Introduction
Cardiac hypertrophy, the thickening of heart muscle,
is a compensatory response to physical stimuli or pathological
insults. These pathological remodeling responses are often
accompanied by fibrosis, pump failure, myocyte degeneration and
apoptosis, which may culminate in heart failure and sudden death
(1). Heart failure occurs when the
heart is unable to pump blood at a rate proportional to the body's
requirement for oxygen, or when this function leads to cardiogenic
pulmonary edema. With >1 million hospitalizations annually (up
175% in the past 25 years), and costs of ~$15.4 billion, acute
heart failure is a critical health concern. Furthermore, half of
the patients discharged from the hospital are readmitted within 6
months. In-hospital mortality rates remain high, between 4 and 7%
(2,3). Heart failure is a significant problem
as the population ages. The prevalence in the US is 2.5% of the
population, or 5 million patients (3) Cardiac hypertrophy is a major
determinant of congestive heart failure. Angiotensin II is known to
participate in cardiac hypertrophy by binding to angiotensin II
receptor subtype 1 (AGTR1). In addition,
AGTR1 overexpression has been closely associated with
cardiac hypertrophy (4).
MicroRNAs (miRs) are endogenous, highly conserved,
small non-coding RNAs that negatively regulate the expression of
target genes at the post-transcriptional level (5). miR-155 is encoded by the human miR-155
host gene and is abundantly expressed in the lungs, heart and
kidneys (6). Seok et al
(7) and Heymans et al
(8) have revealed that
gain-of-function mutations in miR-155 exacerbate myocardial
hypertrophy, whereas loss-of-function mutations ameliorate cardiac
hypertrophy. Sethupathy et al (9) experimentally investigated the target
sites for hsa-miR-155 within the 3′-untranslated region of the
human AGTR1 gene and demonstrated that hsa-miR-155
downregulated the expression of AGTR1. These previous
findings suggest that miR-155 ameliorates hypertension by
modulating AGTR1 expression, as AGTR1
signaling occurs upstream of cardiac hypertrophy (10). The present study therefore aimed to
test the hypothesis that miR-155 promotes cardiac hypertrophy by
targeting AGTR1, and to determine the underlying
molecular behavior of miR-155 in cardiac hypertrophy.
Materials and methods
Cell culture and reagents
Rat H9C2 (2–1) cardiomyocytes were purchased from the
China Center for Type Culture Collection, Wuhan University (Wuhan,
China). Lipofectamine 2000, TRIzol® and Platinum
SYBR® Green qPCR SuperMix-UDG were purchased from
Invitrogen (Thermo Fisher Scientific, Inc., Waltham, MA, USA),
angiotensin II was purchased from Sigma-Aldrich (St. Louis, MO,
USA), and the mirVana PARIS RNA and Native Protein Purification kit
was from Ambion (Thermo Fisher Scientific, Inc.). Rabbit
anti-AGTR1 polyclonal antibody (cat. no. ab9391) was
purchased from Abcam (Cambridge, UK). Rabbit anti-calcineurin-β
(CaN-β; cat. no. BS6114), nuclear factor of activated T-cells
(NFAT-4; cat. no. BS1762) and glyceraldehyde-3-phosphate
dehydrogenase (GAPDH; cat. no. BS60630) polyclonal antibodies were
purchased from Bioworld Technology, Inc. (St. Louis Park, MN, USA).
Goat anti-rabbit polyclonal antibody labeled with horseradish
peroxidase (HRP) (cat. no. BA1125) were purchased from Boster
Biological Technology, Co., Ltd., (Wuhan, China). miR-155 analogue
and inhibitor, with or without fluorescein (FAM) conjugation, were
synthesized by GenePharma (Shanghai, China), in accordance with the
miR-155 sequence provided in the miRBase database (www.mirbase.org/; accession no. MIMAT0030409;
5′-UUAAUGCUAAUUGUGAUAGGGGU-3′). The sequences were as follows:
miR-155 analogue forward, 5′-UUAAUGCUAAUUGUGAUAGGGGU-3′ and
reverse, 5′-CCCUAUCACAAUUAGCAUUAAUU-3′; and miR-155,
5′-ACCCCUAUCACAAUUAGCAUUAA-3′. The analogues and inhibitor were
diluted with sterile water to a final concentration of 20 µmol/l
and stored at −80°C until use.
miR-155 transfection
Rat cardiomyocytes were seeded into 6-well plates at
a density of 1×108 cells/ml in Dulbecco's modified
Eagle's medium supplemented with 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.). Cells were incubated at 37°C and
5% CO2 until 30–50% confluency was reached, after which
the cardiomyocytes were transfected with either 8µl miR-155
analogue or inhibitors using lipofectamine, followed by incubation
for 24 h in serum-free medium. Cells were then stimulated with
1×10−7 mmol/l angiotensin II for 48 h in various groups.
FAM fluorescence was assessed at an excitation wavelength of 480 nm
and an emission wavelength of 520 nm using the LAS X Widefield
Systems (Leica Microsystems GmbH, Wetzlar, Germany).
Experimental groups
The cardiomyocytes were divided into six groups, as
follows: i) Untreated control cells, subjected to no transfection
or chemical regents; ii) treated with 1×10−7 mmol/l
angiotensin (Ang II) only; iii) transfected with 80 nmol/l miR-155
analogue; iv) transfected with 80 nmol/l miR-155 inhibitors; v) 80
transfected with nmol/l miR-155 analogue and treated with
1×10−7 mmol/l AngII; and vi) transfected with 80 nmol/l
miR-155 inhibitors and treated with 1×10−7 mmol/l AngII.
