Introduction
Biliary and duodenal obstruction is a common
complication in patients with gastroduodenal or pancreatobiliary
malignancies. Stent implantation has been widely used in clinical
practice, which is the preferred method for palliative management
of malignant biliary and duodenal obstruction (1–6).
Obstructive jaundice accompanied by duodenal obstruction is mainly
caused by periampullary or pancreatic head carcinoma, malignant
duodenal tumor and lymph node metastasis. Biliary and duodenal
obstruction causes cholestasis and hepatic insufficiency. Patients
with malignant obstructive jaundice and duodenal obstruction are in
a poor condition or the tumor has already invaded the surrounding
tissue or organs, thus tumor excision in no longer possible
(7).
The most commonly used surgical approach for biliary
and duodenal obstruction is palliative cholangioenteric
anastomosis, gastroenterostomy or jejunostomy; however, these
approaches are not considered safe for patients who are weak, have
electrolyte imbalance or experience multiple organ system failure
(8,9). In comparison to the surgical
approaches, percutaneous transhepatic biliary stenting (PTBS)
combined with peroral duodenal stent implantation has certain
advantages: Ease and safety of the procedure, minimum invasiveness,
rapid recovery with less complications and smaller effect on the
gastrointestinal function. The results of studies that focused on
the combination of these two stenting methods reported high
technical and clinical success rates (8,9).
The present study investigated the short-term
therapeutic effectiveness of combined metallic stenting under
fluoroscopic guidance in 20 patients with malignant biliary and
duodenal obstruction.
Materials and methods
Patients
The present study was approved by the Institutional
Review Board of the Third Affiliated Hospital of Harbin Medical
University (Harbin, China) and informed consent was provided by all
participants. In total, 20 patients (male, 13; female, 7; age
range, 35–72 years; mean age, 63.1±8.2 years) with malignant
obstructive jaundice and duodenal obstruction were enrolled in the
study between June 2004 and June 2013. Computed tomography (CT),
magnetic resonance imaging, ultrasound and/or gastroenterography
were performed in all patients prior to the interventional
procedures. Among the patients, 6 cases were diagnosed with
pancreatic carcinoma, 11 with cholangiocarcinoma, 1 with duodenal
carcinoma and 2 with abdominal metastatic lymph nodes. In total, 16
patients initially presented with bile duct obstruction, which was
manifested as skin and sclera yellowing, dark urine and pale
stools, with 7 complaining of skin itching. The patients were
