Introduction
There is an increasing interest in the activation
mechanisms of cannabinoid (CB) receptors as experimental or
clinical therapeutic strategies with recent reviews (1). The main receptors of the
endocannabinoid system are the CB1 and CB2 cannabinoid receptors:
Although CB1 receptors are highly expressed in the brain (2), and also present nerve terminal of
peripheral tissues (3), the CB2
receptors are expressed in inflammatory and immune cells, and in
various peripheral tissues under normal physiological or
pathophysiological conditions (4).
2-arachidonoylglycerol (2-AG) is the endogen ligand of CB2,
arachidonic acid-derived endogenous bioactive lipid, its production
from cell membrane lipid precursors is activity-dependent and its
action is terminated by specific lipase, monoacylglycerol lipase
(MAGL) (3). The high levels of CB2
express within the immune system including B cells, natural killer
cells, monocytes, neutrophils, CD8 lymphocytes, CD4 lymphocytes
(5,6).
Inflammation is a pathophysiological response to
disturbed homeostasis caused by infection, injury and trauma. Cell
migration into the site of injury, erythema, oedema, pain, and
fever are clinical signs of inflammation and symptoms. The release
of pro-inflammatory cytokines, such as tumour necrosis factor-α
(TNF-α), activated by macrophages, leads to vascular endothelial
cell activation and causes leukocytes to migrate to the inflamed
region (7).
It is well known that carrageenan-induced paw oedema
model result in oxidative stress with the increase lipid
peroxidation or the depletion of antioxidant sulfhydryl pool such
as glutathione, during the acute phase of inflammation in cells
(8–10).
A lot of studies suggest an involvement of CB2
receptors in inflammatory process (11,12). A
recent study demonstrates that the selective MAGL inhibitor
inhibits carrageenan inflammation with attenuated the development
of paw oedema in mice; the activation of CB2 receptors mediated its
anti-oedematous actions (13). The
selective MAGL inhibitor, URB602, is to prevent an acute
inflammatory disease without producing adverse psychoactive
effects, so the data contributed to clarify the physiological role
of 2-AG in respect to inflammatory reactions, suggesting its
protective role in the body (14).
Studies have identified an anti-inflammatory role for the
endocannabinoid system by its inhibition of TNF-α release. The
other researches indicate the controversial results. Lunn et
al (15), interestingly suggest
that CB2 antagonism by means of inverse agonist of CB2 receptor is
inhibit leukocyte trafficking in vivo rodent model induced
by chemokines or by antigen challenge, so it may provide
immunotherapeutic treatment of inflammatory diseases.
We hypothesized that the administered CB2 specific
receptor agonist could induce an anti-inflammatory effect in a
carrageenan-induced inflammatory model by stimulating CB2 receptor
activity on peripheral cells, decreasing the release of cytokines,
and regulating the oxidant/anti-oxidant balance. This model of
inflammatory was chosen because it is widely used to study acute
inflammation in rodents. Therefore, the aim of this study was to
investigate: i) presence of CB2 receptor in paw tissue; ii) a
pharmacologic role of CB2 receptor; iii) effects of CB2 receptor
agonist and antagonist exogenous administration on inflammation,
and iv) a distinctive mechanism for inflammatory processes via CB2
receptors of cannabinoid system in carrageenan induced inflammatory
paw oedema in rats.
Materials and methods
Chemicals
Chemicals including Carrageenan, GW405833, AM630,
and other reagents including dimethyl sulfoxide (DMSO),
trichloroacetic acid (TCA), thiobarbituric acid (TBA),
dithiobisnitrobenzoate (DTNB), hexadecyltrimethylammonium bromide
(HETAB), and o-dianisidine were obtained from Sigma-Aldrich; Merck
KGaA (Darmstadt, Germany). All other chemicals were obtained from
analytical grade and standard commercial suppliers. Diclofenac was
obtained from Abdi Ibrahim Ilaç San Tic AŞ (Turkey). GW405833, and
AM 630 were dissolved in 2% DMSO. TNF-α and IL-1β Elisa (Novex)
kits useful for rat were purchased from Thermo Fisher Scientific,
Inc., Waltham, MA, USA.
Animals
Wistar-albino rats weighing 200–250 g were randomly
housed in appropriate cages at 22±2°C under a 12/12-h dark/light
cycle with free access to tap water and commercial rat chow. Ethics
committee approval was obtained from Mustafa Kemal University with
protocol no. 2013/7-17. Procedures were in accordance with the
Guide for Care and Use of Laboratory Animals.
Experimental Protocol and Carrageenan
induced paw oedema
Both CB2 agonist (GW 405833) and CB2 antagonist
(AM630) were given in doses of 3 and 1 mg/kg, respectively, by an
intravenous route from rat tail after dissolving in 2% DMSO. The
doses of CB2 agonist and antagonist compounds were chosen according
to literature (16,17). After non-selective cyclooxygenase
(COX) inhibitor drug, diclofenac, and carrageenan were dissolved in
1% sterile saline, diclofenac was administered at a dose of 10
mg/kg. The rats were randomly assigned 7 groups, with 6 animals in
each group (n=6): 1: Saline control, 2: CAR (carrageenan), 3:
CAR+DIC (carrageenan plus diclofenac), 4: CAR+AGO (carrageenan plus
CB2 receptor agonist), 5: CAR+ANTA (carrageenan plus CB2 receptor
antagonist), 6: CAR+AGO+ANTA (carrageenan plus CB2 receptor agonist
plus CB2 receptor antagonist), 7: CAR+Vehicle (carrageenan plus
DMSO). Antagonist was administered at 5 min. prior to agonist.
Diclofenac was applied intraperitoneally, carrageenan was applied
as intraplantar injection.
Ten minutes after the last drug administration, into
the plantar surface of the right hind paw of all rats, except
saline control group, were given a 0.1 ml volume of the previously
prepared 1% carrageenan solution, resulting in the include of the
paw oedema. All local injections were made at same volume. The
oedema formation was assessed as an increase in paw thickness at
the dorsal-planter axis at the metatarsal level using a calliper.
Basal thickness was determined before animals were given the drug.
The changes in the paw thickness were measured at 0, 1, 2, 3, and 4
h. Relation between paw thickness and basal values was found and
noted. To determine the anti-inflammatory potency, the relation
between all drug-given groups and 1% carrageenan group was
expressed as %.
Animals were anesthetized with ketamine (50 mg/kg,
IP) and xylazine (5 mg/kg, IP). They were monitored for loss of the
tail reflex, which was defined as loss of the twitching or movement
of the tail pinched using the fingers.
Blood was collected by cardiac puncture and
centrifuged at 3000 rpm for 10 min to obtain serum. Serum samples
were stored at −80°C up to analysis. Animals were killed by neck
dislocation and soon paw tissue and skin samples were taken for
histopathological evaluation and biochemical analyses.
Cytokine measurements in serum
IL-1β and TNF-α levels were determined using an
ELISA kits for rats and this process was performed 2 times,
according to the procedures supplied by the manufacturer (Thermo
Fisher Scientefic, Inc.) and the results expressed as pg/ml of
serum.
Malondialdehyde (MDA) measurements in
paw tissue
The lipid peroxide levels, as MDA concentrations, in
the paw tissues were according to the method previously described
(18). Briefly the tissue samples
were homogenized in an ice bath, ice-cold TCA by adding 10 ml of
10% TCA per g of tissue, with an ultrasonic tissue homogenizer.
After two consecutive centrifugations at 3,000 g for 15 min, 500 µl
supernatant was mixed with equal volume of 0.67% TBA and heated to
100°C for 15 min. The absorbances of the samples were then measured
spectrophotometrically at 535 nm. Each assay was performed in
duplicate.
