Introduction
Lower respiratory tract infection (LRTI) is mostly
induced by infection or invasion of outside pathogenic
microorganisms, such as bacteria, virus and chlamydia, into the
human body. LRTI is the most common frequently occurring disease
(1) and it is often complicated by
pneumonia, bronchiectasis, bronchitis and other clinical symptoms,
posing a severe threat to body health and quality of life of
patients (2).
Research shows that white blood cell (WBC) count and
C-reactive protein (CRP) increase abnormally when there is an
infection in the human body. Subsequently, WBC and CRP are two
important reference indexes for diagnosing microbial infection and
judging treatment effect, which are closely related to the severity
degree of patients' illness (3,4). With
the continuous development of economy, the needs for health are
increasing daily, while an increasing variety of antibiotics for
treating LRTI and patients' wish for a speedy recovery lead to very
severe clinical abuse of antibiotics. Injection of huge amounts of
antibiotics into infected persons by medical workers results in a
gradual enhanced drug resistance to LRTI pathogens, which not only
influences the treatment effect on patients, but also gives rise to
a continuous growth in the number of patients with nosocomial
infection (5). Moreover, antibiotics
are expensive, causing certain economic burden to patients and
their families. LRTI is dominated by gram-negative bacterial
infections, which are sensitive to broad-spectrum antibiotics, such
as penicillins, ceftizoxime sodium, levofloxacin and azithromycin
(6).
In the present study, four different antibiotic
regimens including ceftriaxone sodium, ceftizoxime sodium,
levofloxacin and azithromycin were applied to treat 200 LRTI
patients, to determine the effects and costs of different
antibiotic regimens in the treatment of LRTI.
Materials and methods
General data
A total of 200 patients with LRTI treated in the
Department of Respiratory Medicine of Dongying People's Hospital
(Dongying, China) from February 2015 to May 2017 were selected and
retrospectively analyzed. The patients were randomly divided into
group A (n=50), group B (n=50), group C (n=50) and group D (n=50).
In group A, there were 21 males and 29 females aged 16–59 years,
with an average age of (35.44±16) years and an average course of
disease of 8.46±6.62 days. In group B, there were 23 males and 27
females aged 18–65 years, with an average age of 35.14±2.01 years
and an average course of disease of 7.87±4.49 days. In group C,
there were 19 males and 31 females aged 20–63 years, with an
average age of 34.52±2.35 years and an average course of disease of
7.73±5.96 days. In group D, there were 24 males and 26 females aged
19–66 years, with an average age of 34.68±2.46 years and an average
course of disease of 8.33±6.34 days.
Inclusion and exclusion criteria
Inclusion criteria were: patients with definite
symptoms of LRTI and diagnosed via chest X-ray examination,
patients without receiving treatment within 1 month, patients
without a history of antibiotic therapy, and patients who were
informed, agreed to participate in the clinical study and signed
the informed consent. Exclusion criteria were: patients with
unconsciousness, confusion, complicated with severe heart, liver,
kidney or hematopoietic dysfunction, mental diseases or a family
history of mental illness or allergy to drugs, or pregnant or
breastfeeding women. The present study was approved by the Ethics
Committee of Dongying People's Hospital (Shandong, China) and
signed written informed consents were obtained from the patients
and/or guardians
Drug therapy
Patients in the 4 groups were treated with
ceftriaxone sodium (cat. no. H10983036; Shanghai Roche
Pharmaceutical Co., Ltd., Shanghai, China), ceftizoxime sodium
(cat. no. H20060339; Suzhou Erye Pharmaceutical Co., Ltd., Suzhou,
China), levofloxacin (cat. no. H20060508; Shandong Luoxin
Pharmaceutical Group Ltd., Shandong, China), azithromycin (cat. no.
