Introduction
Parkinson's disease (PD) is a progressive
neurodegenerative disorder that commonly leads to the loss of motor
function (akinetic-rigid, tremor and postural instability gait
disorder), non-motor symptoms (NMS; sleep disorder and autonomic
dysfunction) and cognitive decline (1). Currently available treatments for PD do
not delay or prevent disease progression. Strong evidence has
emerged that mitochondrial dysfunction, inflammation and increased
oxidative stress have pivotal roles in the pathogenesis of PD and
these processes provide a scientific basis for testing potential
‘neuroprotective’ agents.
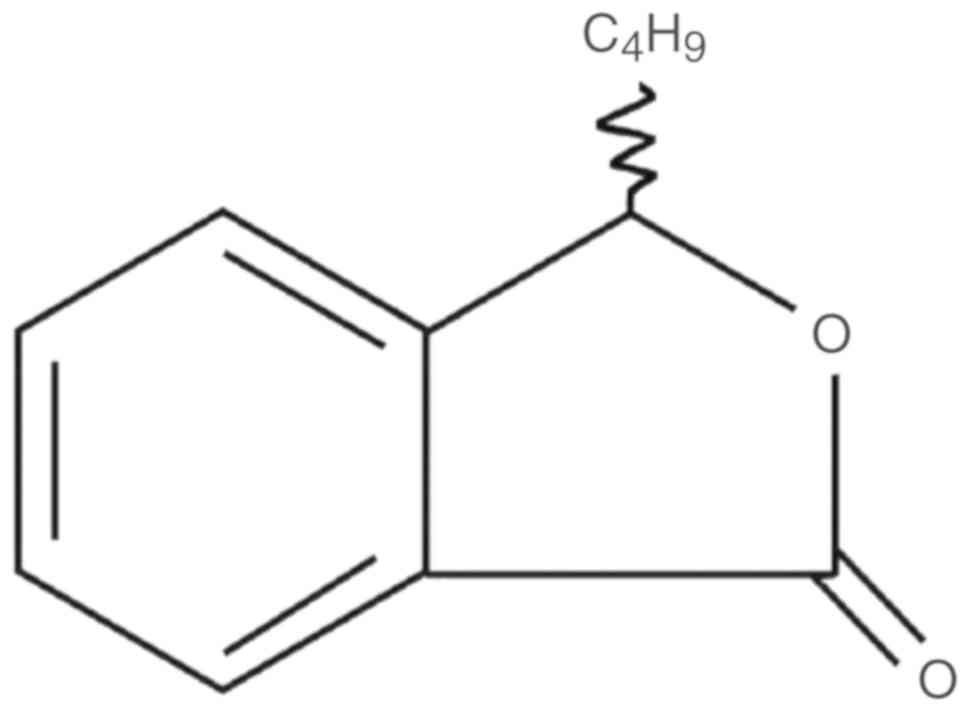
DL-3-n-butylphthalide (NBP) (Fig. 1) is a synthetic chiral compound
developed from L-3-n-butylphthalide that contains the L- and
D-isomers of butylphthalide. NBP was initially isolated as a pure
component from the seeds of Apium graveolens in 1978 and was
first synthesized in 1980 by researchers at the Institute of
Medicine of the Chinese Academy of Medical Sciences (2). Pharmacological tests at that time
indicated that NBP had significant central nervous system (CNS)
effects, including anti-convulsant and sedative activities, but the
dose at which NBP had anti-epileptic effects was close to its
maximum tolerated dose. The pharmacological effects of NBP on
stroke were demonstrated in whole animals, organs, tissues, cells
and at the molecular level, and included a significant reduction in
cerebral infarction, protection of mitochondrial function and
improvement of brain metabolism (2,3). NBP was
approved by the State Food and Drug Administration of China as a
therapeutic drug for the treatment of ischemic stroke in 2005 based
on the results of multicenter phase 2 and 3 randomized controlled
clinical trials and was consistently reported to improve neurologic
function after stroke, with good safety and tolerability (4,5). NBP has
also been reported to prevent oxidative damage and mitochondrial
dysfunction, protect against neurotoxicity via an autophagic
mechanism in a 1-methyl-4-phenylpyridinium (+)-induced cellular
model of PD (6), and protect
dopamine neurons in rotenone-induced models of PD (7). Preliminary experiments performed in our
group indicated that NBP significantly improved dyskinesia, reduced
microglial activation, decreased nuclear α-synuclein deposition and
increased the survival of tyrosine hydroxylase-positive cells in
the substantia nigra of the lipopolysaccharide (LPS)-induced PD
mouse model. Therefore, it was hypothesized that NBP may have
therapeutic efficacy in patients with PD, and the present study was
designed to evaluate this.
Materials and methods
Study design, participants and
oversight
The present study was a prospective, single-center,
parallel-group, randomized controlled trial. Patients were
self-referred or recruited from the First Affiliated Hospital of
Bengbu Medical College (Bengbu, China) between September 2014 and
December 2016. The final follow-up evaluation was in December 2016.
