Introduction
Lung cancer is the leading cause of
cancer-associated mortality in both men and women worldwide, and
>80% of lung cancer cases are diagnosed as non-small cell lung
cancer (NSCLC) (1). Although great
achievements have been made in improving NSCLC diagnosis and
therapeutic approaches, including the development of targeted
therapies, the overall survival rate remains very low (with a
5-year survival rate of ~15%), which is accompanied with a high
recurrence rate (2). Thus, it is of
importance to uncover the molecular mechanism of cancer cell
proliferation and metastasis in NSCLC to facilitate the development
of novel drugs.
microRNAs (miRNAs or miRs) are small, non-coding
RNAs that function by binding to the 3′untranslated regions
(3′UTRs) of target mRNAs to induce mRNA degradation or suppression
of transcription (3). miRNAs have
been reported to be associated with cancer development via
targeting oncogenes or tumor suppressors in various cancer types,
including NSCLC (4–7). However, the role of miR-382 in NSCLC
development remains to be fully elucidated.
LIM-only protein 3 (LMO3) was initially discovered
based on the sequence homology with LMO1 (8). As a member of the LMO family, LMO3 is
an important regulator of cell growth and differentiation during
embryonic development (9). Later
research revealed that LMO3 acts as an oncogene through interacting
with nescient helix-loop-helix 2 in neuroblastoma (10). In NSCLC, LMO3 was previously reported
to promote cancer cell proliferation and metastasis, and was
negatively regulated by miR-630 (11).
In the present study, a decrease in miR-382
expression was observed in the tumor tissues of patients with NSCLC
compared with adjacent normal tissues. Additionally, overexpression
of miR-382 significantly inhibited NSCLC cell growth and migration.
Furthermore, elevated miR-382 expression decreased LMO3 mRNA and
protein expression levels. The luciferase assay confirmed that LMO3
was a direct target of miR-382. More importantly, in LMO3-silenced
A549 cells, miR-382 mimics did not exhibit anti-cancer effects. A
negative association was also identified between LMO3 and miR-382
expression in tumor tissues from patients with NSCLC. Taken
together, the present findings demonstrated a tumor suppressor role
of miR-382 in NSCLC.
Materials and methods
Patient samples
A total of 30 tumor tissues and matched adjacent
normal tissues were collected from 30 patients (10 males and 20
females; age range, 31–68 years) who had undergone surgery to treat
NSCLC at the Zhangzhou Hospital Affiliated to Fujian Medical
University (Zhangzhou, China) between January 2014 and January
2015. Fresh tissue samples were confirmed via histopathological
examination and then immediately stored in a −80°C refrigerator
prior to subsequent experimentation. Patients were graded and the
tumor sizes and the metastatic activity were assessed according to
American Joint Committee on Cancer Staging V7 (12). Diagnosis was confirmed by
pathological analysis. The patients included in the present study
did not receive radiation or chemotherapy prior to surgery. All
patients provided written informed consent and the Ethics Committee
of Zhangzhou Hospital Affiliated to Fujian Medical University
approved the present study.
Cell culture
The normal human bronchial epithelial cell line
BEAS-2B, 293 cells, and human lung cancer cell lines, H1299, H23
and A549 were purchased from the American Type Culture Collection
(Manassas, VA, USA). All cell lines were maintained in Dulbecco's
modified Eagle's medium (Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS;
Hyclone; GE Healthcare Life Sciences, Logan, UT, USA) and 1%
penicillin-streptomycin solution (Gibco; Thermo Fisher Scientific,
Inc.) in a humidified atmosphere containing 5% CO2 at
37°C.
Target prediction for miRNAs
Bioinformatics prediction of the target genes of
miRNA and miRNA binding sites was performed using the online
database, miRanda (http://www.microrna.org/). LMO3 was identified as a
target gene of miR-382 and was verified by experimental
analysis.
Plasmid construction
The LMO3 3′UTR was amplified from the cDNA of A549
cells using PrimeSTAR® Max DNA Polymerase (Takara Bio,
Inc., Otsu, Japan) and cloned into pGL3 plasmids (Invitrogen;
Thermo Fisher Scientific, Inc.) to construct pGL3-LMO3 3′UTR-wild
type (WT). The LMO3 primer sequences were as follows: Forward:
5′-GCTCTAGAAACACTTTAAGTTTGATGTG-3′; Reverse:
5′-GCTCTAGATAAGATCCTGACAATTATAAT-3′. The thermocycling conditions
were as follows: Denaturation at 98°C for 10 sec; annealing at 55°C
for 10 sec; elongation at 72°C for 60 sec for 35 cycles. A549 cDNA
was prepared from extracted RNA using the TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc.). Samples were the
reverse-transcribed into cDNA using the RevertAid RT Reverse
Transcription kit (Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. pGL3-LMO 3′UTR-mutant (mut) was
constructed using a QuikChange Site-Directed Mutagenesis kit
(Agilent Technologies, Inc., Santa Clara, CA, USA), according to
the manufacturer's protocol.