The concentration of AngII was determined in accordance with a
previous study by Zheng et al (11). A previous study by Cheng et al
(12) was used to determine the
concentration of miR-155 analogue and miR-155 inhibitor used.
Cell area measurement
Cardiomyocytes were fixed with 4% paraformaldehyde
for 15 min and imaged using phase contrast microscopy. The LAS X
Widefield Systems was used to count cardiomyocytes and measure the
diameter of single cells. For each group, 10 images were captured
under different perimeters, and 20 cardiomyocyetes were counted
within each perimeter. ImageJ 1.45 software (National Institutes of
Health, Bethesda, MA, USA) was used to calculate the number of
cardiomyocetes and measure the average surface area. Three
replicates were used in each experimental group.
Intracellular calcium
([Ca2+]i) measurements
Calcineurin and intracellular Ca2+
concentration are focal to the development of angiotensin
II-induced cardiac hypertrophy (13,14). To
determine intracellular calcium levels, 5 µmol/l Fura-2/AM
(Biotium, Inc., Hayward, CA, USA) solution was added to the cell
suspension in all groups for 30 min at 37°C. Cells were then washed
twice with Hank's buffered salt solution supplemented with 0.2%
bovine serum albumin (Roche Diagnostics-Basel, Switzerland).
Measurements were then obtained with an excitation wavelength of
340 nm and an emission wavelength of 510 nm using a fluorescent
spectrophotometer (Lengguang, Shanghai, China).
Reverse transcription-polymerase chain
reaction (RT-PCR) and quantitative (PCR)
To detect atrial natriuretic peptide (ANP), myosin
heavy chain-β (β-MHC), calcineurin-β (CaN-β), AGTR1,
miR-155 and Rnu6 expression, cDNA was synthesized using Platinum
SYBR Green qPCR SuperMix-UDG, according to the manufacturer's
protocol. The RT-PCR primer sets are reported in Table I. Quantitative Stem-Loop RT-qPCR was
used to detect the expression levels of miR-155, according to a
previous study (15). These primers
are listed in Table II. The RT-PCR
cycling conditions were as follows: 94°C for 3 min; 35 cycles at
94°C for 30 sec; and 72°C for 1 min. Amplifications were performed
in an ABI 7300 Real Time PCR System, under the following thermal
cycling conditions: 50°C for 2 min, 95°C for 2 min, followed by 40
cycles at 95°C for 15 sec and 60°C for 30 sec, and finally 20°C for
2 min. miR-155 expression levels were normalized to Rnu6 expression
levels. Expression ratios were calculated by the 2−ΔΔCq
method (16).
 | Table I.Reverse transcription-polymerase
chain reaction primers. |
Table I.
Reverse transcription-polymerase
chain reaction primers.
| Gene | Primers
(5′-3′) | Sequence length
(bp) | Melting temperature
(°C) |
|---|
| β-MHC |
|
|
|
| F |
AGGAAGAACCTACTGCGACTG | 264 | 53 |
| R |
CATCCTTAGGGTTGGGTAGCAC |
|
|
| ANP |
|
|
|
| F |
GGCTCCTTCTCCATCACC | 412 | 53 |
| R |
CTCCAATCCTGTCAATCCTAC |
|
|
| CaN-β |
|
|
|
| F |
CCATCTGGTAAAAGAAGGTCG | 198 | 53 |
| R |
AGGTATCGTGTATTAGCAGGTGA |
|
|
| GAPDH |
|
|
|
| F |
CAAGGTCATCCATGACAACTTTG | 496 | 58 |
| R |
GTCCACCACCCTGTTGCTGTAG |
|
|
 | Table II.Rnu 6 and miR-155 primers. |
Table II.
Rnu 6 and miR-155 primers.
| Gene | Reverse
transcription-PCR primers (5′-3′) | Quantitative PCR
primers (5′-3′) |
|---|
| Rnu 6 |
CGCTTCACGAATTTCCGTGTCAT | F:
CTCGCTTCGGCAGCACA |
|
|
| R:
AACGCTTCACGAATTTGCGT |
| M-miR-155-5p |
CTCAACTGGTGTCGTGGAGTCG | F:
ACACTCCAGCTGGGTTAATGCTAATTGTGAT |
|
|
GCAATTCAGTTGAGACCCCTAT | R:
TGGTGTCGTGGAGTCG |
Western blot analysis
Total protein was obtained from the rat
cardiomyocytes using radioimmunoprecipitation assay lysis buffer
(Boster Biological Technology, Co., Ltd.). Protein samples (30 µg
total protein) were separated by 10% SDS-PAGE, transferred to
polyvinylidene fluoride membranes, and blocked with 5% skimmed milk
for 1 h. The membranes were then incubated overnight at 4°C with
rabbit polyclonal antibodies against AGTR1, GAPDH,
CaN-β, and NFAT-4 (1:500 dilution). After washing the membranes
with Tris-buffered saline supplemented with 0.05% Tween-20 (Boster
Biological Technology, Co., Ltd.), the membranes were incubated
with HRP-conjugated goat anti-rabbit IgG (1:15,000 dilution) at
room temperature for 1 h. The protein bands were visualized using
enhanced chemiluminescence, after which images of the gels were
captured and protein band intensities were quantified using
Quantity One software, version 4.4 (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA).
Statistical analysis
Data were analyzed with SPSS 13.0 software (SPSS,
Inc., Chicago, IL, USA). One-way analysis of variance and Tukey's
post-hoc test were used to compare data among the various groups.
