Introduction
Endogenous neural stem cells (eNSCs) have been
proposed as a potential replacement of cells lost due to central
nervous system injury, and the source of tropic molecules to
minimize damage and promote recovery (1). eNSCs respond actively to injury
stimuli, including brain ischemia-reperfusion (2–4).
Furthermore, eNSCs may proliferate to maintain their self-renewal
and differentiate into neurons, astrocytes and oligodendrocytes,
and migrate from the subventricular zone (SVZ) or dentate gyrus
(DG) to injured regions in order to repair damage (4). The rate of proliferation and
differentiation will affect the number of eNSCs and neurons in
different brain regions (5). The
sufficient activation of neurogenesis and improving the
neurological outcomes in a pathological condition are important for
disease treatment.
Electroacupuncture (EA) has been used in East Asian
countries, particularly in China, for >2,000 years to treat
various diseases, including stroke (6). Recently, EA has been demonstrated as a
good alternative therapeutic strategy compared with medicinal
treatment, acupuncture, massage and physiotherapy to improve the
symptoms of stroke (7) and has been
indicated to effectively exert neuroprotective effects on patients
who have experienced a stroke (8). A
recent meta-analysis of a random, controlled study concluded that
EA effectively improved ischemic stroke in patients (9). Additionally, animal studies have
demonstrated that EA stimulation protects the brain against
ischemia-reperfusion injury (10,11).
Mechanistically, the neuroprotective role of EA is associated with
anti-inflammatory, anti-oxidative and anti-apoptotic signaling
pathways (11,12). Our previous study also identified
that EA attenuated neuronal injury in the cervical spinal cord
following brain ischemia in stroke-prone renovascular hypertensive
rats (13).
EA has been indicated to stimulate neurogenesis,
which may be another potential mechanism that elucidates why EA
improves the neurological outcome in clinical and preclinical
settings (14). EA has been reported
to promote neurogenesis in the striatum (15), SVZ (16) and hippocampal DG (17) in animals with stroke. In-depth
understanding of the time course of the neurogenesis effect induced
by EA and the relevant mechanisms would benefit stroke therapy in
the future.
NogoA is an inhibitor of neurite growth and is
regulated by two inhibitory domains: 66-amino acid region (Nogo-66)
and the N-terminal region (amino-Nogo) (18–20).
Nogo-66 inhibits activity-dependent axonal growth by binding to the
Nogo-66 receptor-1 (NgR1) (20). In
turn, the NgR1 competitive antagonist, NEP1-40 (Nogo-66, residues
1–40), and antibodies against NogoA block this inhibitory effect
and improve the neurological outcomes following ischemic stroke in
adult rats (21,22). Lysophosphatidic acid (LPA) is a
bioactive lipid (23). LPA and NogoA
are associated with the RhoA-Rho kinase signaling pathway, and
regulate neurogenesis, although they act on different receptors
(24). Plasticity-related gene 5
(PRG5) is regulated during brain and spinal cord development and is
exclusively allocated within the nervous system (24,25).
PRG5 has been demonstrated to impede LPA/RhoA and NogoA/RhoA
signaling (24,25), which suggests that PRG5 may function
as a positive regulator of neurogenesis. Previously, we have
indicated that EA stimulation reduced brain ischemia-induced
elevation of NogoA and NgR1 protein expression levels at day 14 and
28 in renal vascular hypertensive rats (13). However, whether PRG5/RhoA signaling
is involved in the neurogenesis of EA is unknown.
The present study aimed to investigate the
proliferation and differentiation of eNSCs following EA stimulation
in brain ischemia-reperfused rats in the chronic phase. The dynamic
expression levels of neurogenesis factors, including PRG5, RhoA,
NogoA, LPA, were also detected to explore the potential mechanisms
involved.
Materials and methods
Ethics statement
All animal treatments were conducted in strict
accordance with international ethical guidelines and the National
Institutes of Health Guide for the Care and Use of Laboratory
Animals (eighth edition, 2011). Experiments were performed with the
approval of the Institutional Animal Care and Use Committee of
Guangzhou University of Chinese Medicine (Foshan, China).
Animals
A total of 170 male Sprague-Dawley rats (weighing
200±20 g; 12 weeks old), were used. Rats were acclimatized for 3
days prior to experimentation to laboratory conditions
(temperature, 25±1°C; humidity, 65±5%) and were maintained on a
12-h light/dark cycle and had free access to food and water. All
efforts were made to minimize the number of animals used and their
suffering. Animals were randomly divided into the sham, MCAO and
MCAO + EA groups.
Focal cerebral ischemia-reperfusion
and EA treatment
Middle cerebral artery occlusion (MCAO) was employed
to induce focal ischemia-reperfusion, as we previously described
(26). The rats were anesthetized
with 5% isoflurane (Pharma Handelsgesellschaft mbH, Burgdorf,
Germany) and maintained with 2% isoflurane in an oxygen/air mixture
in a stereotaxic frame (flow rate, 2 l/min; Stoelting Co., Wood
Dale, IL, USA). Once ischemia had been established for 2 h, the
monofilament nylon suture was removed to induce reperfusion on days
1, 7 and 14. Rats in the sham-operated group underwent the same
procedures without occluding the MCA. Notably 8 rats succumbed to
surgical complications and were therefore not included in further
analysis.
Rats were randomly divided into three groups (n=54
per group): The sham group, where rats received sham stimulation
(no EA current stimulation following MCAO); the MCAO group, where
rats received MCAO and sham stimulation; and the MCAO + EA group,
where rats received MCAO and EA stimulation. Each group was further
divided into three subgroups with reperfusion times of 1, 7 and 14
days following MCAO (n=18 in each group). EA treatment was
conducted at the GV20 (Baihui) and GV14 (Dazhui) acupoints for 30
min daily following the induction of MCAO. During EA
administration, rats were maintained within a cloth bag. Sterilized
disposable stainless-steel needles (0.2×25.0 mm, Huan Qiu Brand,
manufactured by Suzhou Medicine Co., Ltd., Suzhou, China) were
inserted obliquely as deep as 5 mm at GV20 (Baihui) and GV14
(Dazhui) points. Electric stimulation was generated using an
electrical stimulator (Hua Tuo SDZ-II; Hua Tuo Medical Instruments
Co., Ltd., Suzhou, China). EA treatment commenced 30 min per
session. The stimulation parameter exhibited disperse-dense waves
of a frequency of 5/20 Hz (28.5/15 msec pulse duration) and a
current density of 2–4 mA.