treated by PTBS, and gastrointestinal obstruction symptoms,
including nausea and vomiting, were observed 1–8 month(s) following
the procedure. Gastroenterography showed duodenal obstruction. In
addition, 3 patients initially presented with duodenal obstructive
symptoms, which were followed by biliary obstruction. In 1 case,
bile duct obstruction and duodenal obstruction occurred
simultaneously.
Procedure
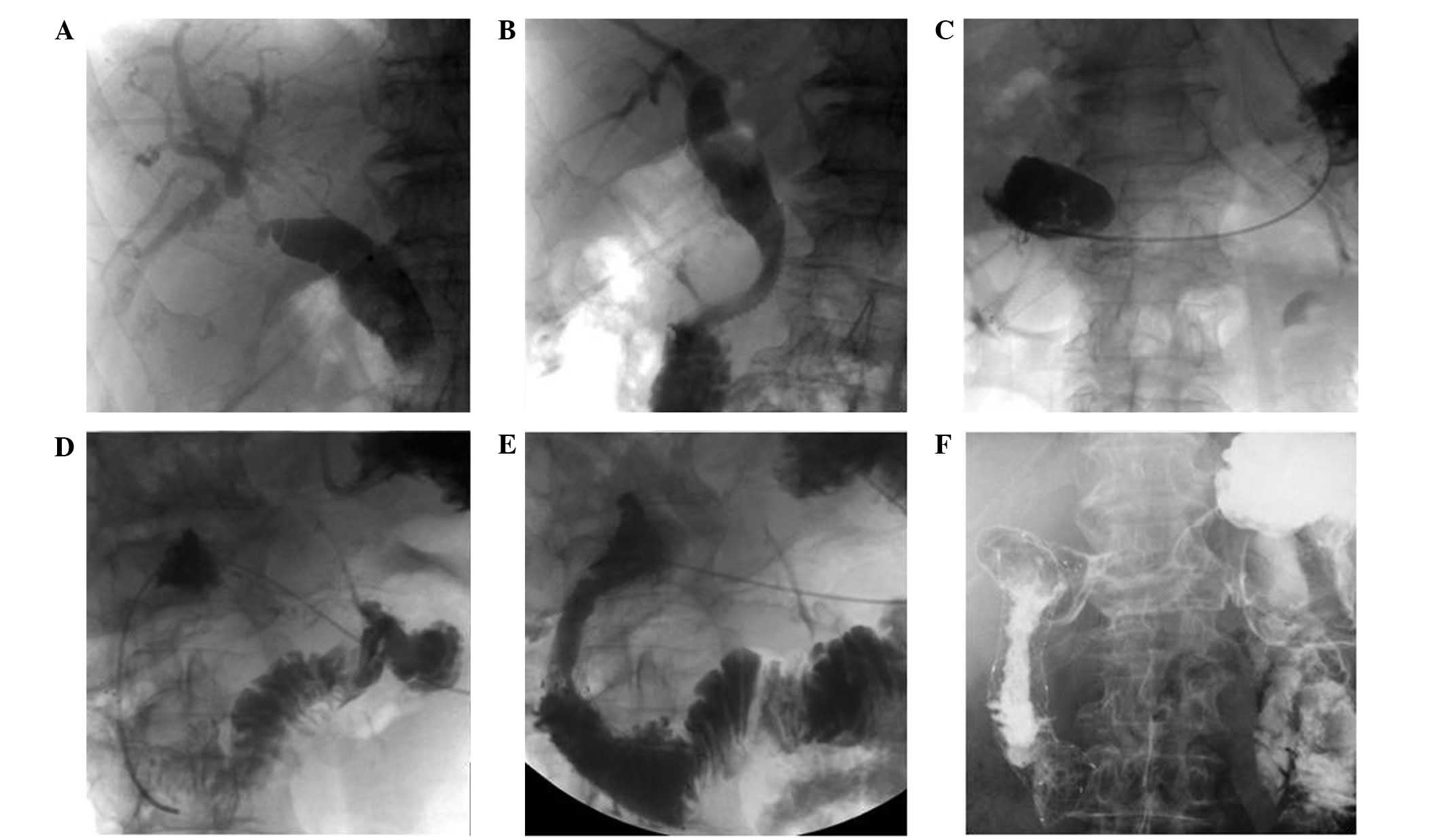
Under monitoring with X-ray fluoroscopy, the
puncture site was selected at an intercostal space under the right
costophrenic angle. The puncture site was locally anesthetized and
a 22 G MReye® Chiba needle (Cook Medical, Inc., Bloomington, IN,
USA) was used to puncture the skin at the right midaxillary line at
T11 level. The depth of the puncture was ~2 cm to the right border
of the vertebra. The needle was slowly withdrawn with simultaneous
infusion of the contrast agent [Omnipaque™; GE Healthcare
(Shanghai) Co., Ltd., Shanghai, China]. When the bile duct became
visible, a Radifocus® guidewire (Terumo, Tokyo, Japan) was
inserted, the Chiba needle was removed and a sheath was positioned
using the guidewire. A cholangiography was performed to detect the
location of the stenosis and a stent (SMART® or Zilver®; Cordis
Corp., Hialeah, USA or Cook Medical, Inc., respectively) was
implanted at the site of the stenosis via the guidewire. A balloon
catheter, 6mm in diameter, would be used for the dilation of the
stenosis, if the rate of the remaining stenosis was 30% following
PTBS.
With regard to the duodenal obstruction, a
nasogastric tube (Freka; Frensenius Kabi AG, Miekinia, Poland) was
inserted to decompress the stomach overnight, prior to the
procedure. A duodenal stent (Hanarostent®; M.I. Tech Co., Ltd.,
Seoul, Korea) was implanted through the mouth, under the guidance
of X-ray fluorescence. A local anesthetic spray was used to numb
the pharynx, and a Cobra angiographic catheter (Radiofocus®;
Terumo) was placed into the proximal segment of stenosis via a
guidewire (Radifocus; Terumo, Tokyo, Japan) and an infused contrast
agent was used to confirm the duodenal obstruction. The guidewire
and the catheter were then advanced to the distal segment of the
stenosis with further radiography to demonstrate the length of the
stenosis. An Amplatz Super Stiff™ guidewire (Boston Scientific, MA,
USA) was used to exchange the catheter and position an intestinal
stent at the stenotic segment, and was then deployed. In cases
where the rate of stenosis was >40% following stent
implantation, further balloon dilation was performed (Fig. 1). Subsequent to biliary stenting, the
guidewire should be carefully used, in order to avoid going through
the biliary stent mesh.