Small interfering (si)RNA
transfection
LMO3 siRNAs and control siRNA were purchased from GE
Healthcare Dharmacon, Inc., Lafayette, CO, USA. Knockdown of LMO3
was achieved by transfection of LMO3-specific siRNAs into A549
cells using Lipofectamine RNAiMAX (Invitrogen; Thermo Fisher
Scientific, Inc.). Briefly, 30 pmol LMO3 siRNAs or control siRNA
were mixed with Lipofectamine RNAiMAX in OptiMEM (Invitrogen;
Thermo Fisher Scientific, Inc.), incubated for 15 min at 37°C and
subsequently added into each well containing culture medium in
6-well or 96-well plates. Cells were cultured for a further 48 h
prior to experimentation. The siRNA sequences were as follows: LMO3
siRNA1, CGUCCCUACUCGAAUGAUG; LMO3 siRNA2, CCGUUACACCAAAGAGCCUC;
LMO3 siRNA3, GUAGUCCGUCUGGCAAAGGA; control siRNA,
UAAGGCUAUGAAGAGAUAC.
Luciferase assay
miR-382 mimics and miR-negative control (NC) mimics
were synthesized and purchased from Guangzhou RiboBio Co., Ltd.
(Guangzhou, China). The dual luciferase assay was performed using a
Dual-Luciferase® Reporter Assay System (Promega
Corporation, Madison, WI, USA). Briefly, 293 cells were cultured in
24-well plates and cotransfected with miR-382 mimics or miR-NC
mimics, pGL3-LMO3 3′UTR-WT or pGL3-LMO3 3′UTR-mut and
Renilla luciferase vector using Lipofectamine 2000
(Invitrogen; Thermo Fisher Scientific, Inc.). Following 48 h, cells
were collected and the dual-luciferase activity was examined with
Renilla luciferase as the internal control. The sequences
were of the miRs were as follows: miR-382 mimic,
5′GAAGUUGUUCGUGGUGGAUUCG3′ and miR-NC mimic,
5′CAUGUAGUACGCGUUGAGUACC3′.
Western blot analysis
Anti-LMO3 antibody (cat. no 517019, 1:1,000) was
purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA) and
anti-GAPDH antibody (cat. no G8795; 1:5,000) was obtained from
Sigma-Aldrich; Merck KGaA (Darmstadt, Germany). Cell lysates were
prepared using radioimmunoprecipitation assay lysis buffer
(Beyotime Institute of Biotechnology, Haimen, China). The protein
concentration was determined using a Pierce BCA Protein Assay kit
(Thermo Fisher Scientific, Inc.). Proteins (20 µg) were separated
using 8% SDS-PAGE and transferred to polyvinylidene difluoride
membranes. Membranes were subsequently blocked with 5% non-fat milk
at room temperature for 1 h and incubated with the indicated
primary antibodies (1:1,000) overnight at 4°C. The next day,
membranes were washed with TBS-Tween 20 and incubated with
horseradish peroxidase-conjugated secondary antibody for 1 h at
room temperature (cat. no. 516102; Santa Cruz Biotechnology, Inc.;
1:10,000). Subsequently, the membranes were developed using ECL
Prime Western Blotting Detection Reagents (GE Healthcare Life
Sciences). Images were captured and analyzed using ImageQuant TL
7.0 (GE Healthcare Life Sciences). GAPDH served as a loading
control.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
An miRNeasy Mini Kit (Qiagen, Inc., Valencia, CA,
USA) was used to extract total RNA from patient tissue and cells
(BEAS-2B, 293, H1299, H23 and A549) in accordance with the
manufacturer's instructions. Following this, a NanoDrop 2000 kit
(Thermo Fisher Scientific, Inc.) was used to measure the
concentration and quality of RNA. TransScript First-Strand cDNA
Synthesis SuperMix (Beijing Transgen Biotech Co., Ltd., Beijing,
China) was used to reverse transcribe RNA into cDNA following
manufacturer's protocol. qPCR was performed with a CFX96 Touch™
Real-Time PCR Detection System (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA) using SYBR Premix Ex Taq (Takara Bio, Inc.). The
thermocycling conditions were as follows: Pre-denaturation at 95°C
for 30 sec; denaturation at 95°C for 5 sec; annealing and
elongation at 60°C for 30 sec for 40 cycles. The relative
expression of genes was calculated using the 2−ΔΔCq
method (13). U6 and GAPDH were used
as internal controls for miRNA and mRNA, respectively. Sequences
for primers used were as follows: miR-382, forward
5′-CTGCAATCATTCACGGACAAC-3′ and reverse
5′-GTGTCGTCGAGTCGGCAATTC-3′; LMO3, forward
5′-ATGCTCTCAGTCCAGCCAGA-3′ and reverse 5′-TCAGCGAACCTGGGGTGCAT-3′;
U6, forward 5′-CCTGCTTCGGCAGCACA-3′ and reverse
5′-TGGAACGCTTCACGAA-3′; and GAPDH, forward
5′-CCACTCCTCCACCTTTGAC-3′ and reverse 5′-ACCCTGTTGCTGTAGCCA-3′.
Cell proliferation assay
Cell growth was measured using a Cell Counting Kit
(CCK)-8 (Dojindo Molecular Technologies, Inc., Kumamoto, Japan)
according to the manufacturer's protocol. Briefly, cells were
seeded in 96-well plates at 37°C. On the following day, 10 µl CCK-8
solution was added into each well and the cells were incubated for
2 h at 37°C. The absorbance at 450 nm was detected using a
microplate reader (Bio-Rad Laboratories, Inc.). At 24, 48 and 72 h
following transfection with miR-382 mimics or miR-NC mimics, the
cell number was analyzed using CCK-8.
Wound-healing assay
Cell migration ability was measured using a
wound-healing assay. A549 cells were cultured in 6-well plates at
37°C. On the following day, a wound was made by introducing a
scratch at the center of each well with a 10-µl pipette tip.
