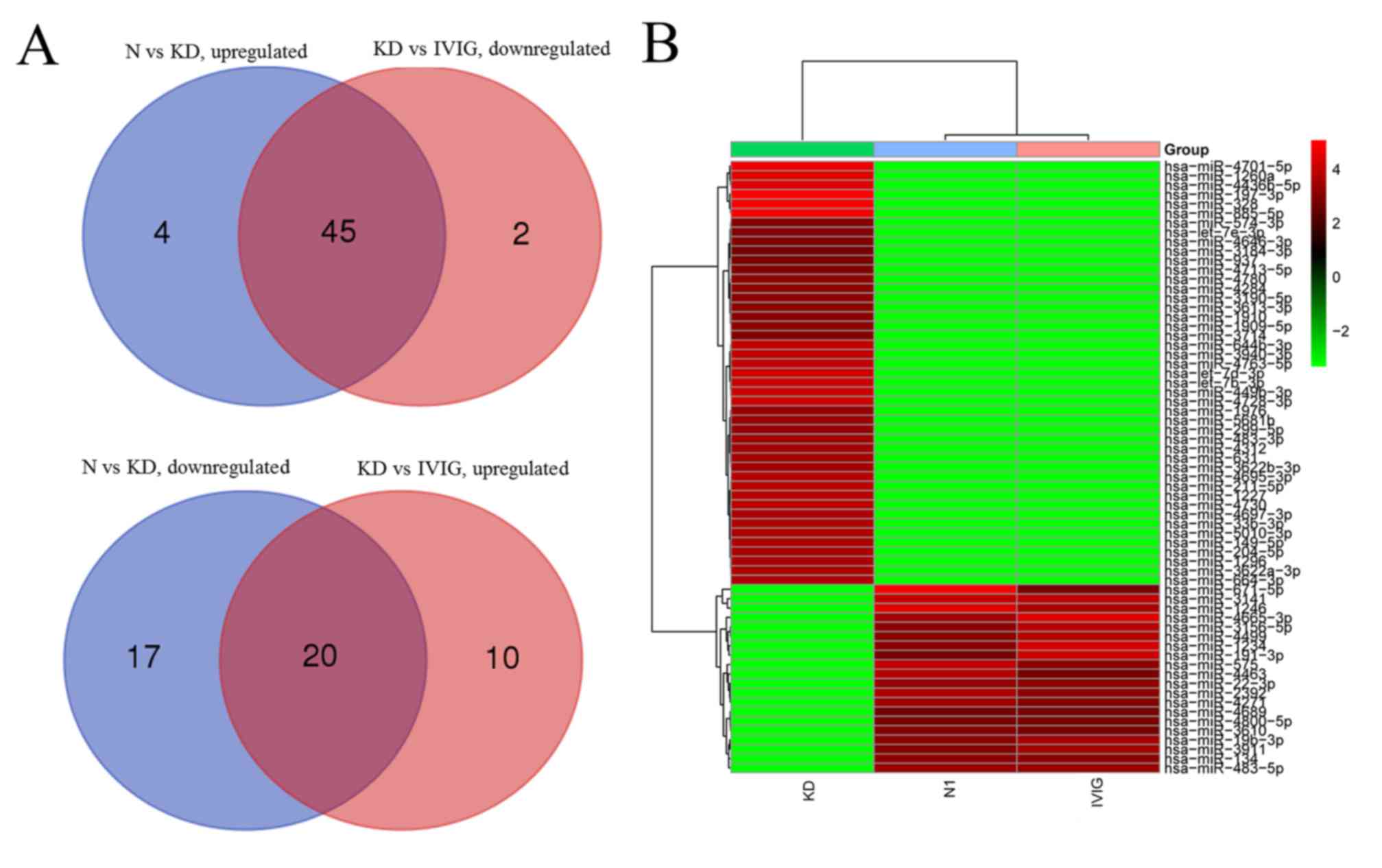
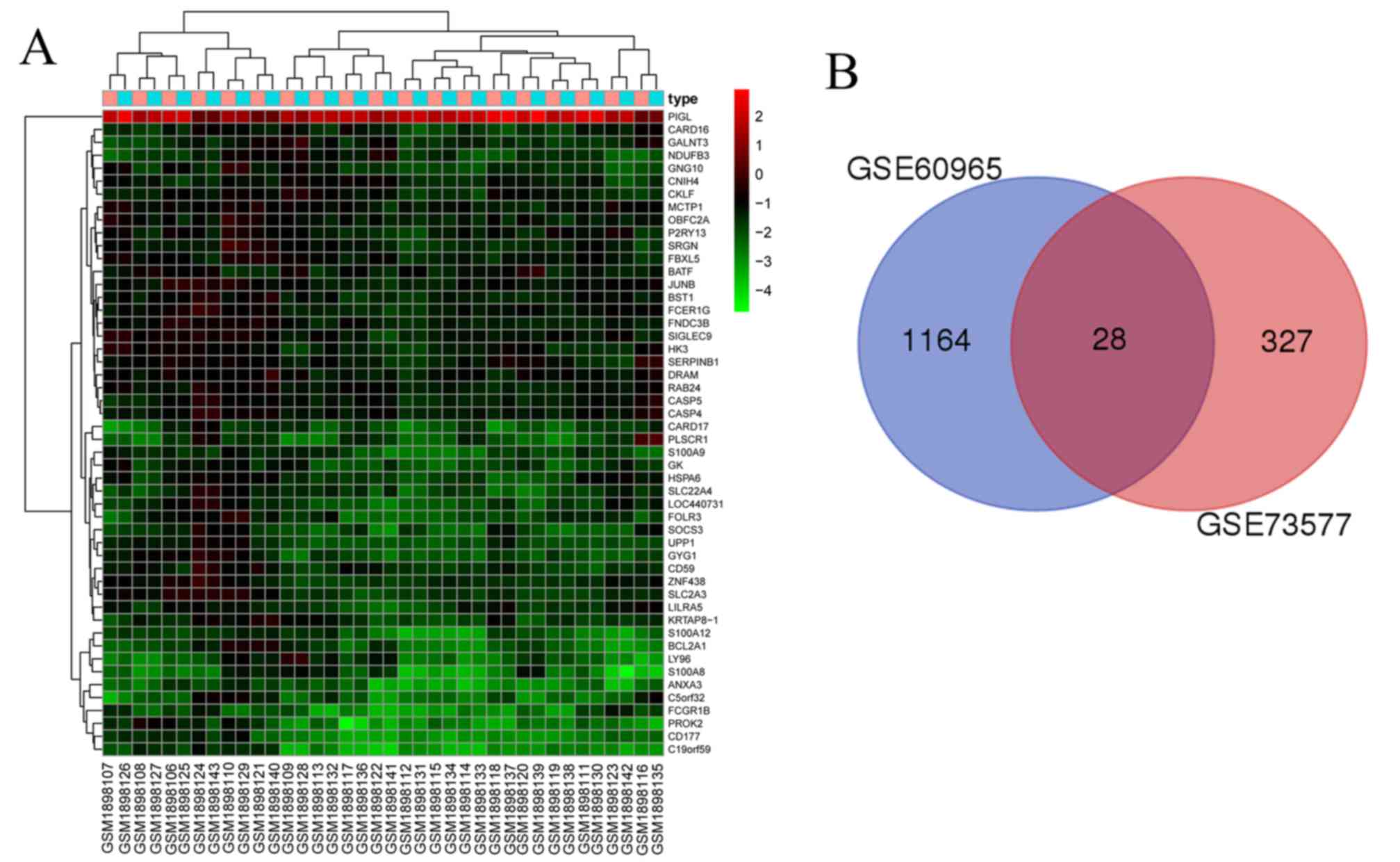
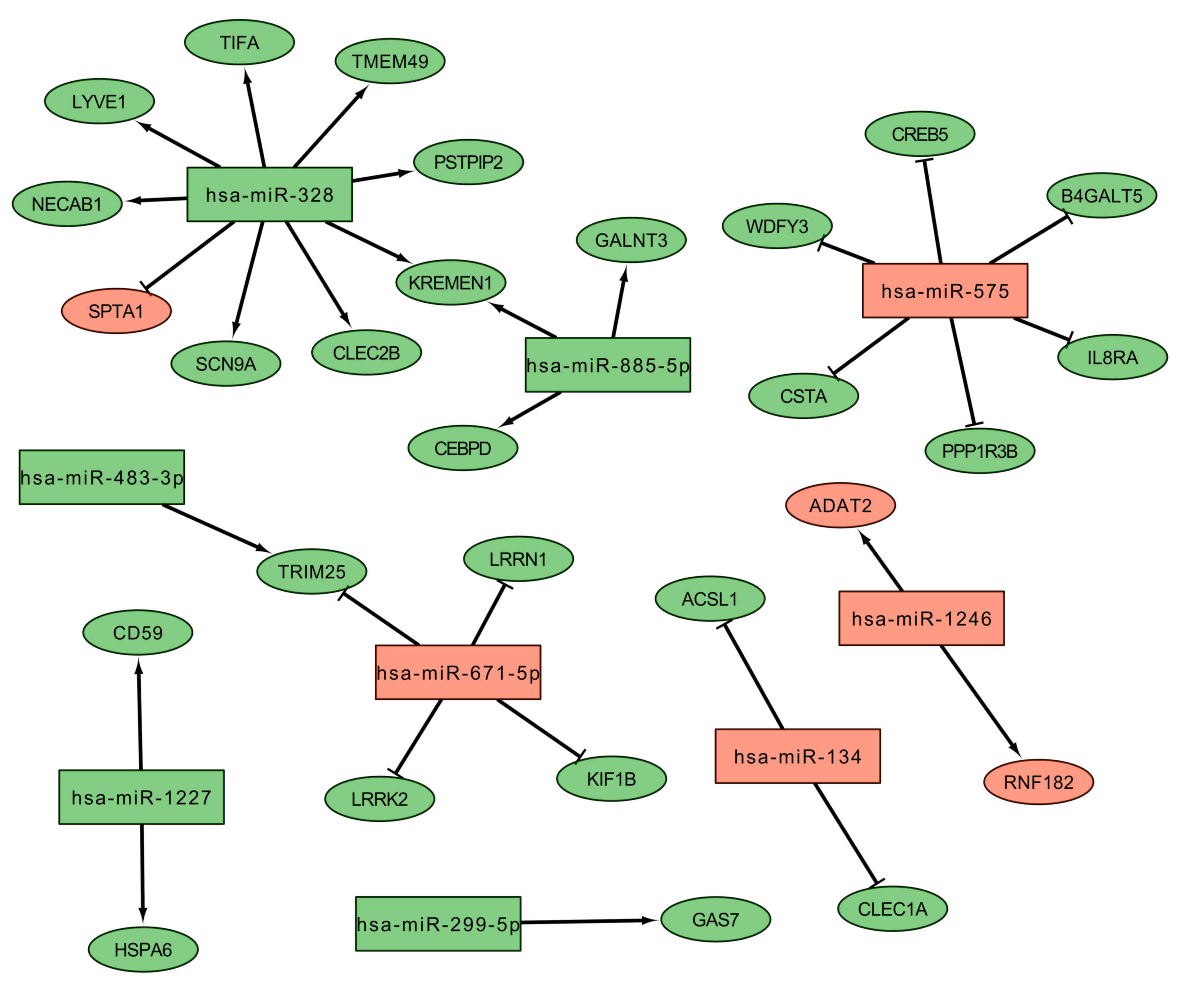
| GO:0045893,
positive regulation of transcription, DNA-templated |
1.39×10−5 | PPARD, GDF2, MITF,
ZXDC, TGFB3, NFKB1, CTCF, GLI3, LGR4, ZIC3, WNT4, RRN3, ZNF281,
TBL1XR1, EGR2, FOXJ2, SOX11, MED14, RB1, ESR2, MED13, SIX4, HIPK2,
MAPK3, ZNF711, TFAP2B, ERBB4, HOXA11, SOX2, EHF, CDH1, NFYA, TFAM,
NR1D2, NPAT, CREBL2, KLF6, IL5, TRIP4, TBX3, SMAD4, IGF1, MSTN,
CREB5, ATMIN, FZD4, RLF, YWHAH, SFRP1, SP1, SETD7, NEUROD1,
TP53INP2, F2R |
| GO:0045944,
positive regulation of transcription from RNA polymerase II
promoter |
1.05×10−3 | CCNT2, HLF, GDF2,
RNASEL, E2F8, STAT5B, MITF, EDN1, ARID4B,TGFB3, CTCF, NFKB1, GLI3,
ZIC3, ZBTB38, CRX, PGR, PCGF5, ZNF304, SERPINE1, OGT, FGF1,
TBL1XR1, SATB2, EXOSC9, EGR2, FOXJ2, SOX11, MTA2, RXRA, MED14, RB1,
SIX4, MED13, CD40, GRHL2, GTF2H1, ACVR2A, VEGFA, MAPK3, HIPK2,
TFAP2B, TFAP2C, CRTC3, FGFR2, SOX2, ONECUT2, TAF9B, EHF, EGLN1,
CDC73, NR2C2, ATF1, TFAM, CHD7, NIPBL, DDX3X, PKD2, MYF6, IKZF4,
IKZF1, CEBPD, SMAD4, IGF1, EN2, CSRP3, TET1, PARK7, DDX58, RLF,
ATRX, SP1, TRPS1, NEUROD1, IRF2, NHLH2, PBX3, PBX2, FOXI1 |
| GO:0043200,
response to amino acid |
1.36×10−3 | ICAM1, SLC1A2,
GLRB, MTHFR, CDKN1B, GLRA3, EDN1, CDO1 |
| GO:0043406,
positive regulation of MAP kinase activity |
1.49×10−3 | RASGRP1, EDN1,
VEGFA, PDE5A, ADRA2A, KITLG, PDGFC, KIT, CD40, FGF1, LRRK2 |
| GO:0043268,
positive regulation of potassium ion transport |
1.57×10−3 | DRD1, KIF5B,