H20010554; Xi'an Daheng Pharmaceutical Co., Ltd., Shaanxi, China)
as follows: intravenous infusion of 2.0 g ceftriaxone sodium
injection + 100 ml 9% sodium chloride injection (2 times/day),
intravenous infusion of 2.0 g ceftizoxime sodium + 100 ml 9% sodium
chloride injection (2 times/day), intravenous infusion of 0.3 g
levofloxacin + 100 ml 9% sodium chloride injection (2 times/day),
and intravenous infusion of 1.0 g azithromycin + 100 ml 9% sodium
chloride injection (2 times/day). All patients were treated for 1
week as 1 course of treatment.
Detection of WBC and CRP
Venous blood was drawn (4 ml) from patients at 1 day
before treatment and at 1, 4 and 7 days after treatment for WBC
counting and CRP detection. WBC in the venous blood was detected
using the DxH800 blood analyzer (Beckman Coulter, Inc., Shanghai,
China), while CRP in the venous blood was detected via latex
immunoturbidimetry using the AU5800 full-automatic biochemical
analyzer (Beckman Coulter, Inc.), strictly according to
instructions of the instrument and kit.
Observation indexes
After treatment for 7 days, the therapeutic effect
of patients was evaluated and scored. Cured: lesions and clinical
symptoms completely disappeared according to X-ray examination,
there were no adverse reactions, and the quality of life returned
to normal. Effective: Lesions and clinical symptoms were greatly
improved according to X-ray examination, there were no adverse
reactions, and the quality of life was obviously improved.
Ineffective: lesions and clinical symptoms were not improved or
exacerbated according to X-ray examination, there were adverse
reactions, and the quality of life was poor. Total effective rate =
(cured cases + effective cases)/no. of cases × 100%. Treatment
costs were recorded.
Statistical analysis
Statistical analysis was performed using SPSS v.17.0
(SPSS, Inc., Chicago, IL, USA). Measurement data were presented as
mean ± standard deviation (mean ±SD). One-way analysis of variance
followed by post hoc test (Least Significant Difference) was used
for the comparison of means among groups, and repeated measures
analysis of variance was used for the data comparison at different
time-points within the group. The Chi-square test was used for
enumeration data. P<0.05 was considered to indicate a
statistically significant difference.
Results
General clinical data of patients in
the 4 groups
There were no statistically significant differences
in sex, age, course of disease, red blood cell (RBC) count,
platelet (PLT) count and hemoglobin (Hb) among the 4 groups
(P>0.05; Table I).
 | Table I.General clinical data of patients in
the 4 groups (mean ± SD). |
Table I.
General clinical data of patients in
the 4 groups (mean ± SD).
| Item | Group A (n=50) | Group B (n=50) | Group C (n=50) | Group D (n=50) | F/χ2 | P-value |
|---|
| Sex [n(%)] |
|
|
|
| 1.200 | 0.752 |
| Male | 21 (42.00) | 23 (46.00) | 19 (38.00) | 24 (48.00) |
|
|
|
Female | 29 (58.00) | 27 (54.00) | 31 (62.00) | 26 (52.00) |
|
|
| Age (years) | 35.44±2.16 | 35.14±2.01 | 34.52±2.35 | 34.68±2.46 | 1.755 | 0.157 |
| Course of disease
(days) | 8.46±6.62 | 7.87±4.49 | 7.73±5.96 | 8.33±6.34 | 0.177 | 0.911 |
| RBC
(×1012/l) | 4.64±0.43 | 4.61±0.38 | 4.51±0.31 | 4.53±0.53 | 1.102 | 0.349 |
| PLT
(×109/l) | 183.72±29.45 | 188.7±32.84 | 191.53±28.77 | 185.16±33.67 | 0.635 | 0.592 |
| Hb (g/l) | 134.57±6.93 | 133.75±7.65 | 135.59±6.11 | 132.46±6.01 | 1.946 | 0.123 |
WBC counts in the 4 groups of patients
before and after treatment
No statistically significant differences were found
in the WBC count in patients among the 4 groups at 1 day before
treatment and at 1, 4 and 7 days after treatment (P>0.05).