P<0.05 was considered to represent a statistically significant
difference. Data are presented as the mean ± standard error of the
mean.
Results
miR-155 transfection increases miR-155
expression levels
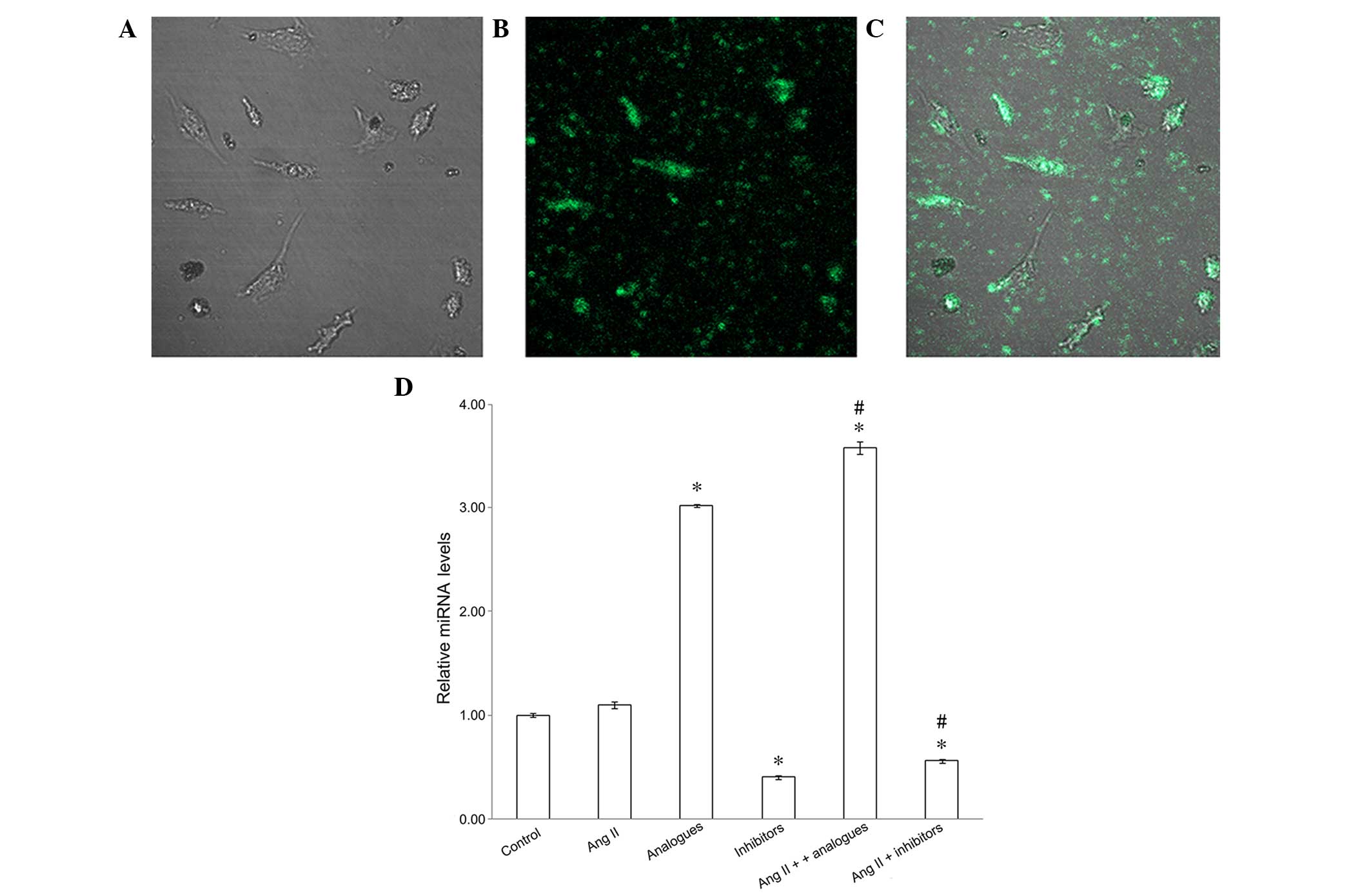
To confirm that the miR-155 analogues had been
successfully transfected into the cardiomyocytes, the fluorescence
of FAM-labeled miR-155 analogues was observed and miR-155 mRNA
expression levels were detected by RT-PCR (Fig. 1). miR-155 analogues labeled with FAM
were successfully transfected into H9C2 (2–1) cells
using lipofectamine (Fig. 1A-C), and
miR-155 expression was significantly increased in cells transfected
with FAM-labeled analogues, as compared with the control cells
(P<0.05; Fig. 1D). Furthermore,
downregulated miR-155 expression was demonstrated in the miR-155
inhibitor and angiotensin II + miR-155 inhibitor groups, as
compared with the control group (P<0.05) In addition, the
expression levels of miR-155 were significantly increased in the
angiotensin II + miR-155 analogues group, as compared with the
cells transfected with miR-155 analogues alone (P<0.05; Fig. 1D).