Glutathione (GSH) levels in paw
tissue
GSH contents of the paw tissue samples were measured
by a modified Elman method (19). To
the 0.5 ml of supernatant obtained by using the same homogenization
procedure as described above, 2 ml 0.3 M Na2HPO4 solution was
added. A 0.2 ml solution of DTNB was added into the mixture, and
the absorbance at 412 nm was measured immediately after vortexing.
Each assay was performed in duplicate.
Myeloperoxidase (MPO) activity
To determine MPO activity, an index of
polymorphonuclear cell accumulation, exactly 100 mg of tissue was
weighed and homogenized in potassium buffer containing 0.5%
hexadecyltrimethylammonium bromide (HETAB). To obtain tissue
pellet, homogenates were centrifuged at 4,500 g for 15 min at 4°C
and then they were resuspended, and MPO activity was assayed by
measuring the change in absorbance at 460 nm using
o-dianisidinedihydrochloride and hydrogen peroxide (20). Each assay was performed in duplicate.
One unit of MPO activity was defined as that degrading 1 µmol of
peroxide per min at 25°C. The activity was then normalized as unit
per mg of tissue (U/mg).
Histopathological analyses
The paw tissue and skin samples were fixed in 10%
formaldehyde solution for 10 days. The samples were dehydrated,
cleared and embedded in increasing alcohol series (EMD Millipore,
Billerica, MA, USA), in xylene series (EMD Millipore) and in the
paraffin blocks, respectively. To evaluate histological analyses,
before these blocks were stained with hematoxylin and eosin
(H&E) (EMD Millipore) the blocks were cut into 10 µm thick
sections and mounted on glass slides. After than this sections were
evaluated with a camera attachment microscope (Carl Zeiss Axiocam
ERc5s; Carl Zeiss AG, Oberkochen, Germany). All histological
analyses were performed by an observer blinded to the treatment
protocol. To calculate both the results and the mean of the
numerical intensity of inflammation, in each H&E section
approximately 700 inflammatory cells were evaluated. The degree of
inflammation was evaluated with a score from 0 to 5, defined as
follows: 0=no inflammation, 1=weak inflammation, 2=mild
inflammation, 3=moderate inflammation, 4=moderate/severe
inflammation and 5=severe inflammation.
Statistical analysis
For 6 animals per group, the data are expressed as
mean value SEM. For the present study, one-way analysis of variance
(ANOVA) was used and Bonferroni post hoc test for multiple
comparisons was followed. The pharmacological roles of CB2 receptor
agonist and/or antagonist on carrageenan-induced inflammatory
processes were compared with that in the diclofenac and carrageenan
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
Macroscopic results of anti-oedematous
activity
The anti-inflammatory activities of CB2 receptor
agonist, CB2 receptor antagonist, and diclofenac on
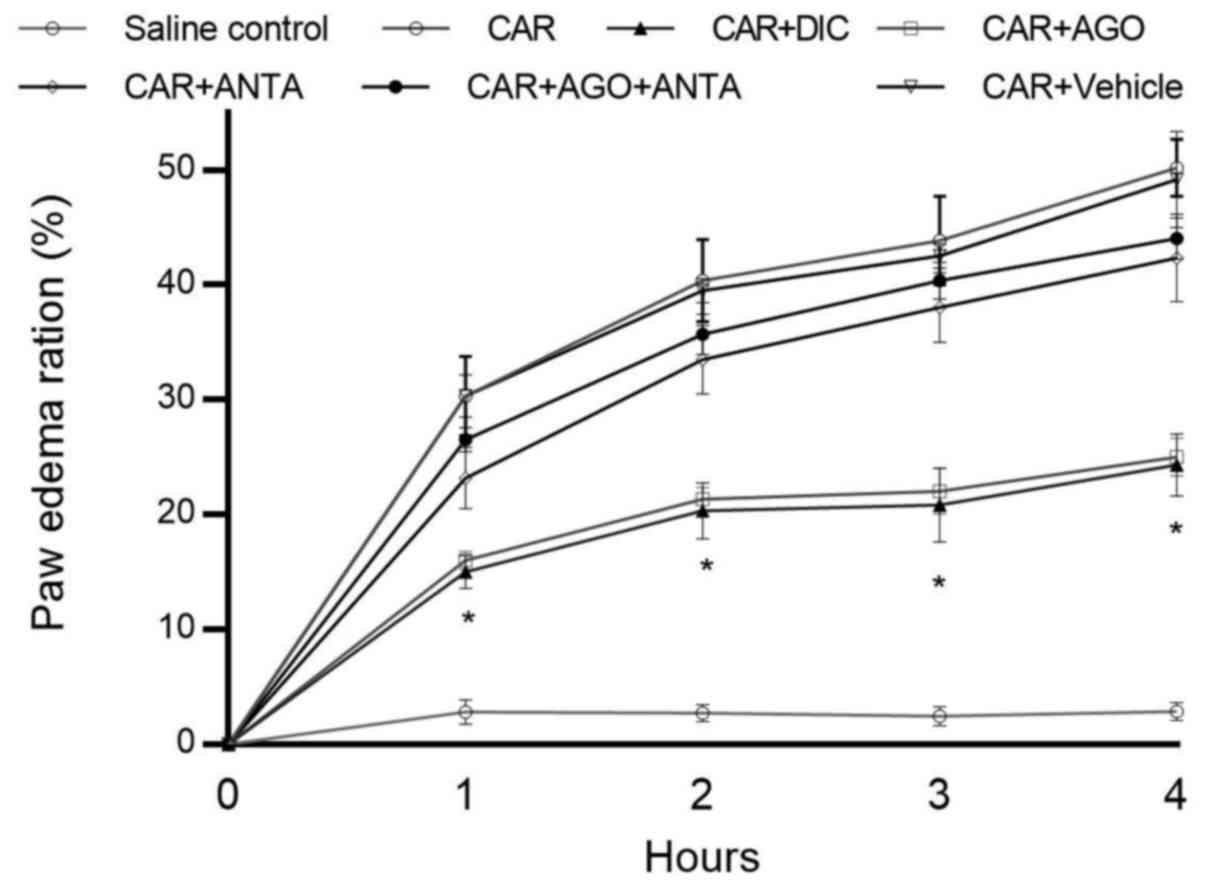
carrageenan-induced acute paw oedema were shown in Fig. 1. Intraplantar injection of
carrageenan into the rat paws caused the clear thickening of the
paw at 4 h after the administration. Carrageenan-induced paw oedema
was been reduced in the diclofenac and CB2 agonist given group. The
anti-oedematous activity of CB2 receptor agonist was comparable to
that of diclofenac. When the antagonist was given alone, it had no
any anti-inflammatory activity. Moreover, when administered in
combination with the agonist, antagonist reversed anti-inflammatory
effect of the agonist.
Cytokine, IL-1β, and TNF-α, serum
measurements
Serum cytokine levels in the group treated with
carrageenan were seen to significantly increase (Fig. 2). These levels were observed to
significantly decrease in the CB2 agonist or diclofenac given
groups. However, these decreases in cytokine levels were not seen
in the antagonist group. As reversed anti-inflammatory effect, the
effects of agonist were reversed by CB2 antagonist on serum
cytokine levels.