ADRA2A, STK39, DLG1 |
| GO:0051091,
positive regulation of sequence-specific DNA binding transcription
factor activity |
2.15×10−3 | IL5, EDN1, TRIM27,
TRIM14, TRIM25, ESR2, KIT, FZD4, TRIM21, PARK7, DDX58, TRIM32,
HIPK2, NEUROD1, NHLH2 |
| GO:0019228,
neuronal action potential |
3.97×10−3 | DRD1, SCN1A, SCN3A,
GRIK2, ANK3, KCNA1, SCN9A |
| GO:0014066,
regulation of phosphatidylinositol 3-kinase signaling |
4.01×10−3 | FGFR2, C3ORF58,
EREG, ERBB4, ERBB3, RASGRP1, MAPK3, KITLG, KIT, FGF1, PIP4K2A,
PIP4K2C |
| GO:0050890,
cognition |
5.41×10−3 | MAGT1, CHD7, NIPBL,
PTCHD1, NF1, CHRNA4, CHRNB2, CHMP2B |
| GO:0007265, Ras
protein signal transduction |
5.42×10−3 | ZNF304, PLD1,
PLCE1, RASGRP1, NF1, ADRA2A, IGF1, RB1, SHC3, KSR1, PARK7 |
| GO:0014070,
response to organic cyclic compound |
5.50×10−3 | ICAM1, CD83, ACSL1,
SFRP1, TRPA1, ABCC4, ABCD3, COMT, ATF1 |
| GO:0047496, vesicle
transport along microtubule |
6.24×10−3 | DYNC1I1, NDEL1,
KIF5B, HTT, RASGRP1 |
| GO:0010842, retina
layer formation |
6.48×10−3 | PROM1, HIPK2, FJX1,
TFAP2B, CALB1, DSCAM |
| GO:0061024,
membrane organization |
7.89×10−3 | YWHAH, RAB14,
YWHAB, TBC1D4, PMP2, RAB10, YWHAE |
| GO:0008585, female
gonad development |
8.14×10−3 | WNT4, COL9A3,
SFRP1, ZFP42, TIPARP |
| GO:0060021, palate
development |
9.66×10−3 | WFIKKN2, ACVR2B,
SATB2, CHD7, TBX3, TIPARP, TGFB3, SMAD4, COL2A1, C5ORF42, GLI3 |
| GO:0090073,
positive regulation of protein homodimerization activity |
1.14×10−2 | CRBN, TIRAP, PARK7,
TRAF4 |
| GO:0048565,
