood cell lysate preparation, packed
erythrocytes were diluted with distilled water (1:1 v/v), vortexed
vigorously, and centrifuged at 4,000 × g for 15 min at 4°C. The
supernatant was also aliquoted and stored at −80°C for later
analysis of catalase activity and GSH levels. Finally, more blood
was collected in separate tubes containing clot activator, left at
room temperature for 20 min to clot, and centrifuged at 1,370 × g
for 10 min at 4°C in order to obtain serum. The supernatant was
aliquoted and stored at −80°C for later determination of the UA,
total bilirubin, γ-GT, AST and ALT levels.
Methods
Each variable was analyzed in duplicate on the same
day. Samples went through only one freeze-thaw cycle.
Assays in plasma
TAC determination was based on the scavenging of
1,1-diphenyl-2-picrylhydrazyl (DPPH), as previously described in
the study by Janaszewska and Bartosz (22). Plasma (20 µl) was added to 480 µl of
10 mM sodium potassium phosphate (pH 7.4) and 500 µl of 0.1 mM DPPH
free radical. The samples were incubated in the dark for 30 min at
room temperature. Subsequently, the samples were centrifuged at
20,000 × g for 3 min at 25°C. The absorbance of the samples was
read at 520 nm. TAC is presented as mM of DPPH reduced to
1,1-diphenyl-2-picrylhydrazine (DPPH:H) by the antioxidants of
plasma.
The TBARS levels were measured as previously
described in the study by Keles et al (23). For TBARS determination, 100 µl of
plasma were added to 500 µl of 35% TCA and 500 µl of Tris-HCl (200
mM, pH 7.4). The samples were incubated for 10 min at room
temperature. Subsequently, 1 ml of 2 M Na2SO4
and 55 mM thiobarbituric acid solution were added, and the samples
were incubated for 45 min at 95°C. The samples were then cooled on
ice for 5 min and 1 ml of 70% TCA was then added. The samples were
vortexed and centrifuged at 15,000 × g for 3 min at 25°C. The
absorbance of the supernatant was read at 530 nm. A baseline
absorbance was taken into account by running a blank along with all
samples during the measurement. The calculation of the TBARS
concentration was based on the molar extinction coefficient of
malondialdehyde, as previously described (23).
The PC levels were measured as previously described
in the study by Patsoukis et al (24). For the determination of PC levels, 50
µl of 20% TCA were added to 50 µl of plasma. The samples were
incubated in an ice bath for 15 min and centrifuged at 15,000 × g
for 5 min at 4°C. Subsequently, the supernatant was discarded, and
500 µl of 10 mM 2,4-dinitrophenylhydrazine (in 2.5 N HCl) for the
sample or 500 µl of 2.5 N HCl for the blank was added to the
pellet. The samples were incubated in the dark at room temperature
for 1 h, with intermittent vortexing every 15 min. The samples were
then centrifuged at 15,000 × g for 5 min at 4°C. The supernatant
was discarded, and 1 ml of 10% TCA was added, vortexed and
centrifuged at 15,000 × g for 5 min at 4°C. The supernatant was
discarded once again, and 1 ml of ethanol:ethyl acetate (1:1 v/v)
was added, vortexed and centrifuged at 15,000 × g for 5 min at 4°C.
This washing step was repeated twice. Finally, the supernatant was
discarded, and 1 ml of 5 M urea (pH 2.3) was added, vortexed and
incubated for 15 min at 37°C. The samples were centrifuged at
15,000 × g for 3 min at 4°C, and the absorbance was read at 375 nm.
The calculation of the protein carbonyl concentration was based on
the molar extinction coefficient of dinitrophenylhydrazine, as
previoulsy described (24).
Assays in red blood cell lysate
Catalase activity was determined as previously
described in the study by Aebi (25). For the determination of catalase
activity, 4 µl of erythrocyte lysate (diluted 1:1) were added to
2991 µl of 67 mM sodium potassium phosphate (pH 7.4). The samples
were incubated at 37°C for 10 min. Subsequently, 5 µl of 30%
hydrogen peroxide were added to the samples, and the change in
absorbance was immediately read at 240 nm for 2 min. The
calculation of catalase activity was based on the molar extinction
coefficient of H2O2, as previously described
(25).
The GSH levels were determined as previously
described by Reddy et al (26). A total of 20 µl of erythrocyte lysate
treated with 5% TCA was mixed with 660 µl of 67 mM sodium potassium
phosphate (pH 8.0) and 330 µl of 1 mM 5,5′-dithiobis-2
nitrobenzoate. The samples were then vortexed and incubated in the
dark at room temperature for 45 min. The absorbance of the samples
was read at 412 nm, as previously described (26). The GSH concentration was calculated
by calibration curves constructed using commercial standards.
Hemoglobin in red blood cell lysate was determined
using a commercially available kit (Dutch Diagnostics BV, Zutphen,
The Netherlands), in order to estimate the final levels of GSH and
catalase activity. For the determination of hemoglobin, 10 µl of
erythrocyte lysate treated with 5% TCA were mixed with 2500 µl of
working reagent (pH 7.3; diluted 1:10). The samples were
immediately vortexed and left for at least 3 min at 25°C.