Patients were eligible for participation in the study if they i)
had idiopathic PD confirmed by a movement disorder specialist,
according to the Parkinson's Disease Society Brain Bank criteria
(8); ii) had been receiving a stable
medication regimen for a duration of ≥6 weeks or were early PD
patients who had no prior medication treatment and were receiving
their first consultation; iii) were between 40 and 80 years of age;
and iv) had a family member or friend who was willing to also
participate. Exclusion criteria included any factor that may have
precluded completion of neuropsychological testing; clinically
significant gastrointestinal, hepatic, renal, respiratory or
cardiovascular system disease; infection; alcoholism; drug
addiction; or known hypersensitivity to celery. Patients were
evaluated at the Department of Neurology of the First Affiliated
Hospital of Bengbu Medical College (Bengbu, China).
Interventions
NBP was selected, as this additional treatment has
been used in the clinic for patients with brain infarction and
vascular cognitive impairment but no dementia with good safety and
tolerability (2,3). In addition, NBP protected dopamine
neurons in in vivo and in vitro models of PD. All
participants continued their stable medical regimens under the care
of their physicians. Patients were matched by age, gender and
off-medication Unified Parkinson's Disease Rating Scale motor
subscale (UPDRS-III) scores, and randomly assigned at a 1:1 ratio
to the NBP group, which received oral NBP 200 mg three times daily
based on the dose approved for stroke that is known to exert CNS
effects for 24 weeks (5), or the
control group. All patients continued their originally prescribed
medication regimen during the 48 weeks of the study. Furthermore,
in this time, no new PD treatment was provided to the study
participants except for the experimental treatment with NBP.
Study procedures
All assessments were performed at the Department of
Neurology of the First Affiliated Hospital of Bengbu Medical
College (Bengbu, China) after a 12-h overnight withdrawal of
dopaminergic medication. Off-medication assessments were completed
in the morning (9). Motor signs were
evaluated using the UPDRS-III and, based on the predominant motor
features in the UPDRS III motor examination section score, the
scores were subtyped into tremor scores and non-tremor
(bradykinesia plus rigidity) scores. Tremor scores were derived
from the sum of UPDRS III items 20 (tremor at rest) and 21 (action
or postural tremor of hands). Non-tremor scores were derived from
the sum of UPDRS III items 18 (speech), 19 (facial expression), 22
(rigidity), 23 (finger taps), 24 (hand movement), 25
(diadochokinesia), 26 (leg flexibility), 27 (arising from the
chair), 28 (posture), 29 (gait), 30 (postural stability), and 31
(body bradykinesia and hypokinesia) (10). The quality of sleep was also assessed
using the Pittsburgh sleep quality index (PSQI), and the quality of
life using the Parkinson's disease 39-items questionnaire (PDQ-39),
following the methods described in previous studies (11).
Follow-up
Patients were examined at 12 (midpoint), 24 (end of
treatment) and 48 weeks (follow-up evaluation). The entire baseline
assessment procedure was repeated. The same evaluator who evaluated
the baseline indicators performed all subsequent off-medication
evaluations and recorded the current medications and adverse events
at each visit.
Randomization and blinding
As patients were assigned, the statistician informed
the assigned treatment and study coordinator that randomization had
occurred. Only the statistician and the coordinator who recruited
the patients knew the treatment assignment, but none of these
individuals had a role in data collection. The research personnel
involved in data collection were blinded to the treatment
assignments. The patients knew their treatment assignment, but did
not know the study hypothesis.
Statistical analysis
The qualitative information in the baseline data was
analyzed using the χ2 test and the quantitative data
were analyzed using the independent-samples t-test. The primary
outcome measures were the change in the off-medication non-tremor
score, tremor score, PSQI and PDQ-39 scores from baseline to 12, 24
and 48 weeks. To estimate the treatment effects, comparisons
between groups were performed using the Mann-Whitney U-test for two
sets of independent samples, as the data did not follow a normal
distribution. Statistical analyses were performed using SPSS 17.0
software (SPSS, Inc., Chicago, IL, USA). All statistical tests were
two-sided P<0.05 was considered to indicate a statistically
significant difference.