AGTR1 mRNA and protein
expression
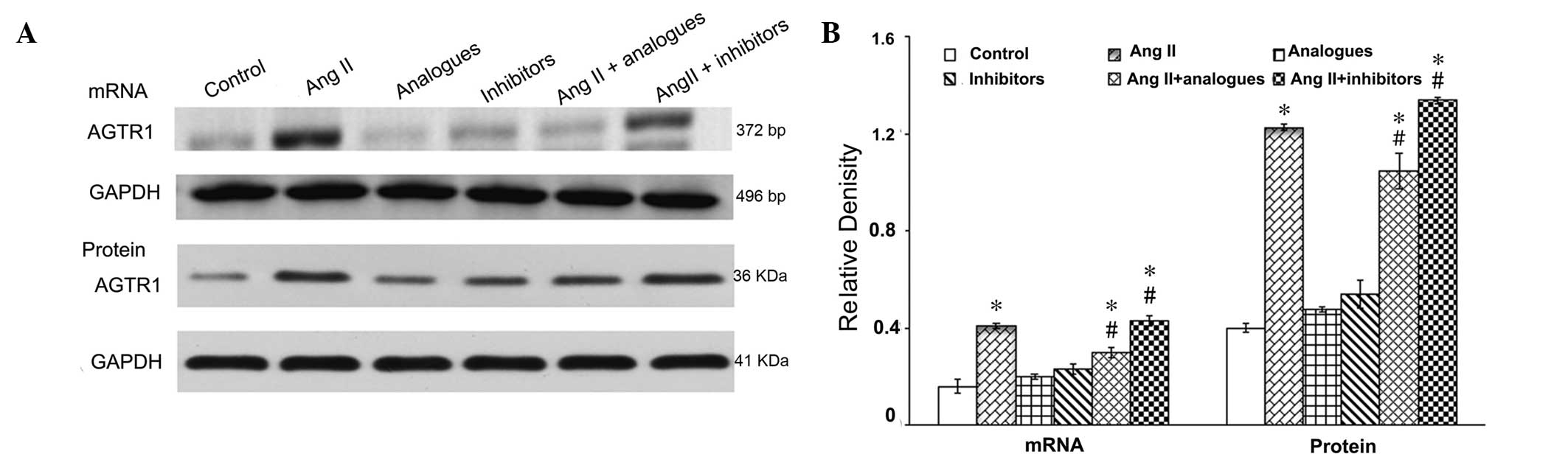
Angiotensin II-AGTR1 signaling was shown
to be markedly activated in cardiac hypertrophy (17). Therefore the effects of miR-155 on
the mRNA and protein expression levels of AGTR1 were
evaluated in the presence of angiotensin II (Fig. 2). Transfection with miR-155 analogues
or inhibitors alone had no effect on the AGTR1 mRNA and
protein expression levels (P>0.05), as detected by RT-PCR and
immunoblotting, respectively. Angiotensin II treatment
significantly increased the mRNA and protein expression leves of
AGTR1, as compared with the control cells (P<0.05).
Notably, miR-155 analogues significantly reduced the effects of
angiotensin II on miR-155 expression in cardiomyocytes (P<0.05),
but these were not restored to control levels. The mRNA and protein
expression levels of AGTR1 were significantly increased
in the angiotensin II + miR-155 inhibitor group, as compared with
the angiotensin II alone group (P<0.05).
Myocardiocyte surface area and the