Assays in serum
The UA, total bilirubin, γ-GT, AST and ALT levels
were measured on a Clinical Chemistry Analyzer Z 1145 (Zafiropoulos
Diagnostica, Athens, Greece) using commercially available kits
(Zafiropoulos Diagnostica). For the determination of UA levels, 6
µl of serum were added to 600 µl of working reagent. The samples
were incubated for 1 min at 37°C and then their absorbance was read
at 340 nm. For the determination of γ-GT levels, 70 µl of serum
were added to 600 µl of working reagent. The samples were incubated
for 1 min at 37°C and then their absorbance was read at 405 nm. For
the determination of AST levels, 70 µl of serum were added to 600
µl of working reagent. The samples were incubated for 1 min at 37°C
and their absorbance was then read at 340 nm. For the determination
of ALT levels, 70 µl of serum were added to 600 µl of working
reagent. The samples were incubated for 1 min at 37°C and their
absorbance was then read at 340 nm.
The intra-assay coefficients of the variation for
GSH, catalase, TAC, UA, total bilirubin, TBARS, PC, γ-GT, AST and
ALT levels were 2.21, 3.38, 2.44, 2.75, 3.81, 2.10, 1.53, 1.15,
1.76 and 2.19, respectively.
Statistical analysis
Two-way (time × group) repeated measures ANOVA was
conducted to examine the differences in the indices of liver
function and blood redox status. If a significant interaction was
noted, pairwise comparisons were performed through simple contrasts
and simple main effects analysis using the Bonferroni correction.
Moreover, an independent t-test was conducted to examine whether
there were any differences between the baseline values of the
anthropometric and physiological parameters. A p-value <0.05 was