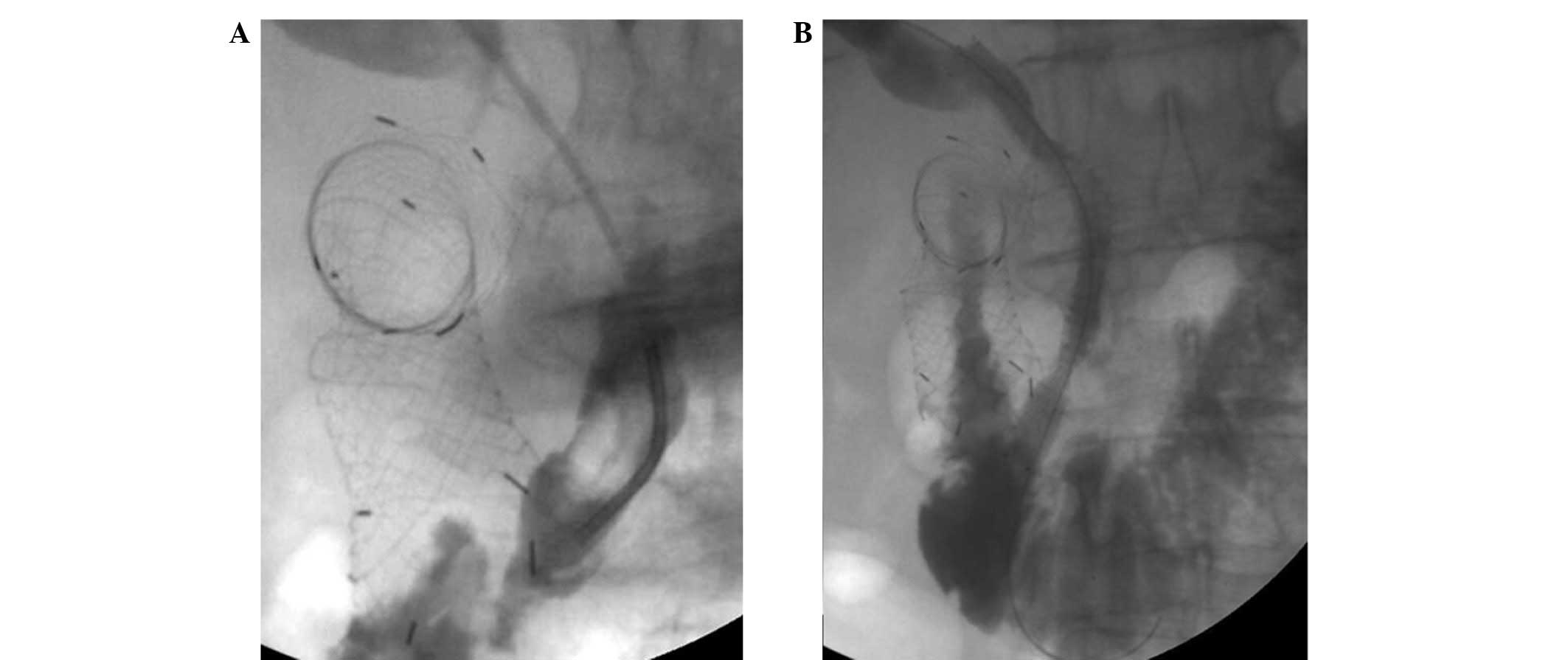
In cases that developed duodenal obstruction
following PTBS, duodenal stenting was performed. Similarly, PTBS
was attempted in the patients who developed biliary obstruction
following duodenal stent placement (Fig.