Evaluation of neurological
deficits
Neurological deficits were scored 48 h following
reperfusion by another investigator who was blinded to the
experimental groups. The scoring criteria for neurological deficits
were as follows: 0 points, no neurological deficit; 1 point,
difficulty in fully extending the contralateral forelimb; 2 points,
unable to fully extend the contralateral forelimb; 3 points,
circling to the contralateral side; and 4 points, no consciousness
or ambulation. The higher score of the neurological deficits
represented greater impairment of motor function.
Bromodeoxyuridine (BrdU) labeling
The S-phase marker BrdU (50 mg/kg; Sigma-Aldrich;
Merck KGaA, Darmstadt, Germany) (27) was intraperitoneally injected twice a
day for 5 consecutive days prior to ischemic injury. Sham surgery
rats received the same dose of BrdU, which was also administered 5
days before rats were subjected to the same procedures without
occluding the MCA. Following injection, BrdU was incorporated into
the DNA of dividing cells and its signaling was observed using a
fluorescence microscope (Olympus Corporation, Tokyo, Japan).
Immunofluorescence
Brains were removed and hippocampal tissues were
fixed with formaldehyde-sucrose for 24 h at 20°C. Brains were
removed and fixed with 4% paraformaldehyde at 4°C for 24 h.
Following dehydration in graded ethanol and xylene, brain slices
were embedded in paraffin, sectioned to 4 µm in thickness, dewaxed,
rehydrated and stained with 1% toluidine blue at 37°C for 30 min.
Following rinsing, sections were dehydrated in increasing
concentrations of ethanol, cleared in xylene and mounted with
Permount cover slips. Brain sections were washed twice with PBS,
fixed with 4% paraformaldehyde at 4°C for 1 h, permeated with 0.1%
Triton for 30 min at 4°C. Samples were then blocked with 10% normal
goat serum (Vector Laboratories, Inc, Burlingame, CA, USA) for 30
min at 4°C and incubated with primary antibodies overnight at 4°C.
The primary antibodies used included mouse anti-BrdU (1:100; EMD
Millipore, Billerica, MA, USA), mouse anti-Nestin (1:200; EMD
Millipore), mouse anti-Doublecortin (DCX; 1:200; Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA). The sections were then
incubated with fluorescein-conjugated secondary antibodies
rhodamine-conjugated donkey anti-mouse IgG antibody (1:200; Jackson
ImmunoResearch Laboratories Inc., West Grove, PA, USA) were used as
secondary antibodies. The secondary antibody solution (1:200) was
then added and incubated at room temperature for 1 h. Immunostained
sections were visualized using an inverted Olympus fluorescence
microscope (LSM 700; Carl Zeiss GmbH; Jena, Germany). For
immunohistochemistry, the number of positive cells or staining
intensity in the peri-ischemic zone was measured at magnification
of ×200. In all of the slices, five fields per sample and three to
six tissues were quantified in each group. Expression was analyzed
using Image-Pro Plus 6.0 software (Media Cybernetics Inc,
Buckinghamshire, UK). For BrdU, DCX and Nestin, positive cells were
counted in five different fields per rat by an observer blind to
the present study, and the quantitation was performed by
calculating the positive cells per mm2.
Nissl staining
Isolated brain sections from rats in each group were
dehydrated, dipped into paraffin and, using a microtome, sliced
into 5 µm tissue sections. Samples were sectioned into three
slices, starting 3 mm from the anterior tip of the frontal lobe in
the coronal plane. The second set of slices were embedded in
paraffin, cut into 5-µm sections, and then mounted on
poly-L-lysine-coated slides.
Nissl staining was performed using 1% cresyl violet
solution (Sigma-Aldrich; Merck KGaA) for 6 min at room temperature.
Primary antibodies were applied using a biotinylated anti-rabbit or
anti-mouse secondary antibody (ab23673; 1:200; Jackson
ImmunoResearch Laboratories, Inc.) at room temperature for 1 h at
room temperature for 1 h and detected using a
streptavidin-peroxidase kit (Maixin, Fuzhou, China) at room
temperature for 1 h, according to the manufacturer's instructions.
Images were captured using a fluorescence microscope. The soma
diameter (in µm) of pyramidal neurons was assessed from the images
of the CA1 pyramidal layer using a magnification of ×40. To
quantify the soma diameters, a pyramidal-like neuron was randomly
selected from the pyramidal layer of the CA1 region from each
slice. Normal cells were identified by the presence of Nissl
substance in the cytoplasm, loose chromatin and prominent nucleoli.
Damaged neurons were identified by the loss of Nissl substance,
cavitation around the nucleus and by the presence of pyknotic
homogenous nuclei. Neural-like cells were evaluated based on
presence of Nissl bodies. All experiments were repeated five times
to obtain the percentage of healthy cells. For images obtained
using a 40×10 optical microscope, Image-Pro Plus 6.0 software
(Media Cybernetics Inc., Buckinghamshire, UK). was for
analysis.
Western blot analysis
The rat ischemic cerebral hemispheres and bilateral
hippocampi were isolated at day 1, 7 and 14. Protein concentration
was determined using a BCA Protein Assay Reagent kit (Pierce;
Thermo Fisher Scientific, Inc., Waltham, MA, USA), according to the
manufacturer's instructions. Once proteins were denatured in
SDS-loading buffer, protein samples (50 µg/lane) were separated
using 8–12% SDS-PAGE and then transferred to polyvinylidene
fluoride membranes (EMD Millipore). Following blocking in PBS
blocking solution containing 3% non-fat powdered milk and 0.3%
Tween for 1 h at room temperature, the membranes were incubated
overnight at 4°C with primary antibodies, including mouse NogoA
monoclonal antibody (AB5888; 1:1,000; EMD Millipore), mouse PRG5
monoclonal antibody (AB15172; 1:500; EMD Millipore), mouse RhoA
monoclonal antibody (sc-28565; 1:1,000; Santa Cruz Biotechnology,
Inc.) and anti-β-actin (TA-09; 1:500; ZSGB-Bio, Inc., Beijing,
China), followed by incubation with anti-rabbit or anti-mouse
horseradish peroxidase-conjugated secondary antibodies (sc-2371;
1:5,000; Santa Cruz Biotechnology, Inc.) for 1 h at room
temperature. Bands were detected by chemiluminescence technology
using an ECL advanced western blot analysis detection reagent (EMD
Millipore), quantified and normalized to β-actin using ImageJ
software (version 1.38, National Institutes of Health, Bethesda,
MD, USA).