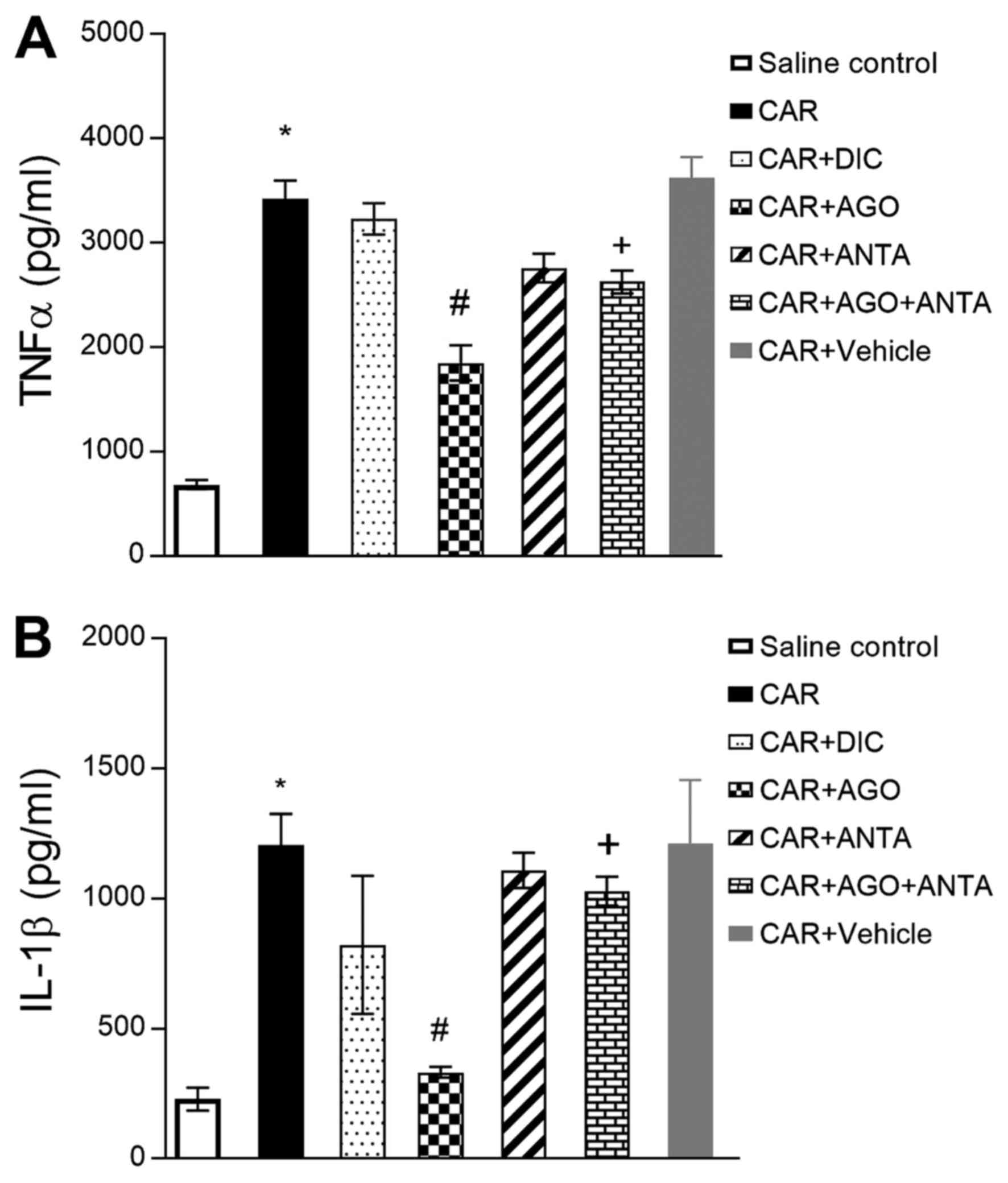 | Figure 2.Effects of GW405833 (CAR+AGO), AM630
(CAR+ANTA), their combination (CAR+AGO+ANTA) and diclofenac
(CAR+DIC) on serum (A)TNF-α and (B) IL-1β activities in
carrageenan-induced paw inflammation of rat (4th hour). Each point
represents the mean ± SEM of rats (n=6). *Statistically significant
compared with saline control group, #statistically
significant compared with carrageenan group, and
+statistically significant compared with agonist-treated
group at P<0.05. TNF-α, tumor necrosis factor-alpha; IL-1β,
interleukin 1β. AGO, agonist; ANTA, antagonist; DIC, diclofenac;
CAR, carrageenan. |
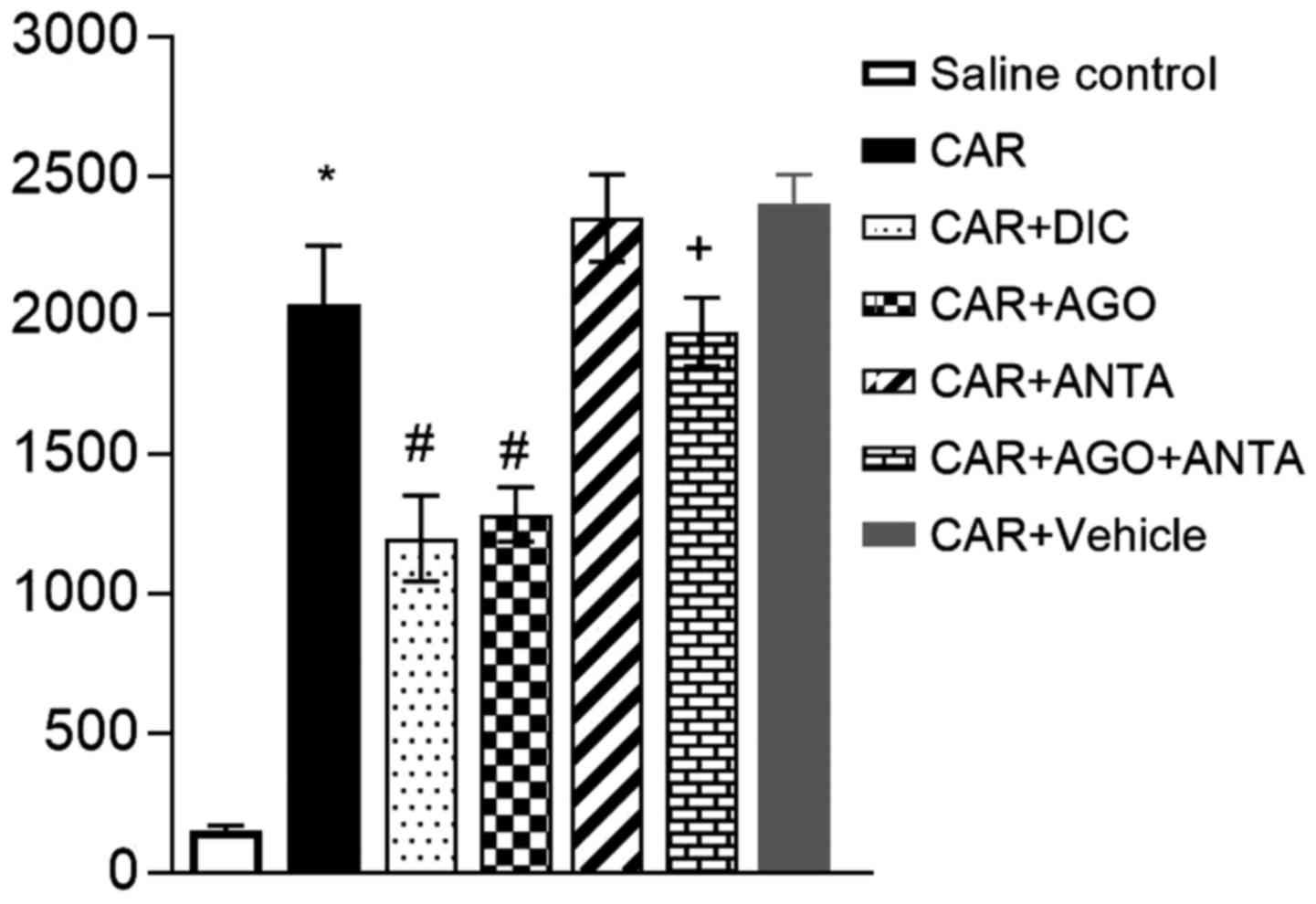
Oxidant and antioxidant activity in
paw tissue
Carrageenan application was found to increase the
MDA concentration in rat paw tissues. The MDA concentration was
significantly lower in the diclofenac and agonist group than in the
carrageenan group. An increase in the inducer effect of carrageenan
in the antagonist group alone was no found to alteration, while the
beneficial effect of agonist was found to be caused by the
combination of antagonist and agonist. It was decreased GSH level
in inflammatory tissue, while level of GSH was increased in both
diclofenac and agonist-given group (Fig.
