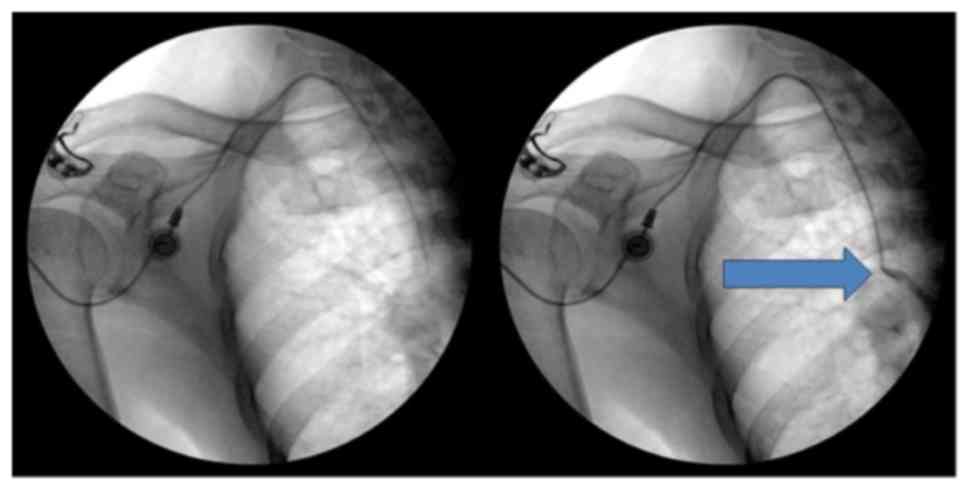
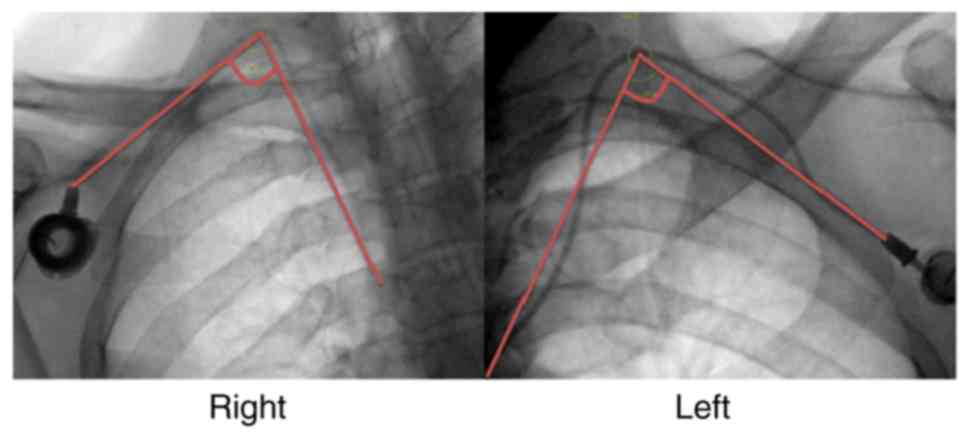
|
1
|
Niederhuber JE, Ensminger W, Gyves JW,
Liepman M, Doan K and Cozzi E: Totally implanted venous and
arterial access system to replace external catheters in cancer
treatment. Surgery. 92:706–712. 1982.PubMed/NCBI
|
|
2
|
Kock HJ, Pietsch M, Krause U, Wilke H and
Eigler FW: Implantable vascular access systems: Experience in 1500
patients with totally implanted central venous port systems. World
J Surg. 22:12–16. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schenck M and Jäger T: What is practically
important when carrying out a chemotherapy? Urologe A. 45:572,
574–576. 578–579. 2006.(In German).