2). In cases with simultaneous biliary and duodenal obstruction
without any previous interventions, a duodenal stent (Hanarostent;
M.I. Tech, Co., Ltd.) was inserted several days after the placement
of a biliary drainage catheter (Ultrathane; Cook Medical, Inc.),
which was followed by implantation of the biliary stent (SMART;
Cordis Corp.).
The clinical outcomes, including complications
associated with the procedures and patency of the stents, were
evaluated. Cholangitis was diagnosed in the case of abdominal pain
and fever without any other infection outside the hepatobiliary
system that required antibiotic treatment within 24 h of the
procedure. Pancreatitis was diagnosed following a 3-fold increase
relative to the normal levels of serum amylase, which was
accompanied by persistent abdominal pain for >24 h
postoperatively. Duodenal stenting was suggested when recurrent
vomiting was observed in the absence of features suggestive of
peritoneal carcinomatosis, and was confirmed using imaging methods.
Biliary stent occlusion was diagnosed upon recurrence of the
typical symptoms of biliary tract obstruction, such as yellowing of
skin and sclera, dark urine and pale stool. Follows-ups that
included clinical examinations and assessments of stent patency
continued until the death of the patients.
Statistical analysis
All analyses were conducted using SPSS software,
version 20.0 (SPSS, Inc., Chicago, IL, USA). The paired Student's
t-test was used to compare the mean values of serum bilirubin pre-
and post-stent implantation. P<0.05 was considered to indicate a
statistically significant difference.
Results
Stenting
A total of 39 stents were implanted in 20 patients
with a technical success rate of 90% (18/20). Of these stents, 21
were biliary, including 11 SMART (Cordis Corp.) and 10 Zilver (Cook
Medical Inc.) stents. In addition, 1 patient underwent an
implantation of two biliary stents due to stent sliding, while 5
patients received subsequent balloon inflation, following which
adequate stent expansion was achieved. The mean serum bilirubin
level prior to PTBS was 326.5±105.7 mol/l and dropped to 35.9±12.2
mol/l at 7 days after stenting (P<0.05). The skin and sclera
jaundice gradually vanished and the skin itching was relieved in
all 7 patients 10–25 days after PTBS (median, 13.3 days). No
aggravating jaundice was identified in the patients who had
undergone duodenal stent implantation, which suggested that the
biliary stent was not blocked by duodenal stent implantation.
Furthermore, 18 duodenal stents (Hanarostent; M.I.
Tech Co., Ltd.) were implanted, all of which were successfully
deployed and inflated, according to gastroenterography. However,
50–60% of stenosis remained following implantation of 2 stents, as
observed by the slow passing of the contrast agent. Following the
secondary balloon inflation, the remaining in-stent stenosis
decreased by ~20%. At 1 week after the duodenal stent implantation,
radiography confirmed the normal flow of the contrast agent through
the stent. All patients were able to have a liquid or semi-liquid
diet immediately following stent implantation and were allowed a
solid diet 1 week later. The symptom of bloating after the meals
persisted in 3 patients, but vomiting was not observed.
Gastroenterography confirmed that the stent remained open, but
gastrointestinal movement was decreased.
Duodenal obstruction occurred in 2 patients, 4 and 8
months after PTBS. Duodenal stent implantation was then planned.
Transcatheter radiography revealed that the length of the free part
of the biliary stent was relatively long and contained in the
duodenal carcinoma.
Complications
The duodenal stent would have to pass through the
mesh of the biliary stent, which could destroy its structure;
therefore, the duodenal stent placement was not performed.
A total of 5 patients exhibited a temporary increase
in serum amylase with no abdominal pain. The patients were treated
with fasting, fluid infusion and heteropathy and serum amylase
returned to its normal range after 24–48 h. In addition, 9 patients
suffered from mild abdominal pain following stent implantation,
which was relieved following heteropathy. Furthermore, 4 patients
were found to be positive or strong positive for occult blood in
the stool, which disappeared in 3–5 days.
Survival time
Additionally, 1 patient exhibited cough and fever,
which, according to a CT scan, were considered to be symptoms of
aspiration pneumonia. The patient succumbed to the disease 5 days
after the procedure, with no cholangitis or pancreatitis
detected.
The survival time of the 18 patients was 5–21 months
(median, 9.6 months). In total, 6 of those patients survived for
>12 months.