Statistical analysis
Statistical analysis was performed using SPSS for
Windows (v. 14.0; SPSS, Inc., Chicago, IL, USA). Experiments were
repeated in triplicate independently. Data were expressed as the
mean ± standard error of the mean. One-way analysis of variance
with Dunnett's post hoc testing was used to compare multiple
western blot and BrdU labeling datasets. P<0.05 was considered
to indicate a statistically significant difference.
Results
EA stimulation improves neurological
outcome and reduces hippocampal CA1 neuronal injury in rats
subjected to brain ischemia-reperfusion
The neuroprotective role of EA stimulation was
evaluated. The modified neurological score was used to evaluate
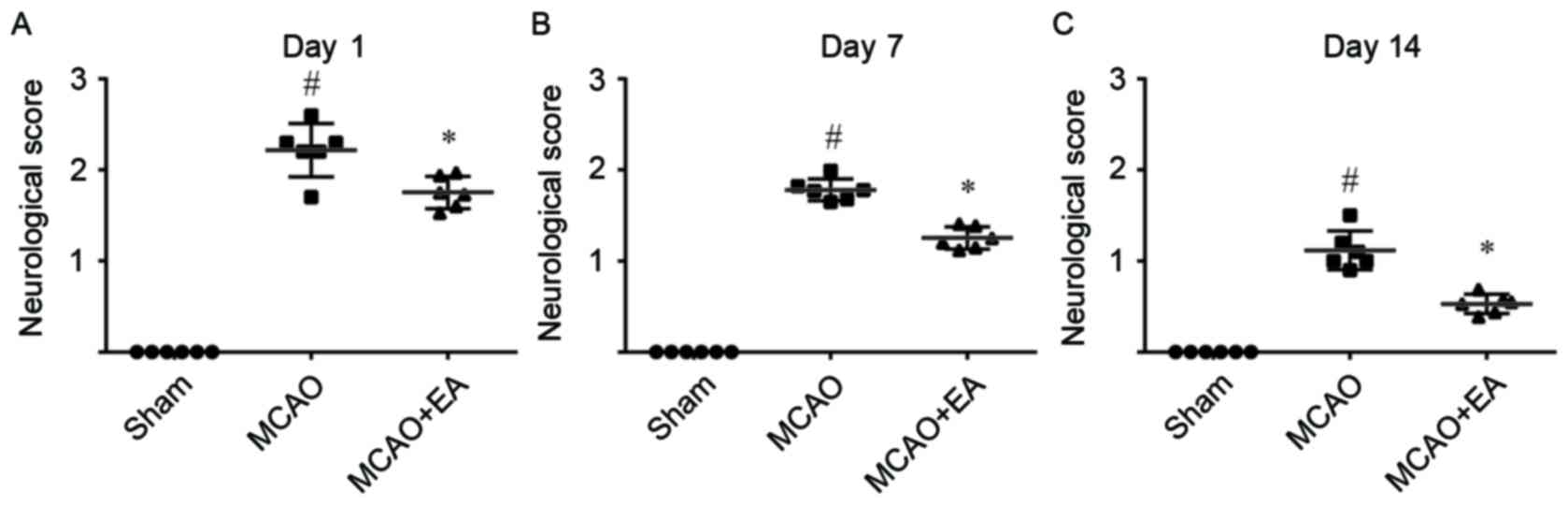
neurologic deficits of rats at 1, 7 and 14 days after MCAO. As
indicated in Fig. 1, the neurology
score of rats in the MCAO group was significantly increased
compared with that in the sham group on day 1, 7 and 14
(P<0.05). However, EA treatment at the GV20 (Baihui) and GV14
(Dazhui) acupoints significantly reduced the neurology score
(P<0.05) at 1, 7 and 14 days after the induction of ischemia
compared with the MCAO group, indicating EA treatment ameliorated
brain injury and improved the neurological outcome.