3).
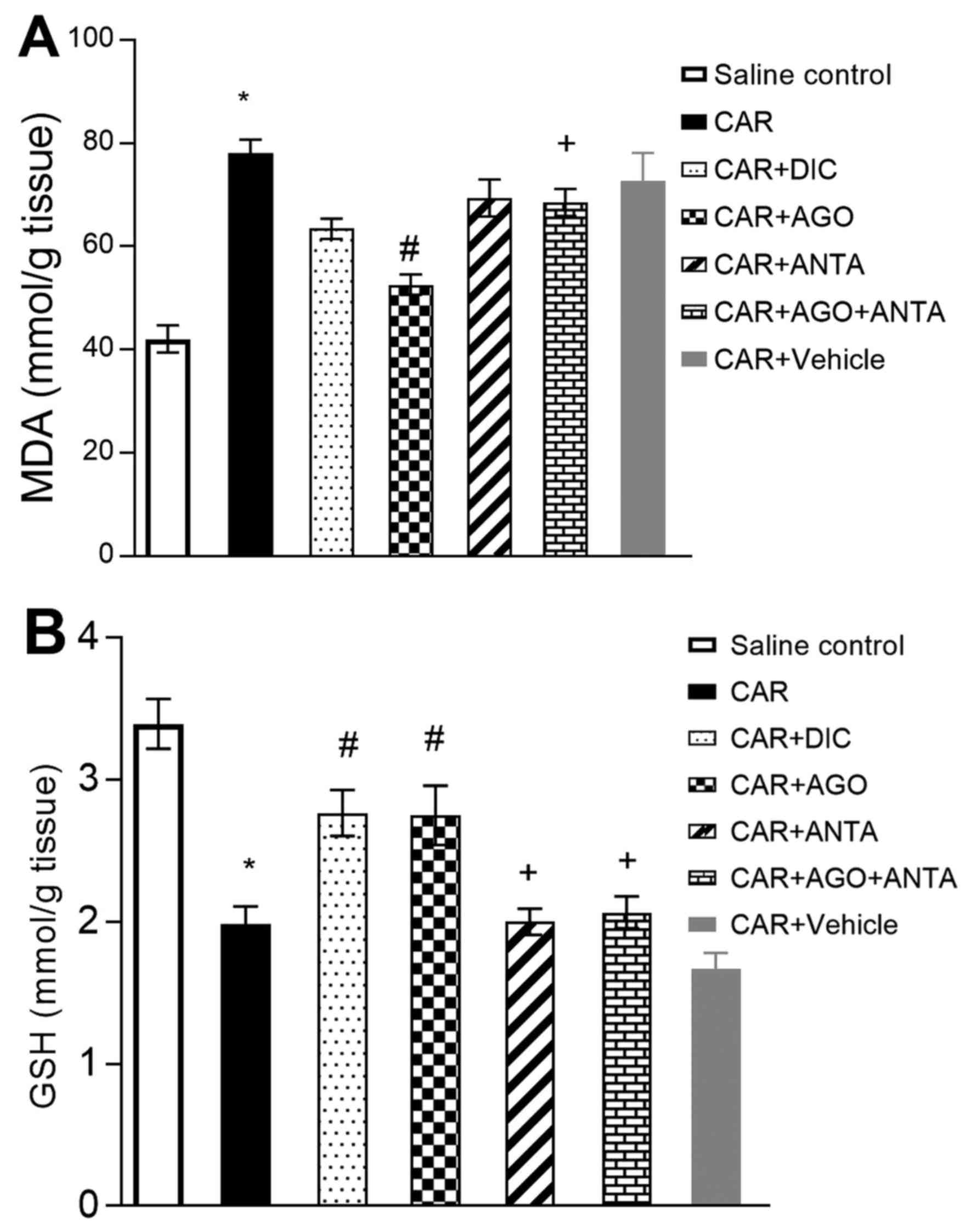 | Figure 3.Effects of GW405833 (CAR+AGO), AM630
(CAR+ANTA), their combination (CAR+AGO+ANTA) and diclofenac
(CAR+DIC) on (A) MDA and (B) GSH levels in carrageenan-injected paw
tissues (4th hour). Each point represents the mean ± SEM of rats
(n=6). *Statistically significant compared with saline control
group, #statistically significant compared with
carrageenan group, and +statistically significant
compared with agonist-treated group at P<0.05. GSH, glutathione;
MDA, malondialdehyde. AGO, agonist; ANTA, antagonist; DIC,
diclofenac; CAR, carrageenan. |
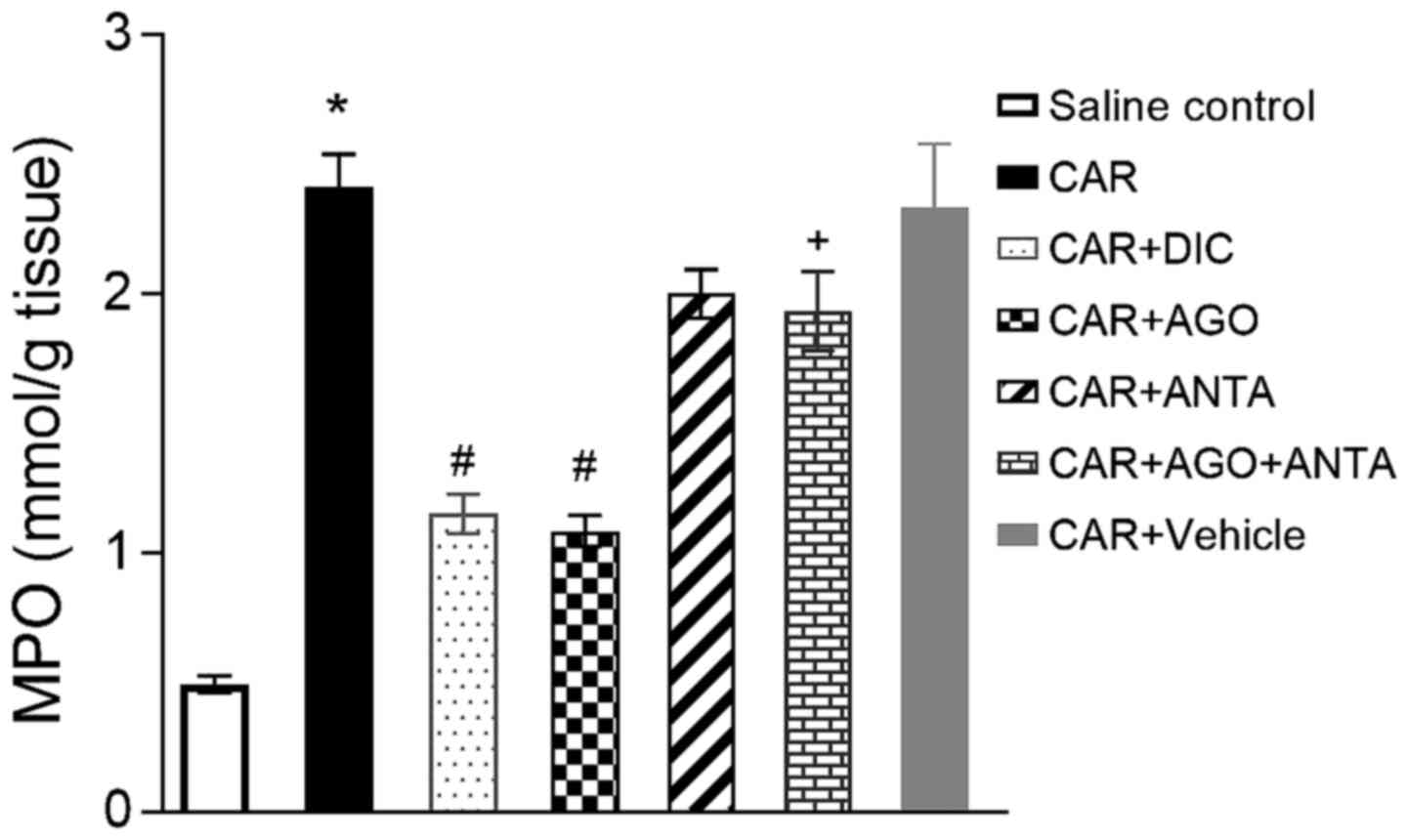
MPO activity in paw tissue
The development of histological damage was
associated with increased infiltration of neutrophils as shown by
an increase in MPO activity, a peroxidase enzyme released by
neutrophils and considered a marker of neutrophilic infiltration.