digestive tract development |
1.22×10−2 | FGFR2, TRPS1,
TGFB3, PDGFC, RB1, KIT, LGR4 |
| GO:0007059,
chromosome segregation |
1.32×10−2 | CIAO1, NDEL1,
DDX3X, PPP1R7, SLC25A5, USP9X, NEK9, CTCF, SRPK1, MIS12 |
| GO:0042552,
myelination |
1.34×10−2 | EGR2, TSPAN2, MAL2,
ATRN, CMTM8, XK, QKI, ACSBG1 |
| GO:0001764, neuron
migration |
1.44×10−2 | SATB2, TUBB2B,
USP9X, CELSR1, PCM1, YWHAE, SEMA6A, NDEL1, NAV1, CCR4, NEUROD4,
DCX, MYH10 |
| GO:0043154,
negative regulation of cysteine-type endopeptidase activity
involved in apoptotic process |
1.45×10−2 | ARL6IP1, LAMP3,
DDX3X, TNFAIP8, VEGFA, TFAP2B, USP47, RAG1, BIRC5, YWHAE |
| GO:0035136,
forelimb morphogenesis |
1.56×10−2 | NIPBL, TBX3,
RNF165, TFAP2B |
| GO:0007156,
homophilic cell adhesion via plasma membrane adhesion
molecules |
1.59×10−2 | PCDHA6, ME2,
PCDHA2, CLSTN2, CADM2, PCDH9, CDH1, PTPRT, CELSR1, CDH2, IGSF9B,
PCDHAC2, PCDHAC1, CDH9, PCDHA10, ROBO2, DSCAM |
| GO:0043372,
positive regulation of CD4-positive, alpha-beta T cell
differentiation |
1.74×10−2 | CD83, TNFSF4,
SASH3 |
| GO:0010951,
negative regulation of endopeptidase activity |
1.80×10−2 | WFIKKN2, C5, CD109,
PAPLN, FURIN, A2ML1, WFDC8, SERPINE2, SERPINE1, TFPI, PEBP1, ITIH5,
CSTA, CRIM1 |
| GO:0005975,
carbohydrate metabolic process |
1.83×10−2 | GALNT3, GANAB,
FUT9, GNPDA2, ST8SIA1, GPD1L, MAN2A2, PGM2, PGM3, GANC, ALDH1B1,
SLC2A2, AKR1B1, ST8SIA5, FUT4, SPAM1, B4GALT5, PYGB |
| GO:0040007,
growth |
1.83×10−2 | OPA3, BMP3, GDF2,
VEGFA, BMP8B, FOXP2 |
| GO:0006513, protein