mRNA expression of β-MHC and ANP
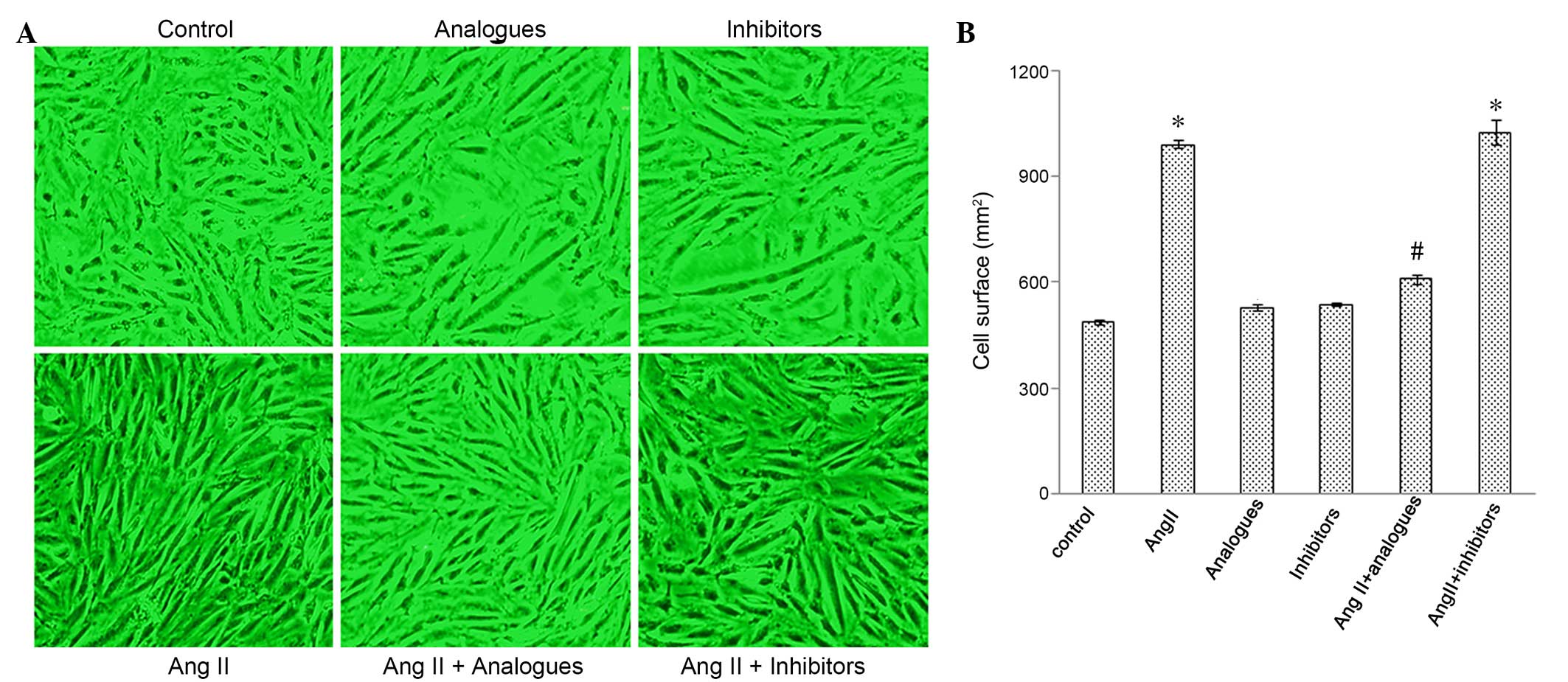
Cardiac hypertrophy is characterized by an increase
in the size of cardiomyocytes, and in the synthesis of ANP and
β-MHC (18,19). Cardiomyocytes incubated with
angiotensin II, or transfected with miR-155 analogues or inhibitors
and then treated with angiotensin II, had greater surface areas, as
compared with control cells (Fig. 3A and
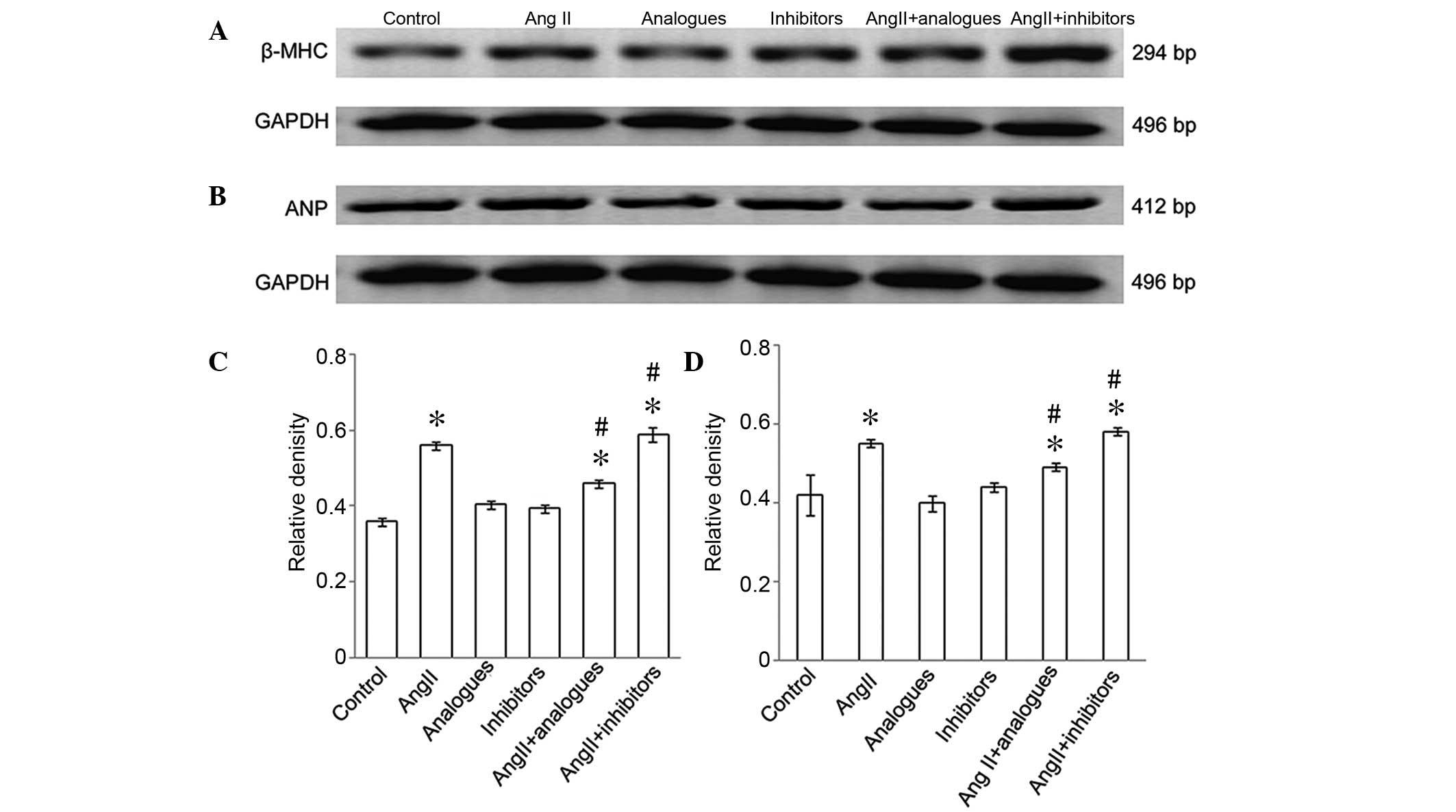
B). The mRNA expression levels of β-MHC (Fig. 4A-C) and ANP (Fig. 4B-D) were significantly increased in
the angiotensin II-treated group, as compared with the control
cells (P<0.05), whereas the mRNA expression levels of ANP and
β-MHC in cells transfected with miR-155 inhibitors or analogues
alone were not significantly different from the control cells
(P>0.05). Furthermore, β-MHC and ANP levels were significantly
decreased in the angiotensin II-treated, miR-155
analogue-transfected group, as compared with the angiotensin II
only group (P<0.05; Fig. 4B).
