Introduction
The incidence of recurrent abortion is approximately
10–15% among all pregnancies. The rate of recurrent pregnancy loss
can be as high as 50%, and recurrent spontaneous abortion brings
serious injury and distress to the pregnant woman and her family
(1). There are a number reasons for
the medical condition including genetic defects, reproductive
system abnormalities, endocrine disorders, immune disorders,
infection, thrombosis and environmental factors (2). In the clinic, the cause of pregnancy
loss is unable to be determined in more than 55% of the patients,
which makes treatment difficulty (3). The mechanism of recurrent abortion is
considered to be related to the proliferation and apoptosis of
human decidual cells and cytotrophoblasts (4). TP53 protein can induce cell growth
arrest, apoptosis, differentiation and DNA repair, and activate or
inhibit the expression of downstream genes such as Bax and CDKN1A
(5). In the present study, we
comparatively analyzed the cell apoptosis and cell signaling
pathways of healthy patients and those with recurrent spontaneous
abortion, providing a theoretical basis for clinical treatment.
Patients and methods
Patients
A total of 34 cases diagnosed with recurrent
spontaneous abortion in our hospital were continuously selected
from January 2015 to January 2016. Exclusion criteria included: a)
no previous surgery and drug therapy; b) possibility of genetic
defects; c) reproductive system abnormalities or endocrine
disorders; d) immune system disorders or infections; e) thrombosis
and environmental factors with normal function of male reproductive
system; and f) semen and no fetal haemolytic disease. The age of
patients ranged from 23 to 37 years with a mean age of 26.6±5.2
years. Gestational weeks were 5–16 weeks, with an average of
9.2±3.7 weeks. Thirty healthy pregnant women planning an artificial
abortion were chosen as control. The controls had an age range of
20 to 35 years and an average age of 24.3±5.6 years. Gestational
age ranged from 4.5 to 12 weeks, and an average age of 8.3±3.9
weeks. The age and gestational weeks of the women were compared
between the two groups and the differences were not statistically
significant (p>0.05).
Specimen collection
The tissues were obtained through negative pressure
aspiration biopsy. Each sample was divided into two parts. One part
was frozen and stored in liquid nitrogen at −196°C to prepare for
polymerase chain reaction (PCR) experiments. Another part was fixed
at room temperature with 10% formalin and embedded with paraffin.
The section thickness was 5 µm, and was placed on a glass slide
with APES for immunohistochemistry and apoptotic staining.
Apoptosis rate as detected with
terminal deoxynucleotidyl-transferase-mediated dUTP nick end
labelling (TUNEL) method
Reagents and equipment
DeadEnd™ Fluorometric TUNEL system kit (Promega,
Madison, WI, USA), proteinase K (Biotechnology Engineering and
Technical Services Co., Shanghai, China), propidium iodide
(Sigma-Aldrich, St. Louis, MO, USA), and Slowfade® Gold
Antifade reagent (Invitrogen Life Technologies, Carlsbad, CA, USA)
were used to determine the apoptotic rate. Inverted fluorescence
microscope (Olympus, Tokyo, Japan) was used for imaging.
Procedure
Preprocessing of the tissue sections was performed
before paraffin embedding [dewaxing with xylene, graded alcohol and
phosphate-buffered saline (PBS) washing]. The tissues were
incubated in polyformaldehyde solution at room temperature for 15
min, followed by washing with PBS. The proteinase K solution (20
µg/ml) of 100 µl was added and incubation was carried out at room
temperature for 8 min, followed by another PBS washing. The
poly-formaldehyde solution was incubated at room temperature for 15
min and then washed with PBS. For apoptosis detection, each sample
was covered using 100 µl equilibrium buffer to cover the cells. The
nucleoside mixture was thawed at room temperature for 10 min and
soaked with bibulous paper. Fifty microliters of rTdT incubation
buffer was added to every 5 cm2 area of the cells,
followed by covering with a cover glass. The sections were
incubated in a humidified chamber to avoid light for incubation at
37°C for 60 min for the tailing reaction. The 20X SSC was diluted
with deionized water to 1:10, and added to cover the dye vat.