monoubiquitination |
1.83×10−2 | TSG101, DTL,
KLHL12, RAD18, TRIM25, TRIM21 |
| GO:0001894, tissue
homeostasis |
1.93×10−2 | AKR1B1, TRIM32,
COL2A1, RB1, TP53INP2 |
| GO:0048745, smooth
muscle tissue development |
2.06×10−2 | NF1, TIPARP, DLG1,
FOXP2 |
| GO:0046622,
positive regulation of organ growth |
2.06×10−2 | ARX, IL7, RAG2,
SASH3 |
| GO:0061045,
negative regulation of wound healing |
2.06×10−2 | WNT4, HMGCR,
SERPINE1, CD109 |
| GO:0007519,
skeletal muscle tissue development |
2.28×10−2 | MYF6, CCNT2, CFL2,
NF1, SIX4, FLNB, CSRP3, FOXP2 |
| GO:0048839, inner
ear development |
2.56×10−2 | CDKN1B, CXCL14,
CEBPD, DUOX2, SOX2, TGFB3, NEUROD1 |
| GO:0031954,
positive regulation of protein autophosphorylation |
2.73×10−2 | RAP2B, VEGFA,
PDGFC, RAD50, CALM2 |
| GO:0097150,
neuronal stem cell population maintenance |
2.73×10−2 | SOX2, CDH2, PCM1,
HOOK3, MMP24 |
| GO:0033157,
regulation of intracellular protein transport |
2.80×10−2 | NDEL1, SH3TC2,
LCP1 |
| GO:0021631, optic
nerve morphogenesis |
2.80×10−2 | CHRNB2, GLI3,
EPHB1 |
| GO:0048511,
rhythmic process |
3.03×10−2 | HLF, SP1, NR1D2,
SFPQ, PASD1, PRKAA2, NFYA, FBXL3 |
| GO:0070911, global
genome nucleotide-excision repair |
3.13×10−2 | SUMO3, UBE2N, DDB2,
ERCC4, USP45, GTF2H1 |
| GO:0098609,
cell-cell adhesion |
3.13×10−2 | ZC3HAV1, KIF5B,
CKAP5, RPL15, YWHAB, TRIM25, ARFIP1, FLNB, YWHAE, PARK7, MMP24,
CHMP2B, EIF4G2, GAPVD1, DDX3X, FNBP1L, SERBP1, TMOD3, PCMT1,
DNAJB1, MAPRE1, RAB10, UBAP2, AHNAK |
| GO:0039702, viral
