Introduction
Carbon monoxide (CO) is a colorless, odorless and
tasteless gas harming human health at levels exceeding 100 ppm
(1). Although CO poisoning is
preventable, it remains an important cause of morbidity and
mortality in modern society and a leading cause of death due to
poising (2,3). Common symptoms of CO poisoning include
headache, nausea, vomiting, dizziness, malaise and altered mental
state as well as myocardial ischemia, chest pain and shortness of
breath in some cases (4). The
toxicity of CO is mainly caused by the high affinity of CO for
hemoglobin, which results in tissue hypoxia (1). In addition, various cellular mechanisms
have been implicated in the toxic effects of CO, including binding
of CO to other intracellular proteins (such as myoglobin and
cytochrome), generation of nitric oxide and peroxynitrite, lipid
peroxidation, mitochondrial oxidative stress, immune-mediated
damage and apoptosis (2).
Administration of oxygen is the main treatment of CO poisoning,
which can accelerate the elimination of carboxyhemoglobin (COHb)
and reduce tissue hypoxia.
Several studies have reported that hyperbaric oxygen
therapy is an effective treatment for CO poisoning (4–6).
Furthermore, the addition of dexamethasone (7) or N-butylphthalide (8) to hyperbaric oxygen therapy may also
exert beneficial effects.
Delayed encephalopathy after acute carbon monoxide
poisoning (DEACMP) is a serious and common complication of exposure
to CO at toxic levels (9,10). DEACMP manifests as memory impairment,
cognitive dysfunction and behavioral disorders that develop several
days or weeks after acute exposure to CO (9,10). There
is some evidence that hyperbaric oxygen therapy may be beneficial
in patients with DEACMP (11).
Nonetheless, the prognosis of DEACMP is poor, and the factors
associated with clinical outcomes include age, lucid interval
(between the initial recovery from the acute CO intoxication and
the subsequent appearance of DEACMP symptoms), danger activities of
daily living score, complications (11) and diffusion tensor imaging findings
(12).
Identifying patients with acute CO poisoning that
would go on to develop DEACMP would facilitate decision-making
regarding treatment strategies, but this identification is
challenging. Reported risk factors for DEACMP include age >35
years, CO exposure for >24 h, serious disturbance of
consciousness at emergency admission, prolonged disturbance of
consciousness, elevated levels of creatine kinase, creatine
kinase-MB and lactate dehydrogenase, and low Global Assessment
Scale score (13–16). However, the clinical utility of these
factors in predicting DEACMP is limited. Magnetic resonance imaging
(MRI) has revealed that DEACMP is associated with multiple lesions
in the basal ganglia (mainly the globus pallidus, but also in the
caudate nucleus and putamen alone), subcortical and periventricular
white matter, cerebral cortex and hippocampus (17–19). In
light of this, it is valuable to apply imaging methods to identify
patients at risk of developing DEACMP. Although MRI can identify
cerebral lesions in patients with acute CO poisoning, one study has
found that the presence of MRI lesions at 1 month after CO
intoxication was unable to accurately predict longer-term clinical
outcome (20). Computed tomography
(CT) is a clinically routine and non-invasive imaging technique
that may have the potential to identify patients at risk of DEACMP.
Two previous studies have reported that CT findings of hypoxic
encephalopathy are related to the development of DEACMP (16,21).
However, there remains a paucity of data regarding the utility of
CT for identifying patients at risk of DEACMP.
This study identified the early-stage clinical
features and CT findings that predicted the development of DEACMP
in patients with acute CO poisoning.
Patients and methods
Study design and patients
Patients with acute CO poisoning admitted to Beijing
Chaoyang Hospital, Shenyang Ninth People's Hospital, Shanxi Second
People's Hospital and Shandong Provincial Hospital in China from
January 2013 to January 2016 were retrospectively analyzed.