Discussion
With regard to patients with biliary and duodenal
obstruction, particularly those who present obstruction in the
descending segment of the duodenum, the present results suggest
that the management begins with PTBS and peroral enteral nutrition
using a jejunal feeding tube. The duodenal stent can be implanted
when jaundice has decreased and the patient's general condition has
improved. Another option for these patients is receiving PTBS
following duodenal stent implantation, during which a guidewire is
placed into the duodenum through the mesh of the duodenal stent
using a catheter, under fluoroscopic guidance. A balloon catheter,
8–10 mm in diameter, is placed to dilate the mesh of the duodenal
stent and allow biliary stent expansion. In the case that the
biliary stent cannot fully expand, the balloon catheter is used to
dilate the biliary stent. No interference has been identified
between the biliary and duodenal stents. The duodenal stent is
braided, so the balloon dilation, which is meant to expand the
mesh, will not affect the entire structure of the stent (8).
Profili et al (10) reported 4 cases with pancreatic cancer
treated with combined biliary and duodenal stenting for the
palliative treatment of malignant biliary and duodenal obstruction
under fluoroscopic guidance. The authors suggested that the biliary
stent implantation should be performed prior to the duodenal stent
implantation, since the existence of a stent in the duodenum may
make the placement and expansion of a biliary stent difficult or
even unsuccessful (10). In
addition, the biliary stent can assist the duodenal stent
positioning at the same time. Out of the 20 patients who
participated in the present study, 4 received duodenal stenting
first, and then PTBS was successfully performed. It is therefore
suggested that duodenal stent implantation should be performed
prior to PTBS, since biliary stenting through the duodenal stent
mesh is not challenging. If malignant duodenal obstruction is
detected in patients with a biliary stent, the guidewire should not
pass through the mesh of the biliary stent, since the duodenal
stent may not be sufficiently inflated and the chyme may block the
biliary stent. The biliary stent is small and integrative, thus if
the mesh cannot be inflated, it will lead to severe deformation
(11). In the present study,
duodenal stent implantation following PTBS failed in 2 patients,
since the guidewire passed through the mesh of the biliary stent,
but was unable to pass between the stent and the duodenum.
Combined duodenal and biliary stenting can also be
performed either simultaneously or separately under endoscopic
guidance (7,12,13). Kaw
et al (7) reported the
clinical outcome of combined duodenal and biliary stenting under
endoscopic guidance in 18 patients. All patients had documented
evidence of symptomatic duodenal and biliary obstruction, and
received endoscopic duodenal stenting using enteral Wallstent and
endoscopic biliary stenting with biliary Ultraflex or Wallstent
(7). Combined metal stenting for
malignant biliary and duodenal obstruction was successful in 17
patients, while one procedure failed due to a tortuous duodenal
stricture. From the 17 patients who underwent a successful duodenal
stenting, 16 had a good clinical outcome, with relief of the
obstruction symptoms (7). No
immediate stent-associated complications were noted; therefore,
based on the results of the study by Kaw et al (7), the endoscopic approach can be
considered if the peroral approach fails.
In the present study, duodenal stenting failed in 2
cases due to the fact that the free part of the biliary stent in
the duodenal lumen was relatively long. The existence of a biliary
stent in the duodenum may disturb the insertion of a duodenal
stent, which may then destroy the structure of the biliary stent.
It is possible, however, for the guidewire to pass through the
space between the biliary stent and the duodenal wall under
endoscopic guidance.
PTBS combined with para-oral duodenal stent is
minimally invasive; however, the technique has been associated with
complications, such as bleeding, choleperitonitis, cholangitis,
duodenum perforation and aspiration pneumonia. In the present
study, 1 patient was diagnosed with pancreatic cancer and
obstructive jaundice, and postoperatively suffered from abdominal
distention, nausea and vomiting, which were caused by duodenal
obstruction. Gastroenterography demonstrated obstruction of the
descending segment of the duodenum. An exchange guidewire was
placed into the jejunum, through which a stent (18×120 mm) was
implanted. Following stent implantation, 40% of the stenosis
remained. Considering the local stenosis, it was decided to perform
balloon dilation. A balloon catheter (18×60 mm) was introduced, but
was not able to enter the stenosis segment. Several attempts were
unsuccessful, probably due to the deformation of the stent. The
patient exhibited nausea and vomiting during the 2-h interventional
procedure, as well as cough and fever (maximum temperature, 39°C).