Culture medium was replaced with fresh medium containing 1% FBS,
and the A549 cells were cotransfected with miR-382 mimics or miR-NC
mimics and LMO3 siRNA or control siRNA using the protocol as
aforementioned in the luciferase assay. An image of the scratch was
then captured. Following 40 h, a second image of the scratch was
captured. Subsequently, the percentage of migratory cells was
analyzed using Image Pro Plus 6 (Media Cybernetics, Inc.,
Rockville, MD, USA).
Statistical analysis
All statistical analyses were performed using
GraphPad Prism 5.0 software (GraphPad Software, Inc., La Jolla, CA,
USA). Data are presented as the mean ± standard deviation.
Differences between two groups were analyzed using Student's t-test
and multiple groups were analyzed using one-way analysis of
variance followed by the Newman-Keuls post hoc test. P<0.05 was
considered to indicate a statistically significant difference.
Results
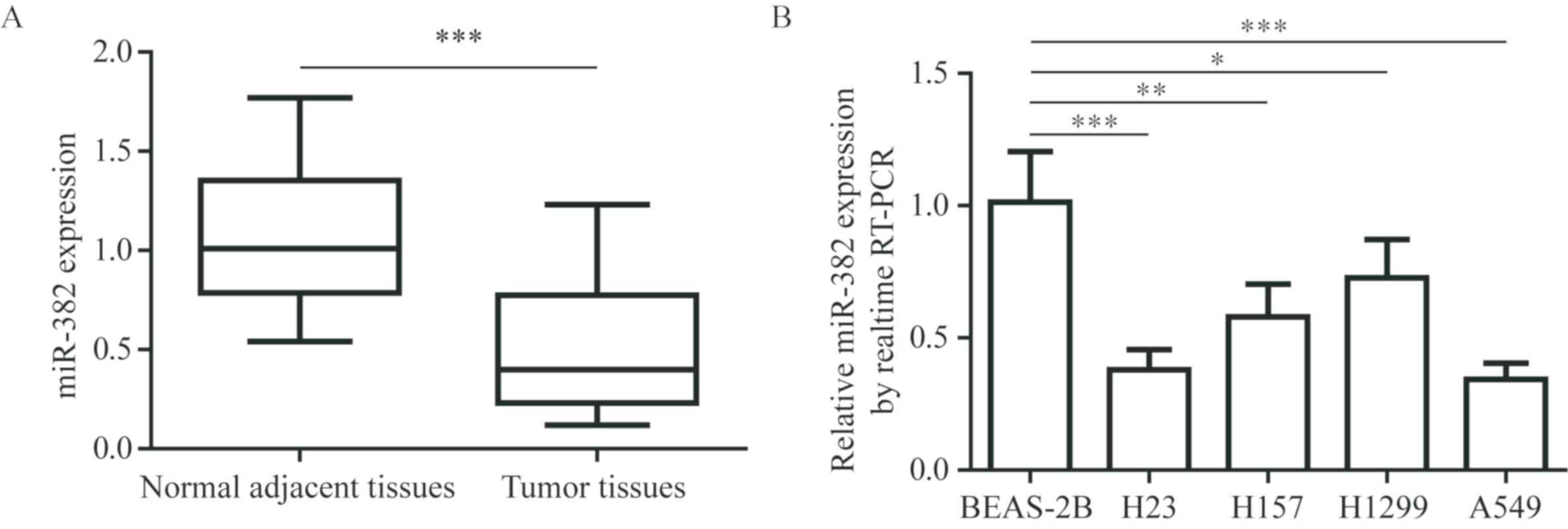
miR-382 expression is reduced in tumor
tissues from patients with NSCLC and NSCLC cell lines
To explore the role of miR-382 during NSCLC cancer
progression, the miR-382 expression levels between tumor tissues
and normal adjacent tissues from 30 patients with NSCLC were
compared. RT-qPCR detected a significant decrease in miR-382
expression in tumor tissues compared with normal adjacent tissues
(Fig. 1A). In addition,
significantly reduced miR-382 expression levels were observed in 3
NSCLC cell lines compared with BEAS-2B, a normal human bronchial
epithelial cell line (Fig. 1B).