Inclusion criteria: i) patients aged 18–75 years; ii) those
diagnosed with acute CO poisoning based on signs, symptoms and COHb
levels (22,23); iii) those imaged by brain CT during
the early stages (i.e., within 24 h of admission to hospital); and
ⅳ) those who were diagnosed or suspected of the disease before the
development of DEACMP. Exclusion criteria: i) patients with history
of severe cerebrovascular disease, epilepsy, encephalitis,
meningitis or congenital neurologic disease; ii) those with history
of severe heart, liver or kidney disease; iii) those who had
visited several hospitals, been ill for several days (with obvious
signs and symptoms), or not received a CT examination at an early
stage; and iv) women who were pregnant or breastfeeding. This study
was approved by the Ethics Committee of the China Center for
Disease Control and Prevention (Beijing, China), but patients did
not sign the informed consent due to the retrospective design of
the study.
Diagnosis of DEACMP
DEACMP was diagnosed if any of the following
clinical abnormalities were observed at 2–60 days after the initial
recovery from the consciousness disorder caused by acute CO
poisoning: i) disturbance of mental state and/or consciousness
manifesting as dementia or delirium; ii) extrapyramidal neuropathy
manifesting as Parkinson's syndrome; iii) pyramidal neurologic
damage (such as hemiplegia, pathologic reflexes or urinary
incontinence); and iv) focal cerebral cortical dysfunction (such as
aphasia, blindness or the occurrence of secondary epilepsy).
Additional evidence of DEACMP included pathologic low-density
regions in brain CT images and moderate or severe abnormalities in
the electroencephalogram.
Brain CT imaging
Brain CT imaging was performed immediately and was
repeated within 24 h after the occurrence or suspected occurrence
of toxic encephalopathy symptoms (to aid in the diagnosis of
DEACMP). The CT scanner used was either a SOMATOM Emotion 16
(Siemens Healthineers, Erlangen, Germany) or a BrightSpeed Elite
(GE Healthcare, Chicago, IL, USA), and the scanning parameters
were: layer thickness, 5 mm; layer interval, 10 mm.
Analysis of CT images
The CT value within a region of interest (2–5
cm2, adjusted according to the size of the site being
analyzed) was measured in the following brain regions: bilateral
centrum semiovale, white matter of the frontal, parietal, occipital
and temporal lobes, basal ganglia, thalamus, capsula interna,
cerebral peduncle, pons and cerebellum (mainly white matter).
Except for special cases, when the original data could not be
provided and the CT value was measured only by the doctor appointed
by the local hospital, all CT values were measured independently by
two senior radiologists (with 22 and 26 years of work experience,
respectively), with each radiologist measuring the CT value of each
brain region 3 times to obtain an average value.
Semi-quantitative analysis of the CT
abnormalities
Three experienced radiologists (each with >20
years of work experience) independently calculated an integrated CT
score, based on the characteristics of the regions of abnormal
density, and an average value was obtained for each patient. This
approach is not subject to the limitations of the CT scanner,
allowing the data to be directly compared between hospitals. The
method used to determine scores was as follows: i) distribution of
lesion for each brain region: 0, no abnormal density; +1,
unilateral density abnormality; +2, bilateral density abnormality.
The scores for the various brain regions analyzed were summated to
provide an overall score for lesion distribution; a higher score
indicated a wider distribution of abnormal density. ii) Extent of
lesion: +1, when the density/abnormality was limited to the deep
white matter; +2, when the density/abnormality was both in deep
white matter and in subcortical white matter. iii) Lesion severity:
+1, when the reduction of CT value was ≤2–5 Hounsfield units (HU);
+2, when the reduction in CT value was >5 HU. The scores for
various brain regions were summarized to provide an overall score
for lesion severity. iv) Swelling of parenchyma: 0, no brain
swelling; +1, sulcal effacement; +2, ventricle is subjected to
compression and becomes narrower. v) Other, including cerebral
cortex involvement, hemorrhagic lesion and hippocampal lesion: +1,
one condition; +2, two conditions; +3, ≥3 conditions. The above
scores were summarized to give a final total score for each
patient; a higher total score was taken to indicate more serious
disease.