budding via host ESCRT complex |
3.20×10−2 | CHMP1A, TSG101,
CHMP6, VPS37C, CHMP2B |
| GO:0060078,
regulation of postsynaptic membrane potential |
3.20×10−2 | SCN1A, SCN3A, PKD2,
SCN9A, SCN4B |
| GO:0006366,
transcription from RNA polymerase II promoter |
3.28×10−2 | CCNT2, NCBP2, HLF,
POLR2E, STAT5B, TAF9B, MITF, SOX2, ONECUT2, NFKB1, EHF, CTCF, NFYA,
GLI3, ATF1, ZIC3, CRX, TFAM, MAX, DDX21, VEZF1, MYF6, ZNF831, EGR2,
FOXJ2, CEBPD, SOX11, SNAPC3, SMAD4, CREB5, SIX4, GRHL2, GTF2H1,
TRPS1, TFAP2B, IRF2, NEUROD1, TFAP2C, PBX3, FOXI1 |
| GO:0006044,
N-acetylglucosamine metabolic process |
3.29×10−2 | CHST7, GNPDA2,
GNPNAT1, MGEA5 |
| GO:0060134,
prepulse inhibition |
3.29×10−2 | DRD1, SLC6A3,
NRXN1, CTNNA2 |
| GO:0032897,
negative regulation of viral transcription |
3.29×10−2 | TRIM32, TRIM14,
TRIM27, TRIM21 |
| GO:0007399, nervous
system development |
3.30×10−2 | PCDHA6, GLRB, FUT9,
MOBP, PCDHA2, ERBB4, CAMK2G, ARID1B, IGSF9B, GAS7, NR2C2, PCDHAC2,
PCDHAC1, SEMA6A, ATXN3, NDEL1, TPP1, VEGFA, MSI1, PCDHA10, DCX,
CRIM1, DLG1, WNT8B, DSCAM |
| GO:0045892,
negative regulation of transcription, DNA-templated |
3.46×10−2 | PPARD, GCLC,
TSG101, CTCF, GLI3, LGR4, ZBTB38, WNT4, ZNF227, NIPBL, NR1D2,
GATAD2A, ZNF425, PASD1, CRY1, BAHD1, MYF6, ZNF281, IKZF4, TNFSF4,
TBX3, IKZF1, CEBPD, YWHAB, SMAD4, BIRC5, RB1, SIX4, FOXP2, CHMP1A,
CDKN1B, SFRP1, TRIM33, EREG, SFPQ, RBAK, USP47, TFAP2B, XCL1 |
| GO:0015758, glucose
transport |
3.52×10−2 | PPARD, SLC2A10,
SLC2A2, EDN1, SLC2A1, HK2 |
| GO:0051402, neuron
apoptotic process |