considered to indicate a statistically significant difference. The
statistical programme used was SPSS version 18.0 (SPSS, Inc.,
Chicago, IL, USA). Data are presented as the means ± standard error
of the mean.
Results
Anthropometric, physiological and
other characteristics
The waist-to-hip ratio (WHR) and systolic blood
pressure (SBP) values were significantly higher (p<0.05) in the
EG compared to the CG (WHR, 0.90±0.03 vs. 0.82±0.02; SBP, 122.1±2,7
vs. 111.7±3.8) and the AUDIT score was also much higher (p<0.05)
in the EG than the CG (17.7±1.3 vs. 2.6±0.4; Table I). The other anthropometric and
physiological characteristics did not differ significantly between
the 2 groups (Table I).
 | Table I.Anthropometric, physiological and
other characteristics of the subjects (mean ± SE). |
Table I.
Anthropometric, physiological and
other characteristics of the subjects (mean ± SE).
| Variables | EG | CG |
|---|
| Age, years |
31.6±3.2 |
33.5±1.3 |
| Height, cm | 175.1±1.9 | 170.3±2.1 |
| Weight, kg |
84.3±3.4 |
76.4±4.9 |
| BMI,
kg/m2 |
27.4±0.8 |
26.1±1.4 |
| WHR |
0.90±0.03a |
0.82±0.02 |
| Systolic BP, mm
Hg |
122.1±2.7a | 111.7±3.8 |
| Diastolic BP, mm
Hg | 80.4±1.9 |
77.4±2.4 |
| Rest HR | 66.2±1.7 |
65.3±1.4 |
| Exercise HR | 128–139 |
126–138 |
| IPAQ | 1322.9±386.9 | 1340.7±139.0 |
| AUDIT score |
17.7±1.3a |
2.6±0.4 |
| Cigarettes/day | 10.7±2.0 |
10.9±3.5 |
Liver function variables
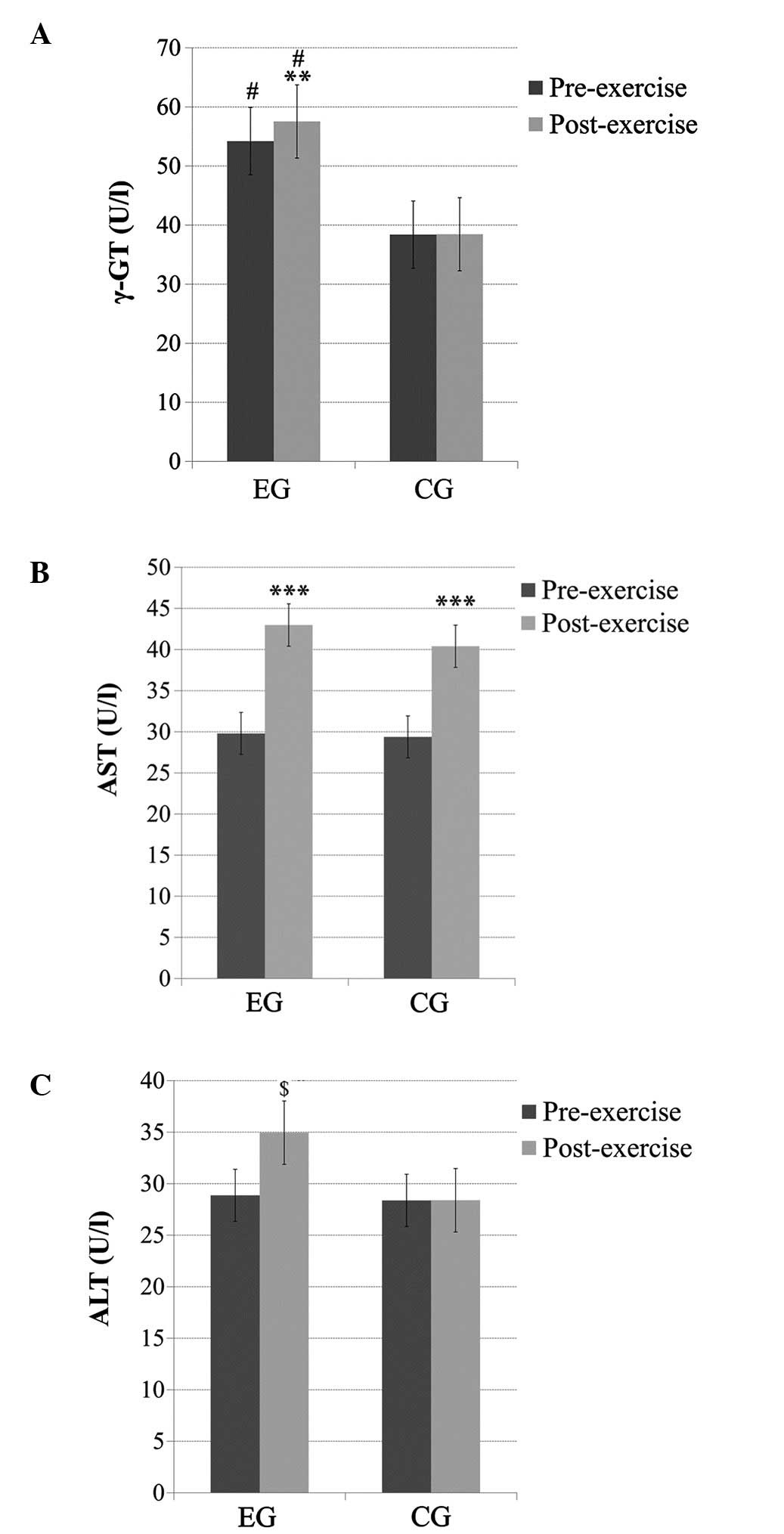
γ-GT
There was a significant main effect of time (pre-
and post-exercise; p<0.05), a significant main effect of group
(p<0.05) and a time × group interaction (p<0.05) for the γ-GT
levels. The subjects in the EG had significantly higher (p<0.05)