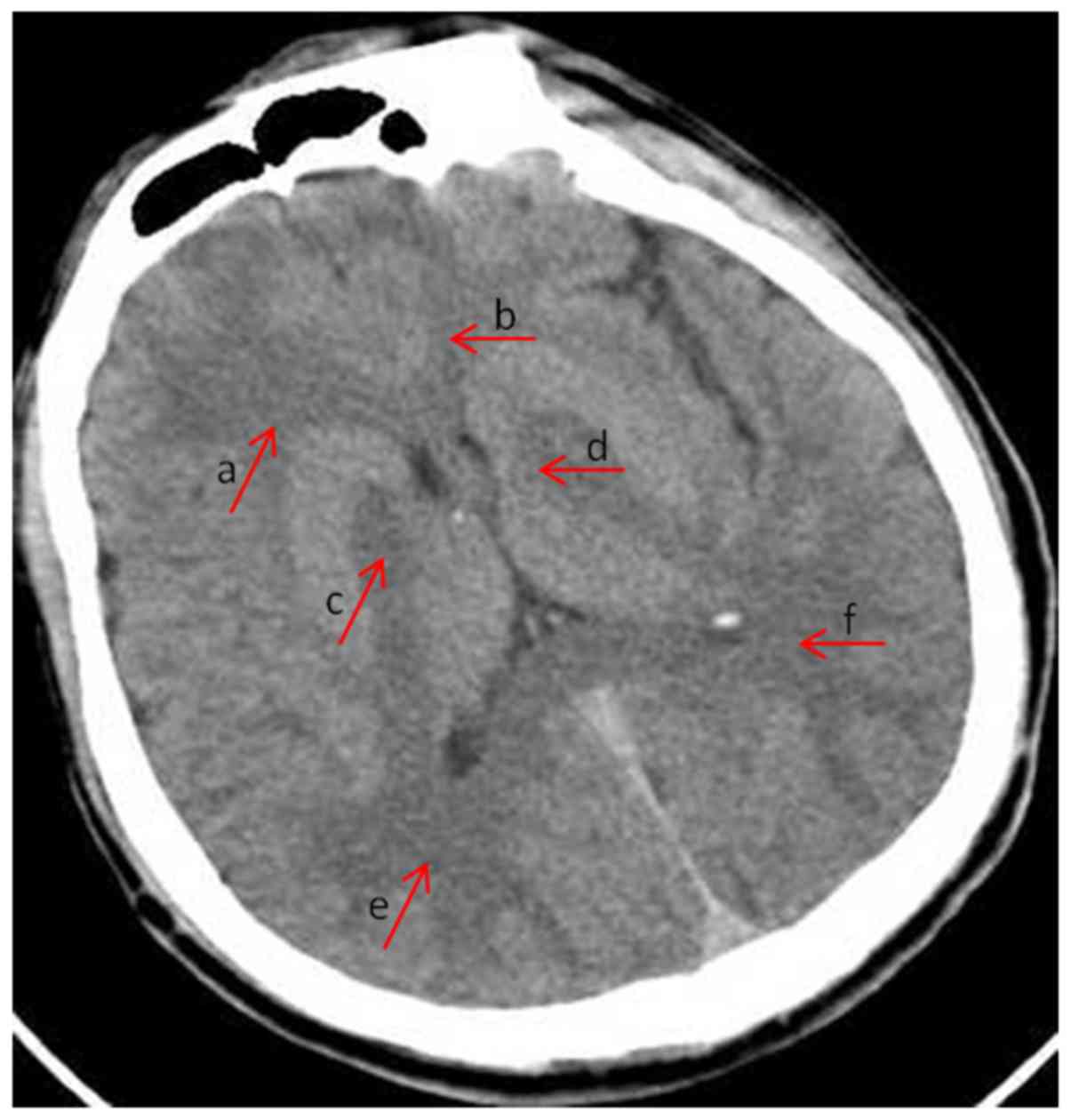
Fig. 1 shows an
example of semi-quantitative analysis of the CT abnormalities. The
patient was diagnosed with acute CO poisoning and the brain CT was
at onset (within 24 h). Distribution of lesion: +2, the areas of
subcortical decreased attenuation in bilateral frontal (arrows a
and b) and occipital lobe (arrows e and f); +2, bilateral globus
pallidus (arrows c and d). Extent of lesion: +1, deep white matter,
no subcortical white matter involvement, frontal and occipital
lobe. Lesion severity: +1, hypodensity of parenchyma, CT value ≤2–5
HU, frontal lobe, occipital lobe and globus pallidus. Swelling of
parenchyma: +1, sulcal effacement; +0, no involvement of other
regions. The total integrated CT score of the patient was 11.