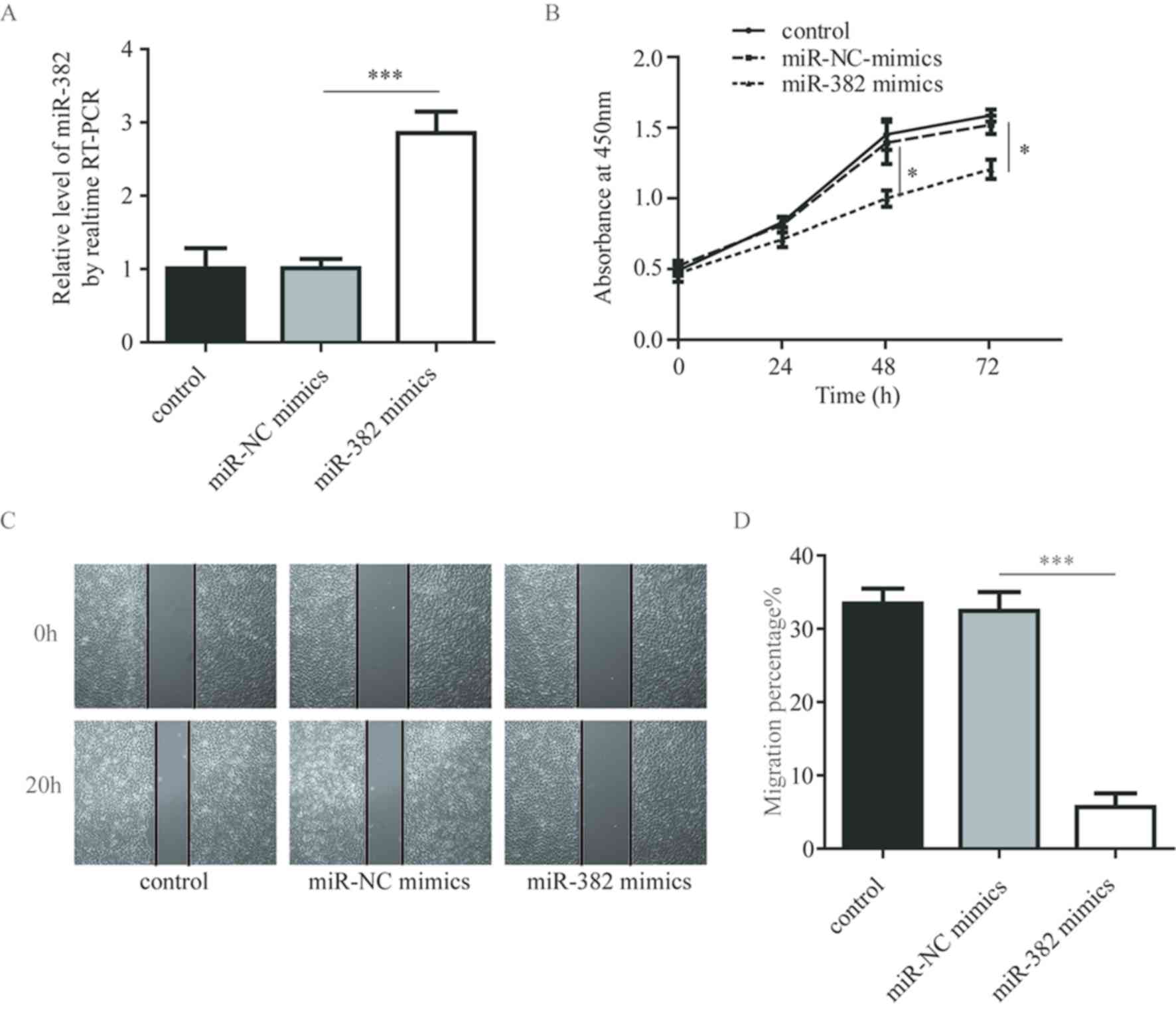
miR-382 suppresses cell proliferation
and migration in A549 cells
To further investigate how miR-382 influences NSCLC
cell behavior, the cell proliferation and migration abilities of
A549 cells were evaluated following the overexpression of miR-382
using miR-382 mimics. Compared with the control and miR-NC mimics,
miR-382 mimics significantly increased the miR-382 expression
levels in A549 cells (Fig. 2A).
Notably, overexpression of miR-382 provoked an inhibitory effect on
A549 cell proliferation and migration (Fig. 2B-D), suggesting that miR-382 may
suppress NSCLC cell growth and metastasis.
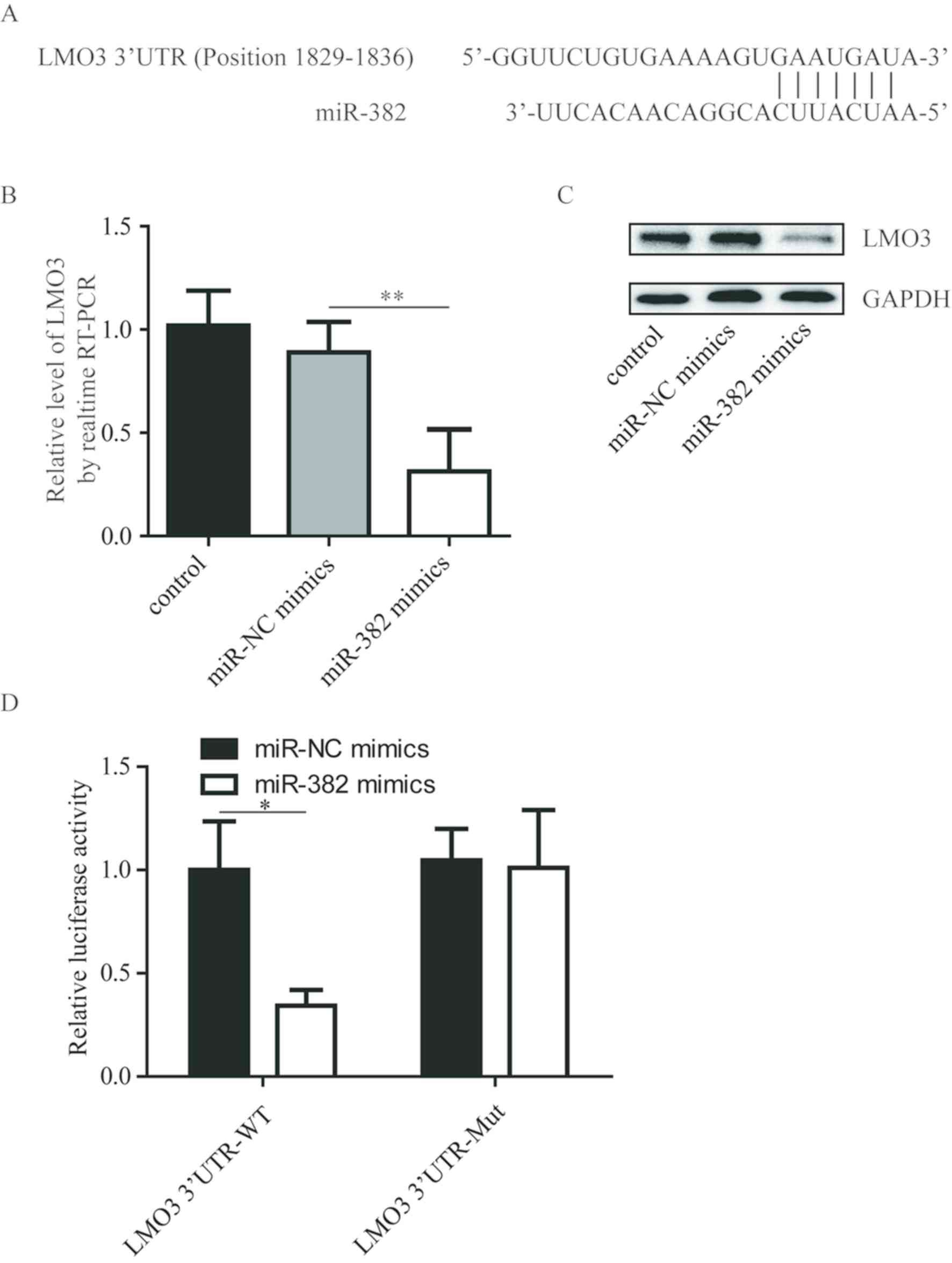
LMO3 is a target gene of miR-382
LMO3 has been reported to promote NSCLC growth and
metastasis (11). miRanda analysis
indicated that LMO3 was a potential target gene of miR-382
(Fig. 3A). In A549 cells,
overexpression of miR-382 decreased LMO3 mRNA and protein
expression levels (Fig. 3B and C).
Subsequently, whether LMO3 was a direct target gene of miR-382 was