3.52×10−2 | MAX, USP53, GRIK2,
ERBB3, RB1, NLRP1 |
| GO:0006914,
autophagy |
3.58×10−2 | TSG101, CHMP6,
VPS41, VPS37C, PARK7, VTI1A, CHMP2B, TBC1D25, ATG5, FNBP1L, RB1CC1,
ATG4A, LRRK2, VPS39 |
| GO:0050680,
negative regulation of epithelial cell proliferation |
3.60×10−2 | FGFR2, PPARD, EREG,
SFRP1, SOX2, CDC73, RB1, DLG1 |
| GO:0006479, protein
methylation |
3.70×10−2 | PCMTD2, BHMT,
PCMT1, ETF1, N6AMT1 |
| GO:0045662,
negative regulation of myoblast differentiation |
3.70×10−2 | PPARD, TBX3,
CXCL14, MSTN, CSRP3 |
| GO:0046854,
phosphatidylinositol phosphorylation |
3.75×10−2 | FGFR2, EREG, ERBB4,
ERBB3, PI4K2A, KITLG, PI4K2B, KIT, FGF1, PIP4K2A, PIP4K2C |
| GO:0045787,
positive regulation of cell cycle |
3.94×10−2 | FGFR2, ANKRD17,
CDKN1B, TBX3, TRIM32, TRIM21 |
| GO:0007585,
respiratory gaseous exchange |
3.94×10−2 | HNMT, TMPRSS11D,
EDN1, CHRNA4, PBX3, TRAF4 |
| GO:0006813,
potassium ion transport |
3.96×10−2 | KCNS3, KCNMA1,
KCNS1, CDKN1B, SLC12A2, ATP4B, SLC24A3, KCNA1, KCNA6, KCNJ12 |
| GO:0051260, protein
homooligomerization |
4.03×10−2 | CCDC88C, GLRA3,
KCNA1, PRND, KCNA6, KCNA7, KCNS3, ANXA6, STOM, KCNS1, CLDN1,
KCTD16, ZBTB1, SLC1A1, EHD3, SPAST, KCTD12 |
| GO:0034454,
microtubule anchoring at centrosome |
4.05×10−2 | KIF3A, PCM1,
HOOK3 |
| GO:0035020,
regulation of Rac protein signal transduction |
4.05×10−2 | SSX2IP, OGT,
CRK |
| GO:0010606,
positive regulation of cytoplasmic mRNA processing body
assembly |
4.05×10−2 | CNOT6L, CNOT2,
CNOT6 |
| GO:0010960,
magnesium ion homeostasis |