Data collection
The following information was extracted from the
patients' medical records: age, sex, onset time of symptoms,
duration of exposure to CO, COHb level at admission, acute
physiology and chronic health evaluation-II (APACHE-II) scale score
(24), Glasgow Coma Scale (GCS)
score (25) and abnormal CT
findings. Besides, the severity of the clinical symptoms was
classified as mild, moderate or severe based on the APACHE-II score
and the diagnosis of CO poisoning based on signs, symptoms and COHb
levels (22,23).
Follow-up
Each patient was followed-up for 6 months from the
occurrence of acute CO poisoning and was reviewed daily for the
first 2 months and then monthly for the remaining period. Follow-up
was conducted in the hospital or clinic. GCS score, APACHE-II score
and brain CT images were evaluated at onset. The occurrence of
DEACMP was determined on the basis of the patient's clinical
symptoms and signs (vide supra).
Statistical analysis
Statistical analysis was performed using SPSS for
Windows, release 19.0 (IBM Corp., Armonk, NY, USA). Data are
presented as the mean ± standard deviation (SD) or n (%) and
compared using the independent samples t-test, Chi-square test or
rank sum test, as appropriate. Non-normally distributed data were
compared with the rank sum test. The utility of the integrated CT
value in the prediction of DEACMP was explored using the receiver
operating characteristic (ROC) curve analysis with calculation of
the area under the curve (AUC). P<0.05 was considered to
indicate a statistically significant difference.
Results
Baseline demographic and clinical
characteristics
A total of 123 patients with acute CO poisoning were
included in the analysis, among whom 27 (22.0%) developed DEACMP
(DEACMP group) and 96 (78.0%) did not (non-DEACMP group). The
baseline demographic and clinical characteristics of the study
participants are presented in Table
I. There were no significant differences between the DEACMP and
non-DEACMP group in age, sex, and COHb level at admission (Table I). However, compared with patients in
the non-DEACMP group, those in the DEACMP group had a longer
duration of exposure to CO (13.11±7.26 vs. 8.12±4.71 h;
P<0.001), higher APACHE-II score (16.48±5.25 vs. 9.02±5.22;
P<0.001), lower GCS score (4.59±1.76 vs. 9.28±4.10; P<0.001),
and there was a higher proportion of patients with severe clinical
symptoms (P=0.001; Table I) and
brain CT abnormalities (81.5 vs. 51.0%; P=0.002).
 | Table I.Baseline demographic and clinical
characteristics of the study participants. |
Table I.
Baseline demographic and clinical
characteristics of the study participants.