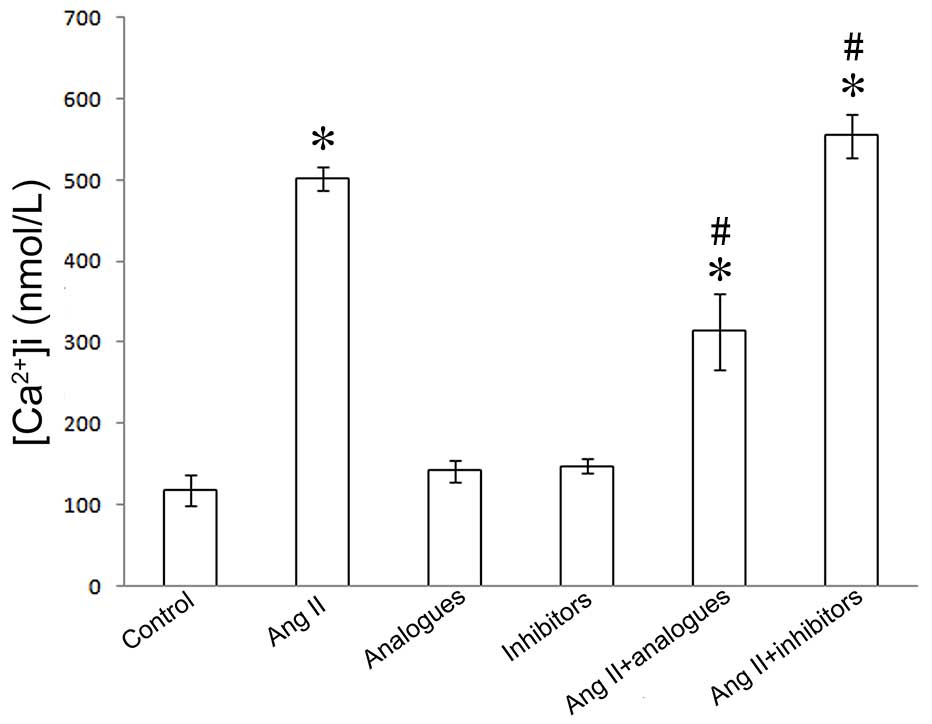
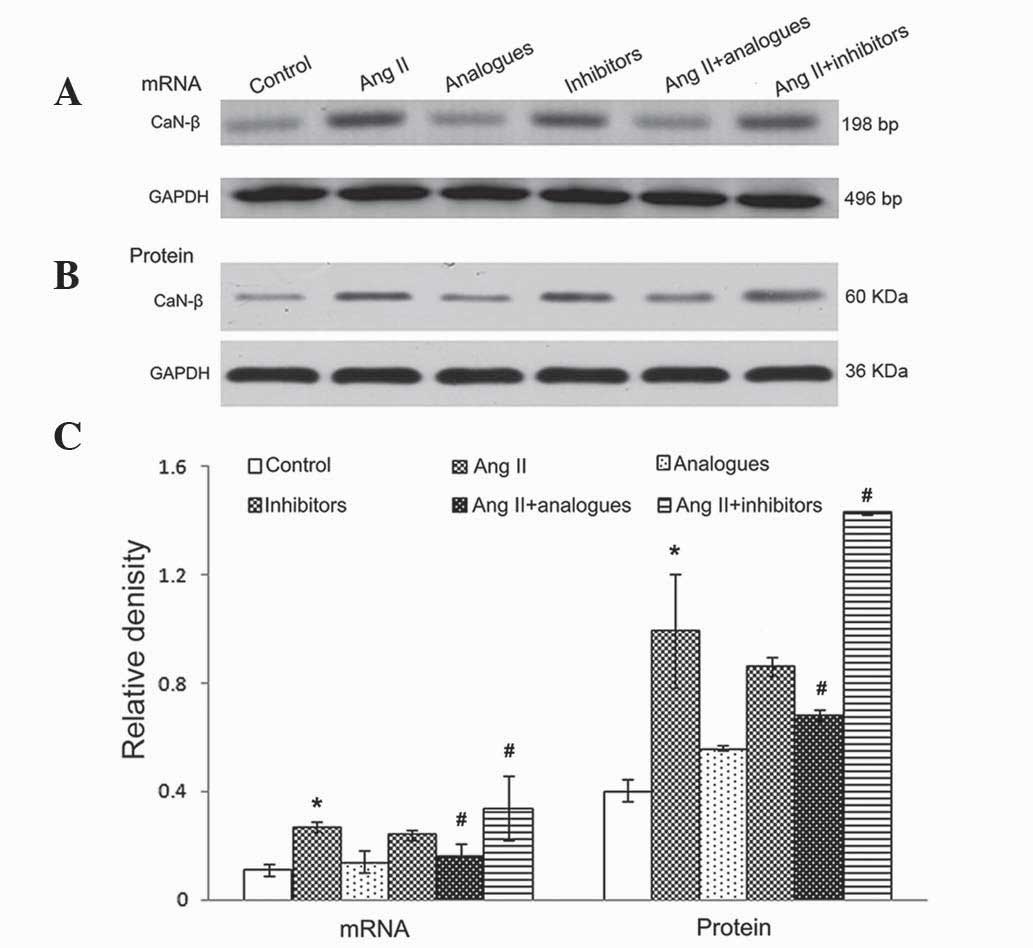
[Ca2 +]i, NFAT-4 and CaN-β
expression
In previous studies, angiotensin II was shown to
promote CaN-β-mediated calcium influx and NFAT-4 translocation into
the nucleus by activation of CaN-β in cardiomyocytes, resulting in
upregulation of the expression of ANP and β-MHC (20). Upon angiotensin II treatment, the
miR-155 analogue-transfected cells had lower intracellular calcium
levels, and NFAT-4 and CaN-β expression levels (P<0.05; Figs. 5–7).