Subsequently, the reaction was stopped by placing at room
temperature for 15 min. The PBS was used again to remove the
redundant fluorescein-deoxyuridine triphosphate. Two drops of
SlowFade® Gold Antifade reagent with DAPI was added to
the sample. Finally, transparent nail polish was used for slide
mounting.
To assess the staining, green staining of the
nucleus was indicative of an apoptotic cell under fluorescence
microscopy. Three fields were selected for each sample (100x
magnification). Each field was photographed under a fluorescence
microscope at 520-nm wavelength (green TUNEL) and at 430-nm
wavelength (bluish violet DAPI). Photoshop CS5 was used to merge
the two images for the final fluorescence analysis with ImagePro
Plus 6.0.
Detection of TP53 protein with
immunohistochemistry (IHC)
Reagents and equipment
Mouse anti-human TP53 monoclonal antibody (1:300;
Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA), goat
anti-mouse antibody (GBI Labs, Mukilteo, WA, USA) and DAB
(ZSGB-BIO, Beijing, China) were used for IHC. Leica RM2245 paraffin
slicing machine (Leica, Shanghai, China) was utilized.
Procedure
The procedure for IHC was followed as described
elsewhere (6). The positive tissue
in the positive control group was used as the control group. The
PBS buffer was used instead of the primary antibody in the negative
control with other operating procedures unchanged. Each sample was
observed by taking images from three horizons at 200x
magnification. The images in each field were calculated from the
gray value by using ImageJ 1.44 software, and then the average
value of three horizons was taken.
Detection of the relative expression
of CDKN1A and BAX mRNA as determined by real-time quantitative PCR
method
Main reagents and instruments
TRIzol (Invitrogen Life Technologies), reverse
transcription kit (Takara, Dalian, China), DNAmarker Marker III,
50-bp ladder, 1-kb ladder, Lamda-HindIII fragment (Fermentas
International, Inc., Burlington, ON, Canada), SYBR-Green Mix
(Roche, Basel, Switzerland). PTC-220 PCR instrument (MJ Research
Inc., Waltham, MA, USA), GIS-1000 Gel imaging system (Shanghai
Quanray Electronics Co., Shanghai, China), real-time PCR instrument
(Applied Biosystems Life Technologies, Foster City, CA, USA), Model
650–60 fluorescence spectrophotometer (Hitachi, Ltd., Tokyo,
Japan), ZF-90 Multifunctional box type UV transilluminators
(Gu-cun, Shanghai, CN) and a 96-well culture dish (Applied
Biosystems Life Technologies) were used for real-time quantitative
PCR method.
Procedure
The conventional TRIzol method was used to extract
RNA. The concentration and purity of the RNA were determined by UV
spectrophotometer, and cDNA was synthesized with the kit. The
primers were designed and synthesized by Takara and were: CDKN1A
forward, 5′-GCAGCGGAACAAGGAGT, 3′-GGAGAAACGGGAACCAG; BAX forward,
5′-CCCCCGAGAGGTCTTTTTCC, 3′-TGTCCAGCCCATGATGGTTC; internal
reference GAPDH forward, 5′-GGCTACAGCAACAGGGTG,
3′-TTTGGTTGAGCACAGGGT.
The PCR reaction system consists of SYBR®
Premix Ex Taq™ [2x (12.5 µl)], primers 10 µM (1 µl),
ddH2O (10.5 µl) to make a final volume of 25 µl. The
reaction conditions consisted of denaturation at 95°C for 30 sec,
95°C for 3 sec, annealing at 60°C for 30 sec, extension at 84°C for
1 sec for up to 40 cycles. The fluorescence intensity was measured
at 84°C to construct the dissolution curve. The relative expression
of mRNA was calculated using the 2−∆∆Ct method.
