|
1
|
Miyamoto Y, Zhang W and Lenz HJ: Molecular
landscape and treatment options for patients with metastatic
colorectal cancer. Indian J Surg Oncol. 8:580–590. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bogach J, Levine O, Parpia S, Valencia M,
Ruo L and Serrano P: Does the addition of biologic agents to
chemotherapy in patients with unresectable colorectal cancer
metastases result in a higher proportion of patients undergoing
resection? A systematic review and meta-analysis. J Gastrointest
Surg. 22:523–528. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kwakman JJM, Vink G, Vestjens JH,
Beerepoot LV, de Groot JW, Jansen RL, Opdam FL, Boot H, Creemers
GJ, van Rooijen JM, et al: Feasibility and effectiveness of
trifluridine/tipiracil in metastatic colorectal cancer: Real-life
data from The Netherlands. Int J ClinOncol. 23:482–489. 2018.
|
|
4
|
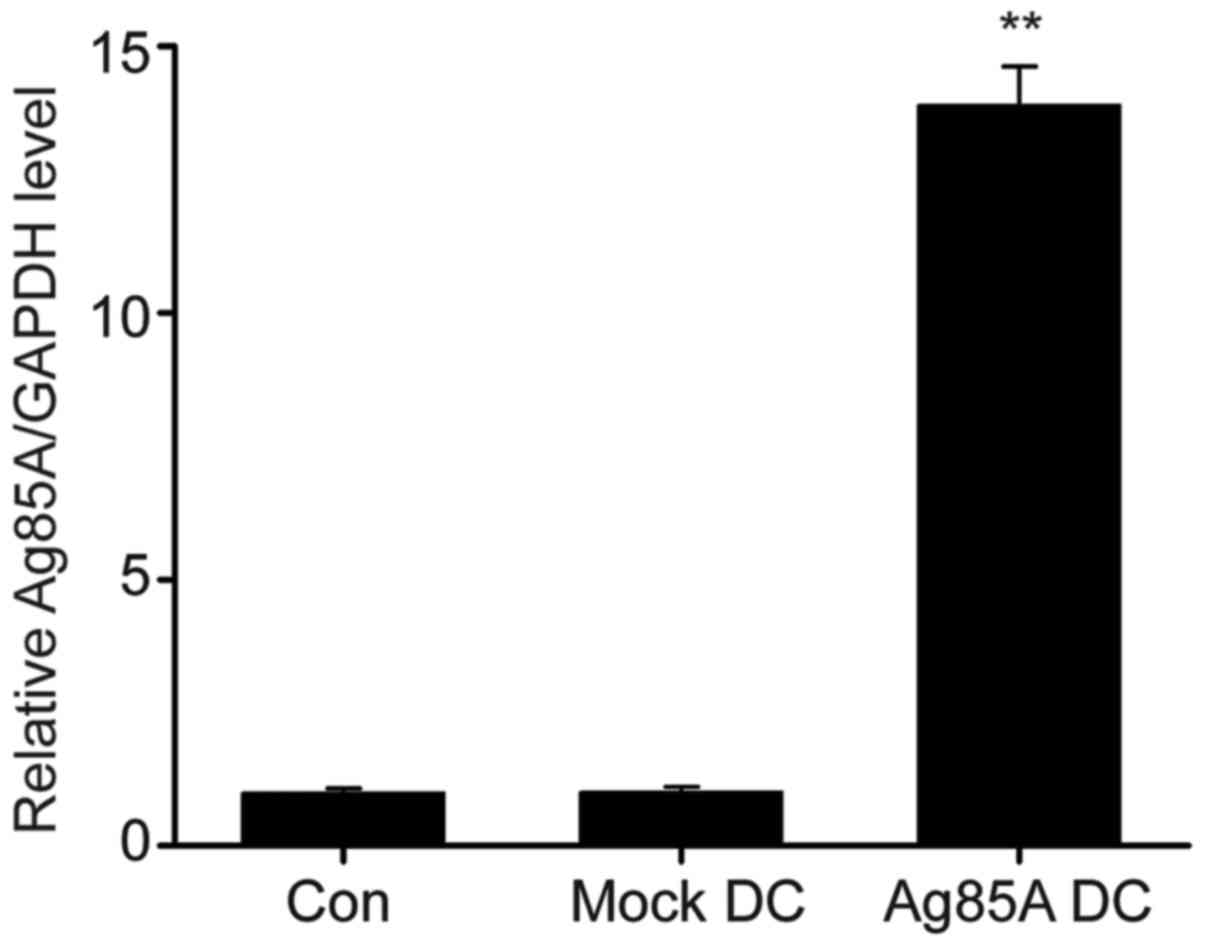
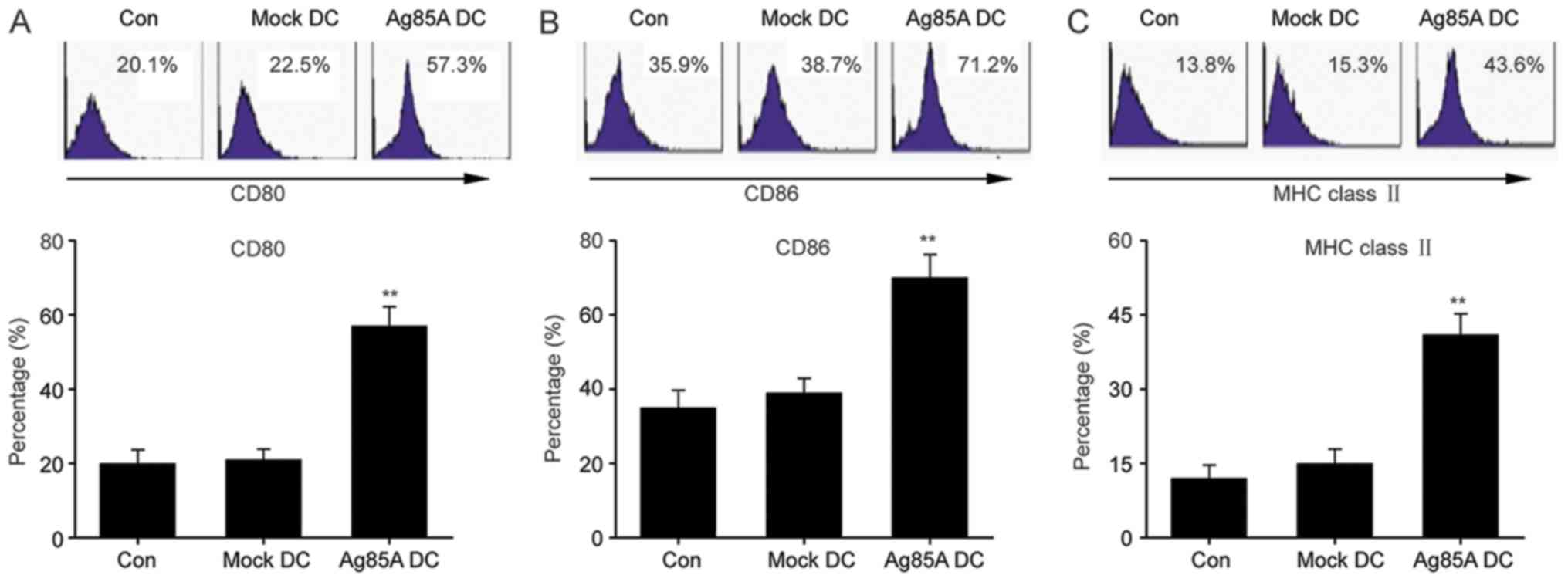
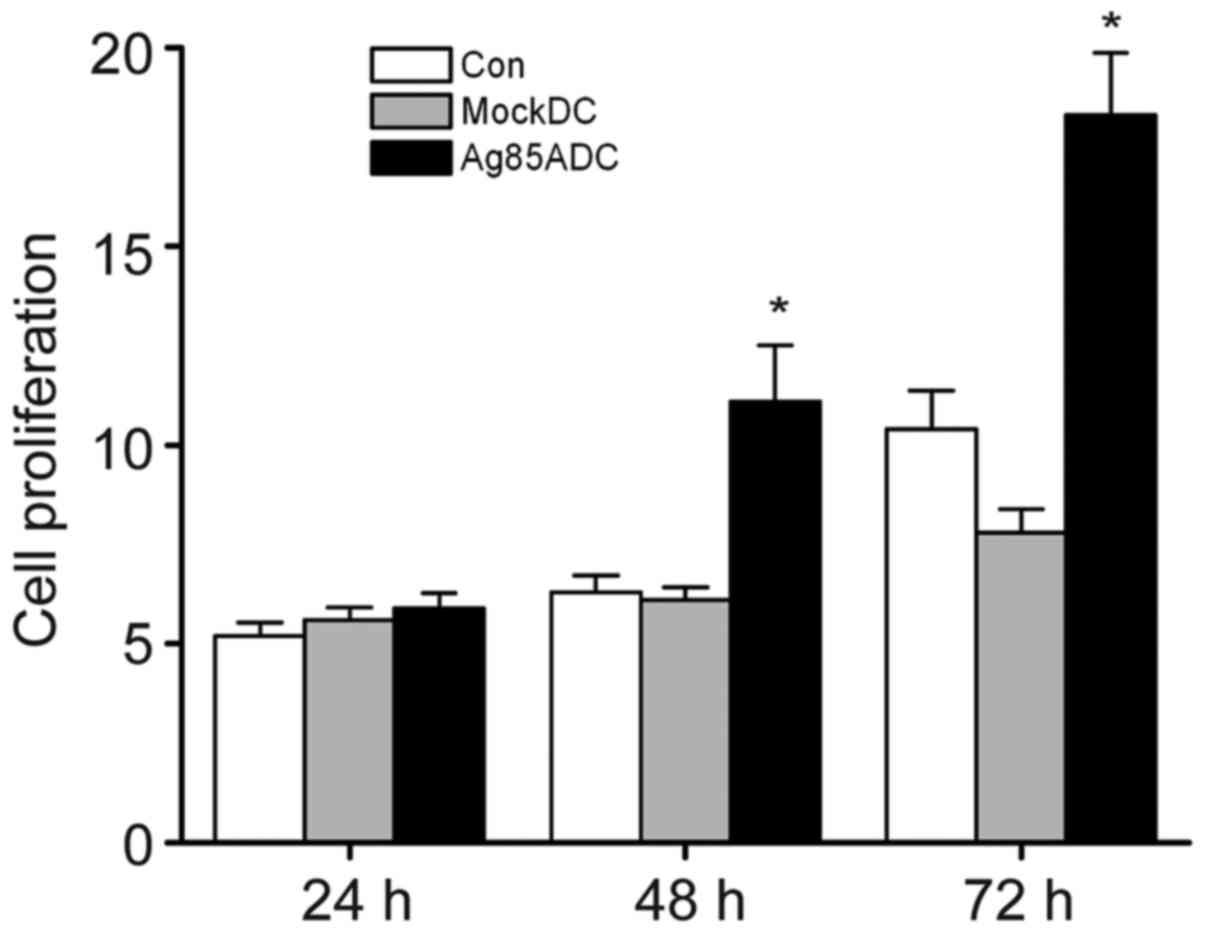
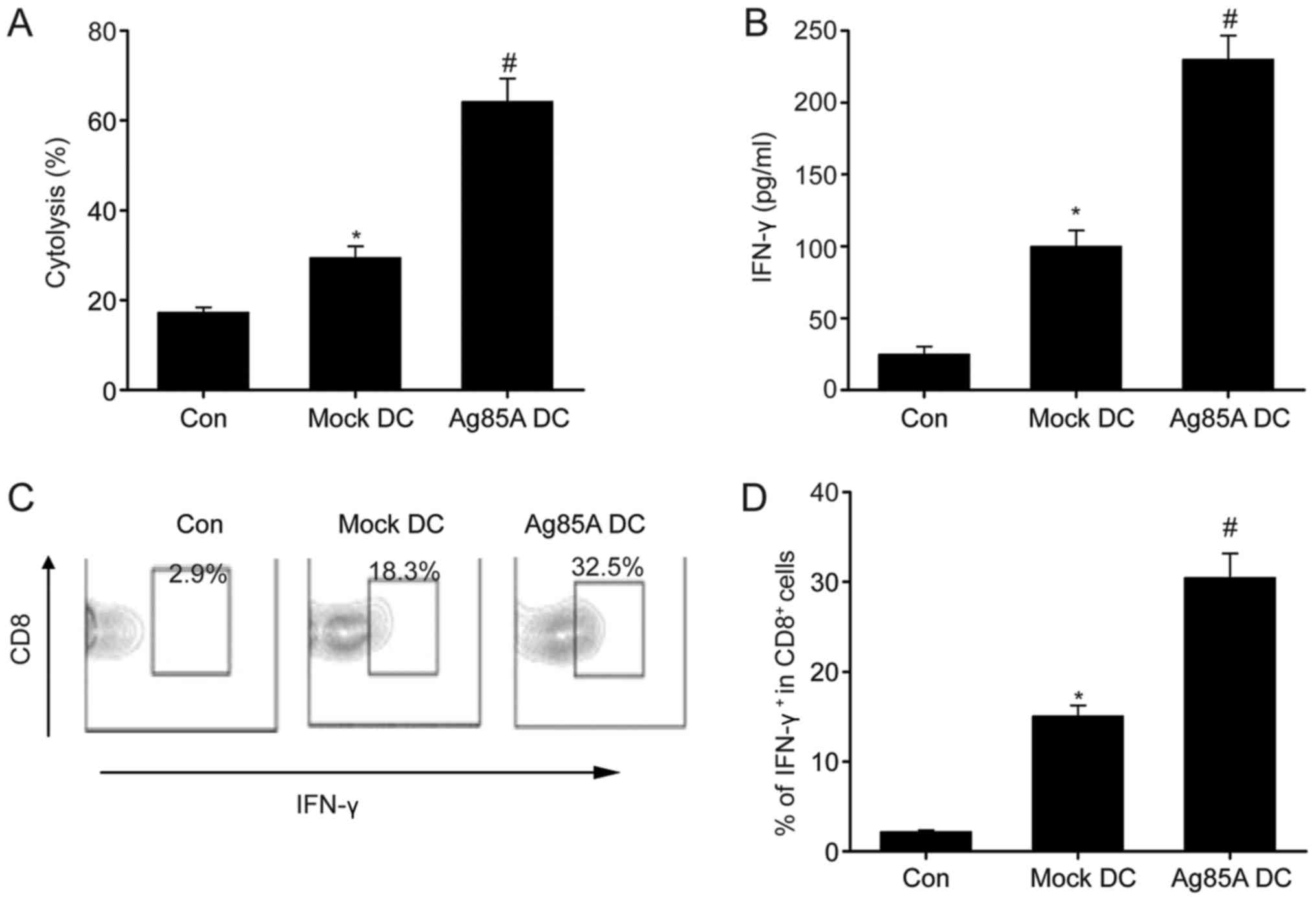
Meyer B and Are C: Current status and
future directions in colorectal cancer. Indian J Surg Oncol.
8:455–456. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mohammadian M, Zeynali S, Azarbaijani AF,
Khadem AM and Kheradmand F: Cytotoxic effects of the
newly-developed chemotherapeutic agents 17-AAG in combination with
oxaliplatin and capecitabine in colorectal cancer cell lines. Res
Pharm Sci1. 12:517–525. 2017. View Article : Google Scholar
|
|
6
|
Sun X, Han X, Xu L, Gao M, Xu J, Yang R
and Liu Z: Surface-engineering of red blood cells as artificial
antigen presenting cells promising for cancer immunotherapy. Small.
13:2017. View Article : Google Scholar
|
|