investigated using the dual luciferase assay. The results
demonstrated that miR-382 mimics significantly reduced the
luciferase activity in 293 cells transfected with LMO3 3′UTR-WT
compared with transfection of miR-NC mimics (Fig. 3D). In addition, miR-382
overexpression did not alter the luciferase activity of 293 cells
transfected with LMO3 3′UTR-Mut (Fig.
3D).
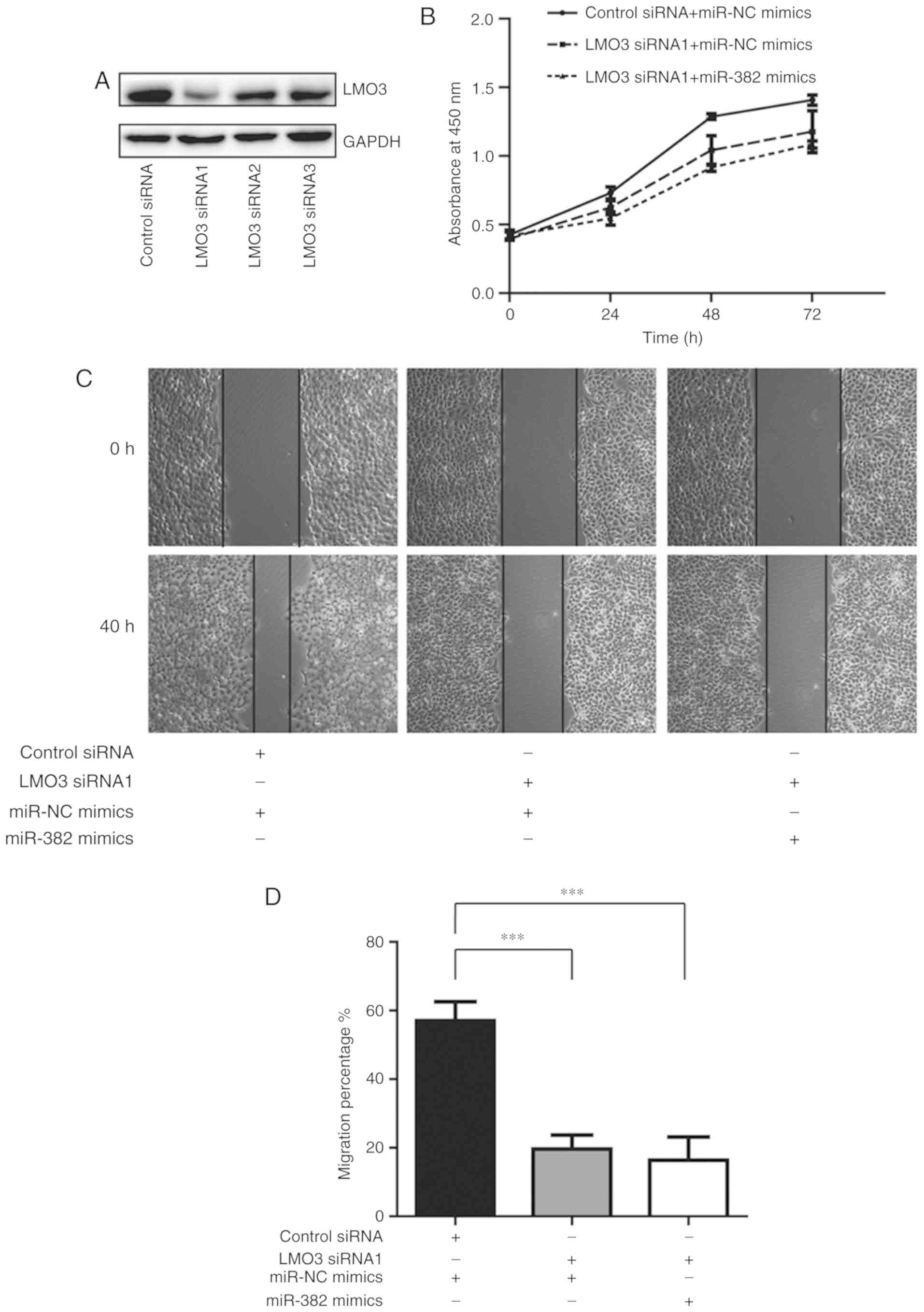
miR-382 inhibits A549 cell
proliferation and migration via LMO3
To further investigate whether miR-382 is associated
with the regulation of A549 cell proliferation and migration,
A549-cell proliferation and migration abilities were detected
following combining miR-382 overexpression with LMO3-knockdown. In
A549 cells, decreased LMO3 protein expression levels were observed
following transfection with LMO3 siRNA1, LMO3 siRNA2 or LMO3 siRNA3
(Fig. 4A). LMO3 siRNA1 exhibited the
highest efficiency in regulating LMO3 protein expression levels and
was therefore used for the following experiments. Results indicated