Compared with those at 1 day before treatment, WBC counts in
patients in the 4 groups were significantly decreased at 1, 4 and 7
days after treatment (P<0.01). Compared with those at 1 day
after treatment, WBC counts in patients in the 4 groups were
significantly decreased at 4 and 7 days after treatment
(P<0.01). Compared with those at 4 days after treatment, WBC
counts in patients in the 4 groups were obviously decreased at 7
days after treatment (P<0.01; Table
II).
 | Table II.Comparisons of WBC count among the 4
groups of patients before and after treatment (×109/l)
(mean ± SD). |
Table II.
Comparisons of WBC count among the 4
groups of patients before and after treatment (×109/l)
(mean ± SD).
| Time | Group A (n=50) | Group B (n=50) | Group C (n=50) | Group D (n=50) | F | P-value |
|---|
| 1 day before
treatment | 15.35±4.39 | 16.23±5.34 | 15.63±4.34 | 16.63±5.33 | 0.703 | 0.551 |
| 1 day after
treatment |
12.96±3.69a |
12.28±3.94a |
12.85±4.01a |
11.98±4.25a | 0.686 | 0.561 |
| 4 days after
treatment |
9.13±2.54a,b |
10.25±2.43a,b |
9.25±2.25a,b |
9.42±2.54a,b | 2.144 | 0.096 |
| 7 days after
treatment |
7.51±2.13a–c |
7.26±2.38a–c |
7.36±2.16a–c |
6.97±2.37a–c | 0.506 | 0.678 |
| F | 58.060 | 50.980 | 61.040 | 58.240 | – | – |
| P-value | P<0.001 | P<0.001 | P<0.001 | P<0.001 | – | – |
CRP in the 4 groups of patients before
and after treatment
There were no statistically significant differences
in the CRP measured value in patients among the 4 groups at 1 day
before treatment and at 1, 4 and 7 days after treatment
(P>0.05). Compared with those at 1 day before treatment, CRP
measured values in patients in the 4 groups were obviously
decreased at 1, 4 and 7 days after treatment (P<0.01). Compared
with those at 1 day after treatment, CRP measured values in
patients in the 4 groups were obviously decreased at 4 and 7 days
after treatment (P<0.01). Compared with those at 4 days after
treatment, CRP measured values in patients in the 4 groups were
significantly decreased at 7 days after treatment (P<0.01;
Table III).
 | Table III.Comparisons of CRP among the 4 groups
of patients before and after treatment (mg/l) (mean ± SD). |
Table III.
Comparisons of CRP among the 4 groups
of patients before and after treatment (mg/l) (mean ± SD).
| Time | Group A (n=50) | Group B (n=50) | Group C (n=50) | Group D (n=50) | F | P-value |
|---|
| 1 day before
treatment | 49.36±19.35 | 54.28±20.27 | 52.17±22.63 | 56.47±18.63 | 1.062 | 0.366 |
| 1 day after
treatment |
44.36±14.53a |
39.85±13.74a |
42.58±14.29a |
41.83±14.09a | 0.868 | 0.458 |
| 4 days after
treatment |
21.26±11.29a,b |
19.67±10.37a,b |
20.87±9.38a,b |
22.42±8.13a,b | 0.661 | 0.577 |
| 7 days after
treatment |
2.19±0.87a–c |
2.23±0.93a–c |
2.11±1.01a–c |
2.35±0.96a–c | 0.561 | 0.641 |
| F | 133.500 | 146.900 | 125.000 | 180.700 | – | – |
| P-value | P<0.001 | P<0.001 | P<0.001 | P<0.001 | – | – |
Therapeutic effects in the 4 groups of
patients
No adverse reactions occurred in the 4 groups of
patients during treatment. In Group A, there were 21 cured cases
(42.00%), 26 effective cases (52.00%) and 3 ineffective cases
(6.00%), with a total effective rate of 94.00%. In Group B, there
were 19 cured cases (38.00%), 27 effective cases (54.00%) and 4
ineffective cases (8.00%), with a total effective rate of 92.00%.