7
|
Richaud M and Bendriss-Vermare N: Cancer
immunotherapy via systemic RNA delivery to dendritic cells. Med Sci
(Paris). 33:852–856. 2017.(In French). View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Brown MC, Holl EK, Boczkowski D, Dobrikova
E, Mosaheb M, Chandramohan V, Bigner DD, Gromeier M and Nair SK:
Cancer immunotherapy with recombinant poliovirus induces
IFN-dominant activation of dendritic cells and tumor
antigen-specific CTLs. Sci Transl Med. 9(pii): eaan42202017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gilboa E: DC-based cancer vaccines. J Clin
Invest. 117:1195–1203. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chang WT, Chen HM, Yin SY, Chen YH, Wen
CC, Wei WC, Lai P, Wang CH and Yang NS: Specific
dioscoreaphytoextracts enhance potency of TCL-loaded DC-based
cancer vaccines. Evid Based Complement Alternat Med.
2013:9320402013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Draube A, Klein-González N, Mattheus S,
Brillant C, Hellmich M, Engert A and von Bergwelt-Baildon M:
Dendritic cell based tumor vaccination in prostate and renal cell
cancer: A systematic review and meta-analysis. PLoS One.
6:e188012011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hsu FJ, Benike C, Fagnoni F, Liles TM,
Czerwinski D, Taidi B, Engleman EG and Levy R: Vaccination of
patients with B-cell lymphoma using autologous antigen-pulsed
dendritic cells. Nat Med. 2:52–58. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nakai N, Asai J, Ueda E, Takenaka H, Katoh
N and Kishimoto S: Vaccination of Japanese patients with advanced
melanoma with peptide, tumor lysate or both peptide and tumor
lysate-pulsed mature, monocyte-derived dendritic cells. J Dermatol.