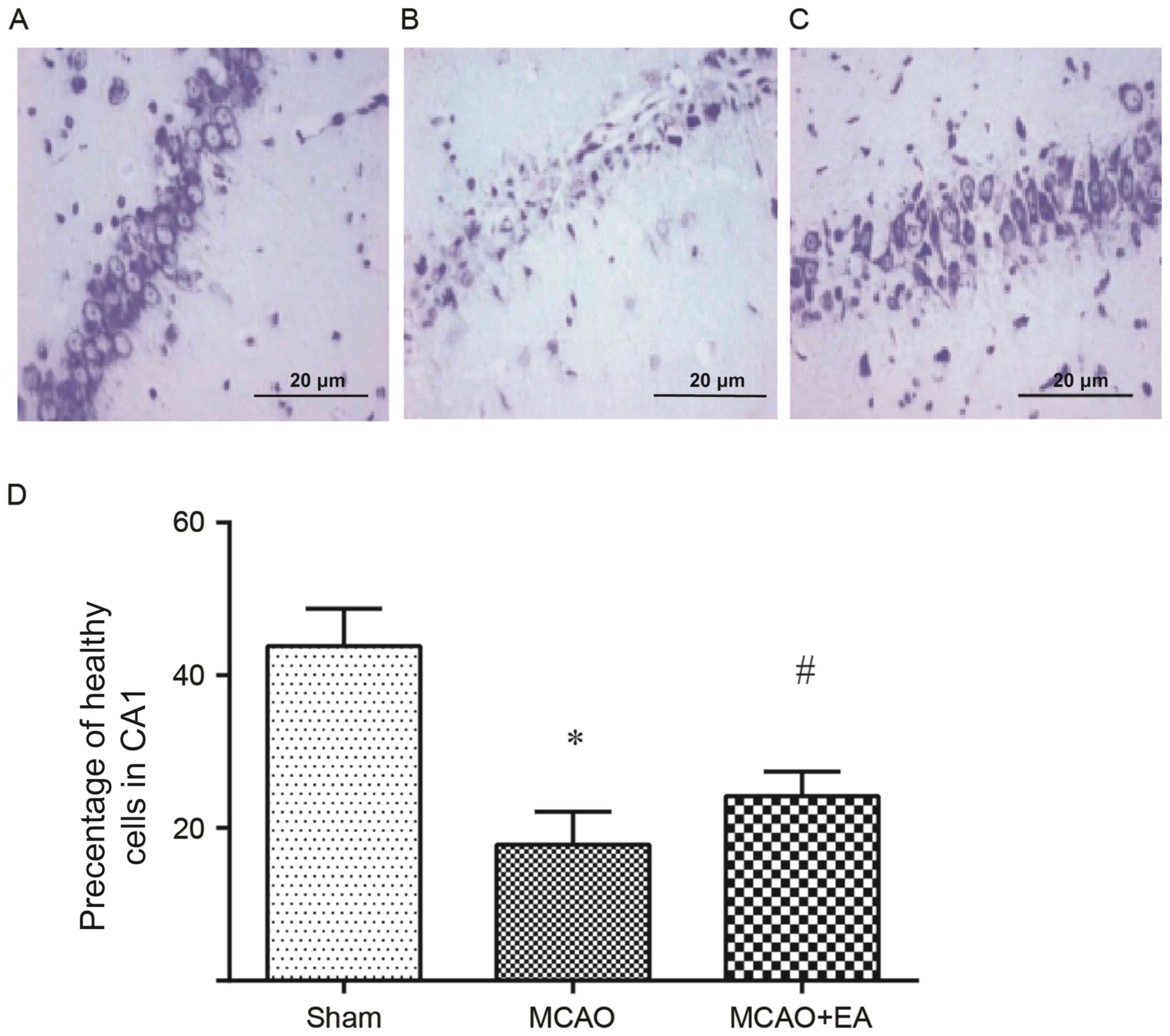
Nissl staining assay was conducted to observe the
neuroprotective effects of EA. Notably, data in Fig. 2 revealed that cerebral
ischemia-reperfusion resulted in a significant hippocampal neuron
loss, which was significantly attenuated by EA stimulation (both
P<0.05). These data were in agreement with previous findings
that indicated EA stimulation has a positive neuroprotective effect
in a model of cerebral ischemia-reperfusion (28).
EA treatment promotes the
proliferation and differentiation of eNSCs in rats with MCAO
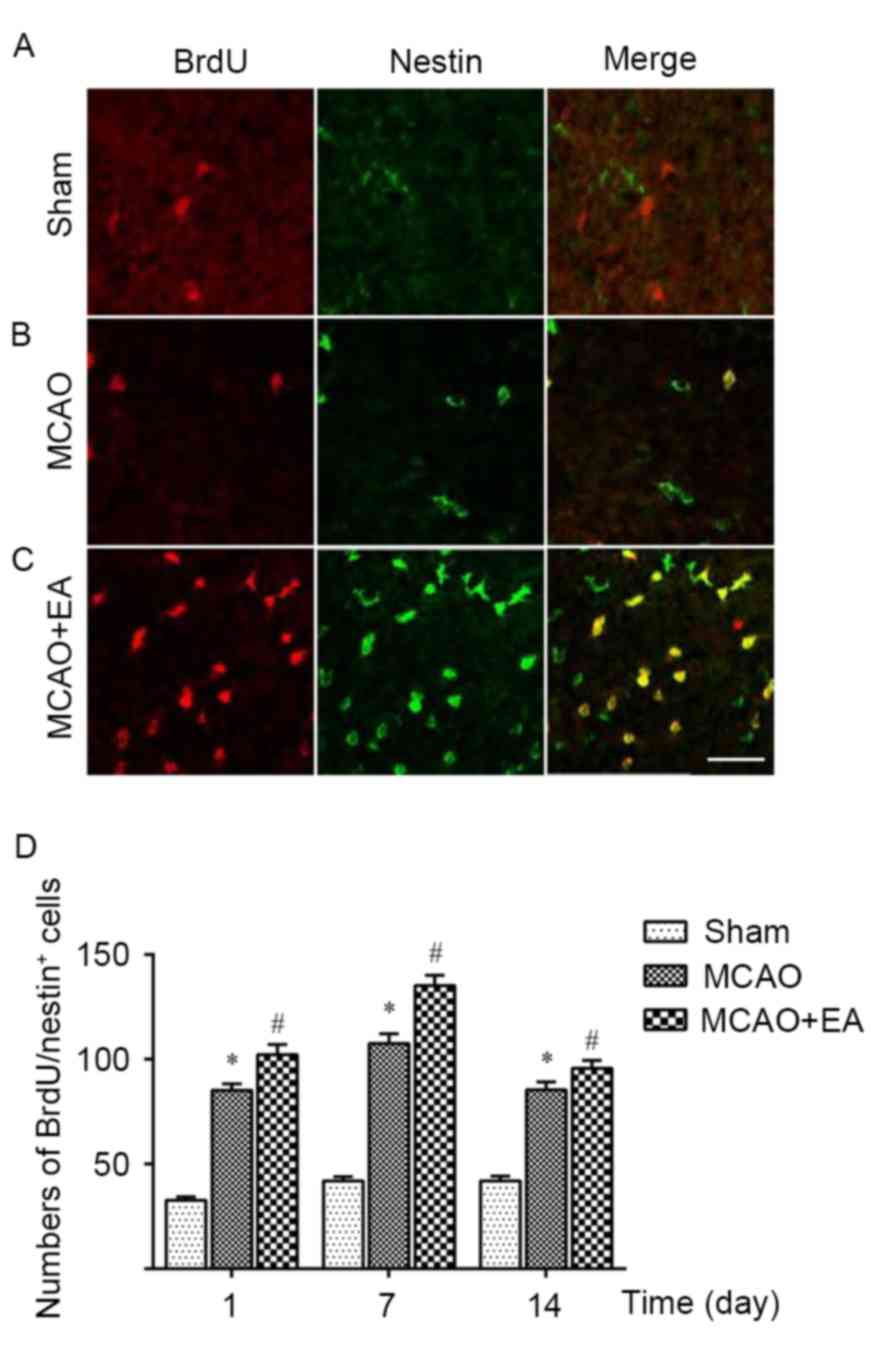
To investigate the neurogenesis effects of EA
treatment, BrdU incorporation and nestin staining assays were
conducted to detect the proliferation of eNSCs. BrdU is a marker of
newly formed cells but not undivided cells. Nestin is a specific
marker protein for neural stem cells (29). BrdU-positive cells (red) represented
the new dividing nerve cells, whereas nestin-positive
immunoreactive cells (green) indicated eNSCs. The double-stained
cells (yellow) in the merged Brdu/Nestin images revealed the newly
generated immature neurons and indicated eNSCs that were in the
proliferous period (Fig. 3A-C).