4.05×10−2 | ANK3, KCNA1,
TFAP2B |
| GO:0071910,
determination of liver left/right asymmetry |
4.05×10−2 | PKD2, CCDC39,
ZIC3 |
| GO:0045165, cell
fate commitment |
4.22×10−2 | FGFR2, WNT4, ERBB4,
TRPS1, ONECUT2, NEUROD4, WNT8B |
| GO:0071456,
cellular response to hypoxia | 4.24
×10−2 | ICAM1, PPARD,
PTGIS, TBL2, STC2, CPEB2, SFRP1, EDN1, VEGFA, BNIP3L, HIPK2 |
| GO:0032456,
endocytic recycling |
4.25×10−2 | STX6, VPS52, RAB14,
ARL4C, EHD3 |
| GO:0030307,
positive regulation of cell growth |
4.52×10−2 | EIF4G2, EXTL3,
EXOSC9, DDX3X, SFRP1, TRIM32, TAF9B, USP47, H3F3B, N6AMT1 |
| GO:0042384, cilium
assembly |
4.54×10−2 | KIF3A, DZIP1,
ONECUT2, PCM1, C5ORF42, ACTR2, TTC30A, FNBP1L, C10ORF90, ABCC4,
SSX2IP, EXOC5, EHD3 |
| GO:0009636,
response to toxic substance |
4.81×10−2 | GLYAT, MAPK3,
SLC30A4, SLC6A14, SCN9A, CDH1, GUCY2C, HTR1D, NQO1, PON3 |
| GO:2000679,
positive regulation of transcription regulatory region DNA
binding |
4.82×10−2 | NEUROD1, IGF1, RB1,
PARK7 |
| GO:0022408,
negative regulation of cell-cell adhesion |
4.82×10−2 | NF2, TNR, EPB41L5,
CDH1 |
| GO:0007076, mitotic
chromosome condensation |
4.82×10−2 | CHMP1A, NCAPH,
NCAPG, CDCA5 |
| GO:0006351,
transcription, DNA-templated |
4.96×10−2 | IL16, ZNF451, ZXDC,
CNOT2, ZNF250, MED22, CNOT6, ZNF254, PGR, ZNF304, EPC2, MIER3,
ZNF445, CRY1, SAMD4B, ZNF449, IKBKAP, SATB2, RXRA, ARID1B, TRIM33,
MAPK3, TGIF2, VGLL3, CRTC3, ERBB4, HOXA11, NR2C2, ARX, ZNF227,
DDX3X, CNOT6L, ZNF697, ZNF425, ZNF124, CREBL2, IKZF4, KLF6, TRIP4,