In Group C, there were 24 cured cases (48.00%), 24 effective cases
(48.00%) and 2 ineffective case (4.00%), with a total effective
rate of 96.00%. In Group D, there were 20 cured cases (40.00%), 25
effective cases (50.00%) and 5 ineffective cases (10.00%), with a
total effective rate of 90.00%. The total effective rate indicated
no statistically significant difference among the 4 groups of
patients (P>0.05; Table IV).
 | Table IV.Comparisons of therapeutic effects
among the 4 groups of patients [n (%)]. |
Table IV.
Comparisons of therapeutic effects
among the 4 groups of patients [n (%)].
| Group | n | Cured | Effective | Ineffective | Total effective
rate |
|---|
| Group A | 50 | 21 (42.00) | 26 (52.00) | 3 (6.00) | 47 (94.00) |
| Group B | 50 | 19 (38.00) | 27 (54.00) | 4 (8.00) | 46 (92.00) |
| Group C | 50 | 24 (48.00) | 24 (48.00) | 2 (4.00) | 48 (96.00) |
| Group D | 50 | 20 (40.00) | 25 (50.00) | 5 (10.00) | 45 (90.00) |
| χ2 | – | – | – | – | 1.536 |
| P-value | – | – | – | – | 0.664 |
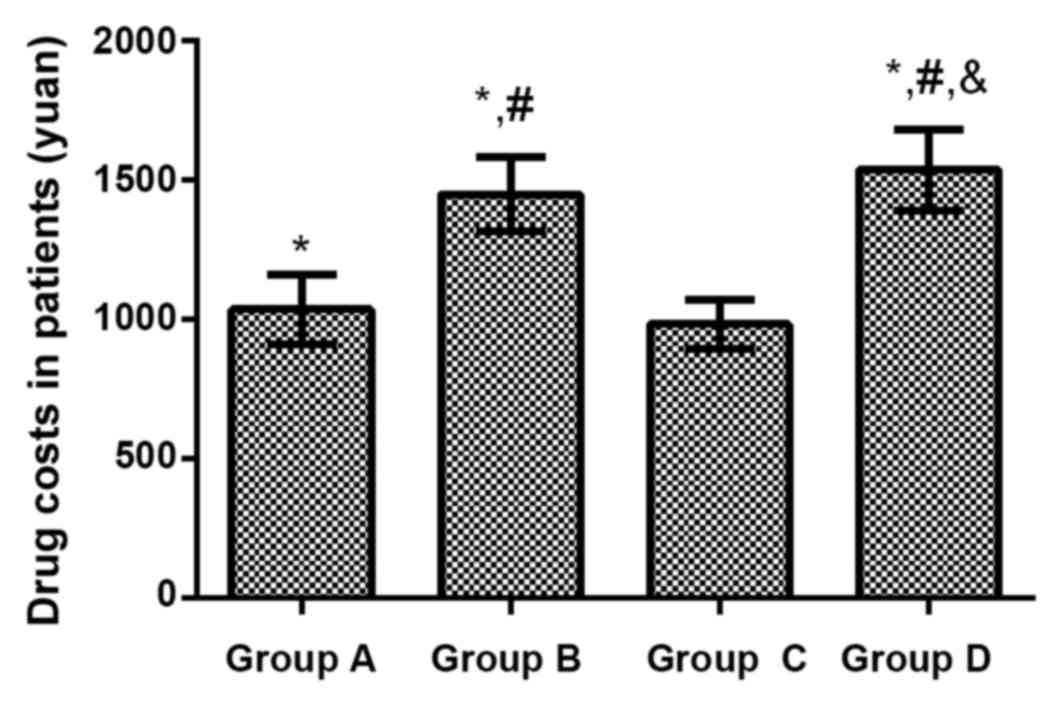
Drug costs in the 4 groups of
patients
The treatment cost was (1037.15±126.51) yuan in
Group A treated with ceftriaxone sodium, (1451.38±134.55) yuan in
Group B treated with ceftizoxime sodium, (983.67±86.37) yuan in
Group C treated with levofloxacin and (1537.45±146.59) yuan in
Group D treated with azithromycin. Compared with that in Group C,
treatment costs of patients were significantly increased in Group
A, Group B and Group D (P<0.01). Compared with that in Group A,
treatment costs of patients were remarkably increased in Group B
and Group D (P<0.01). Compared with that in Group B, the
treatment cost of patients in Group D was increased obviously
(P<0.01; Fig. 1).
