Introduction
Urinary calculus is commonly encountered in urinary
surgery, and its worldwide incidence has been constantly increasing
in recent years (1). The cause of
urinary calculus is complex and the major influencing factors
include individual and environmental factors. Urinary calculus is
likely to cause urinary tract obstruction, accompanied by dull or
colic pain in the waist, hematuria and fever, which severely
affects the quality of life and the health of affected patients
(2–4).
Non-contrast helical computed tomography (NCHCT) is
the gold standard for the diagnosis of urinary calculus, and
provides accurate information, including the size, location, shape
and amount of calculi. NCHCT provides a preliminary estimation of
the hardness and fragility of urinary calculi by measuring the
computed tomography (CT) value, which is the X-ray absorption
coefficient of a certain tissue/material, and the volume of the
calculus may be accurately calculated through the establishment of
a three-dimensional reconstruction model.
After the first successful application of holmium
laser to the urinary calculus (5),
this technique has received increasing attention. Due to its
precise and powerful fragmenting function and high safety, holmium
laser has become one of the most popular treatments for urinary
lithotripsy. Holmium laser is able to fragment the calculus more
effectively than extracorporeal shockwave and other lithotripsy,
and achieve a high calculus-free rate of >90% (6,7).
Compared to other fragmenting equipment, holmium laser generates
smaller fragments of the calculus, which are easier to discharge
(8). Holmium laser treatment causes
minimal injury to the surrounding area and is safer than other
fragmenting operations (9).
Certain studies have demonstrated that the mechanism
of holmium laser lithotripsy maybe a photomechanical effect. When
the laser fiber is exposed to the calculus, the large energy is
immediately absorbed by the internal and external water of the
calculus. Cavitation bubbles are produced at the water-calculus
interface and the shock waves produced by continuous rebound and
countless bursting of the bubbles are transferred to the calculus,
resulting in its fragmentation (10,11).
However, certain studies have refuted this view. Dushinski and
Lingeman (12) proposed that the
mechanism of fragmentation should be explained by the photothermal
effect on urinary calculi. Chan et al (13) also reported that the major mechanism
of holmium laser lithotripsy is the photothermal effect. The large
energy of the holmium laser increases the temperature of the
irradiated region and exceeds the threshold temperature. The heat
causes a chemical breakdown of the calculus and weakens the
structural integrity of the calculus, and it also contributes to
the fragmentation of the interstitial water and vapor
expansion.
Several parameters affecting the effect of
fragmentation of calculi by holmium laser lithotripsy have been
evaluated. Sea et al (14)
reported that with the increase of the frequency of constant pulse
energy, the crushing rate was not increased. Chawla et al
(15) have reported that the
crushing rate rises accompanied by the increase of the pulse
energy, but it does not increase with the pulse frequency.
Kronenberg and Traxer (16)
indicated that the setting of a low pulse repetition frequency and
high pulse energy achieved a higher crushing rate. Bader et
al (7) suggested that with the
same power setting, there was no significant difference in the
fragmentation rate between different pulse durations. Furthermore,
Kronenberg and Traxer (16) observed
that at the same power level, a setting with a low frequency and
high pulse energy is more efficient than high a frequency and low
pulse energy. They also identified a linear correlation of the
pulse energy with the size of the fragment, as well as with the
width and depth of the fissure.
Previous theoretical and clinical studies on holmium
laser lithotripsy mainly focused on the mechanism, influencing
factors, applied range and therapeutic efficacy of the operation.
Only a few studies have assessed the correlation between the
parameters of NCHCT and the total energy of holmium laser
lithotripsy (17). Few studies
investigated the guidance of the energy use in holmium laser
lithotripsy (18). The purpose of
the present study was to investigate the correlation between the
parameters of NCHCT and the total energy of lithotripsy (TEL), and
to establish a correlative mathematical model.