Results
Patients
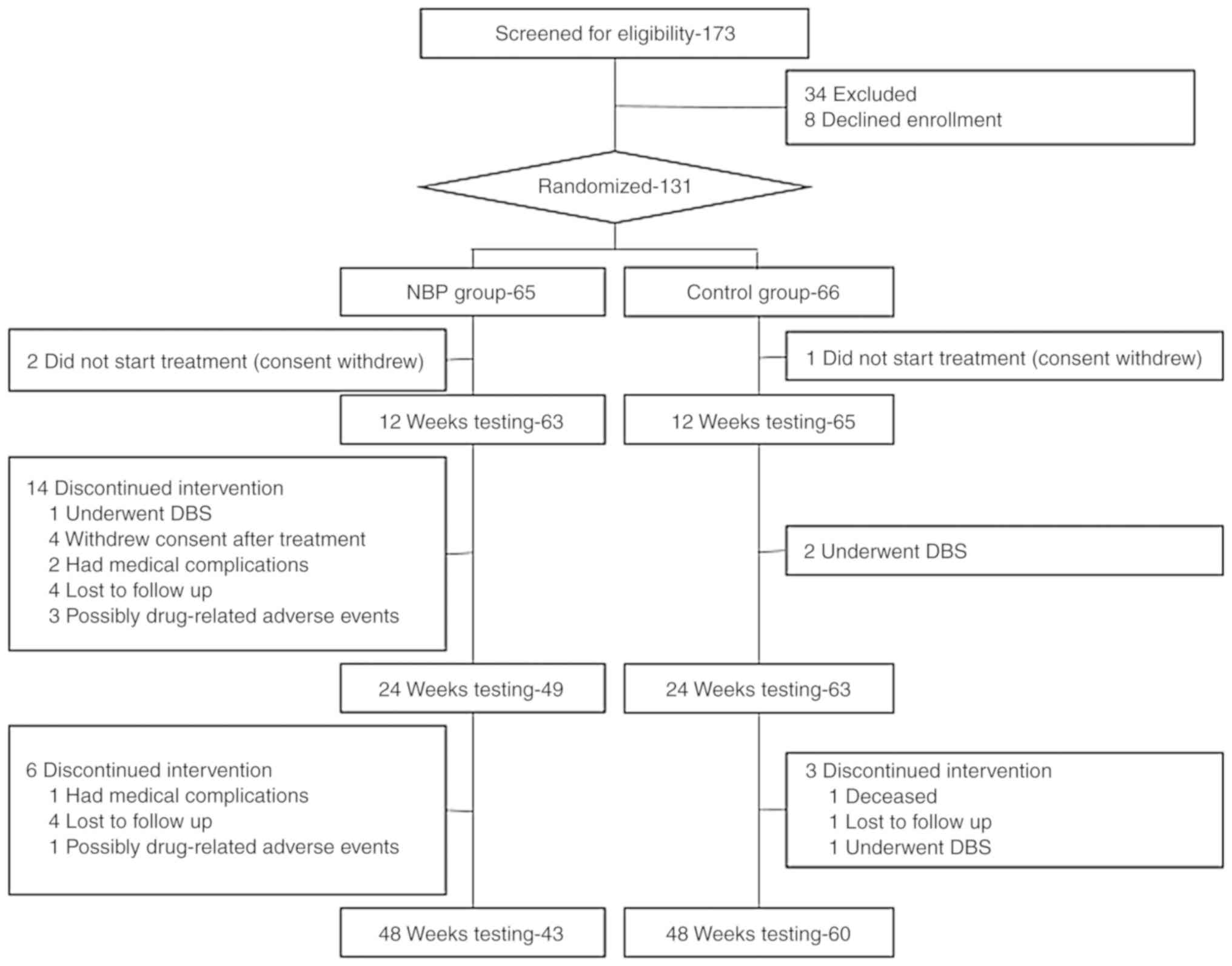
Between September 2014 and December 2016, 173
patients with PD were screened for study participation and 131
patients were randomly assigned to the NBP group or the control
group. A total of 103 patients were followed up until the end of
the study, including 43 patients in the NBP group and 60 patients
in the control group (Fig. 2). The
baseline characteristics were not statistically different between
the study groups (Table I). The
clinical profiles of the enrolled patients were consistent with the
diagnosis of PD. Most enrolled patients were taking concomitant
medications, with the most common one being the dopamine receptor
agonist L-dopamine, but there were no significant differences in
medication usage between the treatment group and the control
group.
 | Table I.Characteristics of patients at
baseline, by treatment group. |
Table I.
Characteristics of patients at
baseline, by treatment group.