Discussion
LRTI is the most common infectious disease in
clinic, which is often caused by bacterial, viral and chlamydia
infections (7). LRTI often seriously
threatens the physical health of patients, leading to low immune
function and abnormal changes in the body's inflammatory indexes.
Both WBC and CRP are indexes closely related to LRTI, and the
effective detection indexes for the diagnosis of patients with
respiratory tract infection (8,9).
Antibiotics are dominated in the clinical treatment of LRTI, and
there are a variety of different therapeutic regimens. Injection
therapy of large-dose antibiotics lead to enhanced drug resistance
of pathogens (10,11). During treatment, the increase in the
drug costs often lays a heavy economic burden on patients.
Therefore, selecting the appropriate drug therapeutic regimen and
reducing the drug costs can greatly reduce the economic burden
while improving the physical health of patients, which is of vital
significance in clinic.
Ceftriaxone sodium, ceftizoxime sodium, levofloxacin
and azithromycin are often applied in the clinical treatment of
LRTI (12). As the third-generation
cephalosporin antibiotic drugs, ceftriaxone sodium and ceftizoxime
sodium are characterized by strong therapeutic effects and broad
antibacterial spectrum. Ceftriaxone sodium has always been a kind
of safe and reliable antibacterial drug in the treatment of LRTI,
but it has high drug resistance (13). Ceftizoxime sodium is too expensive to
be accepted by most patients. As a quinolone antibacterial drug,
levofloxacin is characterized by a potent antibacterial effect and
broad antibacterial spectrum, which can inhibit the activity of DNA
gyrase in bacteria with a good antibacterial efficacy (14). Azithromycin, as a macrolide
antibiotic, can inhibit a variety of bacteria with a broad
antibacterial spectrum, which can reduce the incidence rate of
adverse reactions in patients, and has been widely applied in
clinic (15). In the present study,
the total effective rate was 94.00% in Group A in the treatment of
LRTI with ceftriaxone sodium, 92.00% in Group B, 96.00% in Group C
and 90.00% in Group D. There were no differences in the total
effective rate of treatment among the 4 groups of patients, and
there were no adverse reactions during treatment. The above results
indicate that the 4 kinds of antibacterial drugs have good
antibacterial efficacy, and their curative effects are well
consistent in the treatment of LRTI. Di Marco et al
(16), studied and showed that
curative effects of ceftriaxone sodium, ceftizoxime sodium,
levofloxacin and azithromycin are satisfactory in the treatment of
LRTI, which are similar to results in the present study. However,
studies have revealed that azithromycin has a better therapeutic
effect (12,15). Such a difference is possibly due to
different objects and courses of disease in the present study, so
the conclusion needs further verification.
The WBC count in the body is changed due to a
variety of inflammatory responses and bacterial infections. WBC
includes monocytes, lymphocytes and granulocytes, and changes in
its count reflect the body's immune function and bacterial
infection (17). CRP is a kind of
globulin synthesized by the liver, as well as a reactive protein
produced when there are concurrent inflammations or injuries in the
body. When inflammation or infection occurs in the body, the
synthesis of CRP will be rapidly increased and can reach the peak
within 50 h. CRP is sensitive to, not only bacterial infection, but
also chronic inflammatory response, acute trauma, burns and viral
infection, whose level often has a positive correlation with the
severity of disease (18,19). Results in the present study
demonstrated that both WBC count and CRP measured values
significantly declined in the 4 groups of patients at 1, 4 and 7
days after treatment compared with those at 1 day before treatment.