baseline γ-GT levels compared to subjects in the CG. Exercise thus
resulted in significantly higher γ-GT levels (p<0.01) only in
the EG (Fig. 1A).
AST
No significant main effect of group or time × group
interaction was observed for the AST levels; however, there was a
significant main effect of time (p<0.001). Pairwise comparisons
indicated that the AST levels significantly increased (p<0.001)
post-exercise in both groups (Fig.
1B).
ALT
No significant main effect of group was observed for
the ALT levels; however, a significant main effect of time
(p<0.05) and a time × group interaction (p<0.05) was
observed. Pairwise comparisons indicated that the ALT levels
increased significantly (p<0.01) post-exercise only in the EG
(Fig. 1C).
Redox status variables
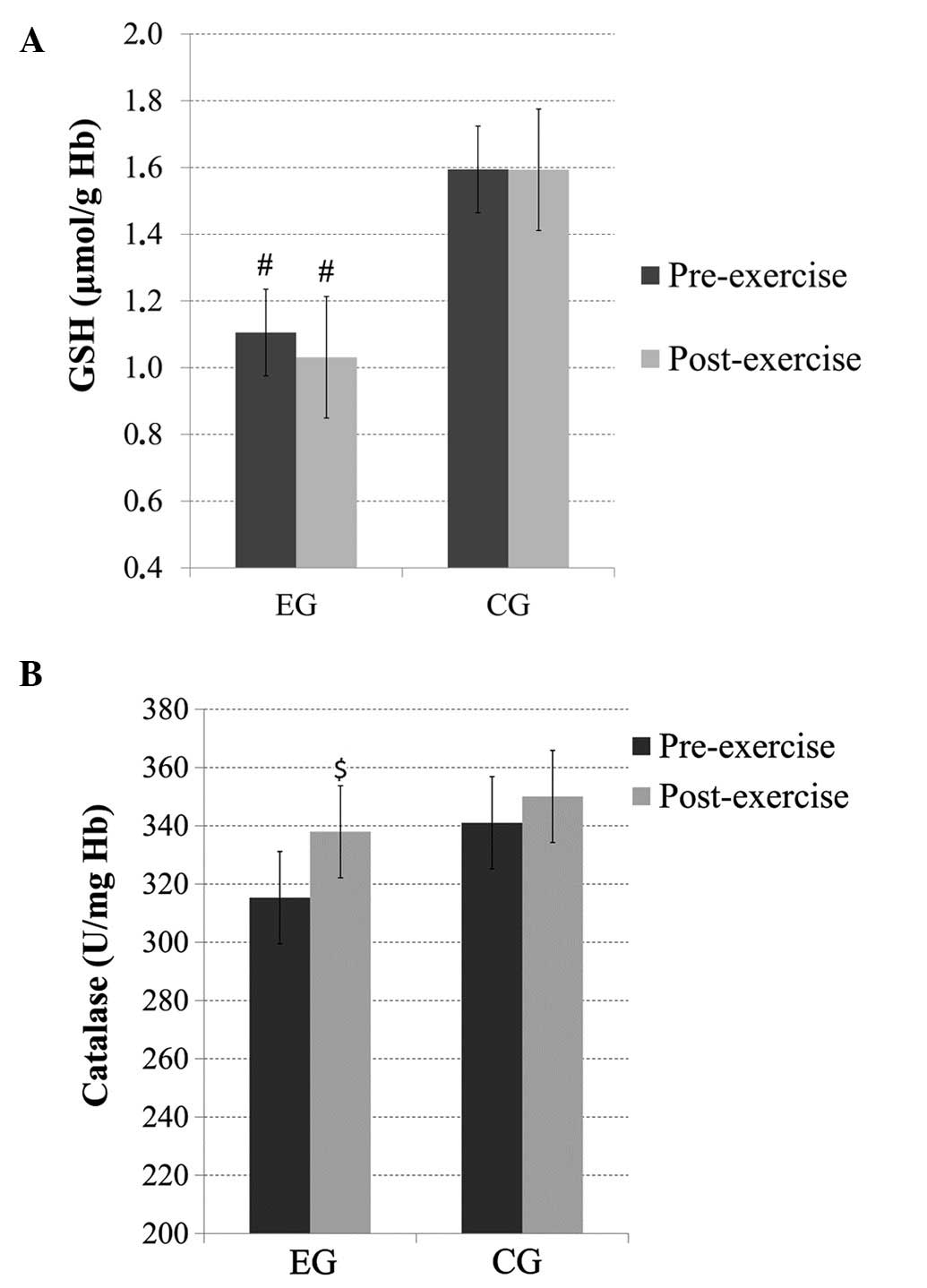
GSH
No significant main effect of time or time × group
interaction was observed for the GSH levels; however, there was a
significant main effect of group (p<0.05), with subjects in the
EG exhibiting significantly lower GSH levels than the subjects in
the CG before and after exercise (Fig.
2A).
Catalase
There was no significant main effect of group or
time × group interaction for catalase; however, there was a trend
(p=0.07) for a main effect of time. Pairwise comparisons revealed
increased post-exercise catalase levels in the EG (Fig. 2B).
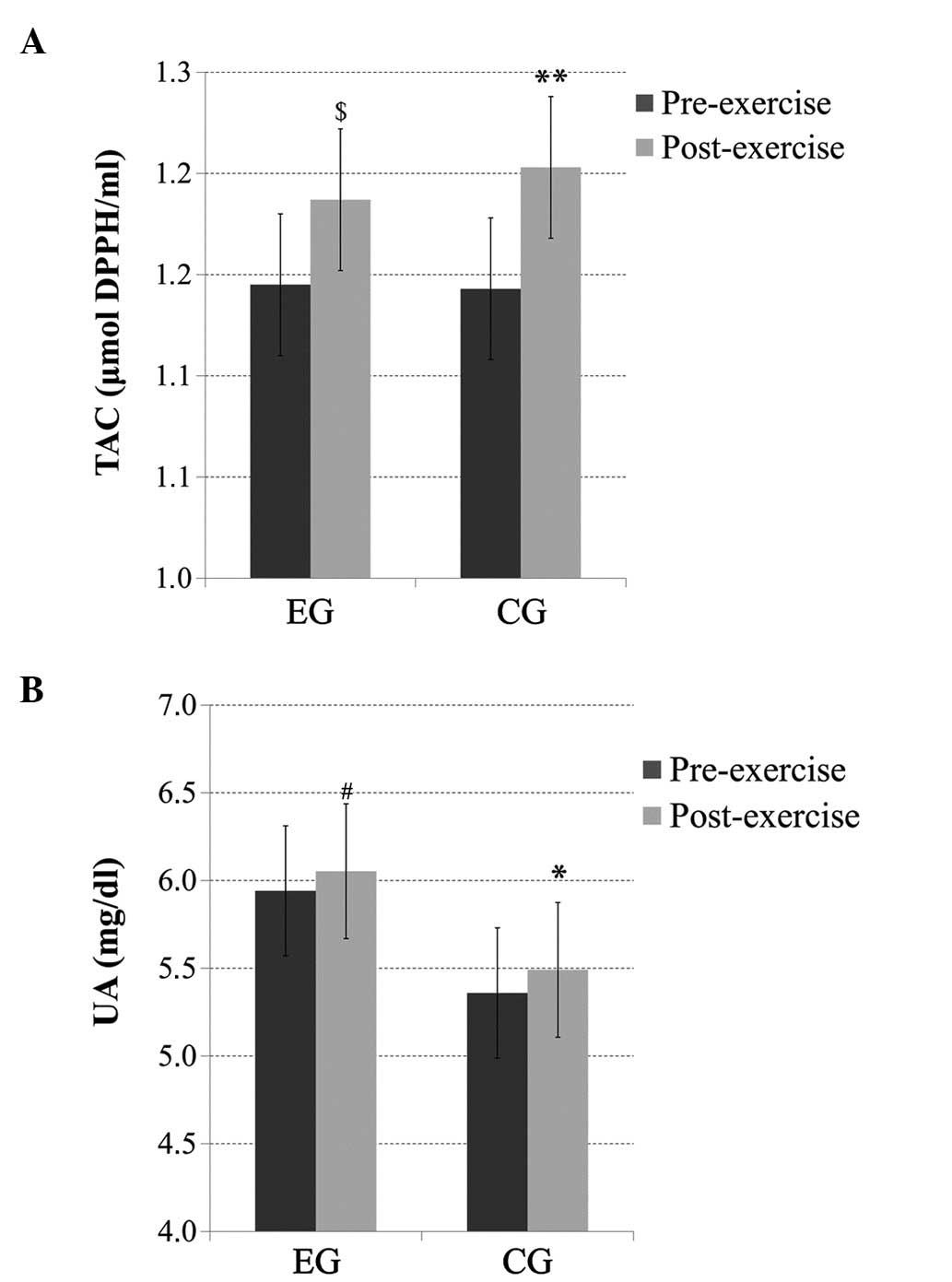
TAC
There was no significant main effect of group or
time × group interaction for the TAC levels; however, there was a
significant main effect of time, with significantly increased
(p<0.01) post-exercise TAC levels in the CG and, similarly, a
trend for increased (p=0.06) post-exercise TAC levels in the EG
(Fig. 3A).