| Characteristic | NBP (n=43) | Control (n=60) | χ2/t | P-value |
|---|
| Age (years) | 60.00±9.82 | 61.92±9.77 | −0.980 | 0.330 |
| Duration of disease
(months) | 38.30±26.84 | 48.08±31.36 | −1.655 | 0.101 |
| UPDRS, off
medication | 47.40±23.96 | 53.32±26.76 | −1.156 | 0.250 |
| UPDRS III, off
medication | 30.72±14.87 | 34.42±17.20 | −1.164 | 0.247 |
| MMSE | 26.51±5.84 | 24.53±6.90 | 1.572 | 0.119 |
| ESS | 5.37±4.81 | 5.52±5.30 | −0.142 | 0.887 |
| PSQI | 8.63±6.94 | 6.72±4.18 | 1.608 | 0.113 |
| FSS | 3.28±2.02 | 3.73±2.12 | −1.093 | 0.277 |
| HRSD | 14.16±8.87 | 15.43±8.28 | −0.745 | 0.458 |
| HAMA | 8.16±5.91 | 9.02±5.50 | −0.753 | 0.453 |
| PDQ-39, off
medication | 40.49±27.80 | 48.37±29.41 | −1.372 | 0.173 |
| Gender |
|
| 0.434 | 0.510 |
| Male | 23 (53.5) | 36 (60.0) |
|
|
|
Female | 20 (46.5) | 24 (40.0) |
|
|
| Levodopa equivalent
dose |
|
| 1.131 | 0.288 |
| No | 18 (41.9) | 19 (31.7) |
|
|
| Yes | 25 (58.1) | 41 (68.3) |
|
|
| H-Y stage, off
medication |
|
| 5.072 | 0.407 |
| 1 | 12 (27.9) | 14 (23.3) |
|
|
| 1.5 | 10 (23.3) | 12 (20.0) |
|
|
| 2 | 6 (14.0) | 5 (8.3) |
|
|
| 3 | 6 (14.0) | 17 (28.3) |
|
|
| 4 | 9 (20.9) | 10 (16.7) |
|
|
| 5 | 0 (0.0) | 2 (3.3) |
|
|
Motor signs and medication status
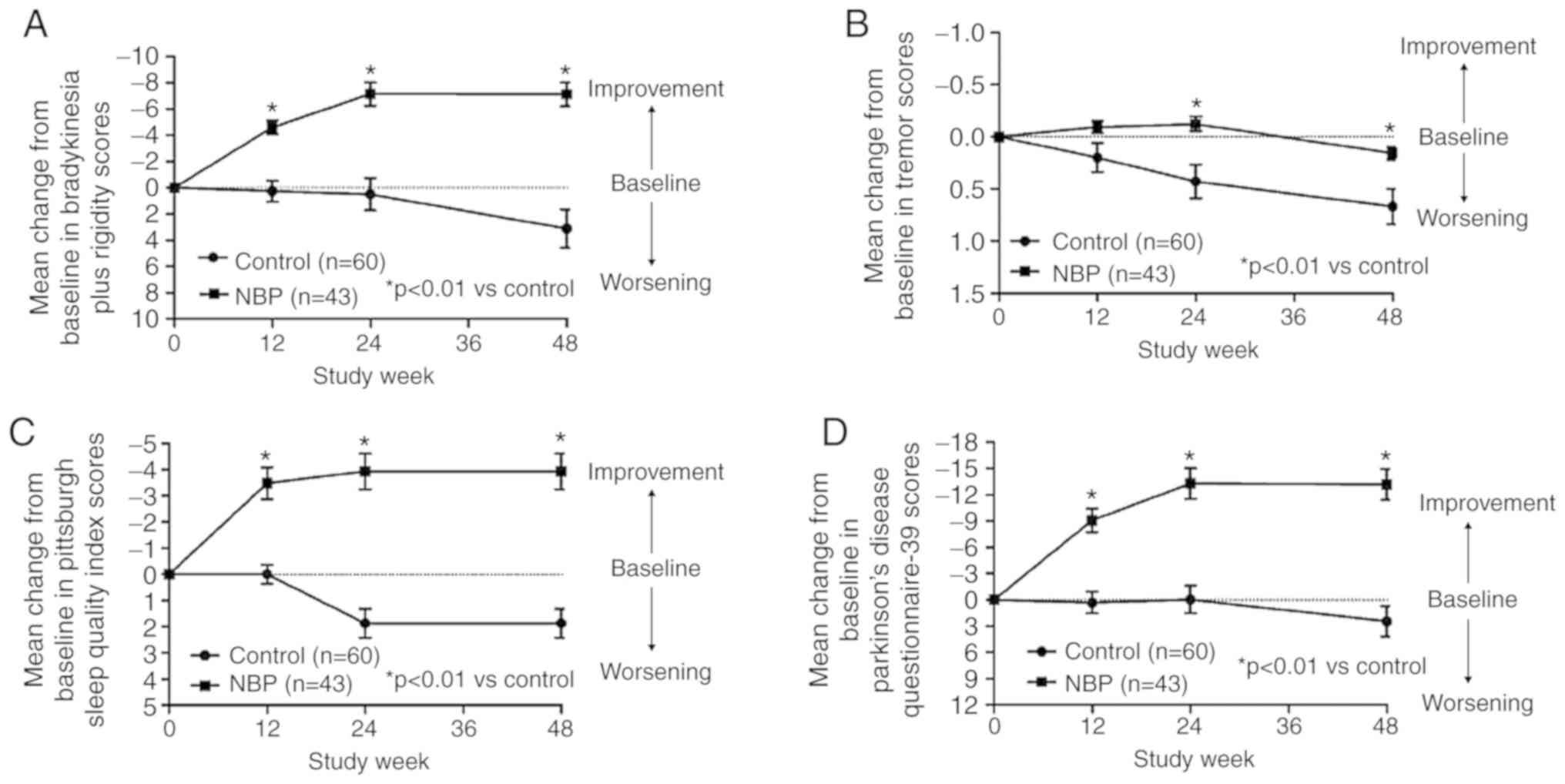
The mean off-medication, bradykinesia plus rigidity
score and tremor score decreased from baseline at 12, 24 and 48
weeks for the patients in the NBP treatment group, The mean
off-medication tremor score decreased slightly at 12 and 24 weeks,
but increased at 48 weeks. The change from the baseline in the
bradykinesia plus rigidity and tremor scores were statistically
significant when compared with the control group. (Table II; Fig.