A thoracic CT scan showed scattered patchy-like shadows in the
lungs and aspiration pneumonia was diagnosed. The patient suffered
from continuous hyperpyrexia and succumbed 5 days later.
Based on the results and observations of the present
study, the following conclusion were made: i) Sufficient
gastrointestinal decompression is necessary to drain the gastric
contents; ii) a long sheath should be placed into the pylorus in
order to reduce the pharyngeal stimulation from the surgery; and
iii) the duration of the surgery should be reduced.
In conclusion, dual stent implantation into the bile
duct and duodenum can successfully treat biliary and duodenal
obstruction. This technique as effective in treating these
obstructions as palliative bypass surgery, while it improves the
quality of life of the patients by promptly alleviating the
symptoms.
References
|
1
|
de Baere T, Harry G, Ducreux M, Elias D,
Briquet R, Kuoch V and Roche A: Self-expanding metallic stents as
palliative treatment of malignant gastroduodenal stenosis. AJR Am J
Roentgenol. 169:1079–1083. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pinto IT: Malignant gastric and duodenal
stenosis: Palliation by peroral implantation of a self-expanding
metallic stent. Cardiovasc Intervent Radiol. 20:431–434. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lee BH, Choe DH, Lee JH, Kim KH and Chin
SY: Metallic stents in malignant biliary obstruction: Prospective
long-term clinical results. AJR Am J Roentgenol. 168:741–745. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yasumoto T, Yokoyama S and Nagaike K:
Percutaneous transcholecystic metallic stent placement for
malignant obstruction of the common bile duct: Preliminary clinical
evaluation. J Vasc Interv Radiol. 21:252–258. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jung GS, Song HY, Kang SG, Huh JD, Park
SJ, Koo JY and Cho YD: Malignant gastroduodenal obstructions:
Treatment by means of a covered expandable metallic stent-initial
experience. Radiology. 216:758–763. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Oikarinen H, Leinonen S, Karttunen A,
Tikkakoski T, Hetemaa T, Mäkelä J and Päivänsalo M: Patency and
complications of percutaneously inserted metallic stents in
malignant biliary obstruction. J Vasc Interv Radiol. 10:1387–1393.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kaw M, Singh S and Gagneja H: Clinical
outcome of simultaneous self-expandable metal stents for palliation
of malignant biliary and duodenal obstruction. Surg Endosc.
17:457–461. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kim KO, Kim TN and Lee HC: Effectiveness
of combined biliary and duodenal stenting in patients with
malignant biliary and duodenal obstruction. Scand J Gastroenterol.
47:962–967. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Maire F, Hammel P, Ponsot P, Aubert A,
O'Toole D, Hentic O, Levy P and Ruszniewski P: Long-term outcome of
biliary and duodenal stents in palliative treatment of patients
with unresectable adenocarcinoma of the head of pancreas. Am J
Gastroenterol. 101:735–742. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Profili S, Feo CF, Meloni GB, Strusi G,
Cossu ML and Canalis GC: Combined biliary and duodenal stenting for
palliation of pancreatic cancer. Scand J Gastroenterol.
38:1099–1102. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Akinci D, Akhan O, Ozkan F, Ciftci T,
Ozkan OS, Karcaaltincaba M and Ozmen MN: Palliation of malignant
biliary and duodenal obstruction with combined metallic stenting.
Cardiovasc Intervent Radiol. 30:1173–1177. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tonozuka R, Itoi T, Sofuni A, Itokawa F
and Moriyasu F: Endoscopic double stenting for the treatment of
malignant biliary and duodenal obstruction due to pancreatic
cancer. Dig Endosc. 25(Suppl 2): 100–108. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Katsinelos P, Kountouras J, Germanidis G,
Paroutoglou G, Paikos D, Lazaraki G, Pilpilidis I, Chatzimavroudis
G, Fasoulas K and Zavos C: Sequential or simultaneous placement of
self-expandable metallic stents for palliation of malignant biliary
and duodenal obstruction due to unresectable pancreatic head
carcinoma. Surg Laparosc Endosc Percutan Tech. 20:410–415. 2010.
View Article : Google Scholar : PubMed/NCBI
|