The administration of either cannabinoid 2 agonist (GW405833, 3
mg/kg) or diclofenac (10 mg/kg) each significantly reduced MPO
activity (Fig. 4), although
cannabinoid 2 antagonist, AM630, was not significantly effect.
Histopathological results
Histological changes in oedema paws 4 h after
injection of carrageenan. Light micrograph of rat paw in all
studied groups. Paws were harvested 4 h after injection of
carrageenan and subjected to histochemical staining of paw tissues.
Saline control group was showing no histopathological changes. The
inflammation score for the experimental groups (carrageenan,
antagonist, and vehicle) was more than that of the saline control
group and treatment groups (agonist or diclofenac).
Carrageenan-treated and Vehicle group showing marked (+++++)
inflammatory reaction associated with intermuscular infiltration
with massive number of neutrophils. CAR+DIC group and CAR+AGO group
are showing weak (+) inflammatory reactions. CAR+ANTA group is
showing (++++) inflammatory reaction. CAR+AGO+ANTA group is showing
mild (++) inflammatory reaction (Fig.
5).
Histopathological pictures of all groups are
summarized in Fig. 6. In the paw
samples of the CAR, CAR+ANTA, CAR+AGO+ANTA, and CAR+Vehicle groups,
the inflammatory infiltrates were abundant. In the all these
groups, collagen and elastic bundles, more fibroblast profiles,
capillaries, dense lymphocyte, white blood cells (especially
neutrophil), macrophage-like cells were found, as compared with
saline control or groups treated with agonist or diclofenac.
 | Figure 6.Histological changes in oedema paws 4
h after injection of carrageenan. Light micrograph of rat paw in
all studied groups. Paws were harvested 4 h after injection of
carrageenan and subjected to histochemical staining of paw tissues.
Histopathological pictures of all groups are summarized in Fig. 6. Saline control group was showing no
histopathological changes. The amount of inflammatory cells in the
carrageenan group was higher than that of the saline control group
and agonist or diclofenac groups. There is a slight difference
between saline control and treatments group in term of tissue
content. The amount of inflammatory cells in the antagonist and
agonist+antagonist groups were higher than that of the saline
control group and agonist or diclofenac groups. The amount of
inflammatory cells in the vehicle group was higher than that of the
saline control group and agonist or diclofenac groups. H&E
staining, the magnifications of images a, b, and c are at ×4, ×10
and ×40, respectively. Epi, epidermis; Der, dermis; AT, adipose
tissue; SG, Sweet gland; Fb, fibroblast; bv, blood vessel; Ef,
elastic fiber; Cf, collagen fiber. IA, inflammation area; Rbc, red
blood cell; Ic, inflammatory cell; MC, macrophage like cell. Ac,
adipose cell; MT, muscle tissue. (A) Saline control group; (B)
carrageenan model group; (C) diclofenac group; GW405833, (D) CB2
agonist group; (E) AM630, CB2 antagonist group; (F) CB2
agonist+antagonist group; and (G) Vehicle group. AGO: Agonist,
ANTA, antagonist; DIC, diclofenac; CAR, Carrageenan, Vehicle,
dimethyl sulfoxide. |
Saline control group results: Histologically, the
structures of the epidermis, stratified squamous epithelial cells
and dermis were normal. The fibers and connective tissue cells also
were normal.
CAR group results: Observation of inflammatory cells
in the dermis and hypodermis was evident. The inflammation score
for this group was more than saline control, CAR+AGO, and CAR+DIC
groups. In addition, macrophage like cells was observed in the
hypodermis, around the vessels.
CAR+DIC group results: The appearance of the dermis,
hypodermis, epidermis and other tissue components, including fibers
and connective tissue cells were similar to those in the saline
control group.
CAR+AGO group results: The tissues in this group
were similar to those in the healthy group. In term of epidermis,
dermis, hypodermis and other tissue components, including fibers
and connective tissue cells, no significant difference was found
between the tissues in this group and either the saline control or
diclofenac group.
CAR+ANTA group results: Compared with the
carrageenan group, dermis and hypodermis were found to have fewer
inflammatory cells, but the inflammation cell density in this group
was higher than the saline control and treatment group.
CAR+AGO+ANTA group results: The appearance of the
epidermis, dermis, hypodermis and other tissue components,
including fibers and connective tissue cells were similar to the
carrageenan group. There was a significant difference compared to
the saline control and diclofenac group in term of inflammatory
cells density. In addition, but in the epidermis around the
vessels, macrophage-like cells were observed.
CAR+Vehicle group results: This group is associated
with a large number of inflammatory cells in dermis and hypodermis
when compared with the saline control and treatment groups.
Moreover, in the hypodermis around the vessels, macrophage-like
cells were observed.
Discussion
Although the role of the CB2 receptor in the
regulation of inflammation has been recently researched, its
mechanism has not been fully understood. The results of this study
confirm the effects of exogenous CB2 agonist, and a pharmacologic
role for CB2 receptor on carrageenan-induced paw oedema in rats. We
demonstrate that pre-treatment of rat with CB2 selective receptor
agonist GW405833 decreases paw oedema, tissue inflammatory cell
infiltration and increased MPO level, tissue lipid peroxidation,
and serum TNF-α level. CB2 receptor activation also increases the
tissue GSH level.
The process of inflammation is actually a response
to various stimuli against those who have given organism. Some of
these stimuli are trauma, infection, ischemia/reperfusion and
immunological reactions (21,22).