However, treatment of miR-155 inhibitor-transfected cells with
angiotensin II caused increased intracellular calcium, NFAT-4 and
CaN-β expression (P<0.05; Fig.
5–7).
Discussion
The angiotensin II-AGTR1 and calcineurin
signal transduction pathways have vital roles in the development of
myocardial hypertrophy (21).
Angiotensin II has previously been reported to regulate the
expression of numerous miRNAs, including miR-29b, miR-129-3p,
miR-132, miR-132 and miR-212 (22).
Sayed et al (23) reported in
an in vivo model that numerous miRNAs are involved in the
regulation of calcineurin signaling, and miR-155 was central to
this cardiac hypertrophy model. Cheng et al (12) revealed that miR-155 inhibited
AGTR1 expression, possibly through binding to the 3′
non-coding region of the AGTR1 gene, resulting in
AGTR1 gene silencing.
The present study demonstrated that, compared with
normally growing cardiomyocytes, miR-155 mRNA expression increased
3–4 fold upon transfection with miR-155 analogues when combined
with angiotensin II treatment. However, transfection with miR-155
inhibitors had the opposite effect in cells treated with
angiotensin II. Overexpression of miR-155 was able to effectively
inhibit AGTR1 expression at the mRNA and protein level
in hypertrophic cardiomyocytes. When hypertrophic H9C2 (2–1) cells
were incubated with miR-155 analogues, intracellular calcium
concentrations were reduced, and those of CaN-β and NFAT-4 were
upregulated. Furthermore, cell area was increased in hypertrophic
rat cardiomyocytes, and β-MHC and ANP levels, which are indicative
of the degree of hypertrophy, were reduced. It was therefore
speculated that miR-155 may inhibit angiotensin II-induced cardiac
hypertrophy by silencing the AGTR1 and the calcineurin
signal transduction pathways. However, transfection with miR-155
inhibitors did not alter ANP or β-MHC levels and cell area in
hypertrophic H9C2 (2–1) cells, despite an increase in [Ca2+]i,
CaN-β and NFAT-4 levels.
Two online prediction tools used to determine the
miRNA targets, TargetScan and PicTar, were previously reported
(24). These revealed that
AGTR1 may be a target of miR-155. In addition,
AGTR1 was combined with angiotensin II to enhance CaN-β
(21). Myogenic enhancer factor 2,
which has been reported to be involved in the regulation of CaN-β,
is a regulatory target of miR-155 (25). Suppressor of cytokine signaling 1
from macrophages may also be a target of miR-155 and participate in
cardiac hypertrophy (8). Conversely,
angiotensin II may also elevate intracellular calcium concentration
via the phospholipase C-inositol trisphosphate signaling pathway
(26), and increase
mitogen-activated protein kinase levels to induce cardiac
hypertrophy (27). These data
indicate that multiple factors cause cardiac hypertrophy, including
the effects of AGTR1. miR-155 may therefore represent a
target at multiple sites during the development of cardiac
hypertrophy induced by angiotensin II The present study solely
investigated the role of miR-155 within hypertrophic
cardiomyocytes, however. The calcium signaling pathway may have an
important role in the development of cardiac hypertrophy, and in
the apoptosis of myocardial cells (28). In the current study, miR-155
inhibitors did not attenuate hypertrophy of H9C2 (2–1) cells.
It is possible that when miR-155 is inhibited, activation of the
calcium signaling pathway may lead to the apoptosis of a proportion
of myocardial cells, thereby reducing the overall levels of
myocardial hypertrophy markers.
In conclusion, the results of the present study
demonstrated that miR-155 overexpression effectively inhibits
angiotensin II-induced myocardial hypertrophy by preventing the
effects of AGTR1. As a result, intracellular free
calcium and NFAT-4 nuclear translocation were reduced. Additional
studies are therefore required to determine the role of miR-155 as
an important target in the treatment of cardiac hypertrophy.
Acknowledgements
The present study was supported by a grant from the
Municipal Science and Technology Bureau (grant no. ZD2012014).
References
|
1
|
Hill JA and Olson EN: Cardiac plasticity.
N Engl J Med. 358:1370–1380. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Abraham WT, Fonarow GC, Albert NM, Stough
WG, Gheorghiade M, Greenberg BH, O'Connor CM, Sun JL, Yancy CW and
Young JB: OPTIMIZE-HF Investigators and Coordinators: Predictors of
in-hospital mortality in patients hospitalized for heart failure:
Insights from the Organized Program to Initiate Lifesaving
Treatment in Hospitalized Patients with Heart Failure
(OPTIMIZE-HF). J Am Coll Cardiol. 52:347–356. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yancy CW, Jessup M, Bozkurt B, Butler J,
Casey DE Jr, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi
JL, et al: 2013 ACCF/AHA guideline for the management of heart
failure: Executive summary: A report of the American College of
Cardiology Foundation/American Heart Association Task Force on
practice guidelines. Circulation. 128:1810–1852. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Savoia C, Burger D, Nishigaki N, Montezano
A and Touyz RM: Angiotensin II and the vascular phenotype in
hypertension. Expert Rev Mol Med. 13:e112011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Small EM and Olson EN: Pervasive roles of
microRNAs in cardiovascular biology. Nature. 469:336–342. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Faraoni I, Antonetti FR, Cardone J and
Bonmassar E: miR-155 gene: A typical multifunctional microRNA.