Results demonstrated cell cycle-specific nestin was increasingly
expressed in a time-dependent manner, until day 14, where
expression appeared to decrease (Fig.
3D). Following focal cerebral ischemia at day 1, the number of
nestin immunoreactive cells increased in the DG zone. Furthermore,
an increased expression of immunoreactive cells was indicated in
the infarct area of the DG zone at day 7 compared with day 1 in the
MCAO and the MCAO + EA group. In addition, the number of nestin
immunoreactive cells in the MCAO + EA group was significantly
higher than that in the MCAO group, especially on day 7. Double
staining of BrdU and nestin in the MCAO + EA group demonstrated
that a significantly increased number of the proliferous eNSCs were
expressed compared with those in the MCAO group on days 1, 7 and 14
(P<0.05), suggesting EA stimulation increased eNSC proliferation
in rats with MCAO.
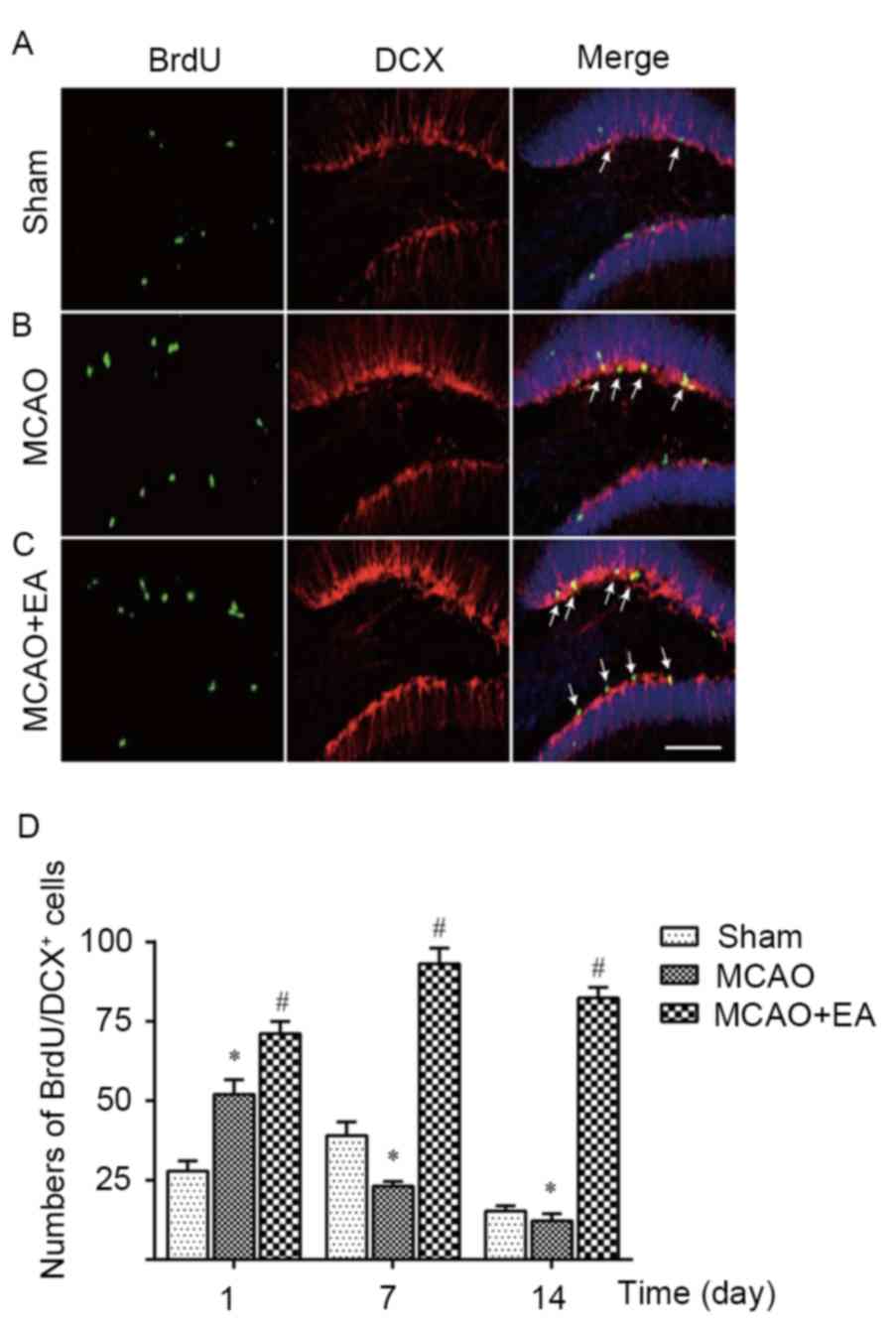
In response to injurious stimuli, eNSCs may
differentiate into neurons (30). To
investigate whether EA stimulates neurogenesis during brain
ischemia-reperfusion, brain sections were co-stained with BrdU and
antibody for DCX, a marker of newborn neurons (31), as indicated in Fig. 4A-C. The number of DCX immune-reactive
cells increased at day 1, peaked at day 7 and declined at day 14
(Fig. 4) in the DG zone of rat
hippocampus. The DCX immunoreactive cells were located in the
infarct DG zone. No DCX immunoreactive cells were detected in the
contralateral hemisphere of the brain in the sham group. Few DCX
immunoreactive cells were indicated in the MCAO group. Notably, the
number of DCX immunoreactive cells in the MCAO + EA group was
significantly higher than that in the MCAO group, particularly on
day 7 (P<0.05; Fig. 4D). These
results suggest that EA stimulation may effectively induce
neurogenesis during brain ischemia and reperfusion.