that silencing LMO3 markedly reduced A549 cell proliferation
ability; however, combined LMO3 knockdown and miR-382
overexpression did not significantly induce further cell growth
inhibition (Fig. 4B). Furthermore,
compared with the LMO3 knockdown group, there was no significant
difference in the cell migration ability in cells with combined
LMO3 silencing and miR-382 overexpression (Fig. 4C and D). These data indicated that,
miR-382 did not show significant effects on the cell proliferation
and cell migration of A549 cells without LMO3, suggesting that
miR-382 may primarily rely on the negative regulation of LMO3 to
suppress NSCLC cell growth and migration.
LMO3 is overexpressed and negatively
associated with miR-382 in tumor tissues from patients with
NSCLC
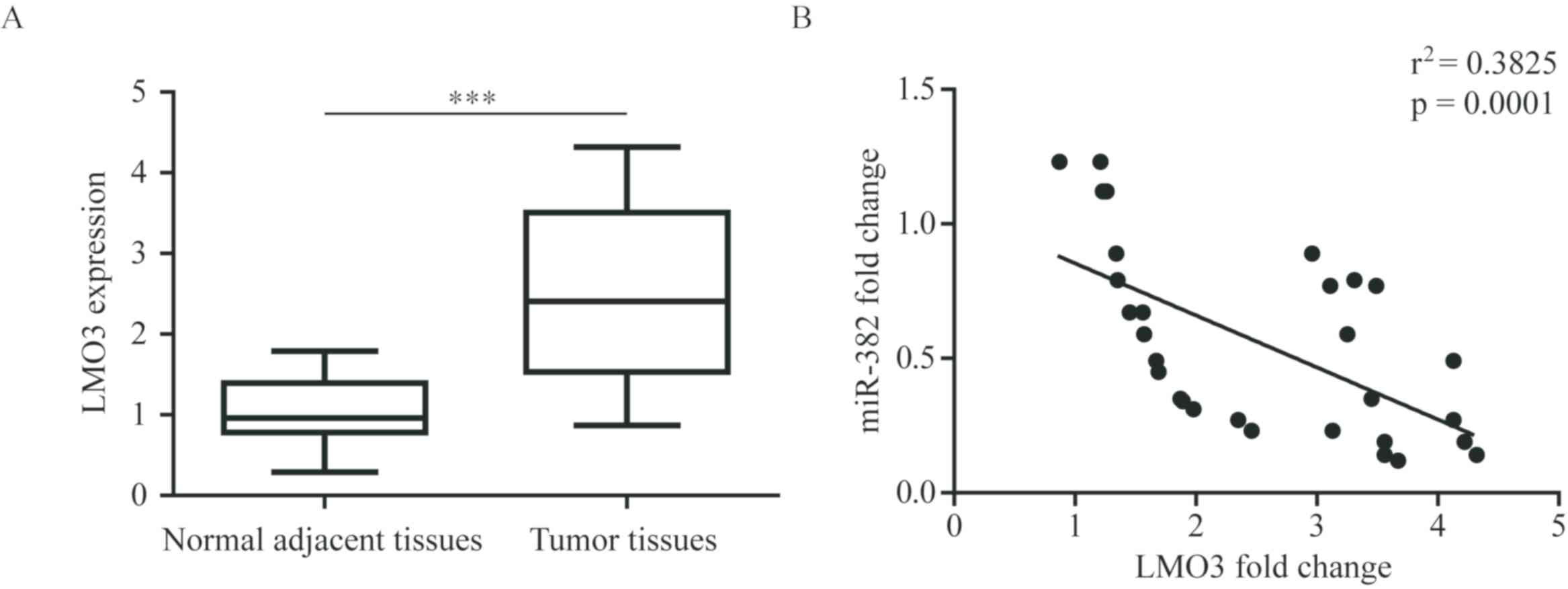
LMO3 expression levels in patients with NSCLC were
investigated. Significant elevation of LMO3 mRNA expression levels
were detected in tumor tissues compared with normal adjacent
tissues in patients with NSCLC (Fig.