Statistical analysis
SPSS statistical software was used for data input
and analysis. The quantitative data are represented as mean ±
standard deviation, and t-test was applied for the intergroup
comparison. Qualitative data are expressed as number or percentage
(%), and the comparison between groups was carried out using the
χ2 test. p<0.05 indicates a statistically significant
difference.
Results
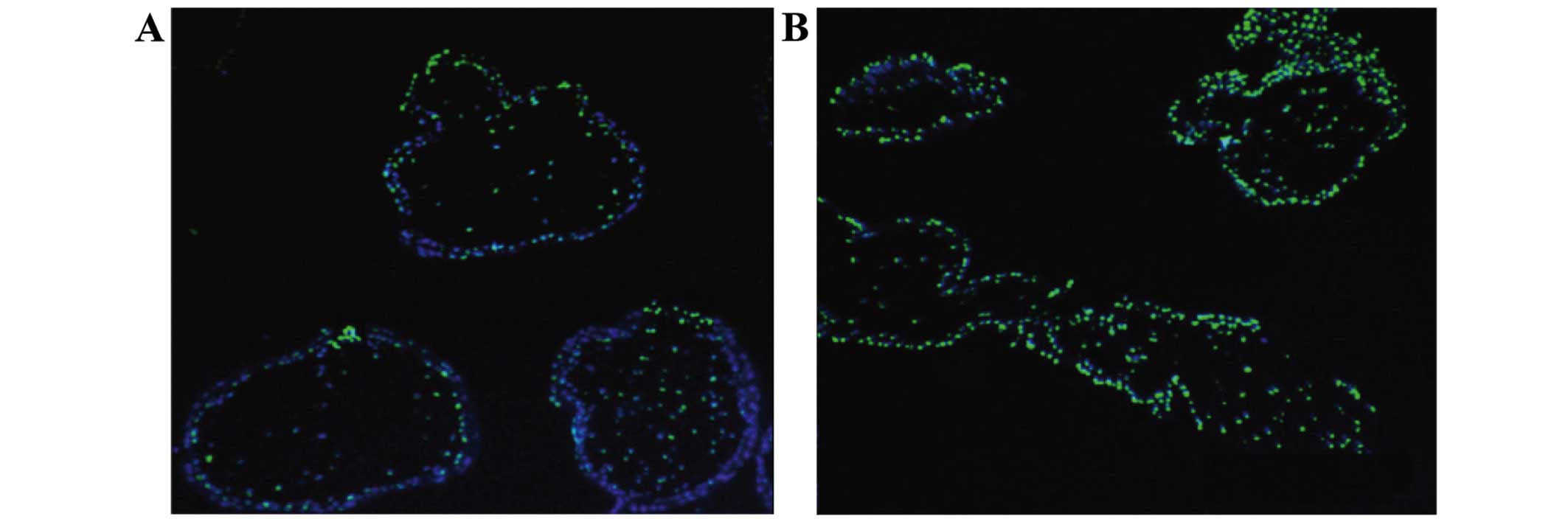
Comparison of the apoptosis rate
The apoptosis rate in the spontaneous abortion group
was significantly increased as compared to the rate noted in the
healthy control group (Fig. 1).