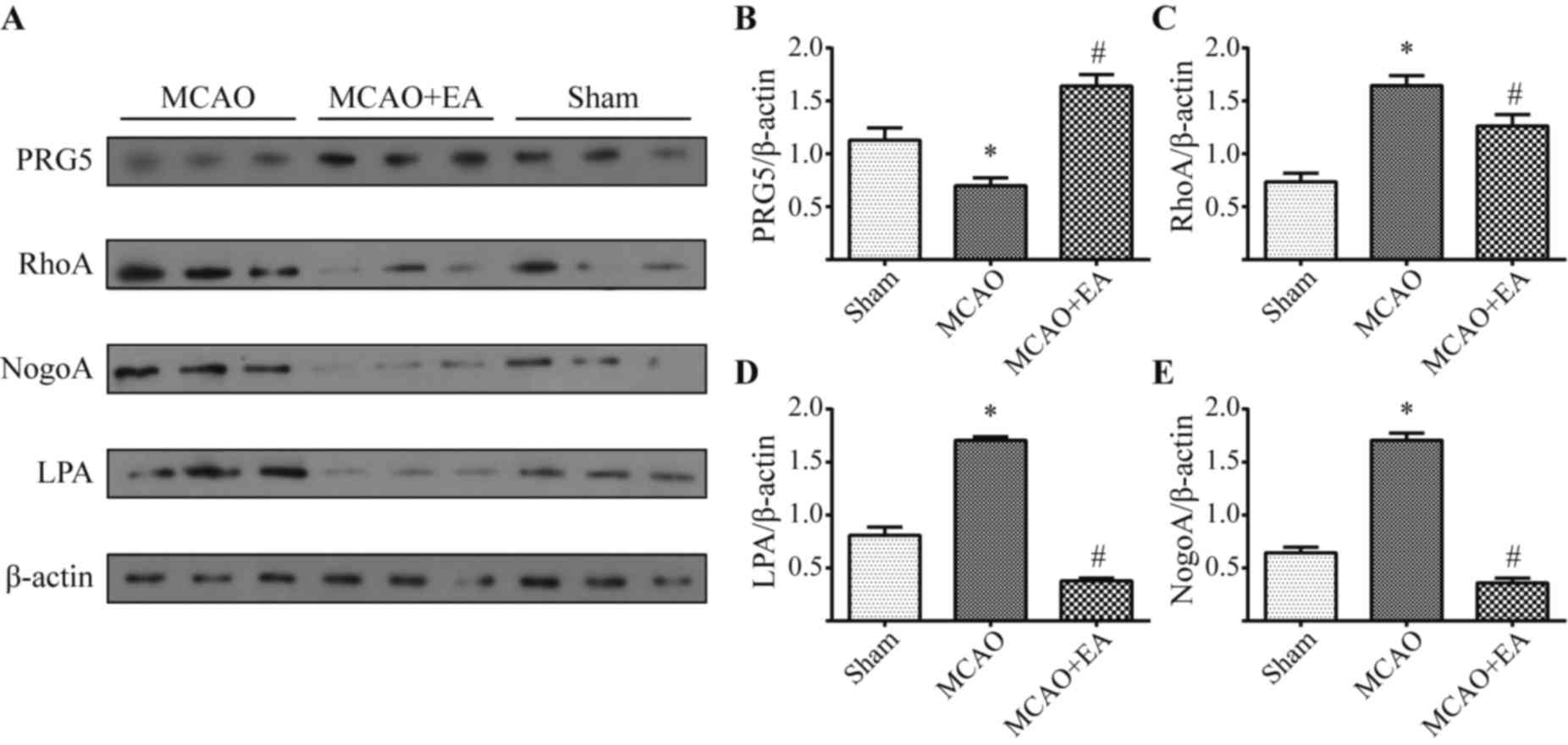
EA treatment increases PRG5 expression
and reduces NogoA-LPA/RhoA signaling in the brain of rats
To study the underlying mechanism of EA-mediated
neurogenesis in the established model of stroke,
neurogenesis-regulated signaling molecules were detected with
western blot analysis. Results in Fig.
5A revealed that MCAO lowered the protein expression levels of
PRG5 and increased RhoA, NogoA and LPA in comparison with the sham
group at day 7, and quantification of these results indicated the
differences were statistically significant (all P<0.05; Fig. 5B-E). However, compared with the MCAO
group, treatment with EA significantly elevated the protein
expression levels of PRG5, whose expression was even higher
compared with the sham group (P<0.05; Fig. 5B). Notably, EA treatment
significantly decreased RhoA, LPA and NogoA protein expression
levels, compared with the MCAO group (all P<0.05; Fig. 5C-E, respectively). These data suggest
that PRG5/NogoA-LPA/RhoA signaling likely mediates EA-induced
neurogenesis in rats with stroke.
Discussion
The present findings indicated that EA effectively
ameliorated neuronal injury and activated the proliferation and
differentiation of eNSCs, which may contribute to the repair of the
central nervous system and improvement of neurological function in
a rat model of brain ischemia-reperfusion.
EA has been used as a treatment for stroke for
>2,000 years in East Asia (32).
The neuroprotective and neurogenesis roles of EA in animals with
stroke have been previously reported in various studies (8,9,11–13). The
present study focused to elucidate EA-stimulated eNSC proliferation
and neuronal differentiation at day 1, 7 and 14 following
reperfusion in rats with MCAO. The time points of EA-induced
neurogenesis were in line with the length of EA treatment, which
suggests the neurogenesis-inducing role of EA contributes to brain
repair during the chronic phase of ischemic stroke.