5A). Additionally, via analysis of the miR-382 and LMO3 mRNA
expression levels in NSCLC tumor tissues, a significant negative
association between miR-382 and LMO3 mRNA expression levels was
revealed (Fig. 5B). Therefore,
miR-382 may promote cancer cell growth and metastasis via targeting
LMO3 in patients with NSCLC.
Expression of miR-382 and LMO3 in lung
carcinoma tissues
The fold changes of miRNA-382 and LMO3 were tested
in the 30 patients in the present study (10 male and 20 female)
with respect to various patient characteristics, including sex,
age, tumor size, histological grade and lymph node metastasis. As
presented in Table I, there was no
significant difference in miRNA-382 expression according to sex
(P=0.938), age (P=0.331) or histological grade (P=0.156); however,
low miRNA-382 expression was significantly associated with large
tumor size (P=0.006) and lymph node metastasis (P=0.027).
Furthermore, there was no significant difference in LMO3 expression
according to sex (p=0.418) or age (P=0.234); however, high LMO3
expression was significantly associated with large tumor size
(P=0.010), low histological grade (P=0.002) and lymph node
metastasis (P=0.002).
 | Table I.Expression of miR-382 and LMO3 in lung
carcinoma patients' tissues. |
Table I.
Expression of miR-382 and LMO3 in lung
carcinoma patients' tissues.
| Factors | Patients (n) | miR-382 (fold
change) | P-value | LMO3 (fold
change) | P-value |
|---|
| Sex |
|
| 0.938 |
| 0.418 |
| Male | 10 | 0.549±0.123 |
| 2.332±0.114 |
|
|
Female | 20 | 0.596±0.071 |
| 2.266±0.242 |
|
| Age (years) |
|
| 0.331 |
| 0.234 |
|
<60 | 17 | 0.610±0.084 |
| 2.454±0.183 |
|
| ≥60 | 13 | 0.485±0.102 |
| 2.363±0.230 |
|
| Tumor size (cm) |
|
| 0.006 |
| 0.010 |
| ≥5 | 16 | 0.401±0.062 |
| 2.314±0.182 |
|
|
<5 | 14 | 0.734±0.103 |
| 2.106±0.234 |
|
| Histological
grade |
|
| 0.156 |
| 0.002 |
|
Well-intermediate
differentiation | 14 | 0.473±0.063 |
| 2.243±0.150 |
|
| Poor
differentiation | 16 | 0.651±0.111 |
| 2.062±0.231 |
|
| Lymph node
metastasis |
|
| 0.027 |
| 0.002 |
| No | 22 | 0.429±0.062 |
| 2.275±0.171 |
|
| Yes | 8 | 0.701±0.113 |
| 2.502±0.133 |
|
Discussion
The expression levels of many miRNAs have previously
been suggested as promising biomarkers for aiding the diagnosis,
prognosis and drug-sensitivity prediction of cancer (14,15). In
the present study, it was demonstrated that miR-382 could inhibit
cell proliferation and migration in NSCLC. In addition, it was
demonstrated that LMO3, an oncogene, was a direct target of
miR-382. Using LMO3 siRNA, it was also demonstrated that the
anti-cancer effect of miR-382 was primarily reliant on the
regulation of LMO3.
Via targeting oncogenes or tumor suppressors,
deregulation of miRNAs may promote or inhibit cancer initiation and
development. Notably, the role of miR-382 during cancer progression
is considered controversial. In a previous study, miR-382 was
demonstrated to downregulate phosphatase and tensin homolog and
impacted the downstream protein kinase B/mechanistic target of
rapamycin signaling pathway to promote angiogenesis in MKN1 human
gastric cancer cells (16). However,
another study indicated that miR-382 targeted tumor suppressor
Ras-like, estrogen-regulated, growth inhibitor to activate the
Ras/extracellular signal-regulated kinase signaling pathway and
promoted cancer proliferation and metastasis in breast cancer
(17). In addition, miR-382 was
demonstrated to inhibit cancer cell growth in osteosarcoma
(18). It was demonstrated in the
present study that the miR-382 expression level was decreased in
NSCLC tumor tissues and NSCLC cell lines. Furthermore, in
vitro experiments indicated that miR-382 mimics greatly
inhibited A549 cell growth and migration. These data were
consistent with a recent study, which indicated that miR-382
inhibits NSCLC progression via targeting SET domain-containing 5
(19).
LMO3 is a well-characterized oncogene and has been
reported to be associated with many types of cancer, including
NSCLC (10,11). Several miRNAs have been reported to
directly or indirectly regulate LMO3 to confer anti-cancer effects.
For example, low levels of miR-101 have been suggested to lead to
hypomethylation of the LMO3 promoter via targeting eEnhancer of
zeste homolog 2, EED and DNA (cytosine-5)-methyltransferase 3A,
thus contributing to LMO3 overexpression in glioma cells (20). In the present study, it was
demonstrated that LMO3 was negatively associated with miR-382 in
tumor tissues from patients with NSCLC. Notably, overexpression of
miR-382 decreased LMO3 mRNA and protein expression in A549 cells.