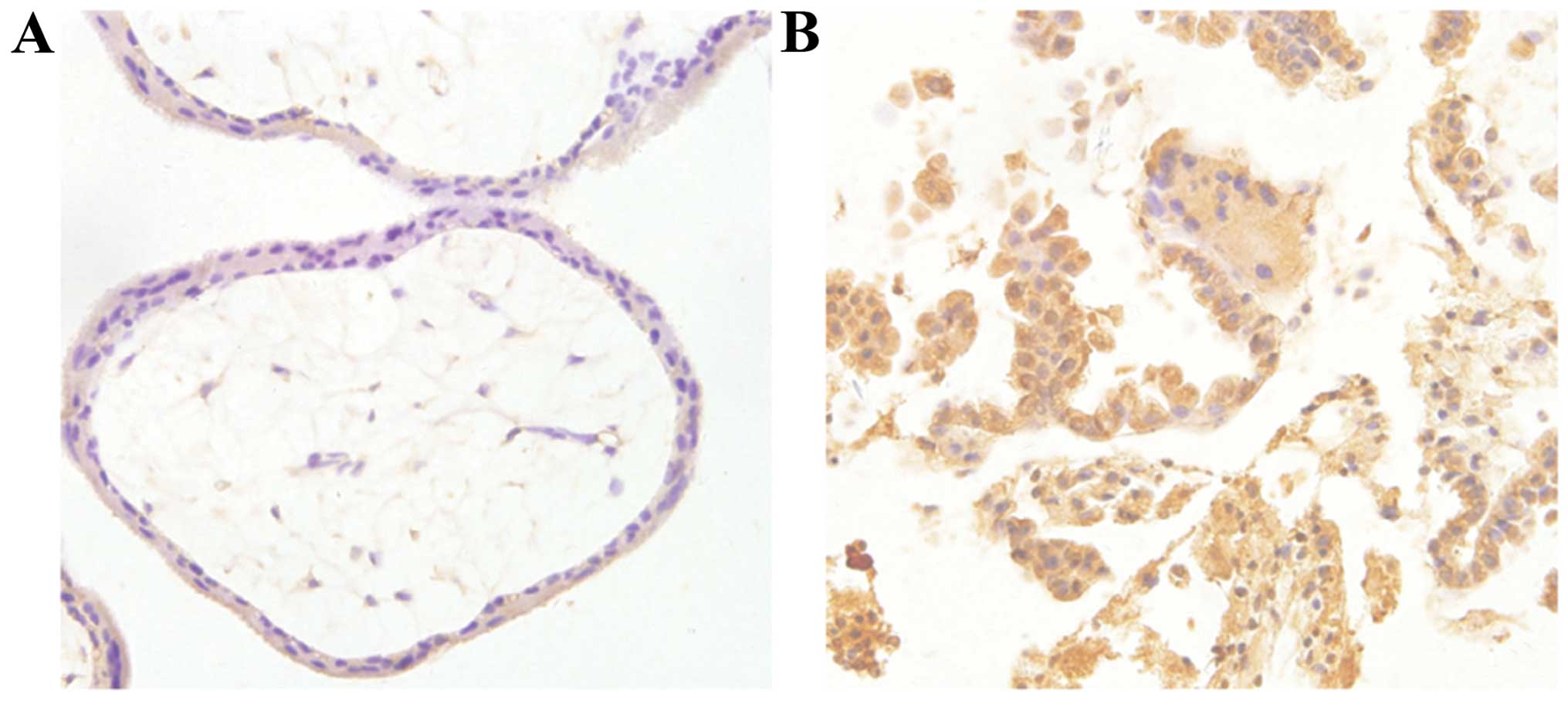
Comparison of TP53 protein
expression
TP53 protein was mainly expressed in the nucleus,
shown as brown or brownish yellow granules. The TP53 protein
expression level in the recurrent abortion group was significantly
increased as compared to the expression noted in the healthy
control group (Fig. 2).
Comparison of the relative expression
of CDKN1A and BAX mRNA
The expression levels of CDKN1A and BAX mRNA were
significantly upregulated in the recurrent abortion group
(p<0.05) as compared with levels in the healthy control group
(Table I).
 | Table I.Comparison of the relative expression
of CDKN1A and BAX mRNA. |
Table I.