3A and B).
 | Table II.Bradykinesia plus rigidity, tremor,
sleep quality and quality of life at each follow-up visit by
treatment group. |
Table II.
Bradykinesia plus rigidity, tremor,
sleep quality and quality of life at each follow-up visit by
treatment group.
|
| Score at follow-up
visit | Change from
baselinea |
|
|
|---|
|
|
|
|
|
|
|---|
| Time-point | Control (n=60) | NBP (n=43) | Control (n=60) | NBP (n=43) | Z-value | P-valueb |
|---|
| Bradykinesia plus
rigidity score, off medication |
|
Baseline | 33.05±17.57 | 28.86±15.85 |
|
|
|
|
| 12
weeks | 33.33±18.24 | 24.26±13.57 | 0.28±6.21 | −4.60±3.51 | −7.562 | <0.001 |
| 24
weeks | 33.55±18.02 | 21.72±11.71 | 0.50±9.48 | −7.14±5.81 | −7.625 | <0.001 |
| 48
weeks | 36.18±19.38 | 21.74±11.77 | 3.13±11.28 | −7.12±5.96 | −7.611 | <0.001 |
| Tremor score, off
medication |
|
Baseline | 48.10±29.22 | 40.49±27.80 |
|
|
|
|
| 12
weeks | 48.43±28.88 | 31.42±22.66 | 0.33±9.54 | −9.07±8.82 | −8.432 | <0.001 |
| 24
weeks | 48.08±28.07 | 27.23±18.15 | −0.02±12.26 | −13.26±11.52 | −8.022 | <0.001 |
| 48
weeks | 50.55±28.61 | 27.33±18.18 | 2.45±13.52 | −13.16±11.53 | −7.963 | <0.001 |
| Pittsburgh sleep
quality index scores, off medication |
|
Baseline | 15.43±8.28 | 14.16±8.87 |
|
|
|
|
| 12
weeks | 15.43±8.28 | 9.93±5.94 | 0.00±2.82 | −3.47±3.99 | −3.945 | <0.001 |
| 24
weeks | 15.60±8.15 | 9.70±5.78 | 1.88±4.22 | −3.93±4.48 | −5.361 | <0.001 |
| 48
weeks | 15.60±8.15 | 9.70±5.78 | 1.88±4.22 | −3.93±4.48 | −5.361 | <0.001 |
| Parkinson's disease
39-items questionnaire scores, off medication |
|
Baseline | 48.10±29.22 | 40.49±27.80 |
|
|
|
|
| 12
weeks | 48.43±28.88 | 31.42±22.66 | 0.33±9.54 | −9.07±8.82 | −8.432 | <0.001 |
| 24
weeks | 48.08±28.07 | 27.23±18.15 | 0.02±12.26 | −13.26±11.52 | −8.022 | <0.001 |
| 48
weeks | 50.55±28.61 | 27.33±18.18 | 2.45±13.52 | −13.16±11.53 | −7.963 | <0.001 |
Sleep quality and medication
status
The changes in the mean PSQI score for the
NBP-treatment group from baseline to 12 and 24 weeks were all
statistically significant compared with those in the control group.
The scores remained constant at 48 weeks (Table II; Fig.
3C). It was indicated that NBP had a beneficial effect on the
sleep quality of PD patients. During the first six months of the
study, two patients in the NBP group and 15 patients in the control
group required an increase in medication, however, this study did
not add other drugs according to the experimental design.
Quality of life
The mean PDQ-39 score decreased from baseline for
the NBP treatment group at 12 and 24 weeks, and remained constant
at 48 weeks. The difference in the PDQ-39 score changes in the NBP
group from the control group was significant (Table II; Fig.
3D). It was therefore indicated that NBP had a beneficial
effect on the quality of life of PD patients.
Adverse events
Only three adverse events directly associated with
the treatment were noted in the NBP group: NBP allergy
characterized by itching and skin rash one week after treatment (1
patient, drug rash improved after discontinuing), slight elevation
of ALT 24 weeks after treatment (1 patient, ALT returned to normal
1 week after stopping the drug) and a mild gastrointestinal
reaction (2 patients, reactions improved 30 min after taking the
medicine).
Discussion
The pathogenesis of PD includes synaptic
transmission, the cytoplasmic cycle, lysosomal autophagy and
multiple molecular mechanisms, including mitochondrial metabolism.