Materials and methods
Patients
A total of 125 patients with urinary calculi who
presented at Shandong Provincial Third Hospital (Jinan, China)
between March 2016 and February 2017, and who were scheduled for
holmium laser lithotripsy, were enrolled in the present study. The
present study was approved by the institutional review board of
Shandong Provincial Third Hospital. All patients provided written
informed consent prior to enrollment. Of all of the patients, 5
were excluded from the final analyses due to failure of their
lithotripsy: In three cases, the calculi in the subrenal calyx were
too secluded to be reached and fragmented effectively, and two
cases of residual fragments sized >4 mm after the procedure were
encountered. Therefore, the present study collected valid data from
120 patients. Of the 120 patients, 77 were female and 43 were male,
the age range was 26–78 years and the average age was 47.9±12.7
years.
NCHCT examination
NCHCT examination was performed with a Philips
Brilliance iCT 256 scanner (Philips Healthcare, Eindhoven, The
Netherlands). All patients were examined one day prior to
lithotripsy. The patients were fasted for at least 8 h to avoid any
interference from intestinal gas and chyme on the results of the
examination. The patients were requested to drink 500–1,000 ml
water at 1prior to the examination to moderately fill the
bladder.
The patients were scanned in a supine position with
their hands on their head, and were requested to hold their breath
during the scan. Image archiving and communication systems were
used to extract the CT images of the patients. First, the position
of each calculus was determined and the calculus was classified as