33:462–472. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fong L, Hou Y, Rivas A, Benike C, Yuen A,
Fisher GA, Davis MM and Engleman EG: Altered peptide ligand
vaccination with Flt3 ligand expanded dendritic cells for tumor
immunotherapy. Proc Natl Acad Sci USA. 98:8809–8814. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Brody JD and Engleman EG: DC-based cancer
vaccines: Lessons from clinical trials. Cytotherapy. 6:122–127.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Engleman EG: Dendritic cell-based cancer
immunotherapy. Semin Oncol. 30(3): Suppl 8:S23–S29. 2003.
View Article : Google Scholar
|
|
17
|
Jakob T, Walker PS, Krieg AM, Udey MC and
Vogel JC: Activation of cutaneous dendritic cells by CpG-containing
oligodeoxynucleotides: A role for dendritic cells in the
augmentation of Th1 responses by immunostimulatory DNA. J Immunol.
161:3042–3049. 1998.PubMed/NCBI
|
|
18
|
Sparwasser T, Koch ES, Vabulas RM, Heeg K,
Lipford GB, Ellwart JW and Wagner H: Bacterial DNA and
immunostimulatory CpG oligonucleotides trigger maturation and
activation of murine dendritic cells. Eur J Immunol. 28:2045–2054.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Belisle JT, Vissa VD, Sievert T, Takayama
K, Brennan PJ and Besra GS: Role of the major antigen of
Mycobacterium tuberculosis in cell wall biogenesis. Science.
276:1420–1422. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lozes E, Huygen K, Content J, Denis O,
Montgomery DL, Yawman AM, Vandenbussche P, Van Vooren JP, Drowart
A, Ulmer JB and Liu MA: Immunogenicity and efficacy of a
tuberculosis DNA vaccine encoding the components of the secreted
antigen 85 complex. Vaccine. 15:830–833. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Huygen K, Content J, Denis O, Montgomery
DL, Yawman AM, Deck RR, DeWitt CM, Orme IM, Baldwin S, D'Souza C,
et al: Immunogenicity and protective efficacy of a tuberculosis DNA
vaccine. Nat Med. 2:893–898. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yuan W, Dong N, Zhang L, Liu J, Lin S,
Xiang Z, Qiao H, Tong W and Qin C: Immunogenicity and protective
efficacy of a tuberculosis DNA vaccine expressing a fusion protein
of Ag85B-Esat6-HspX in mice. Vaccine. 30:2490–2497. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tanghe A, Denis O, Lambrecht B, Motte V,
van den Berg T and Huygen K: Tuberculosis DNA vaccine encoding
Ag85A is immunogenic and protective when administered by
intramuscular needle injection but not by epidermal gene gun
bombardment. Infect Immun. 68:3854–3860. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Denis O, Tanghe A, Palfliet K, Jurion F,
van den Berg TP, Vanonckelen A, Ooms J, Saman E, Ulmer JB, Content
J and Huygen K: Vaccination with plasmid DNA encoding mycobacterial
antigen 85A stimulates a CD4+ and CD8+ T-cell epitopic repertoire
broader than that stimulated by Mycobacterium tuberculosis H37Rv
infection. Infect Immun. 66:1527–1533. 1998.PubMed/NCBI
|
|
25
|
Nakano H, Nagata T, Suda T, Tanaka T,
Aoshi T, Uchijima M, Kuwayama S, Kanamaru N, Chida K, Nakamura H,
et al: Immunization with dendritic cells retrovirally transduced
with mycobacterial antigen 85A gene elicits the specific cellular
immunity including cytotoxic T-lymphocyte activity specific to an
epitope on antigen 85A. Vaccine. 24:2110–2119. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tarrant JP, Walsh MJ, Blanchard MC, Lee
TD, Hoskin DW and Giacomantonio CA: Reduced tumorigenicity of
B16-F10 mouse melanoma cells transfected with mycobacterial antigen
85A. Int J Oncol. 25:1693–1699. 2004.PubMed/NCBI
|
|
27
|
Shen Z, Reznikoff G, Dranoff G and Rock
KL: Cloned dendritic cells can present exogenous antigens on both
MHC class I and class II molecules. J Immunol. 158:2723–2730.