Thus far, no effective medication has been developed to interfere
with this specific pathogenesis. Clinical trials, including those
that evaluated adeno-associated virus serotype 2-nerve growth
factor, coenzyme Q10, creatine, pramipexole and pioglitazone, all
reported negative results (12,13). As
the first randomized, controlled, blinded clinical trial of NBP for
PD, the present study should be considered exploratory. The methods
and results of the present study may provide an important reference
for patient selection, outcome measures and study duration of
future PD drug trials. Of note, the results of the present study
indicated that NBP may be a promising treatment option for PD.
The end-points of the present study were
off-medication tremor score and non-tremor (bradykinesia plus
rigidity) score based on the predominant motor features in the
UPDRS III motor examination section score, rather than generally
assessing the patients' motor symptoms using the UPDRS III
scores.
According to previous reports, 50% of patients with
PD complained of slowness of movement, including the initiation and
execution of movements, particularly sequential and volitional
actions, and 40% of patients are likely to complain of
tremulousness of the hand at rest (14). Based on the different abovementioned
clinical symptoms, patients with PD were subtyped into one of two
clinical groups, non-tremor-dominant subtype (ART) and
tremor-dominant subtype (TDT), and evaluated using the non-tremor
score and tremor score (15).
ART and TDT have different clinical signs, markers
and pathogeneses. In ART, PD progresses more rapidly (16), and in the early stage, the
dopaminergic system is more severely impaired (17). Patients with the ART of PD have
consistently been reported to exhibit a broader array of NMS than
TDT, involving earlier autonomic features and later cognitive
impairment (1). Furthermore,
previous studies have indicated that the disease in NMS-dominant
phenotypes may begin peripherally in the gastrointestinal tract
(18) and link with the
heterogeneity of the initiating neuropathology to reflect the
convergence of deficits in multiple transmitter systems and
pathways, including the cholinergic, serotonergic and noradrenergic
systems, occurring in PD (19).
The present results demonstrated a favorable effect
of NBP in the treatment of patients with bradykinesia plus rigidity
symptoms. The efficacy of NBP in PD may be mediated by multiple
targets involved in the pathogenesis of this disorder. Data from
animal and cell models suggest that NBP protects dopaminergic
neurons by blocking the upregulation of intracellular reactive
oxygen species production, inhibiting a decrease in cleaved
caspase-3/caspase-3 and B-cell lymphoma 2 (Bcl-2)/Bcl-2-associated
X protein ratios (20), scavenging
free radicals (21), increasing the
number of synapses and apical dendritic thorns, attenuating neural
inflammatory responses by reducing oxidative stress, decreasing
nuclear fragmentation, retaining the mitochondrial membrane
potential (7), and activating
autophagy-mediated α-synuclein degradation (22).
Previous animal experiments performed by our group
indicated that NBP reduces microglial activation in mice with
LPS-induced PD. LPS is closely associated to the pathogenesis of PD
(23,24) and is derived from the components of
Gram-negative bacterial cells, which are common in the intestinal
flora. The intestinal flora is closely linked to the immune system,
including the nervous system (25),
and intestinal flora imbalances are associated with PD (23,26). A
dysregulation of the intestinal flora and intestinal inflammation
lead to increased intestinal mucosal permeability, resulting in LPS
entering the body and causing neuroinflammation. During this
process, the intestinal nerve is the first to be affected and
constipation and other non-motor symptoms appear at the early
stages. As these lesions move along the vagus nerve, the brain
stem, midbrain and cerebral cortex are involved, leading to sleep
disorders, symptoms of mobility disorders and cognitive impairment
(23,26,27). The
clinical manifestations for the PD subtype of this pathogenesis are
NMS at the early stage and primarily non-tremor (bradykinesia plus
rigidity) movement symptoms at the later stage (1). As expected, the results of the present
study indicated that NBP mainly improved the bradykinesia plus
rigidity symptom in patients with PD. Therefore, it is likely that
the mechanism of NBP in PD may be the attenuation of neural
inflammatory responses, which elicits a neuroprotective effect and
improves and prevents PD development.
The present results demonstrated a favorable effect
of NBP in the treatment of PD patients with sleep disorders. Sleep
disorders are a common NMS of PD that occurs due to lesions located
in the medulla oblongata in the brain stem. There are two possible
mechanisms by which NBP may improve sleep disorders in patients
with PD. First, similar to how NBP improves the bradykinesia plus
rigidity symptom of PD, NBP may exert neuroprotective effects
through attenuation of neuroinflammatory responses. Furthermore,
NBP was first purified from the seeds of Apium graveolens
(Water celery) in 1978 at the Institute of Medicine of the Chinese
Academy of Medical Sciences, inspired by a folk prescription for
‘boiling of sailboat canvas and celery seeds together in water for
the treatment of epilepsy’, and in 1980, the compound was
synthesized at the same institute for the first time (28). The pharmacological tests at that time
demonstrated that NBP exhibited significant effects on the CNS,
including anti-convulsant and sedative actions. This sedative
effect may be one of the mechanisms by which NBP improves sleep
disorders in PD.