either a renal calculus and a ureteral calculus according to its
position, and the number of calculi was recorded. Furthermore, the
three-dimensional reconstruction model of the calculus was
established based on the CT images capturedon an Extended
Brilliance Workspace workstation (Philips Healthcare), and its
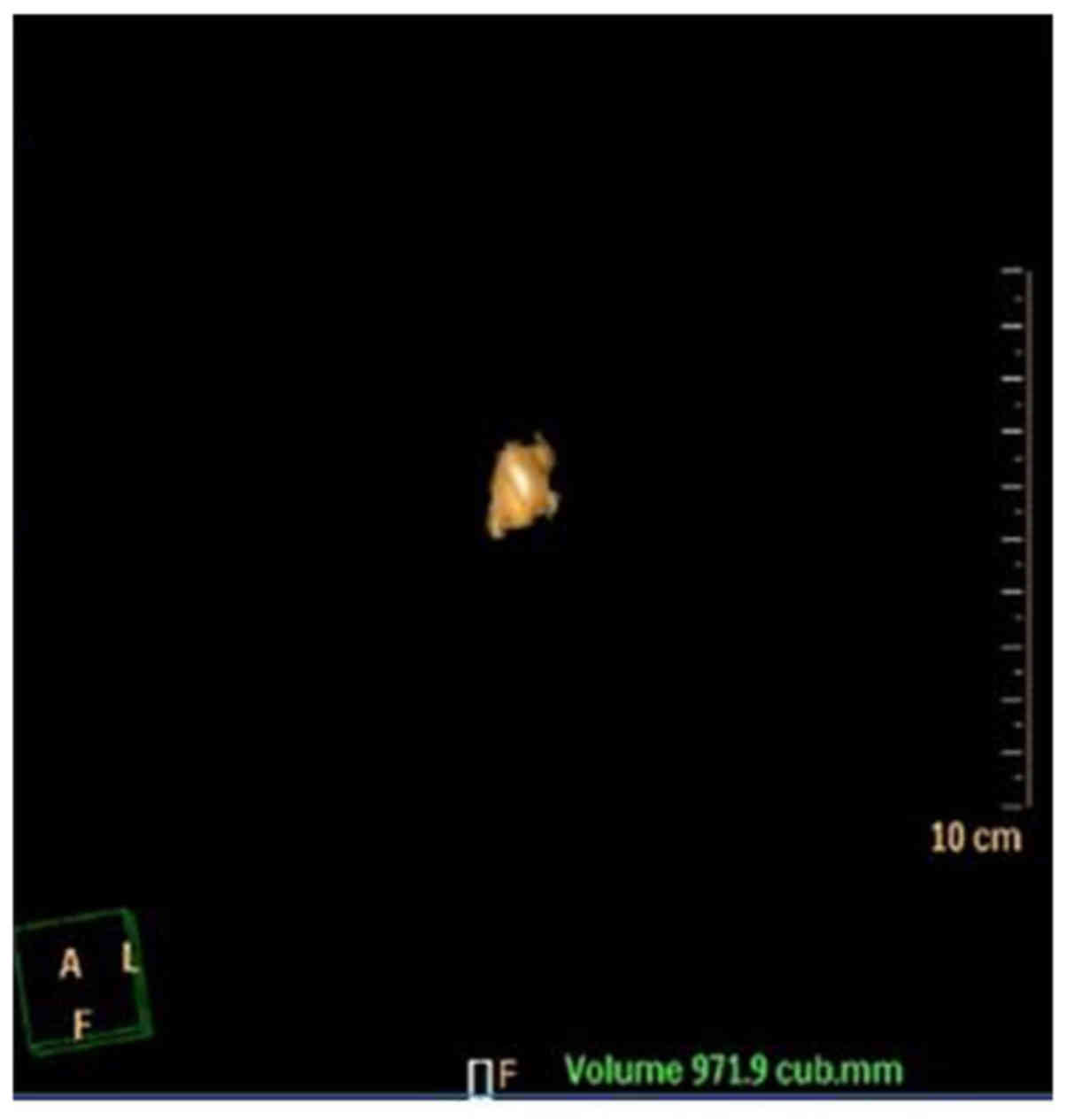
volume was calculated. The detailed procedures were as follows: i)
The calculus was manually colored, ii) the remnant was incised
around the calculus manually, iii) the three-dimensional image of
the calculus was reconstruct, iv) the calculus volume was
calculated using a computer (Fig.
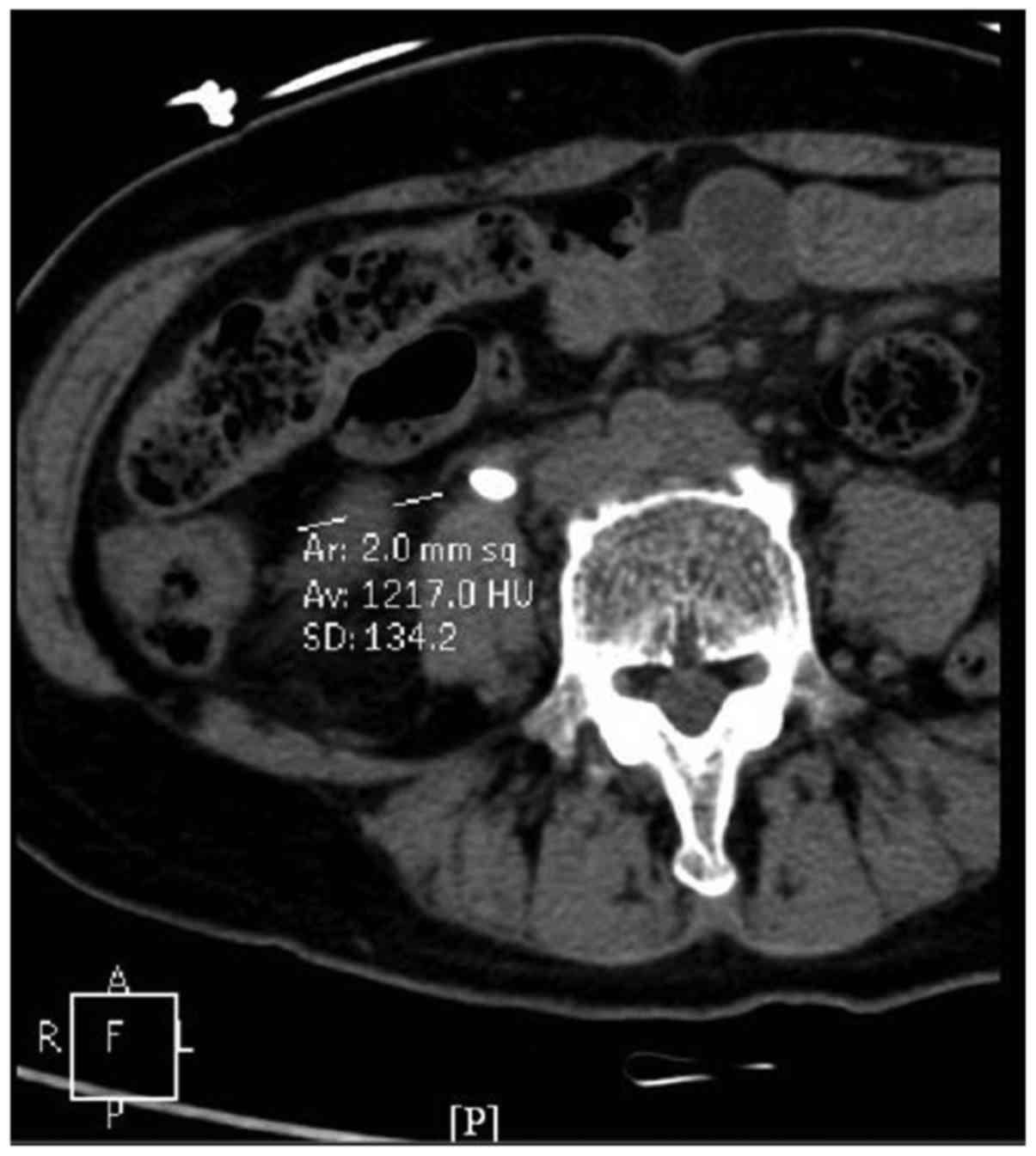
1). Third, the images were magnified by three-fold and the
image of the calculus displaying the longest diameter was selected,
in which the central region of 0.02 cm2 was selected as
the region of interest and the CT value was measured. Another two
adjacent images were used to measure the CT value of the region of
interest (Fig. 2). The average value
of the three measurements was used as the final CT value of the
calculus. It was divided into four grades: 1, CT <400 Hounsfield
units (Hu); 2, Ct=400–799 Hu; 3, CT=800-1,199 Hu; 4, CT ≥1,200
Hu.
Lithotripsy
All lithotripsy treatments were performed by two
surgical urologists (one with 5 years of experience and the other
with 7 years of experience), and with a dual-wavelength holmium
laser therapeutic machine (Power Suite 80/100w; Lumenis Ltd.,
Yokneam, Israel). The laser settings were as follows:
Excitation/emission wavelengths of the laser fiber at 200/365 µm
with an output energy of 0.5/0.6J and a pulse repetition rate of
20/35 Hz. During lithotripsy, the calculus was targeted and