| Characteristics | DEACMP group
(n=27) | Non-DEACMP group
(n=96) | P-value |
|---|
| Sex |
|
|
0.187 |
| Male | 19 (70.4%) | 54 (56.2%) |
|
|
Female | 8
(29.6%) | 42 (43.8%) |
|
| Age (years) |
48.15±13.25 | 45.23±16.64 |
0.403 |
| Onset time of
symptoms (h) | 75.57 | 58.18 |
0.020 |
| Duration of exposure
to CO (h) | 13.11±7.26 | 8.12±4.71 | <0.001 |
| Abnormal cranial CT
findings at admission | 22 (81.5%) | 49 (51.0%) |
0.002 |
| COHb level at
admission |
27.10±12.32% |
24.36±12.85% |
0.325 |
| APACHE-II score | 16.48±5.25 | 9.02±5.22 | <0.001 |
| GCS score |
4.59±1.76 | 9.28±4.10 | <0.001 |
| Severity of clinical
symptoms |
|
|
0.001 |
| Mild | 3
(11.1%) | 49 (51.0%) |
|
| Moderate | 16 (59.3%) | 34 (35.4%) |
|
| Severe | 8
(29.6%) | 13 (13.5%) |
|
Comparison of CT values between the
DEACMP and the non-DEACMP group
As shown in Table I,
the CT values were compared between the DEACMP and non-DEACMP group
for 69 patients scanned with the Siemens SOMATOM Emotion 16 scanner
and 54 patients scanned with the GE Healthcare scanner. For
patients scanned with the Siemens CT scanner, there were no
significant differences in the CT values of the various brain
regions, with the exception of the globus pallidus, which had a
lower CT value in the DEACMP group than in the non-DEACMP group
(29.93±3.29 vs. 31.98±3.70; P=0.014; Table I). For patients scanned with the GE
Healthcare scanner, there were no significant differences in the CT
values of any of the brain regions between the two groups (Table II).
 | Table II.Comparison of CT values of various
brain regions among patients who developed delayed encephalopathy
and those who did not. |
Table II.
Comparison of CT values of various
brain regions among patients who developed delayed encephalopathy
and those who did not.
| Brain region | DEACMP group | Non-DEACMP
group | P-value |
|---|
| Siemens
Healthineers scanner | 18 | 51 |
|
| Centrum
semiovale (HU) | 27.43±2.82 | 28.12±2.56 | 0.205 |
| Frontal
lobe (HU) | 28.37±3.77 | 29.23±5.29 | 0.386 |
|
Parietal lobe (HU) | 27.86±4.06 | 28.83±5.12 | 0.409 |
|
Occipital lobe (HU) | 28.09±3.79 | 29.67±4.99 | 0.167 |
|
Temporal lobe (HU) | 31.80±4.28 | 31.51±4.29 | 0.719 |
| Globus
pallidus (HU) | 29.93±3.29 | 31.98±3.70 | 0.014 |
|
Thalamus (HU) | 32.68±2.06 | 33.82±3.13 | 0.074 |
| Capsula
interna (HU) | 31.28±4.79 | 30.02±4.55 | 0.291 |
|
Cerebral peduncle (HU) | 30.15±2.30 | 30.49±2.84 | 0.618 |
| Pons
(HU) | 31.49±4.35 | 31.39±4.25 | 0.930 |
|
Cerebellar vermis (HU) | 36.19±2.63 | 36.76±3.82 | 0.521 |
|
Cerebellar dentate nucleus
(HU) | 30.42±4.18 | 31.69±3.74 | 0.578 |
| GE Healthcare
scanner, n | 9 | 45 |
|
| Centrum
semiovale (HU) | 23.21±2.34 | 22.72±3.04 | 0.951 |
| Frontal
lobe (HU) | 23.48±3.03 | 24.03±2.85 | 0.717 |
|
Parietal lobe (HU) | 23.64±2.12 | 23.64±3.02 | 0.997 |
|
Occipital lobe (HU) | 24.77±2.64 | 23.94±3.06 | 0.559 |
| Temporal lobe
(HU) | 23.48±3.37 | 24.02±3.35 | 0.717 |
| Globus pallidus
(HU) | 29.67±2.45 | 28.43±3.94 | 0.544 |
| Thalamus (HU) | 30.93±4.31 | 30.58±2.74 | 0.837 |
| Capsula interna
(HU) | 22.67±4.16 | 23.68±3.40 | 0.625 |
| Cerebral peduncle
(HU) | 26.07±3.30 | 26.60±3.18 | 0.781 |
| Pons (HU) | 27.63±3.51 | 26.93±2.66 | 0.668 |
| Cerebellar vermis
(HU) | 31.50±3.05 | 33.24±3.74 | 0.467 |
| Cerebellar dentate
nucleus (HU) | 27.05±5.46 | 26.84±4.08 | 0.925 |
Comparison of integrated CT scores
between the DEACMP and non-DEACMP group
Integrated CT scores were calculated for 111 out of
the 123 patients (the original images could not be obtained in 12
patients for the determination of the integrated CT scores). As
shown in Table II, the DEACMP and
non-DEACMP group did not differ significantly with regard to the
integrated CT scores of the basal ganglia, white matter or other
regions when these brain regions were considered individually.