Carrageenan-induced paw oedema is a well-established model for
evaluating acute inflammation and anti-inflammatory effect of
candidate drugs (9). That oedema
involves both neurogenic and non-neurogenic mechanisms which have
been associated with arachidonic acid cascade which leads to the
formation of eicosanoids, prostaglandin, thromboxane, leukotriene,
reactive oxygen species, cytokines and other inflammatory
mediators, which were mediated by activity of 5-lipoxygenase and
COX enzymes (8). To date, several
previous studies showed endocannabinoid system have been major
roles in nociception or inflammatory reactions by receptor
activation (23,24) or exogenous agonist administrations
(25–30) or inhibition of metabolized enzyme of
2-AG ligand (13,31,32). In
this study we showed that both CB2 receptor selective agonist
GW405833 and non-specific COX inhibitor drug diclofenac, clearly
inhibited the development of carrageenan-induced oedema. Elmes
et al speculate that CB2 agonist JWH-133 on paw volume been
comparable to the effect of systemic post-administration of
morphine and rofecoxib (25). Ghosh
et al declared that JZL184, selective MAGL inhibitor,
reduces carrageenan-induced paw oedema were mediated through CB2
receptors (13). Findings of a study
can be indirectly support our clear observations (33). Because of that results, related to
CB1 receptor way, said do not support a role for anandamide (AEA),
CB1 receptor ligand, in preventing carrageenan-induced oedema,
while palmitoylethanolamide (PEA), an endogenous fatty acid,
administration and the catabolic enzyme fatty acid amide hydrolase
(FAAH) blockade elicited anti-oedema effects of an equivalent
magnitude as produced by, dexamethasone, and diclofenac. Due to
confirm the occurrence of CB2 receptor, we administered CB2
receptor antagonist AM630 plus to CB2 receptor agonist in
carrageenan-induced inflammatory animals. CB2 receptor antagonist
clearly reversed the anti-oedematous effect of agonist in paw
tissue. However CB2 receptor antagonist has no effect on
inflammation response.
At the end of the histopathological examinations; it
was found that there was a significant decrease in the number of
inflammatory cells in the agonist and diclofenac treated groups,
although there were a large number of inflammatory cells in the
tissue samples in the antagonist and carrageenan groups. As seen in
macroscopic analysis, the effect of the COX inhibitor diclofenac
was similar to anti-inflammatory effect of CB2 agonist. Haruna
et al (34), have showed that
the S-777469, (CB2) agonist, reduced the epidermal thickness and
the number of mast cells infiltrating skin lesions of DNFB-induced
ear swelling in mice. MAGL inhibition in diseased mice increased
2-arachidonoylglycerol levels, leading to a reduction of
macroscopic and histological colon alterations in the
trinitrobenzene sulfonic acid (TNBS)-induced colitis (31). Administration of antagonist alone did
not contribute to anti-inflammatory effect. This data suggests that
the CB2 receptor antagonist reversed the effect of the CB2 receptor
agonist.
In pathological conditions and triggers inflammatory
signals, the ability of diclofenac to control carrageenan-induced
paw oedema is through the inhibition of COX, which is overexpressed
in inflammation. Our data show that CB2 receptor agonist is as
effective as the COX inhibitor to control the development of
carrageenan induced paw oedema. CB2 receptor agonist may be
indirectly and/or directly decreases prostaglandin and other
pro-inflammatory mediator production through enhanced COX-2
expression. This data is supported by recent studies. 2-AG limits
COX-2 elevation in neuronal inflammation (35). MAGL inhibition by KML29 induced a
decrease in prostaglandin levels in most peripheral tissues
(32). On the other hand, COX
activity is induced by the endocannabinoids, specifically by the
ligand AEA for CB1 and by inverse agonism of CB2 (36). Another data is that cannabinoid
receptor 2 suppresses leukocyte inflammatory migration by
modulating the lipoxygenase pathway (37).
Cell migration is an important step of the
inflammatory process of the injured tissue. So, the
anti-inflammatory effects of CB2 agonists to be understood that
involve the inhibition of cell migration especially neutrophil
migration, we measured the MPO activity in the tissue for this
purpose. The prominent enzyme of neutrophils is MPO, which are
released in the extracellular space or with their activation. The
more neutrophil chemotaxis and migration into the inflamed tissue,
the more MPO activity increases (38,39). As
it can be seen from the data we have obtained; the CB2 agonist
inhibits neutrophil infiltration, which was evident from the
reduced MPO activity. This demonstrates that neutrophil migration
to the inflamed site is inhibited by the CB2 agonist. JWH015,
selective CB2 agonist, is decrease leukocytes infiltration in both
submucosa and mucosa, as well as the myeloperoxydase activity, in
LPS treated mice (28).
During acute pancreatitis, an upregulation
especially of CB2 on apoptotic cells is occur (40). Cannabinoids may limit hepatic injury
by modulating the expression of adhesion molecules and the
infiltration and activation of inflammatory cells by CB2-dependent
mechanisms (4). Carrageenan-induced
paw inflammation is dependent on PMN infiltration. TNF-α acts as a
continuous stimulator for neutrophil infiltration in the
inflammatory tissues and it also up-regulates the production of
cell-type specific leukocyte chemo attractants, known as
chemokines, which have also been shown to cause up-regulation of
cell adhesion molecules and neutrophil activation (41). We focused on TNF-α since it is
proposed to play an early and crucial role in the cascade of
pro-inflammatory cytokines and the subsequent inflammatory
processes (39). We have proved
inflammation induction with carrageenan led to an increase in serum
levels of TNF-α and IL-1β. It supported our findings in previous
studies (28,31,42,43).
Pro-inflammatory cytokines such as TNF-α and IL-1β
are important mediators associated with certain inflammatory
diseases such as rheumatoid arthritis etc (44). Thus, an effective way to treat
various inflammatory diseases can be a form of treatment for the
inhibition of these mediators. In our study, pre-treatment with CB2
agonist significantly reversed this significant increase,
suggesting that CB2 agonist exerted an anti-inflammatory action and
inhibited polymorph nuclear cell migration, probably by decreasing
TNF-α and IL-1β levels.
One of the major damages caused by inflammation is
free oxygen radicals. Reactive oxygen species are molecules that
are highly unstable and highly reactive (45). Acute inflammatory processes can cause
leukocyte migration occurs involve inflammation mediators including
neutrophil-derived active oxygen species and free radicals
(45). To understand the working
mechanism of agonist and antagonist, the lipid peroxidation
product, MDA, and antioxidant, GSH, parameter in paw tissue were
studied. The end-product of lipid peroxidation is MDA. It refers to
the imbalance between oxidant and antioxidant system. It is well
known that the tissue lipid peroxidation levels can affected and
the antioxidant levels in paw tissues can reduced in carrageenan
inflammation studies. Previous studies have showed that in
carrageenan-induced paw oedema, the elimination of toxic oxygen
metabolites depends on the efficacy of the antioxidants (8–10). GSH
is actually the main component included in the endogenous
antioxidant system. GSH reduces the cytotoxic effects of these
toxic oxygen metabolites (45). In
the present study, the GSH level was significantly higher in the
agonist group than in carrageenan group. When agonist was given
with carrageenan, it caused high MDA level to drop significantly,
and so we speculated that GW405833 decreases oxidative stress
caused by carrageenan inflammation. We think that the CB2 receptor
activation in peripheral tissues may be decrease the carrageenan
paw inflammation by reducing oxidative stress. It is mean that CB2
receptor agonists may be used as anti-inflammatory agents to
prevent inflammation. This study has shown that GW405833 may
contribute to the reduction of oxidative stress in a variety of
inflammatory diseases due to potential therapeutic effects. The
inflammation of paw tissue by reducing oxidative stress is
inhibited by CB2 agonist when administered to exogenous. So, it can
benefit from the activation of the CB2 receptor in order to prevent
inflammation. It has been suggested in our study that CB2-specific
agonists can be used to inhibit the production of both oxidative
stress and pro-inflammatory cytokines in inhibit of various
inflammatory diseases. JWH133, CB2 agonist, has the potential to
treat bacterial translocation and various relevant abnormalities
through inhibition of systemic/intestinal oxidative stress,
inflammatory cytokines and TNF-α release in cirrhosis (29).
We worked GW405833 as CB2 selective agonist to
control carrageenan-induced paw inflammation in rats. Because it
has highly selective on CB2 receptors, a lot of researchers had
used in the designed inflammatory models as sepsis (46), osteoarthritis (47), and inflammatory pain (48). The presence of CB2 receptors is
continuing to discuss while information about whether they are
present in peripheral tissues is limited. The protective effects of
the CB2 receptor stimulation caused us to hypothesize that the
presence of these receptors in the paw tissue and that the
expression of these receptors increases with inflammatory
stimuli.