fragmented into pieces as small as possible. Taking the laser fiber
as a frame of reference, all fragments sized >4 mm were removed
with a basket catheter. The total energy of the completed laser
lithotripsy was recorded. The criterion for a completed laser
lithotripsy was no residual fragment sized >4 mm, and smaller
fragments were expected to be spontaneously excreted. The efficacy
of the lithotripsy was evaluated 1–2 days after performing an
ultrasound and kidney, ureter and bladder X-ray.
Statistical analysis
All statistical analyses were performed with the
SPSS 17.0 software package (SPSS, Inc., Chicago, IL, USA). After
the normality was determined by the Kolmogorov-Smirmnov test,
Spearman's rank correlation analysis was used to assess the
correlations between the TEL, and the location, the volume and the
CT value of the calculi. Multivariate linear regression (forward
selection method) was performed to formulate a mathematical model
to estimate the TEL. P<0.05 was considered to indicate a
statistically significant difference.
Results
Overview of NCHCT
Concerning the calculus location, 48 patients
(40.0%) had renal calculi and 72 (60.0%) had ureteral calculi. In
terms of the calculus volume, the largest calculus was 1,347
mm3, the smallest was 254 mm3 and the average
volume of the calculi was 485.35±195.349 mm3. The
highest CT value was 1,475 Hu and the lowest CT value was 374 Hu,
and the average CT value of the calculi was 927.27±275.186 Hu.
Among them, 5 calculi (4.2%) were grade 1, 32 (26.7%) were grade 2,
63 (52.5%) were grade 3 and 20 (16.6%) were grade 4 (Table I).
 | Table I.Parameters of non-contrast helical
computed tomography. |
Table I.