Of note, the present study have several limitations.
First, it was not a double-blinded trial, as it was not possible to
perform blinding with respect to regular medication and compliance
with the follow-up questionnaire. Furthermore, the sample size was
small. Finally, the control group was not given a placebo drug.
However, these issues were minor, as the participants did not know
the group assignment until baseline testing was completed and the
evaluators did not know it either until the study was
completed.
In conclusion, the present preliminary study
suggested that 6 months of NBP treatment was effective for
improving bradykinesia plus rigidity and sleep quality in patients
with PD, providing a promising option for early intervention in
this disease. Future experiments with larger sample sizes and a
longer duration are required to further test the efficacy of NBP in
PD and its different subtypes.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China province in 2016 (grant no.
81641050) and The Strategic Priority Research Program (Pilot study)
‘Biological basis of aging and intervention strategies’ of the
Chinese Academy of Sciences (grant no. XDPB10).
Availability of data and materials
The datasets used and/or analysed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
HZ was responsible for drafting manuscript,
critically revising it for important knowledge content,
participating in the experimental design, and agreeing to be
responsible for all aspects of the work to ensure proper
investigation and resolution of issues related to the accuracy or
completeness of any part of the work. MY analyzed baseline data for
PD patients. WX acquired the data. MY analyzed and interpreted the
data. XL was involved in XL assessing the patients for fullfilling
the inclusion criteria, and revising the manuscript critically for
important intellectual content. YC designed the experiments,
drafted the manuscript, and critically modified important content.
All authors read and approved the final version of the
manuscript.
Ethical approval and consent to
participate
The present study received full approval from the
First Affiliated Hospital of Bengbu Medical College Institutional
Review Board (Bengbu, China). Informed consent was obtained from
each participant.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Marras C and Chaudhuri KR: Nonmotor
features of Parkinson's disease subtypes. Mov Disord. 31:1095–1102.
2016.
|
|
2
|
Liu CL, Liao SJ, Zeng JS, Lin JW, Li CX,
Xie LC, Shi XG and Huang RX: dl-3n-butylphthalide prevents stroke
via improvement of cerebral microvessels in RHRSP. J Neurol Sci.
260:106–113. 2007.
|
|
3
|
Zhao Y, Lee JH, Chen D, Gu X, Caslin A, Li
J, Yu SP and Wei L: DL-3-n-butylphthalide induced neuroprotection,
regenerative repair, functional recovery and psychological benefits
following traumatic brain injury in mice. Neurochem Int. 111:82–92.
2017.
|
|
4
|
Jia J, Wei C, Liang J, Zhou A, Zuo X, Song
H, Wu L, Chen X, Chen S, Zhang J, et al: The effects of
DL-3-n-butylphthalide in patients with vascular cognitive
impairment without dementia caused by subcortical ischemic small
vessel disease: A multicentre, randomized, double-blind,
placebo-controlled trial. Alzheimers Dement. 12:89–99. 2016.
|
|
5
|
Cui LY, Zhu YC, Gao S, Wang JM, Peng B, Ni
J, Zhou LX, He J and Ma XQ: Ninety-day administration of
dl-3-n-butylphthalide for acute ischemic stroke: A randomized,
double-blind trial. Chin Med J (Engl). 126:3405–3410. 2013.
|
|
6
|
Huang JZ, Chen YZ, Su M, Zheng HF, Yang
YP, Chen J and Liu CF: dl-3-n-Butylphthalide prevents oxidative
damage and reduces mitochondrial dysfunction in an MPP(+)-induced
cellular model of Parkinson's disease. Neurosc Lett. 475:89–94.
2010.
|
|
7
|
Xiong N, Huang J, Chen C, Zhao Y, Zhang Z,
Jia M, Zhang Z, Hou L, Yang H, Cao X, et al: Dl-3-n-butylphthalide,
a natural antioxidant, protects dopamine neurons in rotenone models
for Parkinson's disease. Neurobiol Aging. 33:1777–1791. 2012.
|
|
8
|
Hughes AJ, Daniel SE, Kilford L and Lees
AJ: Accuracy of clinical diagnosis of idiopathic Parkinson's
disease: A clinico-pathological study of 100 cases. J Neurol
Neurosurg Psychiatry. 55:181–184. 1992.
|
|
9
|
Corcos DM, Robichaud JA, David FJ,
Leurgans SE, Vaillancourt DE, Poon C, Rafferty MR, Kohrt WM and
Comella CL: A two-year randomized controlled trial of progressive
resistance exercise for Parkinson's disease. Mov Disord.
28:1230–1240. 2013.
|
|
10
|
Schiess MC, Zheng H, Soukup VM, Bonnen JG
and Nauta HJ: Parkinson's disease subtypes: Clinical classification
and ventricular cerebrospinal fluid analysis. Parkinsonism Relat
Disord. 6:69–76. 2000.
|
|
11
|
Thomas S, Reading J and Shephard RJ:
Revision of the physical activity readiness questionnaire (PAR-Q).