Furthermore, using the luciferase assay, it was indicated that LMO3
was a direct target of miR-382. The present study also demonstrated
that miR-382 mimics did not significantly enhance the inhibition of
cell proliferation and migration in LMO3-silenced A549 cells.
Therefore, these results suggested that miR-382 may be a tumor
suppressor in NSCLC, functioning via targeting of LMO3.
In conclusion, the present findings uncovered a
tumor suppressor role of miR-382 in NSCLC via directly targeting
the oncogene, LMO3. These findings suggest that miR-382 may be a
promising diagnostic biomarker for NSCLC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JZ conceived, designed and supervised the present
study. JZ and DC developed the methodology, reviewed the manuscript
and analyzed, and interpreted the data. DC, YZ, YL, FS, ZZ and JZ
acquired the data.
Ethics approval and consent to
participate
All patients provided written informed consent and
the Ethics Committee of Zhangzhou Hospital Affiliated to Fujian
Medical University approved the present study.
Patient consent for publication
All patients provided written informed consent.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Toloza EM, Harpole L, Detterbeck F and
McCrory DC: Invasive staging of non-small cell lung cancer: A
review of the current evidence. Chest. 123 (Suppl 1):S157–S166.
2003. View Article : Google Scholar
|
|
3
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hwang HW and Mendell JT: MicroRNAs in cell
proliferation, cell death, and tumorigenesis. Br J Cancer. 96
(Suppl):R40–R44. 2007.PubMed/NCBI
|
|
5
|
Li J, Dong J, Li S, Xia W, Su X, Qin X,
Chen Y, Ding H, Li H, Huang A, et al: An alternative
microRNA-mediated post-transcriptional regulation of GADD45A by p53
in human non-small-cell lung cancer cells. Sci Rep. 7:71532017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ye Y, Zhuang J, Wang G, He S, Ni J, Xia W
and Wang J: microRNA-605 promotes cell proliferation, migration and
invasion in non-small cell lung cancer by directly targeting LATS2.
Exp Ther Med. 14:867–873. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu PL, Liu WL, Chang JM, Chen YH, Liu YP,
Kuo HF, Hsieh CC, Ding YS, Chen WW and Chong IW: MicroRNA-200c
inhibits epithelial-mesenchymal transition, invasion, and migration
of lung cancer by targeting HMGB1. PLoS One. 12:e01808442017.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Foroni L, Boehm T, White L, Forster A,
Sherrington P, Liao XB, Brannan CI, Jenkins NA, Copeland NG and
Rabbitts TH: The rhombotin gene family encode related LIM-domain
proteins whose differing expression suggests multiple roles in
mouse development. J Mol Biol. 226:747–761. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dawid IB, Breen JJ and Toyama R: LIM
domains: Multiple roles as adapters and functional modifiers in
protein interactions. Trends Genet. 14:156–162. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aoyama M, Ozaki T, Inuzuka H, Tomotsune D,
Hirato J, Okamoto Y, Tokita H, Ohira M and Nakagawara A: LMO3
interacts with neuronal transcription factor, HEN2, and acts as an
oncogene in neuroblastoma. Cancer Res. 65:4587–4597. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Song YF, Hong JF, Liu DL, Lin QA, Lan XP
and Lai GX: miR-630 targets LMO3 to regulate cell growth and
metastasis in lung cancer. Am J Transl Res. 7:1271–1279.
2015.PubMed/NCBI
|
|
12
|
Clark JR, Rumcheva P and Veness MJ:
Analysis and comparison of the 7th edition American Joint Committee
on Cancer (AJCC) nodal staging system for metastatic cutaneous
squamous cell carcinoma of the head and neck. Ann Surg Oncol.
19:4252–4258. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ma J, Lin Y, Zhan M, Mann DL, Stass SA and
Jiang F: Differential miRNA expressions in peripheral blood
mononuclear cells for diagnosis of lung cancer. Lab Invest.
95:1197–1206. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jiang Z, Yin J, Fu W, Mo Y, Pan Y, Dai L,
Huang H, Li S and Zhao J: MiRNA 17 family regulates
cisplatin-resistant and metastasis by targeting TGFbetaR2 in NSCLC.
PLoS One. 9:e946392014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Seok JK, Lee SH, Kim MJ and Lee YM:
MicroRNA-382 induced by HIF-1alpha is an angiogenic miR targeting
the tumor suppressor phosphatase and tensin homolog. Nucleic Acids
Res. 42:8062–8072. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ho JY, Hsu RJ, Liu JM, Chen SC, Liao GS,
Gao HW and Yu CP: MicroRNA-382-5p aggravates breast cancer
progression by regulating the RERG/Ras/ERK signaling axis.
Oncotarget. 8:22443–22459. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xu M, Jin H, Xu CX, Sun B, Mao Z, Bi WZ
and Wang Y: miR-382 inhibits tumor growth and enhance
chemosensitivity in osteosarcoma. Oncotarget. 5:9472–9483. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen T, Ren H, Thakur A, Yang T, Li Y,
Zhang S, Wang T and Chen M: miR-382 inhibits tumor progression by
targeting SETD8 in non-small cell lung cancer. Biomed Pharmacother.
86:248–253. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu X, Lei Q, Yu Z, Xu G, Tang H, Wang W,
Wang Z, Li G and Wu M: MiR-101 reverses the hypomethylation of the
LMO3 promoter in glioma cells. Oncotarget. 6:7930–7943.
2015.PubMed/NCBI
|