However, the total integrated CT score in the DEACMP group was
notably higher than that in the non-DEACMP group (73.63 vs. 51.39;
P=0.002).
ROC curve analysis of the utility of
integrated CT score in the prediction of DEACMP
The area under the ROC curve was 0.700, with a 95%
confidence interval of 0.584–0.817 (P<0.01).
Discussion
An important finding of the present study was that
the DEACMP group had a longer duration of CO exposure, higher
APACHE-II score, lower GCS score, greater symptom severity and
higher proportion of patients with brain CT abnormalities.
Furthermore, the CT value in various brain regions could not be
used to distinguish between patients who subsequently developed
DEACMP and those who did not. Importantly, integrated CT score was
significantly higher in the DEACMP group than in the non-DEACMP
group and ROC curve analysis indicated that total integrated CT
score could potentially be used as a predictor of DEACMP. Our novel
findings indicate that total integrated CT score could potentially
be used to identify DEACMP in patients.
In this study, DEACMP subsequently occurred in 22%
of those with acute CO poisoning, consistent with previous
investigations reporting values ranging from 16.5 to 29.5%
(14,15,21). No
difference was found in sex between the two groups of patients in
this study, in consistency with a previous publication (15). Age of patients has been proven to
influence the incidence rate of DEACMP, although there are
inconsistencies among the published data. For example, Choi has
found that DEACMP does not occur in patients aged <30 years
(16), while Weaver et al
have determined that at age >35 years the risk in patients not
treated with hyperbaric oxygen is increased but the risk in those
that have received hyperbaric oxygen therapy is decreased (14). In agreement with our findings, Kudo
et al observed no difference in age between patients that
developed DEACMP and those that did not (15).
In this study, disease severity, regardless of the
score used (APACHE-II score, GCS score or clinical symptom score),
was markedly higher in the DEACMP group than that in the non-DEACMP
group, which is consistent with the findings of Kudo et al
that patients who suffered from DEACMP had a significantly higher
Japan Coma Scale score (15). Other
studies have also suggested that impaired consciousness is
correlated with DEACMP (16). It
would perhaps be expected that the degree of exposure to CO would
influence the severity of the CO poisoning and hence the risk of
DEACMP. It was observed that patients in the DEACMP group had a
longer duration of exposure to CO than those in the non-DEACAMP
group, in agreement with a previous report that exposure for 24 h
or longer is an independent risk factor for cognitive sequelae
(14). Although another study has
found no influence of duration of CO exposure on risk of DEACMP
(15), relevant data were available
for only half of their cohort of patients, and the authors noted
that variations in individual circumstances (particularly size and
ventilation of the location of exposure) could have affected the
interpretation of the results. COHb at first hospital consultation
did not appear to be associated with the development of DEACMP in
our study or clinical outcomes in other published research
(15,26). The lack of an association of COHb at
first hospital consultation with DEACMP may be due to the
relatively short half-life of COHb and hence the rapid fall in COHb
level that occurs after cessation of CO inhalation and
administration of hyperbaric oxygen.
There is very little research exploring whether CT
imaging could be used to predict the risk of DEACMP. An important
finding of the present study was that the DEACMP group had a higher
proportion of patients with brain CT abnormalities (81.5 vs.
51.0%). Similarly, Choi et al have determined that 62.2% of
the patients that developed delayed neurologic sequelae had initial
CT abnormalities, compared with only 27.0% of patients that made a
full recovery (21). Kudo et
al have also found that their DEACMP group had substantially
more patients with brain CT abnormalities than their non-DEACMP
group (76.9 vs. 6.2%) (15).