BrdU was used to label newly-divided cells, which
were observed in the DG zone and ipsilateral hemisphere of the
ischemic brain; however, the proliferation sequence and absolute
BrdU-labeled cell counts were significantly different (33). The DG is a potential source of neural
precursors for neuronal injury (34). BrdU-labeled cells were identified in
the DG zone, which was thought to be the source of neural precursor
cells, and the ipsilateral cortex. Furthermore, the absolute number
of BrdU-labeled cells in the ipsilateral cortex was five times
higher compared with the peak cell count in the DG zone (data not
shown). The migration speed of DG cells in the rostral migration
stream has been indicated to be slow in reaching the lesion
(35). The proximity of stroke
injury to the lateral ventricle wall may determine whether there is
a stem/progenitor cell response from the DG. Alternatively, focal
(mild) cortical stroke may not induce neuroblast migration from the
DG because it does not directly injure the DG itself. The MCAO
model produces stroke by disruption of blood flow to cortical
surface arteries. Other MCA occlusion models are generated by
occlusion of the proximal MCA that disrupts blood flow to various
areas, including the lateral ventricle. Because the neurovascular
relationship is a key factor in maintaining the neurogenic niche at
the DG, DG may impact neurogenesis. In proximal MCA occlusion
models, newly born mature neurons in the striatum, radial
glial-like cells at the DG, and/or migration of neuroblasts to the
infarct zone have been reported (36), suggesting that the disruption of
blood flow in neurogenic niches may induce neurogenesis. This
suggests that the DG zone is the dominant neural stem cell niche
during cerebral ischemia. Furthermore, this finding suggests the
possibility of intraparenchymal progenitors in the neocortex, which
is consistent with a previous report (37).
RhoA inhibits survival and growth of newly developed
neurons, and functions as a negative regulator in neuronal survival
and maturation (38). RhoA (and
other small GTPase family proteins) is the convergent signal of
multiple upstream molecules, including PRG5, LPA and NogoA
(39). Furthermore, PRG5 is a
negative regulator, whereas LPA and NogoA are the positive
regulators of RhoA signaling (24).
In the present study, EA stimulation upregulated PRG5 and
downregulated the protein expression levels of RhoA, LPA and NogoA,
which suggests these neurogenesis molecules may participate in
EA-induced neurogenesis activity, although gene loss-of-function
studies are required to further elucidate their roles in the
future.
In conclusion, the present study indicated that EA
improves neuronal function, reduces neuronal loss and promotes the
proliferation and differentiation of eNSCs. Furthermore, the
present findings suggested that the neurogenesis activating role of
EA is likely through modulating PRG5/RhoA signaling. These
preclinical investigations may provide the experimental basis for
the treatment of ischemic stroke using EA.
Acknowledgements
Not applicable.
Funding
The study was supported by grants from the National
Natural Science of Foundation of China (grant nos. 81072947 and
81473470) and the Guangdong Natural Science Foundation (grant nos.
8152800007000001 and 2014A030311033).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
FT and YG conceived and designed the study. JW and
JXL and CW and ML performed the experiments. JW and CW and JXL
wrote the paper. FT and YG and JW reviewed and edited the
manuscript. All authors read and approved the manuscript.
Ethics approval and consent to
participate
The study was approved by the Institutional Animal
Care and Use Committee of Guangzhou University of Chinese Medicine
(Foshan, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kline AE, Leary JB, Radabaugh HL, Cheng JP
and Bondi CO: Combinationtherapies for neurobehavioral and
cognitive recovery after experimental traumatic brain injury: Is
more better? Prog Neurobiol. 142:45–67. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thored P, Arvidsson A, Cacci E, Ahlenius
H, Kallur T, Darsalia V, Ekdahl CT, Kokaia Z and Lindvall O:
Persistent production of neurons from adult brain stem cells during
recovery after stroke. Stem Cells. 24:739–747. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gregoire CA, Goldenstein BL, Floriddia EM,
Barnabe- Heider F and Fernandes KJ: Endogenous neural stem cell
responses to stroke and spinal cord injury. Glia. 63:1469–1482.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tobin MK, Bonds JA, Minshall RD,
Pelligrino DA, Testai FD and Lazarov O: Neurogenesis and
inflammation after ischemic stroke: What is known and where we go
from here. J Cereb Blood Flow Metab. 34:1573–1584. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu D, Wang Z, Zhan J, Zhang Q, Wang J,
Zhang Q, Xian X, Luan Q and Hao A: Hydrogen sulfide promotes
proliferation and neuronal differentiation of neural stem cells
andprotects hypoxia-induced decrease in hippocampal neurogenesis.
Pharmacol Biochem Behav. 116:55–63. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo F, Song W, Jiang T, Liu L, Wang F,
Zhong H, Yin H, Wang Q and Xiong L: Electroacupuncture pretreatment
inhibits NADPH oxidase-mediated oxidative stress in diabetic mice
with cerebral ischemia. Brain Res. 1573:84–91. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tao J, Zheng Y, Liu W, Yang S, Huang J,
Xue X, Shang G, Wang X, Lin R and Chen L: Electro-acupuncture at
LI11 and ST36 acupoints exerts neuroprotective effects via reactive
astrocyte proliferation after ischemia and reperfusion injury in
rats. Brain Res Bull. 120:14–24. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee S, Kim W, Park J, Jang HH, Lee SM, Woo
JS, Kim HS, Lee KH, Kwon YJ, Lee U, et al: Effects of
electroacupuncture on endothelial function and circulating
endothelial progenitor cells in patients with cerebral infarction.
Clin Exp Pharmacol Physiol. 42:822–827. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu AJ, Li JH, Li HQ, Fu DL, Lu L, Bian ZX
and Zheng GQ: Electroacupuncture for acute ischemic stroke: A
meta-analysis of randomized controlled trials. Am J Chin Med.
43:1541–1566. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang J, Ye X, You Y, Liu W, Gao Y, Yang
S, Peng J, Hong Z, Tao J and Chen L: Electroacupuncture promotes
neural cell proliferation in vivo through activation of the ERK1/2
signaling pathway. Int J Mol Med. 33:1547–1553. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shen MH, Zhang CB, Zhang JH and Li PF:
Electroacupuncture attenuatescerebral ischemia and reperfusion
injury in middle cerebral artery occlusion of rat via modulation of
apoptosis, inflammation, oxidative stress, and excitotoxicity. Evid
Based Complement Alternat Med. 2016:94386502016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang Q, Li X, Chen Y, Wang F, Yang Q, Chen
S, Min Y, Li X and Xiong L: Activation of epsilon protein kinase
C-mediated anti-apoptosis is involved in rapid tolerance induced by
electroacupuncturepretreatment through cannabinoid receptor type 1.