Compared with those at 1 day after treatment, both WBC count and
CRP measured values were significantly decreased in the 4 groups of
patients at 4 and 7 days after treatment. Besides, both WBC count
and CRP measured values were obviously decreased in the 4 groups of
patients at 7 days after treatment compared with those at 4 d after
treatment. The above findings indicate that WBC and CRP can be used
to dynamically monitor the treatment of LRTI and accurately observe
the disease evolution and treatment effect on patients. WBC and CRP
can serve as monitoring indexes for LRTI, which is similar to the
research conclusion of Ben Amar et al (20).
The growth of medical costs has always attracted
social concern, which has an extremely important influence on the
daily life of patients. According to results in the present study,
compared with that in Group C, treatment costs of patients were
significantly increased in Group A, Group B and Group D. Compared
with that in Group A, treatment costs of patients were remarkably
increased in Group B and Group D. Compared with that in Group B,
the treatment cost of patients in Group D was increased obviously.
The curative effects of the 4 kinds of antibiotics were well
consistent in the treatment of LRTI. Under the premise of ensuring
the therapeutic effect, the treatment cost of levofloxacin is the
lowest in economic terms.
Subjects in the present study were screened in
strict accordance with the inclusion and exclusion criteria. There
were no differences in general clinical data, such as sex, age,
course of disease, RBC count, PLT count and Hb, among patients
enrolled, thus ensuring the reliability of the study. However,
etiological monitoring and drug susceptibility test were not
performed for the 4 groups of patients in this study, and the drug
resistance mechanisms of the 4 kinds of antibiotics were not
investigated, so there were certain limitations. Therefore, it is
expected that bacterial culture and drug susceptibility test can be
performed for patients with LRTI in subsequent investigations to
verify the results in the present study.
In conclusion, ceftriaxone sodium, ceftizoxime
sodium, levofloxacin, and azithromycin have excellent antibacterial
efficacy. WBC and CRP can be used to dynamically monitor the
treatment of LRTI and accurately observe the disease evolution and
treatment effect on patients. The treatment cost of levofloxacin is
the lowest in economic terms; thus, it is worthy of clinical
promotion and application.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
LZ drafted the present study. LZ and BL collected
the patient data. LZ, BL and CW analyzed the patient data. All the
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Dongying People's Hospital (Dongying, China). Signed
written informed consents were obtained from the patients and/or
guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Houston MS, Silverstein MD and Suman VJ:
Community-acquired lower respiratory tract infection in the
elderly: A community-based study of incidence and outcome. J Am
Board Fam Pract. 8:347–356. 1995.PubMed/NCBI
|
|
2
|
Jiang W, Yin F, Zhou W, Yan Y and Ji W:
Clinical significance of different virus load of human bocavirus in
patients with lower respiratory tract infection. Sci Rep.
6:202462016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chiricozzi E, Loberto N, Schiumarini D,
Samarani M, Mancini G, Tamanini A, Lippi G, Dechecchi MC, Bassi R,
Giussani P, et al: Sphingolipids role in the regulation of
inflammatory response: From leukocyte biology to bacterial
infection. J Leukoc Biol. 103:445–456. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Honda T, Uehara T, Matsumoto G, Arai S and
Sugano M: Neutrophil left shift and white blood cell count as
markers of bacterial infection. Clin Chim Acta. 457:46–53. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Muloiwa R, Dube FS, Nicol MP, Zar HJ and
Hussey GD: Incidence and diagnosis of pertussis in South African
children hospitalized with lower respiratory tract infection.