UA
There was no significant main effect of group or
time × group interaction for the UA levels; however, there was a
significant main effect of time (p<0.05), with significantly
increased (p<0.05) post-exercise UA levels in the CG and a trend
for increased (p=0.08) post-exercise UA levels in the EG (Fig. 3B).
Total bilirubin
No significant main effect of time, group or time ×
group interaction was detected for the total bilirubin levels (data
not shown).
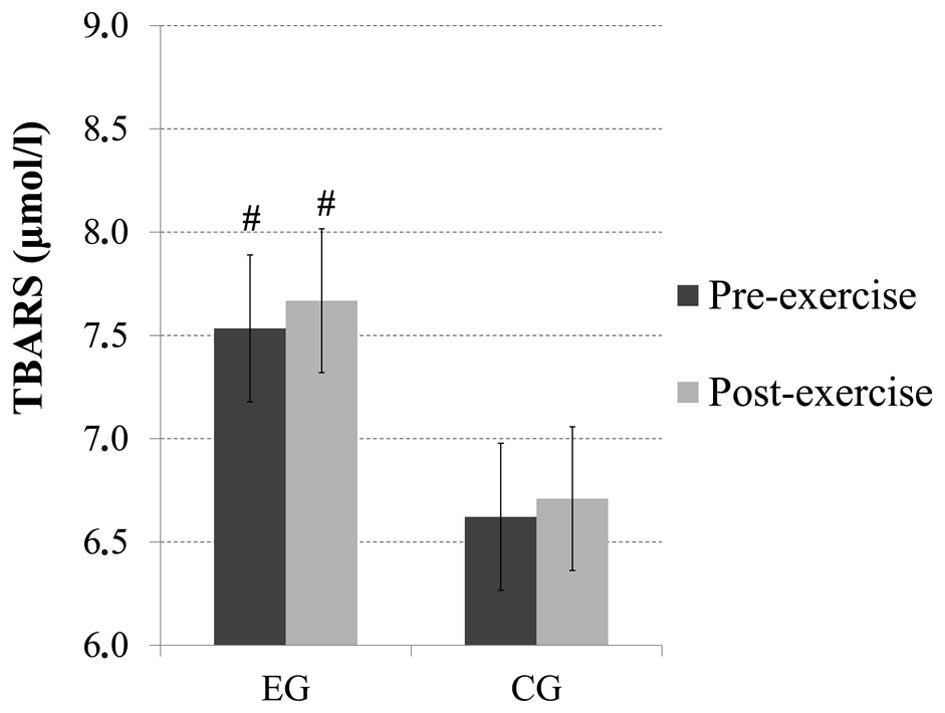
TBARS
No significant main effect of time or time × group
interaction was observed for the TBARS levels; however, there was a
main effect of group (p=0.06). Pairwise comparisons revealed that
there were increased baseline (p=0.08) and post-exercise (p=0.06)
TBARS levels in the subjects from the EG compared with the subjects
from the CG (Fig. 4).
PC
There was no significant main effect of time, group
or time × group interaction observed for the PC levels (data not
shown).
Discussion
Liver function
To the best of our knowledge, this is the first
study to investigate the effects of acute exercise on liver
function and blood redox status in heavy drinkers. It has been well
documented that chronic excessive exposure to alcohol can lead to
liver inflammation, which may eventually impair liver function. A
hypothesis of this study was that heavy drinkers would exhibit
higher baseline levels of liver enzymes (mainly of γ-GT and ALT)
and that exercise would lead to a further increase (mainly in
AST).
γ-GT is a common index used in medicine for the
detection of liver malfunction or issues with the bile ducts.
Excessive alcohol consumption can also result in increased γ-GT
levels. The results of the present study demonstrated that there
were increased baseline levels of γ-GT in heavy drinkers compared
to the controls. It is most likely that this finding is related to
heavy drinking. Another factor that could, independently of alcohol
intake, lead to increased γ-GT levels is cigarette smoking
(27); however, heavy drinkers and
controls had the same smoking habits and therefore most probably
alcohol intake influenced the γ-GT levels.