Stroke. 42:389–396. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tan F, Chen J, Liang Y, Gu M, Li Y, Wang X
and Meng D: Electroacupuncture attenuates cervical spinal cord
injury following cerebral ischemia/reperfusion in stroke-prone
renovascular hypertensive rats. Exp Ther Med. 7:1529–1534. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Naylor M, Bowen KK, Sailor KA, Dempsey RJ
and Vemuganti R: Preconditioning-induced ischemic tolerance
stimulates growth factor expression and neurogenesis in adult rat
hippocampus. Neurochem Int. 47:565–572. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yang ZJ, Shen DH, Guo X and Sun FY:
Electroacupuncture enhances striatal neurogenesis in adult rat
brains after a transient cerebral middle artery occlusion. Acupunct
Electrother Res. 30:185–199. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim YR, Kim HN, Ahn SM, Choi YH, Shin HK
and Choi BT: Electroacupuncture promotes post-stroke functional
recovery via enhancing endogenous neurogenesis in mouse focal
cerebral ischemia. PLoS One. 9:e900002014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhao J, Sui M, Lu X, Jin D, Zhuang Z and
Yan T: Electroacupuncture promotes neural stem cell proliferation
and neurogenesis in the dentate gyrus of rats following stroke via
upregulation of Notch1 expression. Mol Med Rep. 12:6911–6917. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Petrinovic MM, Hourez R, Aloy EM, Dewarrat
G, Gall D, Weinmann O, Gaudias J, Bachmann LC, Schiffmann SN, Vogt
KE and Schwab ME: Neuronal Nogo-A negatively regulates dendritic
morphology and synaptic transmission in the cerebellum. Proc Natl
Acad Sci USA. 110:1083–1088. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang F, Xing S, He M, Hou Q, Chen S, Zou
X, Pei Z and Zeng J: Nogo-A is associated with secondary
degeneration of substantia nigra in hypertensive rats with focal
cortical infarction. Brain Res. 1469:153–163. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pernet V and Schwab ME: The role of Nogo-A
in axonal plasticity, regrowth and repair. Cell Tissue Res.
349:97–104. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhu WW, Ma XL, Guo AL, Zhao HY and Luo HH:
Neuroprotective effects of NEP1-40 and fasudil on Nogo-A expression
in neonatal rats with hypoxic-ischemic brain damage. Genet Mol Res.
10:2987–2995. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tsai SY, Papadopoulos CM, Schwab ME and
Kartje GL: Delayed anti-nogo-a therapy improves function after
chronic stroke in adult rats. Stroke. 42:186–190. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yung YC, Stoddard NC and Chun J: LPA
receptor signaling: Pharmacology, physiology, and pathophysiology.
J Lipid Res. 55:1192–1214. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Broggini T, Nitsch R and Savaskan NE:
Plasticity-related gene 5 (PRG5) induces filopodia and neurite
growth and impedes lysophosphatidic acid- and nogo-A-mediated
axonal retraction. Mol Biol Cell. 21:521–537. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Coiro P, Stoenica L, Strauss U and Brauer
AU: Plasticity-related gene 5 promotes spine formation in murine
hippocampal neurons. J Biol Chem. 289:24956–24970. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gu Y, Zheng G, Xu M, Li Y, Chen X, Zhu W,
Tong Y, Chung SK, Liu KJ and Shen J: Caveolin-1 regulates nitric
oxide-mediated matrix metalloproteinases activity and blood-brain
barrier permeability in focal cerebral ischemia and reperfusion
injury. J Neurochem. 120:147–156. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Attardi LD, de Vries A and Jacks T:
Activation of the p53-dependent G1 checkpoint response in mouse
embryo fibroblasts depends on the specific DNA damage inducer.
Oncogene. 23:973–980. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Feng X, Yang S, Liu J, Huang J, Peng J,
Lin J, Tao J and Chen L: Electroacupuncture ameliorates cognitive
impairment through inhibition of NF-κB-mediatedneuronal cell
apoptosis in cerebral ischemia-reperfusion injured rats. Mol Med
Rep. 7:1516–1522. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wakayama T, Nakata H, Kumchantuek T,
Gewaily MS and Iseki S: Identification of
5-bromo-2′-deoxyuridine-labeled cells during mouse spermatogenesis
by heat-induced antigen retrieval in lectin staining and
immunohistochemistry. J Histochem Cytochem. 63:190–205. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rueger MA, Muesken S, Walberer M, Jantzen
SU, Schnakenburg K, Backes H, Graf R, Neumaier B, Hoehn M, Fink GR
and Schroeter M: Effects of minocycline on endogenous neural stem
cells after experimental stroke. Neuroscience. 215:174–183. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Batailler M, Droguerre M, Baroncini M,
Fontaine C, Prevot V and Migaud M: DCX-expressing cells in the
vicinity of the hypothalamic neurogenic niche: A comparative study
between mouse, sheep, and human tissues. J Comp Neurol.
522:1966–1985. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ren L, Zhang WA, Fang NY and Wang JX: The
influence of electro-acupuncture on neural plasticity in acute
cerebral infarction. Neurol Res. 30:985–989. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kernie SG and Parent JM: Forebrain
neurogenesis after focal Ischemic andtraumatic brain injury.
Neurobiol Dis. 37:267–274. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kempermann G, Song H and Gage FH:
Neurogenesis in the adult hippocampus. Cold Spring Harb Perspect
Biol. 7:a0188122015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Luskin MB and Boone MS: Rate and pattern
of migration of lineally-related olfactory bulb interneurons
generated postnatally in the subventricular zone of the rat. Chem
Senses. 19:695–714. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Villasana LE, Kim KN, Westbrook GL and
Schnell E: Functional integration of adult-born hippocampal neurons
after traumatic brain injury(1,2,3). eNeuro. 2:ENEURO.0056-0015.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shimada IS, Peterson BM and Spees JL:
Isolation of locally derived stem/progenitor cells from the
peri-infarct area that do not migrate from the lateral ventricle
after cortical stroke. Stroke. 41:e552–e560. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Vadodaria KC and Jessberger S: Maturation
and integration of adult born hippocampal neurons: Signal
convergence onto small rho GTPases. Front Synaptic Neurosci.
5:42013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Savaskan NE, Brauer AU and Nitsch R:
Molecular cloning and expression regulation of PRG-3, a new member
of the plasticity-related gene family. Eur J Neurosci. 19:212–220.
2004. View Article : Google Scholar : PubMed/NCBI
|