|
|
4
|
Broviac JW, Cole JJ and Scribner BH: A
silicone rubber atrial catheter for prolonged parenteral
alimentation. Surg Gynecol Obstet. 136:602–606. 1973.PubMed/NCBI
|
|
5
|
Hickman RO, Buckner CD, Clift RA, Sanders
JE, Stewart P and Thomas ED: A modified right atrial catheter for
access to the venous system in marrow transplant recipients. Surg
Gynecol Obstet. 148:871–875. 1979.PubMed/NCBI
|
|
6
|
Torramadé JR, Cienfuegos JA, Hernández JL,
Pardo F, Benito C, González J, Balén E and de Villa V: The
complications of central venous access systems: A study of 218
patients. Eur J Surg. 159:323–327. 1993.PubMed/NCBI
|
|
7
|
Biffi R, de Braud F, Orsi F, Pozzi S,
Mauri S, Goldhirsch A, Nolè F and Andreoni B: Totally implantable
central venous access ports for long-term chemotherapy. A
prospective study analyzing complications and costs of 333 devices
with a minimum follow-up of 180 days. Ann Oncol. 9:767–773. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Funaki B and Zaleski GX: Re: Long-term
follow-up of upper extremity implanted venous access devices in
oncology patients. J Vasc Interv Radiol. 10:12811999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Teichgräber UK, Gebauer B, Benter T and
Wagner J: Long-term central venous lines and their complications.
Rofo. 176:944–952. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yeste S, ánchez L, Galbis Caravajal JM,
Fuster Diana CA and Moledo Eiras E: Protocol for the implantation
of a venous access device (Port-A-Cath System). The complications
and solutions found in 560 cases. Clin Transl Oncol. 8:735–741.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yaacob Y, Nguyen DV, Mohamed Z, Ralib AR,
Zakaria R and Muda S: Image-guided chemoport insertion by
interventional radiologists: A single-center experience on
periprocedural complications. Indian J Radiol Imaging. 23:121–125.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McGee DC and Gould MK: Preventing
complications of central venous catheterization. N Engl J Med.
348:1123–1133. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Beheshti MV: A concise history of central
venous access. Tech Vasc Interv Radiol. 14:184–185. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dede D, Akmangit I, Yildirim ZN, Sanverdi
E and Sayin B: Ultrasonography and fluoroscopy-guided insertion of
chest ports. Eur J Surg Oncol. 34:1340–1343. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Di Carlo I, Pulvirenti E, Mannino M and
Toro A: Increased use of percutaneous technique for totally
implantable venous access devices. Is it real progress? A 27-year
comprehensive review on early complications. Ann Surg Oncol.
17:1649–1656. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sakamoto N, Arai Y, Takeuchi Y, Takahashi
M, Tsurusaki M and Sugimuta K: Ultrasound-guided radiological
placement of central venous port via the subclavian vein: A
retrospective analysis of 500 cases at a single institute.
Cardiovasc Intervent Radiol. 33:989–994. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Teichgräber UK, Kausche S, Nagel SN and
Gebauer B: Outcome analysis in 3,160 implantations of
radiologically guided placements of totally implantable central
venous port systems. Eur Radiol. 21:1224–1232. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ahn SJ, Kim HC, Chung JW, An SB, Yin YH,
Jae HJ and Park JH: Ultrasound and fluoroscopy-guided placement of
central venous ports via internal jugular vein: Retrospective
analysis of 1254 port implantations at a single center. Korean J
Radiol. 13:314–323. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goltz JP, Janssen H, Petritsch B and
Kickuth R: Femoral placement of totally implantable venous power
ports as an alternative implantation site for patients with central
vein occlusions. Support Care Cancer. 22:383–387. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim HJ, Yun J, Kim HJ, Kim KH, Kim SH, Lee
SC, Bae SB, Kim CK, Lee NS, Lee KT, et al: Safety and effectiveness
of central venous catheterization in patients with cancer:
Prospective observational study. J Korean Med Sci. 25:1748–1753.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kumar AH, Srinivasan NM, Thakkar JM and
Mathew S: A prospective observational study of the outcome of
central venous catheterization in 100 patients. Anesth Essays Res.