However, in this investigation, CT values in the various brain
regions examined did not differ between groups, indicating that
this parameter may not be useful for predicting the risk of DEACMP.
In contrast, the integrated CT score differed significantly between
patients who developed DEACMP and those who did not, and ROC curve
analysis suggested that this parameter could potentially be used to
predict the risk of DEACMP. An important advantage of integrated CT
score is that it is independent of the scanner characteristics,
allowing it to be used widely in different Institutions. The
integrated CT score is related to the severity of the patient's
lesions and thus is an indirect evaluation of the pathologic
changes in the brain. Therefore, this parameter may be a novel
approach to risk stratification in patients with acute CO
poisoning.
This study has some limitations. It was a
retrospective study, so it might have been prone to information
bias and selection bias. Acute CO poisoning is relatively rare,
hence the sample size was small despite the inclusion of four study
sites. Therefore, the results could not be generalized to other
regions of China or other countries. Unknown confounding factors
that were not included in the analysis might have influenced the
results. Direct comparisons of CT with other imaging modalities,
such as DWIMRI, were not made. The most commonly used quantitative
indicators of DWI are average diffusion coefficient (ADC) and
fractional nisotropy (FA). In our study, DWI findings were not
involved. The most commonly used quantitative indicators of DWI are
ADC and FA. The associations between CT findings and long-term
clinical outcomes were not examined. Additionally, prospective,
multicenter, large-scale studies are merited to further explore the
utility of CT in predicting the risk of DEACMP. In conclusion,
brain integrated CT score can potentially identify patients at risk
of DEACMP.
Acknowledgements
We would like to thank all patients and
investigators who were involved in this study. We also express our
thanks to Dr Jinsong Zhang and Dr Haishi Wang for their advice on
data analysis and determination of CT values and scores.
Funding
The study was supported by the Special Project for
Scientific Research in the Health Industry of China
(201202006-06).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XD and WH contributed in the data analysis and were
involved in the design of the study. XD, HG and FH were responsible
for determining the CT values and calculating the integrated CT
scores. LG, JW and XZ participated in the data acquisition and the
follow-up management of the patients. CS, HZ and PM were involved
in the design of the methods and the interpretation of the data.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Beijing Chaoyang Hospital (Beijing, China) and the Ethics Committee
of the Chinese Center for Disease Control and Prevention (Beijing,
China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Prockop LD and Chichkova RI: Carbon
monoxide intoxication: An updated review. J Neurol Sci.
262:122–130. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hampson NB, Piantadosi CA, Thom SR and
Weaver LK: Practice recommendations in the diagnosis, management,
and prevention of carbon monoxide poisoning. Am J Respir Crit Care
Med. 186:1095–1101. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hampson NB and Weaver LK: Carbon monoxide
poisoning: A new incidence for an old disease. Undersea Hyperb Med.
34:163–168. 2007.PubMed/NCBI
|
|
4
|
Weaver LK: Hyperbaric oxygen therapy for
carbon monoxide poisoning. Undersea Hyperb Med. 41:339–354.