Can J Sport Sci. 17:338–345. 1992.
|
|
12
|
Volta M, Milnerwood AJ and Farrer MJ:
Insights from late-onset familial parkinsonism on the pathogenesis
of idiopathic Parkinson's disease. Lancet Neurol. 14:1054–1064.
2015.
|
|
13
|
Calabresi P and Di Filippo M: Multitarget
disease-modifying therapy in Parkinson's disease? Lancet Neurol.
14:975–976. 2015.
|
|
14
|
Spiegel J, Möllers MO, Jost WH, Fuss G,
Samnick S, Dillmann U, Becker G and Kirsch CM: FP-CIT and MIBG
scintigraphy in early Parkinson's disease. Mov Disord. 20:552–561.
2005.
|
|
15
|
Ahlskog JE: Diagnosis and differential
diagnosis of Parkinson's disease and parkinsonism. Parkinsonism
Relat Disord. 7:63–70. 2000.
|
|
16
|
Chiaravalloti A, Stefani A, Tavolozza M,
Pierantozzi M, Di Biagio D, Olivola E, Di Pietro B, Stampanoni M,
Danieli R, Simonetti G, et al: Different patterns of cardiac
sympathetic denervation in tremor-type compared to
akinetic-rigid-type Parkinson's disease: Molecular imaging with
¹23I-MIBG. Mol Med Rep. 6:1337–1342. 2012.
|
|
17
|
Schillaci O, Chiaravalloti A, Pierantozzi
M, Di Pietro B, Koch G, Bruni C, Stanzione P and Stefani A:
Different patterns of nigrostriatal degeneration in tremor type
versus the akinetic-rigid and mixed types of Parkinson's disease at
the early stages: Molecular imaging with 123I-FP-CIT SPECT. Int J
Mol Med. 28:881–886. 2011.
|
|
18
|
Shannon KM, Keshavarzian A, Dodiya HB,
Jakate S and Kordower JH: Is alpha-synuclein in the colon a
biomarker for premotor Parkinson's disease? Evidence from 3 cases.
Mov Disord. 27:716–719. 2012.
|
|
19
|
Espay AJ, LeWitt PA and Kaufmann H:
Norepinephrine deficiency in Parkinson's disease: The case for
noradrenergic enhancement. Mov Disord. 29:1710–1719. 2014.
|
|
20
|
Zhao J, Liu J, Xu E, Liu Y, Xie A and
Xiong H: dl-3-n-Butylphthalide attenuation of
methamphetamine-induced neurotoxicity in SH-SY5Y neuroblastoma
cells. Life Sci. 165:16–20. 2016.
|
|
21
|
Zhou J, Zhang YH, Song HZ, Ji H, Wang XL,
Wang L, Qian J, Ling JJ and Ping FF: 5d, a novel analogue of
3-n-butylphthalide, decreases NADPH oxidase activity through the
positive regulation of CK2 after ischemia/reperfusion injury.
Oncotarget. 7:39444–39457. 2016.
|
|
22
|
Liu K, Huang J, Chen R, Zhang T, Shen L,
Yang J and Sun X: Protection against neurotoxicity by an autophagic
mechanism. Braz J Med Biol Res. 45:401–407. 2012.
|
|
23
|
Qin L, Wu X, Block ML, Liu Y, Breese GR,
Hong JS, Knapp DJ and Crews FT: Systemic LPS causes chronic
neuroinflammation and progressive neurodegeneration. Glia.
55:453–462. 2007.
|
|
24
|
Pal GD, Shaikh M, Forsyth CB, Ouyang B,
Keshavarzian A and Shannon KM: Abnormal lipopolysaccharide binding
protein as marker of gastrointestinal inflammation in Parkinson
disease. Front Neurosci. 9:3062015.
|
|
25
|
Hand TW, Vujkovic-Cvijin I, Ridaura VK and
Belkaid Y: Linking the microbiota, chronic disease, and the immune
system. Trends Endocrinol Metab. 27:831–843. 2016.
|
|
26
|
Sampson TR, Debelius JW, Thron T, Janssen
S, Shastri GG, Ilhan ZE, Challis C, Schretter CE, Rocha S,
Gradinaru V, et al: Gut microbiota regulate motor deficits and
neuroinflammation in a model of Parkinson's disease. Cell.
167:1469–1480.e12. 2016.
|
|
27
|
McCann H, Cartwright H and Halliday GM:
Neuropathology of α-synuclein propagation and braak hypothesis. Mov
Disord. 31:152–160. 2016.
|
|
28
|
Yang Q, Hu YD, Wang XF and Zheng FS:
Dl-3n-butylphthalide reduces epileptiform activity through
GluA2-lacking calcium-permeable AMPARs in epilepsy models.
Oncotarget. 8:98242–98257. 2017.
|