1997.PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang P, Wang J, Wang D, Wang H, Shan F,
Chen L, Hou Y, Wang E and Lu CL: Dendritic cell vaccine modified by
Ag85A gene enhances anti-tumor immunity against bladder cancer. Int
Immunopharmacol. 14:252–260. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Okada H, Tahara H, Shurin MR, Attanucci J,
Giezeman-Smits KM, Fellows WK, Lotze MT, Chambers WH and Bozik ME:
Bone marrow-derived dendritic cells pulsed with a tumor-specific
peptide elicit effective anti-tumor immunity against intracranial
neoplasms. Int J Cancer. 78:196–201. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Koirala P, Roth ME, Gill J, Piperdi S,
Chinai JM, Geller DS, Hoang BH, Park A, Fremed MA, Zang X and
Gorlick R: Immune infiltration and PD-L1 expression in the tumor
microenvironment are prognostic in osteosarcoma. Sci Rep.
6:300932016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Eruslanov E, Daurkin I, Vieweg J, Daaka Y
and Kusmartsev S: Aberrant PGE(2) metabolism in bladder tumor
microenvironment promotes immunosuppressive phenotype of
tumor-infiltrating myeloid cells. Int Immunopharmacol. 11:848–855.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu KJ, Wang CC, Chen LT, Cheng AL, Lin
DT, Wu YC, Yu WL, Hung YM, Yang HY, Juang SH and Whang-Peng J:
Generation of carcinoembryonic antigen (CEA)-specific T-cell
responses in HLA-A*0201 and HLA-A*2402 late-stage colorectal cancer
patients after vaccination with dendritic cells loaded with CEA
peptides. Clin Cancer Res. 10:2645–2651. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Meshkat Z, Teimourpour A, Rashidian S,
Arzanlou M and Teimourpour R: Immunogenicity of a DNA vaccine
encoding Ag85a-Tb10.4 antigens from Mycobacterium tuberculosis.
Iran J Immunol. 13:289–295. 2016.PubMed/NCBI
|
|
35
|
Mancha-Agresti P, de Castro CP, Dos Santos
JSC, Araujo MA, Pereira VB, LeBlanc JG, Leclercq SY and Azevedo V:
Recombinant invasive Lactococcus lactis carrying a DNA vaccine
coding the Ag85A antigen increases INF-γ, IL-6 and TNF-α cytokines
after intranasal immunization. Front Microbiol. 8:12632017.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Poecheim J, Barnier-Quer C, Collin N and
Borchard G: Ag85A DNA vaccine delivery by nanoparticles: Influence
of the formulation characteristics on immune responses. Vaccines
(Basel). 4(pii): E322016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Stylianou E, Griffiths KL, Poyntz HC,
Harrington-Kandt R, Dicks MD, Stockdale L, Betts G and McShane H:
Improvement of BCG protective efficacy with a novel chimpanzee
adenovirus and a modified vaccinia Ankara virus both expressing
Ag85A. Vaccine. 33:6800–6808. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Metcalfe HJ, Steinbach S, Jones GJ,
Connelley T, Morrison WI, Vordermeier M and Villarreal-Ramos B:
Protection associated with a TB vaccine is linked to increased
frequency of Ag85A-specific CD4(+) T cells but no increase in
avidity for Ag85A. Vaccine. 34:4520–4525. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chang CN, Huang YC, Yang DM, Kikuta K, Wei
KJ, Kubota T and Yang WK: A phase I/II clinical trial investigating
the adverse and therapeutic effects of a postoperative autologous
dendritic cell tumor vaccine in patients with malignant glioma. J
Clin Neurosci. 18:1048–1054. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lou Q, Conway TJ Jr, Egilmez NK, Loyall
JL, Bernstein SH, Kelleher RJ Jr and Bankert RB: B cell tumor
vaccine enhanced by covalent attachment of immunoglobulin to
surface proteins on dendritic cells. Clin Immunol. 118:66–76. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fields RC, Shimizu K and Mulé JJ: Murine
dendritic cells pulsed with whole tumor lysates mediate potent
antitumor immune responses in vitro and in vivo. Proc Natl Acad Sci
USA. 95:9482–9487. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Dauer M, Herten J, Bauer C, Renner F,
Schad K, Schnurr M, Endres S and Eigler A: Chemosensitization of
pancreatic carcinoma cells to enhance T cell-mediated cytotoxicity
induced by tumor lysate-pulsed dendritic cells. J Immunother.
28:332–342. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Schnurr M, Galambos P, Scholz C, Then F,
Dauer M, Endres S and Eigler A: Tumor cell lysate-pulsed human
dendritic cells induce a T-cell response against pancreatic
carcinoma cells: an in vitro model for the assessment of tumor
vaccines. Cancer Res. 61:6445–6450. 2001.PubMed/NCBI
|