2014.PubMed/NCBI
|
|
5
|
Weaver LK, Hopkins RO, Chan KJ, Churchill
S, Elliott CG, Clemmer TP, Orme JF Jr, Thomas FO and Morris AH:
Hyperbaric oxygen for acute carbon monoxide poisoning. N Engl J
Med. 347:1057–1067. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Annane D, Chadda K, Gajdos P,
Jars-Guincestre MC, Chevret S and Raphael JC: Hyperbaric oxygen
therapy for acute domestic carbon monoxide poisoning: Two
randomized controlled trials. Intensive Care Med. 37:486–492. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xiang W, Xue H, Wang B, Li Y, Zhang J,
Jiang C, Liang F, Pang J and Yu L: Combined application of
dexamethasone and hyperbaric oxygen therapy yields better efficacy
for patients with delayed encephalopathy after acute carbon
monoxide poisoning. Drug Des Devel Ther. 11:513–519. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xiang W, Xue H, Wang B, Li Y, Zhang J,
Jiang C and Pang J: Efficacy of N-butylphthalide and hyperbaric
oxygen therapy on cognitive dysfunction in patients with delayed
encephalopathy after acute carbon monoxide poisoning. Med Sci
Monit. 23:1501–1506. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Goldstein M: Carbon monoxide poisoning. J
Emerg Nurs. 34:538–542. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Guzman JA: Carbon monoxide poisoning. Crit
Care Clin. 28:537–548. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hu H, Pan X, Wan Y, Zhang Q and Liang W:
Factors affecting the prognosis of patients with delayed
encephalopathy after acute carbon monoxide poisoning. Am J Emerg
Med. 29:261–264. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hou X, Ma L, Wu L, Zhang Y, Ge H, Li Z,
Gao Y, Zhou Y and Gao C: Diffusion tensor imaging for predicting
the clinical outcome of delayed encephalopathy of acute carbon
monoxide poisoning. Eur Neurol. 69:275–280. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Weaver LK: Clinical practice. Carbon
monoxide poisoning. N Engl J Med. 360:1217–1225. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Weaver LK, Valentine KJ and Hopkins RO:
Carbon monoxide poisoning: risk factors for cognitive sequelae and
the role of hyperbaric oxygen. Am J Respir Crit Care Med.
176:491–497. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kudo K, Otsuka K, Yagi J, Sanjo K, Koizumi
N, Koeda A, Umetsu MY, Yoshioka Y, Mizugai A, Mita T, et al:
Predictors for delayed encephalopathy following acute carbon
monoxide poisoning. BMC Emerg Med. 14:32014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Choi IS: Delayed neurologic sequelae in
carbon monoxide intoxication. Arch Neurol. 40:433–435. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim JH, Chang KH, Song IC, Kim KH, Kwon
BJ, Kim HC, Kim JH and Han MH: Delayed encephalopathy of acute
carbon monoxide intoxication: Diffusivity of cerebral white matter
lesions. AJNR Am J Neuroradiol. 24:1592–1597. 2003.PubMed/NCBI
|
|
18
|
O'Donnell P, Buxton PJ, Pitkin A and
Jarvis LJ: The magnetic resonance imaging appearances of the brain
in acute carbon monoxide poisoning. Clin Radiol. 55:273–280. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hsiao CL, Kuo HC and Huang CC: Delayed
encephalopathy after carbon monoxide intoxication-long-term
prognosis and correlation of clinical manifestations and
neuroimages. Acta Neurol Taiwan. 13:64–70. 2004.PubMed/NCBI
|
|
20
|
Pavese N, Napolitano A, De Iaco G,
Canapicchi R, Collavoli PL, Lucetti C, Gambaccini G and Bonuccelli
U: Clinical outcome and magnetic resonance imaging of carbon
monoxide intoxication. A long-term follow-up study. Ital J Neurol
Sci. 20:171–178. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Choi IS, Kim SK, Choi YC, Lee SS and Lee
MS: Evaluation of outcome after acute carbon monoxide poisoning by
brain CT. J Korean Med Sci. 8:78–83. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Piantadosi CA: Diagnosis and treatment of
carbon monoxide poisoning. Respir Care Clin N Am. 5:183–202.
1999.PubMed/NCBI
|
|
23
|
Blumenthal I: Carbon monoxide poisoning. J
R Soc Med. 94:270–272. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Knaus WA, Draper EA, Wagner DP and
Zimmerman JE: APACHE II: A severity of disease classification
system. Crit Care Med. 13:818–829. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Teasdale G and Jennett B: Assessment of
coma and impaired consciousness. A practical scale. Lancet.
2:81–84. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tombaugh TN and McIntyre NJ: The
mini-mental state examination: A comprehensive review. J Am Geriatr
Soc. 40:922–935. 1992. View Article : Google Scholar : PubMed/NCBI
|