AST and ALT are commonly used to detect inflammation
and viral infections of the liver. AST is present in the liver and
other tissues, including the skeletal muscles, and increased levels
of AST indicate muscular inflammation. ALT is mainly found in the
liver and in smaller amounts in other tissues, such as the kidneys
and skeletal muscle, and thus increased levels of ALT are mainly
attributable to liver inflammation (28). In this study, the baseline levels of
AST and ALT were within the normal limits for men and women in both
groups. It has previously been suggested that an increased BMI is
an important contributing factor of increased liver enzyme levels
in men (29); however, no
significant difference in BMI between the heavy drinkers and
controls was noted in the present study. On the other hand, a
significantly increased WHR, which is indicative of increased
visceral fat levels, was evident in heavy drinkers. We believe that
differences in physiological (e.g., BMI and SBP) and biochemical
parameters (e.g., γ-GT and ALT) that were found at the baseline are
the result of heavy drinking.
Concerning liver enzyme responses to exercise, the
γ-GT and ALT levels increased significantly only in the EG, whereas
the AST levels increased in both groups after exercise. It is well
known that exercise can result in transient increases in liver
enzymes in healthy individuals, depending on the intensity,
duration and type of exercise performed (30,31).
Thus, we hypothesized that heavy drinkers may be more prone to
increased liver inflammation following moderate-intensity exercise
due to increased oxidative stress.
Finally, the total bilirubin levels did not differ
between groups and did not change after exercise. An increase in
bilirubin levels in individuals at rest can indicate a number of
liver function problems, whereas in healthy individuals it can be
detected following intense exercise, due to hemolysis. Therefore,
it is assumed that moderate-intensity exercise does not cause
hemolysis in heavy drinkers, despite decreased blood GSH levels
that could render erythrocytes more susceptible to lipid
peroxidation and consequently to hemolysis (32).
Taken together, the findings from the present study
indicate that heavy drinking may have resulted in liver
inflammation, which was enhanced by acute exercise. Although acute
exercise can trigger increased liver inflammation in heavy
drinkers, exercise training could lead to decreased levels of liver
enzymes. Previous studies on clinical populations have demonstrated
that exercise can ameliorate metabolic abnormalities. It has been
found that aerobic exercise training may decrease liver enzyme
levels in patients with liver diseases that are not caused by
alcohol (33,34). Training studies to examine the
chronic effects of exercise on liver enzymes in individuals with
AUDs are thus warranted.
Redox status
Excessive alcohol consumption may cause oxidative
stress by both increasing ROS production and decreasing antioxidant
defense mechanisms (7,35), and oxidative stress is also thought
to be involved in the pathogenesis of alcohol-related diseases
(7,8). On the other hand, acute exercise
results in increased production of ROS and also enhances
antioxidant defense mechanisms (11,13).
Based on these facts, we hypothesized that heavy drinkers were more
susceptible to oxidative stress compared to individuals who do not
exceed moderate alcohol consumption, and that exercise leads to
changes in indices of blood redox status in both groups, with heavy
drinkers experiencing greater increases in oxidative stress after
exercise. The results of this study indicate differences in redox
status between heavy drinkers and healthy control, with the former
exhibiting lower GSH and higher TBARS, an index of lipid
peroxidation.
GSH is a major cellular thiol antioxidant with many
functions that protect cells against oxidative stress and its
consequences. Excessive exposure to alcohol can lead to GSH
depletion and decreased antioxidant activity (36–38). It
has been noted that chronic depletion of cytosolic GSH can lead to
decreased levels of mitochondrial GSH (39). Alcohol is thought to contribute to
GSH depletion in the mitochondria of hepatocytes by producing
oxidative agents and also by inhibiting the mitochondrial GSH
transporter (transport of GSH from the cytosol into mitochondria)
(6,40,41).
Mitochondrial GSH may be of greater importance for hepatocyte
survival than cytoplasmic GSH, as its depletion can result in
increased production of H2O2 in mitochondria,
thus causing oxidation of cytoplasmic proteins and affecting cell
signaling (42). However, impaired
redox status also influences changes in erythrocytes and can lead
to decreased levels of blood GSH. It has previously been reported
that individuals with alcohol-related liver diseases exhibit low
levels of blood GSH (36–38). Although heavy drinkers who
participated in this study did not exhibit greater than normal
levels of γ-GT, blood GSH levels were significantly lower than
those of controls. Findings from the present study indicate that
heavy drinkers and individuals with AUDs without clinical signs of
liver dysfunction may experience lower blood GSH levels than
individuals who do not exceed moderate alcohol consumption.
An impaired redox status can lead to DNA damage,
protein modification and lipid peroxidation. Blood redox status
indices usually reflect the overall status of the body, and we
would thus expect to observe differences between the two groups in
the present study. Baseline and post-exercise levels of TBARS,
which act as an index of lipid peroxidation, were higher in heavy
drinkers than the controls in this study. This result has been
reported previously in alcoholics and suggests that alcohol abuse
results in enhanced lipid peroxidation, which in turn leads to
increased fragility of the cell membranes (6,37,43).
In the present study, the exercise-induced
antioxidant response was found to be higher in healthy controls
than heavy drinkers, as indicated by post-exercise changes in TAC
and UA. Heavy drinkers may not respond well to exercise-induced
oxidative stress due to lower antioxidant defenses (35). However, it is not clear as to whether
this antioxidant response in heavy drinkers increases some hours
after exercise. Changes in these indices at more time points after
exercise should therefore be examined.