In summary, our results demonstrate that CB2 agonist
has not only anti-inflammatory activity in the models of acute
inflammation but also the inhibition of neutrophil migration. This
effect, at least in part, is due to reduce IL-1β and TNF-α levels
and oxidative stress.
This results support the conclusion that CB2
receptor activation in the pathogenesis of acute inflammatory in
response to carrageenan. This study demonstrated that CB2 receptors
are present in the paw and that expression of CB2 receptors by
inflammatory stimulation is increased. The CB2 receptor agonist,
GW405833, exerted significant anti-inflammatory effects such as
diclofenac in anti-inflammatory effect. Co-administration of the
CB2 receptor antagonist AM630 with the CB2 agonist reversed this
effect pharmacologically. The anti-inflammatory effect of the CB2
receptor agonist may be due to decrease both in serum cytokine
levels and/or in oxidative stress and increase in unbalanced
antioxidant system. These findings forcefully suggest that
targeting CB2 cannabinoid receptors may represent a novel strategy
in protecting against inflammatory of peripheral cells. In
conclusion, In the prevention of inflammatory diseases, activate of
CB2 receptors may be a promising therapy.
Acknowledgements
The present study was presented orally at the
Pharmacology World Congress on July 20–22, 2015 Brisbane, Australia
(abstract no. 8).
Funding
The present study was supported by the Research Fund
of Adiyaman University (grant no. TIPFBAP/2013-0001).
Availability of data and materials
The datasets used and/or analysed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
AP, SAC, HK and MFD conceived and designed the
experiments, and provided the relevant materials and analytical
tools. AP and MFD performed the experiments. AY and EE performed
histological analysis. FU, MKO and SOA analyzed the data. SOA wrote
the manuscript.
Ethics approval and consent to
participate
Ethics committee approval was obtained from Mustafa
Kemal University with protocol number 2013/7-17. Procedures were in
accordance with the Guide for Care and Use of Laboratory
Animals.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Rom S and Persidsky Y: Cannabinoid
receptor 2: Potential role in immunomodulation and
neuroinflammation. J Neuroimmune Pharmacol. 8:608–620. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zogopoulos P, Vasileiou I, Patsouris E and
Theocharis SE: The role of endocannabinoids in pain modulation.
Fundam Clin Pharmacol. 27:64–80. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Witkamp R and Meijerink J: The
endocannabinoid system: An emerging key player in inflammation.
Curr Opin Clin Nutr Metab Care. 17:130–138. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bátkai S, Osei-Hyiaman D, Pan H, El-Assal
O, Rajesh M, Mukhopadhyay P, Hong F, Harvey-White J, Jafri A, Haskó
G, et al: Cannabinoid-2 receptor mediates protection against
hepatic ischemia/reperfusion injury. FASEB J. 21:1788–1800. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Núñez E, Benito C, Pazos MR, Barbachano A,
Fajardo O, González S, Tolón RM and Romero J: Cannabinoid CB2
receptors are expressed by perivascular microglial cells in the
human brain: an immunohistochemical study. Synapse. 53:208–213.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yao BB, Hsieh G, Daza AV, Fan Y, Grayson
GK, Garrison TR, El Kouhen O, Hooker BA, Pai M, Wensink EJ, et al:
Characterization of a cannabinoid CB2 receptor-selective agonist,
A-836339 [2,2,3,3-tetramethyl-cyclopropanecarboxylic acid
[3-(2-methoxy-ethyl)-4,5-dimethyl-3H-thiazol-(2Z)-ylidene]-amide],
using in vitro pharmacological assays, in vivo pain models, and
pharmacological magnetic resonance imaging. J Pharmacol Exp Ther.
328:141–151. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Marques TH, Marques ML, Medeiros JV, Silva
RO, dos Reis Barbosa AL, Lima TC, de Sousa DP and de Freitas RM:
Cyane-carvone, a synthetic derivative of carvone, inhibits
inflammatory response by reducing cytokine production and oxidative
stress and shows antinociceptive effect in mice. Inflammation.
37:966–977. 2014.PubMed/NCBI
|
|
8
|
Albayrak A, Halici Z, Cadirci E, Polat B,
Karakus E, Bayir Y, Unal D, Atasoy M and Dogrul A: Inflammation and
peripheral 5-HT7 receptors: The role of 5-HT7 receptors in
carrageenan induced inflammation in rats. Eur J Pharmacol.
715:270–279. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Büyükokuroğlu ME: Anti-inflammatory and
antinociceptive properties of dantrolene sodium in rats and mice.
Pharmacol Res. 45:455–460. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Uzkeser H, Cadirci E, Halici Z, Odabasoglu
F, Polat B, Yuksel TN, Ozaltin S and Atalay F: Anti-inflammatory
and antinociceptive effects of salbutamol on acute and chronic
models of inflammation in rats: Involvement of an antioxidant
mechanism. Mediators Inflamm. 2012:4389122012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Conti S, Costa B, Colleoni M, Parolaro D
and Giagnoni G: Antiinflammatory action of endocannabinoid
palmitoylethanolamide and the synthetic cannabinoid nabilone in a
model of acute inflammation in the rat. Br J Pharmacol.
135:181–187. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Iwamura H, Suzuki H, Ueda Y, Kaya T and
Inaba T: In Vitro and in vivo pharmacological characterization of
JTE- 907, a novel selective ligand for cannabinoid CB 2 receptor. J
Pharmacol Exp Ther. 296:420–425. 2001.PubMed/NCBI
|
|
13
|
Ghosh S, Wise LE, Chen Y, Gujjar R,
Mahadevan A, Cravatt BF and Lichtman AH: The monoacylglycerol
lipase inhibitor JZL184 suppresses inflammatory pain in the mouse
carrageenan model. Life Sci. 92:498–505. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Comelli F, Giagnoni G, Bettoni I, Colleoni
M and Costa B: The inhibition of monoacylglycerol lipase by URB602
showed an anti-inflammatory and anti-nociceptive effect in a murine
model of acute inflammation. Br J Pharmacol. 152:787–794. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lunn CA, Fine JS, Rojas-Triana A, Jackson
JV, Fan X, Kung TT, Gonsiorek W, Schwarz MA, Lavey B, Kozlowski JA,
et al: A novel cannabinoid peripheral cannabinoid
receptor-selective inverse agonist blocks leukocyte recruitment in
vivo. J Pharmacol Exp Ther. 316:780–788. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rivers-Auty JR, Smith PF and Ashton JC:
The cannabinoid CB2receptor agonist GW405833 does not ameliorate
brain damage induced by hypoxia-ischemia in rats. Neurosci Lett.
569:104–109. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hayakawa K, Mishima K, Nozako M, Hazekawa
M, Irie K, Fujioka M, Orito K, Abe K, Hasebe N, Egashira N, et al:
Delayed treatment with cannabidiol has a cerebroprotective action
via a cannabinoid receptor-independent myeloperoxidase-inhibiting
mechanism. J Neurochem. 102:1488–1496. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Casini AF, Ferrali M, Pompella A, Maellaro
E and Comporti M: Lipid peroxidation and cellular damage in
extrahepatic tissues of bromobenzene-intoxicated mice. Am J Pathol.
123:520–531. 1986.PubMed/NCBI
|
|
19
|
Sedlak J and Lindsay RH: Estimation of
total, protein-bound, and nonprotein sulfhydryl groups in tissue
with Ellman's reagent. Anal Biochem. 25:192–205. 1968. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bradley PP, Priebat DA, Christensen RD and
Rothstein G: Measurement of cutaneous inflammation: Estimation of
neutrophil content with an enzyme marker. J Invest Dermatol.