Parameters of non-contrast helical
computed tomography.
| Parameters | Value |
|---|
| Calculus
location |
|
|
Renal | 48 (40) |
|
Ureteral | 72 (60) |
| CT value (Hu) | 927.27±275.186 |
| Grade |
|
| 1 (400
Hu) | 5 (4.2) |
| 2
(400–799 Hu) | 32 (26.7) |
| 3
(800–1,199 Hu) | 63 (52.5) |
| 4 (≥1,200
Hu) | 20 (16.6) |
| Calculus volume
(mm3) | 485.35±195.349 |
TEL correlates with the calculus
location, volume and CT value
A strong negative correlation was identified between
the TEL and calculus location (r=−0.819, P<0.001); the TEL for
the renal calculus was higher compared with the TEL for the
ureteral calculus. There was a strong positive correlation between
the volume of the calculus and the TEL (r=0.827, P<0.001);
larger calculus required higher TEL. A moderate correlation between
the CT value of the calculus and the TEL was identified (r=0.468,
P<0.001); a calculus with a higher CT value required higher TEL.
Multivariate linear regression analysis revealed that the location,
the volume and the CT value of the calculus were independently
associated with the TEL (P<0.01; Table II).
 | Table II.Results of Spearman's rank correlation
analysis of the parameters of NCHCT and the total energy of holmium
laser lithotripsy. |
Table II.
Results of Spearman's rank correlation
analysis of the parameters of NCHCT and the total energy of holmium
laser lithotripsy.
| Parameters | r | P-value |
|---|
| Calculus
location | −0.819 | <0.001 |
| Calculus volume | 0.827 | <0.001 |
| CT Value | 0.468 | <0.001 |
Establishment of the mathematical
model
To estimate the TEL of different calculi, a
multivariate linear regression model was established with the
following parameters: Calculus location, volume and CT value
(Table III). After collinearity
was eliminated and normality was tested (Figs. 3 and 4), the following multivariate linear
regression equation was obtained: TEL (J)=753.328–328.835× calculus
location (0=renal calculus; 1=ureteral calculus) + 0.940× calculus
volume (mm3) + 0.421× CT value (Hu) (F=288.858, adjusted
R2=0.879, P<0.01). The adjusted R2
indicated that the energy variation based on the location, the
volume and the CT value of the calculus accounted for 87.9% of the
samples. The equation indicated that more energy was required for
lithotripsy in patients with a renal calculus, a calculus with a
larger volume and a calculus with a higher CT value.
 | Table III.Results of multivariate linear
regression analysis of the variables estimating the total energy of
holmium laser lithotripsy. |
Table III.
Results of multivariate linear
regression analysis of the variables estimating the total energy of
holmium laser lithotripsy.
| Variables | B | SE | t | P-value |
|---|
| Constant | 753.328 | 67.941 | 11.088 | <0.001 |
| Calculus
location | −328.835 | 32.965 | −9.975 | <0.001 |
| Calculus volume | 0.940 | 0.084 | 11.244 | <0.001 |
| CT value | −0.421 | 0.045 | 9.373 | <0.001 |
Discussion
The TEL required to fragment calculi varies
depending on their specific features. Unguided use of energy in
lithotripsy has certain disadvantages. Insufficient energy may not
fragment the calculus effectively, while excessive energy may lead
to a higher incidence of complications and adverse effects. If the
TEL was to be estimated pre-operatively, the urologist would be
able to predict the difficulty of the operation and arrange for a
suitable type of anesthesia, operation monitoring and drug
treatment, and the emergency program for high-risk patientsmay also
be performed in advance. To facilitate the pre-operativeestimation
of the TEL, a mathematical model correlating the parameters of
NCHCT with the TEL was established in the present study, which may
provide a foundation to guide the use of energy in holmium laser
lithotripsy. The safety and efficiency of laser lithotripsy may be
improved by preliminary estimation of the total energy required for
holmium laser lithotripsy.
The present study indicated that the TEL required
for renal calculi exhibited a significant difference from that
required for ureteral calculi, with renal calculi requiring a
higher TEL. A previous retrospective study by Molina et al
(17) indicated that renal calculi
required more energy than ureteral calculi, which was in agreement
with the results of the present study. Although the exact reasons
for this remain elusive, one conceivable explanation is that
hydronephrosis or calyceal hydrocalycosis make the renal calculus
more mobilized, while the location of ureteral calculus is
relatively fixed. In the fragmentation of a renal calculus, the
impact produced by holmium laser may cause the movement of the
calculus, and the total impact of the laser fiber on the calculus
is reduced. As a result, more pulses may be fired inefficiently and
more energy is wasted.