7:71–75. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Babu KG, Suresh Babu MC, Lokanatha D and
Bhat GR: Outcomes, cost comparison, and patient satisfaction during
long-term central venous access in cancer patients: Experience from
a Tertiary Care Cancer Institute in South India. Indian J Med
Paediatr Oncol. 37:232–238. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Son JT, Min SY, Kim JI, Choi PW, Heo TG,
Lee MS, Kim CN, Kim HY, Yi SY, Lee HR and Roh YN: Thrombolytic
therapy using urokinase for management of central venous catheter
thrombosis. Vasc Specialist Int. 30:144–150. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schenck M, Schneider T, Rubben H and
Eisenhardt A: Central venous port implantations via the cephalic
vein applying an intravasal electrographic control of the catheter
tip position: A single-center experience of 316 cases. World J
Urol. 30:399–404. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bishop L, Dougherty L, Bodenham A, Mansi
J, Crowe P, Kibbler C, Shannon M and Treleaven J: Guidelines on the
insertion and management of central venous access devices in
adults. Int J Lab Hematol. 29:261–278. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Brothers TE, Von Moll LK, Niederhuber JE,
Roberts JA, Walker-Andrews S and Ensminger WD: Experience with
subcutaneous infusion ports in three hundred patients. Surg Gynecol
Obstet. 166:295–301. 1988.PubMed/NCBI
|
|
27
|
Whitman ED: Complications associated with
the use of central venous access devices. Curr Probl Surg.
33:309–378. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Harish K: Chemoport-skin erosion: Our
experience. Int J Angiol. 23:215–216. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Almhanna K, Pelley RJ, Thomas Budd G,
Davidson J and Moore HC: Subcutaneous implantable venous access
device erosion through the skin in patients treated with
anti-vascular endothelial growth factor therapy: A case series.
Anticancer Drugs. 19:217–219. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee AY: Cancer and thromboembolic disease:
Pathogenic mechanisms. Cancer Treat Rev. 28:137–140. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chelmow D, Rodriguez EJ and Sabatini MM:
Suture closure of subcutaneous fat and wound disruption after
cesarean delivery: A meta-analysis. Obstet Gynecol. 103:974–980.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hertault A, Sobocinski J, Spear R, Azzaoui
R, Delloye M, Fabre D and Haulon S: What should we expect from the
hybrid room? J Cardiovasc Surg (Torino). 58:264–269.
2017.PubMed/NCBI
|
|
33
|
Frank DA, Meuse J, Hirsch D, Ibrahim JG
and van den Abbeele AD: The treatment and outcome of cancer
patients with thromboses on central venous catheters. J Thromb
Thrombolysis. 10:271–275. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Verso M and Agnelli G: Venous
thromboembolism associated with long-term use of central venous
catheters in cancer patients. J Clin Oncol. 21:3665–3675. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Biffi R, Orsi F, Pozzi S, Pace U, Bonomo
G, Monfardini L, Della Vigna P, Rotmensz N, Radice D, Zampino MG,
et al: Best choice of central venous insertion site for the
prevention of catheter-related complications in adult patients who
need cancer therapy: A randomized trial. Ann Oncol. 20:935–940.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Saber W, Moua T, Williams EC, Verso M,
Agnelli G, Couban S, Young A, De Cicco M, Biffi R, van Rooden CJ,
et al: Risk factors for catheter-related thrombosis (CRT) in cancer
patients: A patient-level data (IPD) meta-analysis of clinical
trials and prospective studies. J Thromb Haemost. 9:312–319. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Di Carlo I, Cordio S, La Greca G,
Privitera G, Russello D, Puleo S and Latteri F: Totally implantable
venous access devices implanted surgically: A retrospective study
on early and late complications. Arch Surg. 136:1050–1053. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Araujo C, Silva JP, Antunes P, Fernandes
JM, Dias C, Pereira H, Dias T and Fougo JL: A comparative study
between two central veins for the introduction of totally
implantable venous access devices in 1201 cancer patients. Eur J
Surg Oncol. 34:222–226. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Safdar N and Maki DG: Risk of
catheter-related bloodstream infection with peripherally inserted
central venous catheters used in hospitalized patients. Chest.
128:489–495. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ballarini C, Intra M, Pisani Ceretti A,
Cordovana A, Pagani M, Farina G, Perrone S, Tomirotti M, Scanni A
and Spina GP: Complications of subcutaneous infusion port in the
general oncology population. Oncology. 56:97–102. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wolosker N, Yazbek G, Nishinari K,
Malavolta LC, Munia MA, Langer M and Zerati AE: Totally implantable
venous catheters for chemotherapy: Experience in 500 patients. Sao
Paulo Med J. 122:147–151. 2004. View Article : Google Scholar : PubMed/NCBI
|