Oxidative stress can alter membrane permeability and
lead to hemolysis (44); however, we
noted that exercise did not lead to hemolysis in heavy drinkers
regardless of the increased oxidative stress. This could be
explained by the intensity of the exercise used in the present
study and also by the fact that antioxidant responses to exercise
were increased in a similar fashion to those of the controls.
In conclusion, taken together, the findings of the
present study suggest that excessive alcohol consumption causes low
baseline GSH and increased γ-GT and TBARS levels. Acute aerobic
exercise increases the responses of liver enzymes in heavy
drinkers, whereas the elevated antioxidant responses following the
aerobic bout of exercise in heavy drinkers are somewhat attenuated
compared to healthy controls. More post-exercise time points would
provide a better understanding of the way individuals with AUDs
respond to exercise. Finally, since exercise training has been
proposed as a useful and safe strategy in the treatment of AUDs,
future research should focus on training exercise interventions
which aim to reduce alcohol consumption and would prevent or
ameliorate alcohol-related liver damage.
Acknowledgements
This study was co-financed by the European Union
[European Social Fund (ESF)] and Greek national funds through the
Operational Program ‘Education and Lifelong Learning’ of the
National Strategic Reference Framework-Research Funding Program:
THALES. Investing in knowledge society through the ESF.
References
|
1
|
Brien SE, Ronksley PE, Turner BJ, Mukamal
KJ and Ghali WA: Effect of alcohol consumption on biological
markers associated with risk of coronary heart disease: systematic
review and meta-analysis of interventional studies. BMJ.
342:d6362011. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ronksley PE, Brien SE, Turner BJ, Mukamal
KJ and Ghali WA: Association of alcohol consumption with selected
cardiovascular disease outcomes: a systematic review and
meta-analysis. BMJ. 342:d6712011. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
National Institute for Alcohol Abuse and
Alcoholism: Drinking Levels Defined. http://www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption-moderate-binge-drinkingRetrieved.
Jan 10–2014.
|
|
4
|
Greenfield TK, Ye Y, Bond J, Kerr WC,
Nayak MB, Kaskutas LA, Anton RF, Litten RZ and Kranzler HR: Risks
of alcohol use disorders related to drinking patterns in the U.S.
general population. J Stud Alcohol Drugs. 75:319–327. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sies H and Jones D: Oxidative stress.
Encyclopedia of Stress. Fink G: 3:(2nd). (Amsterdam). Elsevier.
45–48. 2007. View Article : Google Scholar
|
|
6
|
Das SK and Vasudevan DM: Alcohol-induced
oxidative stress. Life Sci. 81:177–187. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zima T and Kalousová M: Oxidative stress
and signal transduction pathways in alcoholic liver disease.
Alcohol Clin Exp Res. 29(Suppl 11): 110S–115S. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tsukamoto H and Lu SC: Current concepts in
the pathogenesis of alcoholic liver injury. FASEB J. 15:1335–1349.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Davies KJ, Quintanilha AT, Brooks GA and
Packer L: Free radicals and tissue damage produced by exercise.
Biochem Biophys Res Commun. 107:1198–1205. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Finaud J, Lac G and Filaire E: Oxidative
stress: relationship with exercise and training. Sports Med.
36:327–358. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Coffey VG and Hawley JA: The molecular
bases of training adaptation. Sports Med. 37:737–763. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Michailidis Y, Jamurtas AZ, Nikolaidis MG,
Fatouros IG, Koutedakis Y, Papassotiriou I and Kouretas D: Sampling
time is crucial for measurement of aerobic exercise-induced
oxidative stress. Med Sci Sports Exerc. 39:1107–1113. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Steinbacher P and Eckl P: Impact of
oxidative stress on exercising skeletal muscle. Biomolecules.
5:356–377. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Read JP and Brown RA: The role of physical
exercise in alcoholism treatment and recovery. Prof Psychol Res
Pract. 34:49–56. 2003. View Article : Google Scholar
|
|
15
|
Zschucke E, Heinz A and Ströhle A:
Exercise and physical activity in the therapy of substance use
disorders. ScientificWorldJournal. 2012:9017412012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Giesen ES, Deimel H and Bloch W: Clinical
exercise interventions in alcohol use disorders: a systematic
review. J Subst Abuse Treat. 52:1–9. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jamurtas AZ, Zourbanos N, Georgakouli K,
Georgoulias P, Manthou E, Fatouros IG, Goudas M, Koutedakis Y and
Theodorakis Y: Beta endorphin and alcohol urge responses in
alcoholic patients following an acute bout of exercise. J Addict
Res Ther. 5:10001942014.
|
|
18
|
Goldfarb AH and Jamurtas AZ:
Beta-endorphin response to exercise. An update. Sports Med.
24:8–16. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gianoulakis C: Endogenous opioids and
addiction to alcohol and other drugs of abuse. Curr Top Med Chem.