In the present study, a strong correlation between
the calculus volume and the TEL was identified, with larger calculi
requiring more energy for fragmentation. In their retrospective
study, Molina et al (17)
determined a significant correlation between the calculus volume
and the cumulative holmium laser energy. Blomley et al
(19) performed a systematic review
of holmium laser lithotripsy and reported that the required
cumulative energy of lithotripsy was increased with the increase of
the calculus size and mass. These results are consistent with the
conclusion of the present study and it was possible to evaluate the
TEL preferably by the volume of the calculus, which may serve as an
important index of the evaluation of the TEL.
The present study also determined a correlation
between the CT value of the calculus and the TEL, with a calculus
with a higher CT value requiring a higher TEL. Zhang et al
(20) reported that the CT value was
able to effectively predict the fragility of urinary calculi and
the shocking times of extracorporeal shock wave lithotripsy. Gupta
et al (21) came to the same
conclusion that the fragility of a calculus may be estimated based
on the CT value, with a lower CT value of the calculus being
associated with an easier fragmentation. Wang et al
(22) indicated that the CT value
may be used to quantitatively analyze the hardness of urinary
calculi. The study suggested a positive correlation between the CT
value of urinary calculi and their hardness, with a higher CT value
indicating a harder calculus and a more difficult fragmentation.
The CT value is the X-ray absorption coefficient of a certain
tissue/material, and it is an index providing information on the
density, with a higher CT value indicating a larger density.
However, the hardness and fragility of a urinary calculus mainly
depends on its chemical composition and inner structure, which has
a high correlation with the CT value and may be used to estimate
the major composition of calculus. Furthermore, the hardness and
fragility of the calculus have a key role in the efficacy of
lithotripsy, which exhibits marked differences for calculi with
different hardness and fragility. Overall, calculi with different
CT values have a different hardness and fragility, and the TEL is
different.
In conclusion, the present study indicated a
correlation between the parameters of NCHCT and the TEL. A
mathematical model correlating the parameters of NCHCT with the TEL
was established, which may provide a foundation to guide the use of
energy in holmium laser lithotripsy. By providing a preliminary
evaluation of the total energy required in holmium laser
lithotripsy, NCHCT may be used to predict the difficulty of
lithotripsy, and improve the safety and efficiency of
lithotripsy.
Acknowledgements
The authors are particularly grateful to Professor
Yuanyuan Liu (Statistics Department, Shandong Provincial Third
Hospital, Jinan, China) for her assistance with the statistical
analysis. The authors also thank Professor Mingjie Li and Professor
Xiangtao Wang (both Department of Urinary Surgery, Shandong
Provincial Third Hospital) for their technical advice on the
holmium laser lithotripsy.
Funding
The present study was funded by the Science
Foundation of Qilu Hospital of Shandong University and the
Fundamental Research Funds of Shandong University (grant no.
2015QLMS39).
Availability of data and materials
The analyzed data sets generated during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JM, ZY, XZ, CW, XL, ZL, LYu and LYi were responsible
for data collection. LC, JM, ZY, XJ and WH were responsible for
statistical analysis. LC, JM, ZY, XZ and WH wrote the article. The
final version of the manuscript has been read and approved by all
authors, and each author believes that the manuscript represents
honest work.
Ethical approval and consent to
participate
The present study was approved by the institutional
review board of Shandong Provincial Third Hospital (Jinan, China).
All patients provided written informed consent prior to
enrollment.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Brikowski TH, Lotan Y and Pearle MS:
Climate-related increase in the prevalence of urolithiasis in the
United States. Proc Natl Acad Sci USA. 105:9841–9846. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kang HW, Lee SK, Kim WT, Kim YJ, Yun SJ,
Lee SC and Kim WJ: Natural history of asymptomatic renal stones and
prediction of stone related events. J Urol. 189:1740–1746. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schwarzenbach HR and Jenzer S: Diagnosis
and management of suspected nephrolithiasis in a primary care
setting. Praxis (Bern 1994). 101:1187–1192. 2012.(In German).