Biochim Biophys Acta. 1792:497–505. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Seok HY, Chen J, Kataoka M, Huang ZP, Ding
J, Yan J, Hu X and Wang DZ: Loss of microRNA-155 protects the heart
from pathological cardiac hypertrophy. Circ Res. 114:1585–1595.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Heymans S, Corsten MF, Verhesen W, Carai
P, van Leeuwen RE, Custers K, Peters T, Hazebroek M, Stöger L,
Wijnands E, et al: Macrophage microRNA-155 promotes cardiac
hypertrophy and failure. Circulation. 128:1420–1432. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sethupathy P, Borel C, Gagnebin M, Grant
GR, Deutsch S, Elton TS, Hatzigeorgiou AG and Antonarakis SE: Human
microRNA-155 on chromosome 21 differentially interacts with its
polymorphic target in the AGTR1 3′ untranslated region: A mechanism
for functional single-nucleotide polymorphisms related to
phenotypes. Am J Hum Genet. 81:405–413. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Marian AJ: Experimental Therapies in
Hypertrophic Cardiomyopathy. J Cardiovasc Transl Res. 2:483–492.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zheng L, Xu CC, Chen WD, Shen WL, Ruan CC,
Zhu LM, Zhu DL, Gao PJ, et al: MicroRNA-155 regulates angiotensin
II type 1 receptor expression and phenotypic differentiation in
vascular adventitial fibroblasts. Biochem Biophys Res Commun.
400:483–488. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheng W, Liu T, Jiang F, Liu C, Zhao X,
Gao Y, Wang H, Liu Z, et al: microRNA-155 regulates angiotensin II
type 1 receptor expression in umbilical vein endothelial cells from
severely pre-eclamptic pregnant women. Int J Mol Med. 27:393–399.
2011.PubMed/NCBI
|
|
13
|
Gómez AM, Ruiz-Hurtado G, Benitah JP and
Domínguez-Rodríguez A: Ca(2+) fluxes involvement in gene expression
during cardiac hypertrophy. Curr Vasc Pharmacol. 11:497–506. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hogan PG, Chen L, Nardone J and Rao A:
Transcriptional regulation by calcium, calcineurin, and NFAT. Genes
Dev. 17:2205–2232. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Varkonyi-Gasic E1, Wu R, Wood M, Walton EF
and Hellens RP: Protocol: A highly sensitive RT-PCR method for
detection and quantification of microRNAs. Plant Methods. 3:122007.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Saris JJ, 't Hoen PA, Garrelds IM, Dekkers
DH, den Dunnen JT, Lamers JM and Jan Danser AH: Prorenin induces
intracellular signaling in cardiomyocytes independently of
angiotensin II. Hypertension. 48:564–571. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cavallero S, González GE, Puyó AM, Rosón
MI, Pérez S, Morales C, Hertig CM, Gelpi RJ and Fernández BE:
Atrial natriuretic peptide behaviour and myocyte hypertrophic
profile in combined pressure and volume-induced cardiac
hypertrophy. J Hypertens. 25:1940–1950. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Haddad F, Qin AX, Bodell PW, Zhang LY, Guo
H, Giger JM and Baldwin KM: Regulation of antisense RNA expression
during cardiac MHC gene switching in response to pressure overload.
Am J Physiol Heart Circ Physiol. 290:H2351–H2361. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lunde IG, Kvaløy H, Austbø B, Christensen
G and Carlson CR: Angiotensin II and norepinephrine activate
specific calcineurin-dependent NFAT transcription factor isoforms
in cardiomyocytes. J Appl Physiol. 111:1278–1289. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Marian AJ: Hypertrophic cardiomyopathy:
From genetics to treatment. Eur J Clin Invest. 40:360–369. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jeppesen PL, Christensen GL, Schneider M,
Nossent AY, Jensen HB, Andersen DC, Eskildsen T, Gammeltoft S,
Hansen JL and Sheikh SP: Angiotensin II type 1 receptor signalling
regulates microRNA differentially in cardiac fibroblasts and
myocytes. Br J Pharmacol. 164:394–404. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sayed D, Hong C, Chen IY, Lypowy J and
Abdellatif M: MicroRNAs play an essential role in the development
of cardiac hypertrophy. Circ Res. 100:416–424. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kozomara A and Griffiths-Jones S: miRBase:
Annotating high confidence microRNAs using deep sequencing data.
Nucleic Acids Res. 42:D68–73. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Seok HY, Tatsuguchi M, Callis TE, He A, Pu
WT and Wang DZ: miR-155 inhibits expression of the MEF2A protein to
repress skeletal muscle differentiation. J Biol Chem.
286:35339–35346. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rinne A and Blatter LA: Activation of
NFATc1 is directly mediated by IP3 in adult cardiac myocytes. Am J
Physiol Heart Circ Physiol. 299:H1701–H1707. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Izumi Y, Kim S, Zhan Y, Namba M, Yasumoto
H and Iwao H: Important role of angiotensin II-mediated c-Jun
NH(2)-terminal kinase activation in cardiac hypertrophy in
hypertensive rats. Hypertension. 36:511–516. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu Q, Wilkins BJ, Lee YJ, Ichijo H and
Molkentin JD: Direct interaction and reciprocal regulation between
ASK1 and calcineurin-NFAT control cardiomyocyte death and growth.
Mol Cell Biol. 26:3785–3797. 2006. View Article : Google Scholar : PubMed/NCBI
|