IKZF1, RFX5, SMAD4, ZNF521, ZNF320, ZNF585A, ZNF627, CSRP3, TET1,
FOXP2, ZNF419, ZNF417, PNRC1, JAZF1, ZNF318, PHF6, CCNT2, PPARD,
ZNF518B, ZNF81, ARID4B, ZFP42, E2F8, ZNF10, ZBTB38, PCGF5, HIF1AN,
BRD9, ZNF281, NFKBIZ, TBL1XR1, ZNF33A, EGR2, ZNF354A, ZNF354C, ZFY,
ZFX, SF1, RB1, ESR2, ZBTB26, PURB, GTF2H1, CHMP1A, BRWD1, HIPK2,
ZNF711, ZNF480, LIN54, ZNF740, POLR2E, LIN9, SCML2, ZNF660, CHD7,
ZSCAN22, NR1D2, RB1CC1, NPAT, GATAD2A, PRKAA2, ZNF470, BAHD1,
ZNF267, TBX3, PPHLN1, CEBPD, NLK, ZNF770, ZFP1, ZNF667, TRIM27,
BIRC5, ATMIN, ZNF665, RLF, ATRX, ATXN3, SFPQ, RBAK, ZBTB5, NHLH2,
ZNF461, SETD7, NEUROD4, PBX2, ZNF766, TP53INP2 |
| GO:0048661,
positive regulation of smooth muscle cell proliferation |
4.96×10−2 | FGFR2, EREG, HMGCR,
EDN1, AKR1B1, IGF1, ABCC4, CALCRL |
| GO:0045669,
positive regulation of osteoblast differentiation |
4.96×10−2 | ACVR2A, WNT4,
ACVR2B, GDF2, CEBPD, SOX11, IGF1, GLI3 |
| GO:0000122,
negative regulation of transcription from RNA polymerase II
promoter |
4.99×10−2 | PPARD, IMPACT,
E2F8, EDN1, MITF, CNOT2, NFKB1, CTCF, HSBP1, ZNF254, GLI3, CRY1,
DLG1, ZNF281, TBL1XR1, SATB2, SOX11, MTA2, RXRA, HNRNPA2B1, RB1,
ESR2, PURB, ACVR2B, TRIM33, VEGFA, HIPK2, TFAP2B, TGIF2, TFAP2C,
FGFR2, USP9X, TAF9B, SOX2, CDC73, ARX, NIPBL, GATAD2A, IKZF1, TBX3,
RFX5, PTPN2, SMAD4, TRIM27, FOXP2, DLX1, DKK1, SFPQ, TRPS1, JAZF1,
IRF2, ZBTB1 |
|
| B, DEGs
regulated by DE-miRNAs |
|
| Term | P-value | Genes |
|
| GO:0060828,
regulation of canonical Wnt signaling pathway |
2.82×10−2 | KREMEN1, LRRK2 |