Comparison of the relative expression
of CDKN1A and BAX mRNA.
| Group | CDKN1A | BAX |
|---|
| Control | 0.42±0.09 | 0.46±0.08 |
| Recurrent
abortion | 0.13±0.05 | 0.15±0.06 |
| T | 7.528 | 7.629 |
| P-value | 0.009 | 0.006 |
Discussion
Currently, treatment for recurrent spontaneous
abortion includes progesterone and hCG supplementation therapy,
immune therapy for blocking antibody-negative patients, antibody
therapy, coagulopathy for those with anti-phospholipid antibody
syndrome, aspirin and heparin treatment for those with coagulation
disorder. The methods are unsatisfactory, and the mechanisms of
recurrent spontaneous abortion remain unclear.
The apoptosis and proliferation of placenta villi
and decidua during cell normal pregnancy is in a relative state of
balance, and the apoptosis rates of decidual cells in women with
repeated abortion are significantly higher than those of normal
pregnant women. Excessive apoptosis can lead to a series of
cellular dysfunction and eventually threaten the entire process of
pregnancy (7). During a study of the
mechanism of spontaneous abortion in pregnant women exposed to
PCBs, it was found that PCBs induced abnormal gC1qR expression
resulting in trophoblast cell apoptosis eventually leading to the
occurrence of spontaneous abortion (8). The apoptotic phenomena of villus
trophoblast cells and decidual cells were noted in both patients
with recurrent abortion and those with healthy pregnancy and the
apoptosis rate in the recurrent abortion group was significantly
higher. The expression levels of TP53, CDKN1A and Bax mRNA were
significantly increased. Whether the high expression level of the
TP53 protein was caused by the high level of upregulation of the
transcription level or TP53 post-transcriptional modification
warrants further study. TP53 protein is a typical inhibitor protein
of the G1 phase of the cell cycle (9). When cells are damaged, TP53 encodes
transcription activated protein that can be quickly assembled in
the DNA damaged location, resulting in termination of the cell
cycle in the G1 phase and DNA replication. The damaged DNA is then
repaired. If the cell repair cannot be completed, the cell tends to
undergo apoptosis mediated by TP53 protein (10). When the cells are damaged and cannot
complete the TP53-mediated apoptosis, the genetic material of the
cell may change and ultimately lead to malignant transformation.
The TP53 gene is highly correlated with human tumors (11).
The cell signal transduction pathway mediated by
TP53 plays an important role in the regulation of normal cell
activities, and cell contact between other signal transduction
pathways is very complex. Thus, TP53 is included in the regulation
for more than 160 genes (12).
CDKN1A protein is the downstream transcription protein of the TP53
gene. The rpL3 gene was found to regulate TP53-CDKN1A so as to
block cell cycle and promote cell apoptosis (13). Rabdosia rubescens was found to
increase CDKN1A expression and promote autophagy and apoptosis in
prostate cancer cells (14). BAX has
a pro-apoptotic effect that can directly activate the mitochondrial
apoptosis pathway activated by TP53 (15). BCL2 and BAX are two important members
of the BCL2 family, and BCL2 is an anti-apoptotic molecule. The Bax
expression levels in trophoblast cells, endometrial cells, stroma
and decidual cells in patients with early spontaneous abortion were
found to be significantly higher than those in women with voluntary
terminated pregnancy (16). The
expression of Bax in placenta of preeclampsia was also higher than
normal term placenta and Bax expression was lower in placenta of
patients with diabetes that of the normal term placenta. In
addition, melatonin was found to decrease the BCL2 expression level
and increase the expression level of Bax in a study of in
vitro mouse xenograft melatonin therapy, so as to be
therapeutic through apoptosis and necrosis and also to prove the
BCL2/Bax balance relationship (17).
In conclusion, the apoptosis rate of human decidual
cells and cytotrophoblasts in patients with recurrent abortion was
increased, which may be related to the abnormal expression of
CDKN1A and BAX genes in the downstream mediated by TP53
protein.
Acknowledgements
The present study was funded by the Shandong
Province Outstanding Young Scientist Award Fund BS2014YY058.
References
|
1
|
Saravelos SH, Cocksedge KA and Li TC: The
pattern of pregnancy loss in women with congenital uterine
anomalies and recurrent miscarriage. Reprod Biomed Online.