Pediatr Infect Dis J. 35:611–616. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hu S, Jiang T, Zhang X, Zhou Y, Yi Z, Wang
Y, Zhao S, Wang M, Ming D and Chen S: Elizabethkingia anophelis
isolated from patients with multiple organ dysfunction syndrome and
lower respiratory tract infection: Report of two cases and
literature review. Front Microbiol. 8:3822017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mazur NI, Martinón-Torres F, Baraldi E,
Fauroux B, Greenough A, Heikkinen T, Manzoni P, Mejias A, Nair H,
Papadopoulos NG, et al: Respiratory Syncytial Virus Network
(ReSViNET): Lower respiratory tract infection caused by respiratory
syncytial virus: Current management and new therapeutics. Lancet
Respir Med. 3:888–900. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cohen C, Moyes J, Tempia S, Groome M,
Walaza S, Pretorius M, Naby F, Mekgoe O, Kahn K, von Gottberg A, et
al: Epidemiology of acute lower respiratory tract infection in
HIV-exposed uninfected infants. Pediatrics. 137:1372016. View Article : Google Scholar
|
|
9
|
Yoshihara K, Le MN, Okamoto M, Wadagni AC,
Nguyen HA, Toizumi M, Pham E, Suzuki M, Nguyen AT, Oshitani H, et
al: Association of RSV-A ON1 genotype with increased pediatric
acute lower respiratory tract infection in Vietnam. Sci Rep.
6:278562016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lodise TP Jr, Miller C, Patel N, Graves J
and McNutt LA: Identification of patients with Pseudomonas
aeruginosa respiratory tract infections at greatest risk of
infection with carbapenem-resistant isolates. Infect Control Hosp
Epidemiol. 28:959–965. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Volturo GA, Low DE and Aghababian R:
Managing acute lower respiratory tract infections in an era of
antibacterial resistance. Am J Emerg Med. 24:329–342. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kraus EM, Pelzl S, Szecsenyi J and Laux G:
Antibiotic prescribing for acute lower respiratory tract infections
(LRTI)-guideline adherence in the German primary care setting: An
analysis of routine data. PLoS One. 12:e01745842017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shah AJ, Mulla SA and Revdiwala SB:
Neonatal sepsis: High antibiotic resistance of the bacterial
pathogens in a neonatal intensive care unit of a tertiary care
hospital. J Clin Neonatol. 1:72–75. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abdul-Aziz MH, Lipman J, Mouton JW, Hope
WW and Roberts JA: Applying pharmacokinetic/pharmacodynamic
principles in critically ill patients: Optimizing efficacy and
reducing resistance development. Semin Respir Crit Care Med.
36:136–153. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Serisier DJ: Inhaled antibiotics for lower
respiratory tract infections: Focus on ciprofloxacin. Drugs Today
(Barc). 48:339–351. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Di Marco F, Braido F, Santus P, Scichilone
N and Blasi F: The role of cefditoren in the treatment of lower
community-acquired respiratory tract infections (LRTIs): From
bacterial eradication to reduced lung inflammation and epithelial
damage. Eur Rev Med Pharmacol Sci. 18:321–332. 2014.PubMed/NCBI
|
|
17
|
Thompson AA, Dickinson RS, Murphy F,
Thomson JP, Marriott HM, Tavares A, Willson J, Williams L, Lewis A,
Mirchandani A, et al: Hypoxia determines survival outcomes of
bacterial infection through HIF-1alpha dependent re-programming of
leukocyte metabolism. Sci Immunol. 2:eaa128612017. View Article : Google Scholar
|
|
18
|
Kim JH, Lee JY, Cho HR, Lee JS, Ryu JM and
Lee J: High concentration of C-reactive protein is associated with
serious bacterial infection in previously healthy children aged 3
to 36 months with fever and extreme leukocytosis. Pediatr Emerg
Care. 1:2017.
|
|
19
|
Durán A, González A, Delgado L, Mosquera J
and Valero N: Serum level of C-reactive protein is not a parameter
to determine the difference between viral and atypical bacterial
infections. J Med Virol. 88:351–355. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ben Amar J, Zaibi H, Bouzid K, Azzabi S,
Bacca MA, Dahari B and Aouina H: Role of procalcitonin and
c-reactive protein levels: A diagnostic tool in lower respiratory
tract infections. Tunis Med. 94:176–180. 2016.PubMed/NCBI
|