78:206–209. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Arslan SO, Gelir E, Sayan H and Ozacmak
VH: L-Arginine and melatonin interaction in rat intestinal
ischemia-reperfusion. Fundam Clin Pharmacol. 19:533–535. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Arslan SO: Morphine modulates
microvascular leakage dose-dependently in the airway of
ovalbumin-sensitized rats. Turk J Med Sci. 40:279–286. 2010.
|
|
23
|
Ashton JC and Glass M: The cannabinoid CB2
receptor as a target for inflammation-dependent neurodegeneration.
Curr Neuropharmacol. 5:73–80. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Berger ND, Gadotti VM, Petrov RR, Chapman
K, Diaz P and Zamponi GW: NMP-7 inhibits chronic inflammatory and
neuropathic pain via block of Cav3.2 T-type calcium channels and
activation of CB2 receptors. Mol Pain. 10:772014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Elmes SJ, Winyard LA, Medhurst SJ, Clayton
NM, Wilson AW, Kendall DA and Chapman V: Activation of CB1 and CB2
receptors attenuates the induction and maintenance of inflammatory
pain in the rat. Pain. 118:327–335. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Valenzano KJ, Tafesse L, Lee G, Harrison
JE, Boulet JM, Gottshall SL, Mark L, Pearson MS, Miller W, Shan S,
et al: Pharmacological and pharmacokinetic characterization of the
cannabinoid receptor 2 agonist, GW405833, utilizing rodent models
of acute and chronic pain, anxiety, ataxia and catalepsy.
Neuropharmacology. 48:658–672. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Harvey BS, Nicotra LL, Vu M and Smid SD:
Cannabinoid CB2 receptor activation attenuates cytokine-evoked
mucosal damage in a human colonic explant model without changing
epithelial permeability. Cytokine. 63:209–217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tambaro S, Antonietta M, Mastinu A and
Lazzari P: Evaluation of selective cannabinoid CB(1) and CB(2)
receptor agonists in a mouse model of lipopolysaccharide-induced
interstitial cystitis. Eur J Pharmacol. Elsevier. 729:67–74.
2014.
|
|
29
|
Yang YY, Hsieh SL, Lee PC, Yeh YC, Lee KC,
Hsieh YC, Wang YW, Lee TY, Huang YH, Chan CC and Lin HC: Long-term
cannabinoid type 2 receptor agonist therapy decreases bacterial
translocation in rats with cirrhosis and ascites. J Hepatol.
61:1004–1013. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Smoum R, Baraghithy S, Chourasia M, Breuer
A, Mussai N, Attar-Namdar M, Kogan NM, Raphael B, Bolognini D,
Cascio MG, et al: CB2 cannabinoid receptor agonist enantiomers
HU-433 and HU-308: An inverse relationship between binding affinity
and biological potency. Proc Natl Acad Sci USA. 112:8774–8779.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Alhouayek M, Lambert DM, Delzenne NM, Cani
PD and Muccioli GG: Increasing endogenous 2-arachidonoylglycerol
levels counteracts colitis and related systemic inflammation. FASEB
J. 25:2711–2721. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pasquarelli N, Porazik C, Hanselmann J,
Weydt P, Ferger B and Witting A: Comparative biochemical
characterization of the monoacylglycerol lipase inhibitor KML29 in
brain, spinal cord, liver, spleen, fat and muscle tissue.
Neuropharmacology. 91:148–156. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wise LE, Cannavacciulo R, Cravatt BF,
Martin BF and Lichtman AH: Evaluation of fatty acid amides in the
carrageenan-induced paw edema model. Neuropharmacology. 54:181–188.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Haruna T, Soga M, Morioka Y, Imura K,
Furue Y, Yamamoto M, Hayakawa J, Deguchi M, Arimura A and Yasui K:
The inhibitory effect of S-777469, a cannabinoid type 2 receptor
agonist, on skin inflammation in mice. Pharmacology. 99:259–267.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang J and Chen C: Endocannabinoid
2-arachidonoylglycerol protects neurons by limiting COX-2
elevation. J Biol Chem. 283:22601–22611. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kim J and Watkins BA: Cannabinoid receptor
antagonists and fatty acids alter endocannabinoid system gene
expression and COX activity. J Nutr Biochem. 25:815–823. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liu YJ, Fan HB, Jin Y, Ren CG, Jia XE,
Wang L, Chen Y, Dong M, Zhu KY, Dong ZW, et al: Cannabinoid
receptor 2 suppresses leukocyte inflammatory migration by
modulating the JNK/c-Jun/Alox5 pathway. J Biol Chem.
288:13551–13562. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nussbaum C, Klinke A, Adam M, Baldus S and
Sperandio M: Myeloperoxidase: A leukocyte-derived protagonist of
inflammation and cardiovascular disease. Antioxid Redox Signal.
18:692–713. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Winterbourn CC, Kettle AJ and Hampton MB:
Reactive oxygen species and neutrophil function. Annu Rev Biochem.
85:765–1092. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Michler T, Storr M, Kramer J, Ochs S, Malo
A, Reu S, Göke B and Schäfer C: Activation of cannabinoid receptor
2 reduces inflammation in acute experimental pancreatitis via
intra-acinar activation of p38 and MK2-dependent mechanisms. Am J
Physiol Gastrointest Liver Physiol. 304:G181–G192. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Neri M, Fineschi V, Di Paolo M, Pomara C,
Riezzo I, Turillazzi E and Cerretani D: Cardiac oxidative stress
and inflammatory cytokines response after myocardial infarction.
Curr Vasc Pharmacol. 13:26–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Li YY, Li YN, Ni JB, Chen CJ, Lv S, Chai
SY, Wu RH, Yüce B and Storr M: Involvement of cannabinoid-1 and
cannabinoid-2 receptors in septic ileus. Neurogastroenterol Motil.
22:e350–e388. 2010. View Article : Google Scholar
|
|
43
|
Silva RO, Sousa FB, Damasceno SR, Carvalho
NS, Silva VG, Oliveira FR, Sousa DP, Aragão KS, Barbosa AL, Freitas
RM and Medeiros JV: Phytol, a diterpene alcohol, inhibits the
inflammatory response by reducing cytokine production and oxidative
stress. Fundam Clin Pharmacol. 28:455–464. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Gui H, Tong Q, Qu W, Mao CM and Dai SM:
The endocannabinoid system and its therapeutic implications in
rheumatoid arthritis. Int Immunopharmacol. 26:86–91. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Arslan SO, Zerin M, Vural H and Coskun A:
The effect of melatonin on bleomycin-induced pulmonary fibrosis in
rats. J Pineal Res. 32:21–25. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gui H, Sun Y, Luo ZM, Su DF, Dai SM and
Liu X: Cannabinoid receptor 2 protects against acute experimental
sepsis in mice. Mediators Inflamm. 2013:7413032013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Schuelert N, Zhang C, Mogg AJ, Broad LM,
Hepburn DL, Nisenbaum ES, Johnson MP and McDougall JJ: Paradoxical
effects of the cannabinoid CB2 receptor agonist GW405833 on rat
osteoarthritic knee joint pain. Osteoarthr Cartil. 18:1536–1543.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Whiteside GT, Gottshall SL, Boulet JM,
Chaffer SM, Harrison JE, Pearson MS, Turchin PI, Mark L, Garrison
AE and Valenzano KJ: A role for cannabinoid receptors, but not
endogenous opioids, in the antinociceptive activity of the
CB2-selective agonist, GW405833. Eur J Pharmacol. 528:65–72. 2005.
View Article : Google Scholar : PubMed/NCBI
|