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang SJ, Mu XN, Zhang LY, Liu QY and Jin
XB: The incidence and clinical features of acute kidney injury
secondary to ureteral calculi. Urol Res. 40:345–348. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bagley D and Erhard M: Use of the holmium
laser in the upper urinary tract. Tech Urol. 1:25–30.
1995.PubMed/NCBI
|
|
6
|
Lam JS, Greene TD and Gupta M: Treatment
of proximal ureteral calculi: Holmium: YAG laser ureterolithotripsy
versus extracorporeal shock wave lithotripsy. J Urol.
167:1972–1976. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bader MJ, Pongratz T, Khoder W, Stief CG,
Herrmann T, Nagele U and Sroka R: Impact of pulse duration on
Ho:YAG laser lithotripsy: Fragmentation and dusting performance.
World J Urol. 33:471–477. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Johnson DE, Cromeens DM and Price RE: Use
of the holmium: YAG laser in urology. Lasers Surg Med. 12:353–363.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kronenberg P and Traxer O: Update on
lasers in urology 2014: Current assessment on holmium:
Yttrium-aluminum-garnet (Ho:YAG) laser lithotripter settings and
laser fibers. World J Urol. 33:463–469. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Larizgoitia I and Pons JM: A systematic
review of the clinical efficacy and effectiveness of the holmium:
YAG laser in urology. BJU Int. 84:1–9. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Takazawa R, Kitayama S and Tsujii T:
Successful outcome of flexible ureteroscopy with holmium laser
lithotripsy for renal stones 2 cm or greater. Int J Urol.
19:264–267. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dushinski JW and Lingeman JE: High-speed
photographic evaluation of holmium laser. J Endourol. 12:177–181.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chan KF, Vassar GJ, Pfefer TJ, Teichman
JM, Glickman RD, Weintraub ST and Welch AJ: Holmium: YAG laser
lithotripsy: A dominant photothermal ablative mechanism with
chemical decomposition of urinary calculi. Lasers Surg Med.
25:22–37. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sea J, Jonat LM, Chew BH, Qiu J, Wang B,
Hoopman J, Milner T and Teichman JM: Optimal power settings for
Holmium: YAG lithotripsy. J Urol. 187:914–919. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chawla SN, Chang MF, Chang A, Lenoir J and
Bagley DH: Effectiveness of high-frequency holmium: YAG laser stone
fragmentation: The ‘popcorn effect’. J Endourol. 22:645–650. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kronenberg P and Traxer O: In vitro
fragmentation efficiency of holmium: Yttrium-aluminum-garnet (YAG)
laser lithotripsy-a comprehensive study encompassing different
frequencies, pulse energies, total power levels and laser fibre
diameters. BJU Int. 114:261–267. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Molina WR, Marchini GS, Pompeo A, Sehrt D,
Kim FJ and Monga M: Determinants of Holmium:
Yttrium-aluminum-garnet laser time and energy during ureteroscopic
laser lithotripsy. Urology. 83:738–744. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mi J, Li J, Zhang Q, Wang X, Liu H, Cao Y,
Liu X, Sun X, Shang M and Liu Q: Combining ultrasonography and
noncontrast helical computerized tomography to evaluate Holmium
laser lithotripsy. Medicine (Baltimore). 95:e55642016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Blomley MJ, Nicholson DA, Bartal G, Foster
C, Bradley A, Myers M, Man W, Li S and Banks LM: Holmium-YAG laser
for gall stone fragmentation: An endoscopic tool. Gut. 36:442–445.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang P, Tu B, Zhang J, Zhang He, Jiang H
and Jiang N: Forecasting accuracy of dual-energy CT on kidney
calculus components. 33:636–638. 2011.(In Chinese).
|
|
21
|
Gupta NP, Ansari MS, Kesarvani P, Kapoor A
and Mukhopadhyay S: Role of computed tomography with no contrast
medium enhancement in predicting the outcome of extracorporeal
shock wave lithotripsy for urinary calculi. BJU Int. 95:1285–1288.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang Jinfeng ZW, Xinguo Liu and Xiaogang
Li: Prediction of hardness of urinary calculi by computed
tomography in vitro. J Mod Urol. 03:191–193. 2008.(In Chinese).
|