20:416–422. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Franssen MT, Musters AM, van der Veen F,
Repping S, Leschot NJ, Bossuyt PM, Goddijn M and Korevaar JC:
Reproductive outcome after PGD in couples with recurrent
miscarriage carrying a structural chromosome abnormality:
asystematic review. Hum Reprod Update. 17:467–475. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mekinian A, Cohen J, Alijotas-Reig J,
Carbillon L, Nicaise- Roland P, Kayem G, Daraï E, Fain O and Bornes
M: Unexplained recurrent miscarriage and recurrent implantation
failure: is there a place for immunomodulation? Am J Reprod
Immunol. 5:13–15. 2016.
|
|
4
|
Cinar O, Kara F and Can A: Potential role
of decidual apoptosis in the pathogenesis of miscarriages. Gynecol
Endocrinol. 28:382–385. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tang W, Zhou X, Chan Y, Wu X and Luo Y:
p53 codon 72 polymorphism and recurrent pregnancy loss: a
meta-analysis. J Assist Reprod Genet. 28:965–969. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
McNamee K, Dawood F and Farquharson R:
Recurrent miscarriage and thrombophilia: an update. Curr Opin
Obstet Gynecol. 24:229–234. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Seyedhassani S Mohammad, Houshmand M,
Kalantar S Mehdi, Aflatoonian A, Modabber G, Gorgi F Hashemi and
Hadipour Z: BAX pro-apoptotic gene alterations in repeated
pregnancy loss. Arch Med Sci. 7:117–122. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gu PQ, Gao LJ, Li L, Liu Z, Luan FQ, Peng
YZ and Guo XR: Endocrine disruptors, polychlorinated
biphenyls-induced gC1qR-dependent apoptosis in human trophoblast
cell line HTR-8/SVneo. Reprod Sci. 19:181–189. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kaare M, Bützow R, Ulander VM, Kaaja R,
Aittomäki K and Painter JN: Study of p53 gene mutations and
placental expression in recurrent miscarriage cases. Reprod Biomed
Online. 18:430–435. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kruse JP and Gu W: Modes of p53
regulation. Cell. 137:609–622. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Carp HJ: Recurrent miscarriage: genetic
factors and assessment of the embryo. Isr Med Assoc J. 10:229–231.
2008.PubMed/NCBI
|
|
12
|
Jacob T, Hingorani A and Ascher E: p53
gene therapy modulates signal transduction in the apoptotic and
cell cycle pathways downregulating neointimal hyperplasia. Vasc
Endovascular Surg. 46:45–53. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Russo A, Esposito D, Catillo M,
Pietropaolo C, Crescenzi E and Russo G: Human rpL3 induces
G1/S arrest or apoptosis by modulating p21 (waf1/cip1)
levels in a p53-independent manner. Cell Cycle. 12:76–87. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li X, Li X, Wang J, Ye Z and Li JC:
Oridonin up-regulates expression of P21 and induces autophagy and
apoptosis in human prostate cancer cells. Int J Biol Sci.
8:901–912. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cobellis L, De Falco M, Torella M,
Trabucco E, Caprio F, Federico E, Manente L, Coppola G, Laforgia V,
Cassandro R, et al: Modulation of Bax expression in physiological
and pathological human placentas throughout pregnancy. In Vivo.
21:777–783. 2007.PubMed/NCBI
|
|
16
|
ESHRE Capri Workshop Group, . Genetic
aspects of female reproduction. Hum Reprod Update. 14:293–307.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu C, Wu A, Zhu H, Fang H, Xu L, Ye J and
Shen J: Melatonin is involved in the apoptosis and necrosis of
pancreatic cancer cell line SW-1990 via modulating of Bcl-2/Bax
balance. Biomed Pharmacother. 67:133–139. 2013. View Article : Google Scholar : PubMed/NCBI
|