4:39–50. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Moussas G, Dadouti G, Douzenis A, Poulis
E, Tzelembis A, Bratis D, Christodoulou C and Lykouras L: The
Alcohol Use Disorders Identification Test (AUDIT): reliability and
validity of the Greek version. Ann Gen Psychiatry. 8:112009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
World Health Organization: The Alcohol Use
Disorders Identification Test: Guidelines for Use in Primare Care
(2nd). Geneva: WHO Press. 2001.
|
|
22
|
Janaszewska A and Bartosz G: Assay of
total antioxidant capacity: comparison of four methods as applied
to human blood plasma. Scand J Clin Lab Invest. 62:231–236. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Keles MS, Taysi S, Sen N, Aksoy H and
Akçay F: Effect of corticosteroid therapy on serum and CSF
malondialdehyde and antioxidant proteins in multiple sclerosis. Can
J Neurol Sci. 28:141–143. 2001.PubMed/NCBI
|
|
24
|
Patsoukis N, Zervoudakis G, Panagopoulos
NT, Georgiou CD, Angelatou F and Matsokis NA: Thiol redox state
(TRS) and oxidative stress in the mouse hippocampus after
pentylenetetrazol-induced epileptic seizure. Neurosci Lett.
357:83–86. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Aebi H: Catalase in vitro. Methods
Enzymol. 105:121–126. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Reddy YN, Murthy SV, Krishna DR and
Prabhakar M: Role of free radicals and antioxidants in tuberculosis
patients. Indian J Tuberc. 51:213–218. 2004.
|
|
27
|
Whitehead TP, Robinson D and Allaway SL:
The effects of cigarette smoking and alcohol consumption on serum
liver enzyme activities: a dose-related study in men. Ann Clin
Biochem. 33:530–535. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Banfi G, Colombini A, Lombardi G and
Lubkowska A: Metabolic markers in sports medicine. Adv Clin Chem.
56:1–54. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Robinson D and Whitehead TP: Effect of
body mass and other factors on serum liver enzyme levels in men
attending for well population screening. Ann Clin Biochem.
26:393–400. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Halonen PI and Konttinen A: Effect of
physical exercise on some enzymes in the serum. Nature.
193:942–944. 1962. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Parikh DJ and Ramanathan NL: Exercise
induced serum enzyme changes in untrained subjects. Indian J
Physiol Pharmacol. 21:175–180. 1977.PubMed/NCBI
|
|
32
|
Fibach E and Rachmilewitz E: The role of
oxidative stress in hemolytic anemia. Curr Mol Med. 8:609–619.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cho J, Lee I, Kim D, Koh Y, Kong J, Lee S
and Kang H: Effect of aerobic exercise training on non-alcoholic
fatty liver disease induced by a high fat diet in C57BL/6 mice. J
Exerc Nutrition Biochem. 18:339–346. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Keating SE, Hackett DA, Parker HM,
O'Connor HT, Gerofi JA, Sainsbury A, Baker MK, Chuter VH, Caterson
ID, George J and Johnson NA: Effect of aerobic exercise training
dose on liver fat and visceral adiposity. J Hepatol. 63:174–182.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tseng YM, Tsai SM, Lin CC, Jin YR, Yeh WH,
Hsiao JK, Chen CF, Lan WH and Tsai LY: Oxidative stress-related
enzyme polymorphisms associated with the immunological biomarkers
levels in heavy drinkers in Taiwan. J Clin Lab Anal. 27:494–503.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Loguercio C, Blanco FD, De Girolamo V,
Disalvo D, Nardi G, Parente A and Blanco CD: Ethanol consumption,
amino acid and glutathione blood levels in patients with and
without chronic liver disease. Alcohol Clin Exp Res. 23:1780–1784.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Maithreyi R, Janani AV, Krishna R, Shweta
A, Edwin RR and Mohan SK: Erythrocyte lipid peroxidation and
antioxidants in chronic alcoholics with alcoholic liver disease.
Asian J Pharm Clin Res. 3:183–185. 2010.
|
|
38
|
Gupta S, Pandey R, Katyal R, Aggarwal HK,
Aggarwal RP and Aggarwal SK: Lipid peroxide levels and antioxidant
status in alcoholic liver disease. Indian J Clin Biochem. 20:67–71.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Meister A: Mitochondrial changes
associated with glutathione deficiency. Biochim Biophys Acta.
1271:35–42. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Viña J, Estrela JM, Guerri C and Romero
FJ: Effect of ethanol on glutathione concentration in isolated
hepatocytes. Biochem J. 188:549–552. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fernández-Checa JC, García-Ruiz C, Colell
A, Morales A, Marí M, Miranda M and Ardite E: Oxidative stress:
role of mitochondria and protection by glutathione. Biofactors.
8:7–11. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Han D, Hanawa N, Saberi B and Kaplowitz N:
Mechanisms of liver injury. III. Role of glutathione redox status
in liver injury. Am J Physiol Gastrointest Liver Physiol.
291:G1–G7. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Barden A, Zilkens RR, Croft K, Mori T,
Burke V, Beilin LJ and Puddey IB: A reduction in alcohol
consumption is associated with reduced plasma F2-isoprostanes and
urinary 20-HETE excretion in men. Free Radic Biol Med.
42:1730–1735. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lubin B and Chiu D: Properties of vitamin
E-deficient erythrocytes following peroxidant injury. Pediatr Res.
16:928–932. 1982. View Article : Google Scholar : PubMed/NCBI
|